Abstract
Background:
The use of antineoplastic agents is one of the important triggers of tumor lysis syndrome (TLS), but there is still a lack of comprehensive understanding of antineoplastic agents that may trigger TLS and the TLS risk differences between different antineoplastic agents.
Objectives:
This study aims to investigate the TLS risk of different antineoplastic agents and provide reference information for clinical practice.
Design:
Real-world adverse events data in the FDA Adverse Event Reporting System (FAERS) database were used as the basis for the disproportionality analysis.
Methods:
We reviewed the TLS reports in the FAERS database from 2004 to 2022 to summarize an antineoplastic agent list that was reported to trigger TLS, based on which we conducted disproportionality analysis to assess the TLS risk of each antineoplastic agent.
Results:
In all, 164 antineoplastic agents were reported to trigger TLS. On the whole, rituximab was the most reported antineoplastic agent in TLS reports, followed by cyclophosphamide, venetoclax, doxorubicin, and etoposide, while tagraxofusp was the antineoplastic agent with the highest adverse drug reaction (ADR) signal strength in signal detection, followed by floxuridine, pentostatin, tebentafusp, and venetoclax. Integrating ADR signal detection results, 129 of 164 antineoplastic agents showed at least one positive ADR signal, and six antineoplastic agents (bevacizumab, carboplatin, cisplatin, fluorouracil, lenvatinib, and paclitaxel) have the highest total number of positive signals. Further classifying the 164 antineoplastic agents into 46 chemical subgroups to conduct ADR signal detection, nitrogen mustard analogs were the most reported antineoplastic agent subclasses, followed by clusters of differentiation 20 inhibitors, and pyrimidine analogs, while clusters of differentiation 22 inhibitors were the antineoplastic agent subclass with the highest ADR signal strength, followed by podophyllotoxin derivatives and actinomycines.
Conclusion:
Our study showed the TLS risk characteristics of 164 antineoplastic agents by detecting and integrating ADR signals, which may help to optimize clinical practice.
Plain language summary
Background: Antineoplastic agents are medicines that help treat cancer. It is one of the most outstanding achievements of human beings in medicine, which plays an increasingly important role in improving human health and prolonging the life span of cancer patients. However, adverse reactions (ADRs) associated with the use of antineoplastic agents may also cause unexpected harm to patients. Therefore, it is essential to have a comprehensive understanding of antineoplastic-related ADRs to ensure the lives of cancer patients. Tumor lysis syndrome (TLS) is a potentially life-threatening ADR that may occur during antineoplastic agent treatment. However, there is still a lack of comprehensive understanding of antineoplastic agents that may trigger TLS and their risk differences. This study aimed to comprehensively investigate the TLS risk of antineoplastic agents from the pharmacovigilance perspective, providing reference information for patients, health professionals, regulators, and others concerned with antineoplastic agent safety. Methods: Using data from the FDA Adverse Event Reporting System (FAERS) between the years 2004 and 2022, we reviewed TLS reports associated with antineoplastic agent exposure, summarized an antineoplastic agent list that was reported as the potential culprit-drug of TLS, and explored the TLS risk of different antineoplastic agents by disproportionality analysis. Results: Our results showed that 164 antineoplastic agents, involving 64 antineoplastic agent subclasses, were reported as the potential culprit-drug of TLS in the FAERS database, in which 129 antineoplastic agents and 39 antineoplastic agent subclasses were associated with increased TLS risk to varying degrees. Conclusions: Our research expounded the differences in TLS risks of different antineoplastic agents, which helps us pay attention to the occurrence of TLS and give timely treatment when prescribing high-TLS-risk antineoplastic agents to patients.
Introduction
Tumor lysis syndrome (TLS) is a potentially life-threatening metabolic complication during antineoplastic therapy, which results from the rapid destruction of malignant cells and the abrupt release of their cellular contents into the bloodstream, leading to the characteristic findings of hyperuricemia, hyperkalemia, hyperphosphatemia, and hypocalcemia.1–3 These electrolyte and metabolic disturbances can progress to significant clinical toxic effects, including kidney insufficiency, cardiac arrhythmias, seizures, and death due to multiorgan failure.2,4,5 It is reported that the incidence of TLS ranges from 3% to 40% among patients with high-risk hematological malignancies, 6 and the overall in-hospital mortality of TLS can even reach 28% in patients with certain types of cancer. 7 Because of its high mortality, TLS has become a common but challenging emergency in oncologic patient management. 3
In clinical practice, recognition of risk factors, close monitoring of at-risk individuals, and implementing appropriate preventive measures are critical components in TLS medical management because prevention of TLS may be more effective than treatment.5,6,8,9 In this regard, to optimize the identification of high-risk patients and the timely implementation of preventive measures, an international TLS expert consensus panel developed a risk assessment strategy to classify the final TLS risk of tumor patients from three risk factors, including biological signs of TLS, malignant disease type, and renal function. 10 However, although the above-mentioned work has made an excellent critical summary of the literature, standards of practice and clinical experience, the antineoplastic agents, as one of the main triggers of TLS,11–13 is not included in the risk assessment items. The existing literature has shown that even the same class of antineoplastic agents with similar indications may have different risks of triggering TLS.14–16 Therefore, it is necessary to apply feasible methods to fully understand the risk differences of TLS triggered by different antineoplastic agents, providing extra reference information for evaluating the TLS risk of tumor patients.
Pharmacovigilance is the science and activities relating to the detection, assessment, understanding, and prevention of adverse effects or any other possible drug-related problems, in which evaluating the drug safety profile using real-world adverse events (AEs) data in pharmacovigilance database is one of its important contents. 17 Currently, probing for disproportionality between drug use and AE occurrence, namely adverse drug reaction (ADR) signal detection, is one of the main ways to apply the real-world AE data recorded in the pharmacovigilance database. 18 In clinical practice, the ADR signal detection results can be used as a reference tool to provide information for identifying the etiological drugs of specific AE, exploring the drug–drug interaction, and optimizing the drug selection for individual patients. 19 In this regard, TLS is a significant AE during antineoplastic agent use, so real-world AE data in the pharmacovigilance database can provide an unprecedented opportunity to fully understand the risk differences of antineoplastic agents triggering TLS.
In this study, we reviewed the TLS cases associated with the use of antineoplastic agents in the FDA Adverse Event Reporting System (FAERS) database, summarized the antineoplastic agents that may trigger TLS, and evaluated the TLS risk of different antineoplastic agents using well-acknowledged ADR signal detection method, trying to provide extra drug reference information for evaluating the TLS risk of tumor patients.
Materials and methods
Data source
The data of this study come from the FAERS database, which is an important component of the US FDA’s post-marketing safety surveillance program and is used to collect safety data for drug and therapeutic biological products submitted by manufacturers, consumers, and healthcare professionals. openFDA is an official innovation project supported by the US FDA, aiming at realizing easy access to public data. 20 At present, it provides query access and original download access to the structured information of more than 16 million post-marketing AE reports received by the US FDA since 2004. Through the application programming interface (API) provided by openFDA, the structured information of AE reports can be retrieved, including but not limited to the patient’s basic information (e.g., age, sex, and weight), sources of reports (reporter and reporting country), medication information (e.g., drug name, dosage, and duration of medication), AEs submitted, and patient outcomes. Therefore, the TLS reports in the FAERS database can be screened, collected, and downloaded by constructing a reasonable query request through the API provided by openFDA.
Identifying TLS reports related to antineoplastic agents in the FAERS database
In the FAERS database, the AEs that occur in patients are uniformly coded as a standard term called Preferred Terms (PTs) using the Medical Dictionary of Regulatory Activities (MedDRA). Therefore, the AE reports of interest can be identified by selecting appropriate PTs. Standardized MedDRA queries (SMQs) are a collection of PTs that directly or indirectly point to the medical status of interest, which are developed to assist in the retrieval of cases of interest from a MedDRA-coded database. 21 In MedDRA, an SMQ can provide two types of retrieval modes to identify target AE reports, namely broad-scope search and narrow-scope search. Among them, a broad-scope search contains all the PTs that potentially point to the medical condition of interest, while a narrow-scope search only consists of PTs that are closely associated with the medical condition of interest. 22 TLS is one of the SMQs included in MedDRA, but to accurately identify the TLS reports, this study only uses PTs included in the narrow-scope search of “Tumour lysis syndrome (SMQ)” to retrieve the FAERS database (Table 1).
The Preferred Terms included in the narrow-scope search of Tumour lysis syndrome (SMQ).

The MedDRA version used in this study is 23.0.
MedDRA, Medical Dictionary of Regulatory Activities.
For the returned TLS reports, the generic name of the drug recorded in the “patient.drug.openfda.generic_name” field of the reports will be coded according to the Anatomical Therapeutic Chemical (ATC) classification system. If any drug in the TLS report can be classified into “antineoplastic agents (L01),” we considered that the occurrence of TLS is potentially related to the use of antineoplastic agents and included the report in the final analysis.
ADR signal detection method
Disproportionality analysis is a validated statistical approach in drug safety research and surveillance, which is widely used to analyze AE reports and identify potential ADR signals.23,24 In this study, we used the reporting odd ratio (ROR), a well-established disproportionality analysis method, to conduct ADR signal detection. The ROR method includes two main parameters in ADR signal detection, namely the number of cases and the 95% confidence intervals (CIs) of the ROR value. 18 The number of cases is the reporting quantity of the combination of target drug and target AE, which can help us understand the common culprit drugs that may trigger the AE of interest. However, a high reporting quantity of a drug is not exactly equal to a high ADR risk because the difference in the frequency of drug use in the real world will greatly affect this figure. To cope with this problem, the ROR value is introduced as an index to reflect the differences between the real frequency and background frequency of target drug-induced target AE, which can quantitatively represent the strength of statistical correlation between target drug and target AE, thus evaluating the risk of target AE induced by target drug. Based on the classical two-by-two contingency table (Table 2), the ROR value and its corresponding 95% CIs can be calculated by the following formula:
The two-by-two contingency table for ADR signal detection.
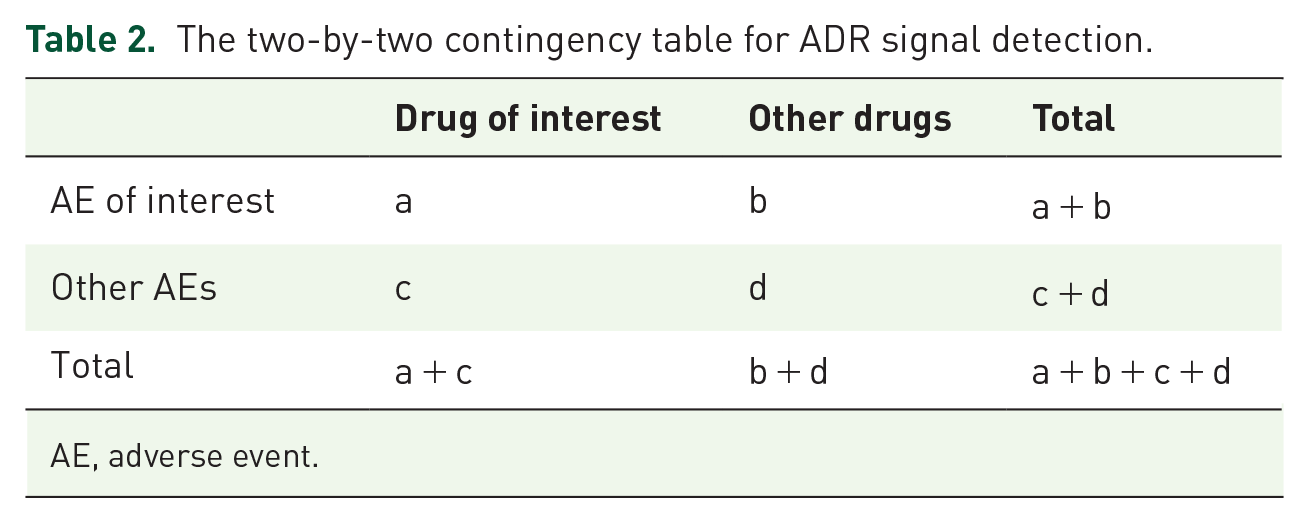
AE, adverse event.


When the lower-limit 95% CI of ROR value is above 1.0, and the number of cases is greater than or equal to 3 (a ⩾ 3 in Table 2), it suggests that the target drug has the potential high risk of causing target AE, namely a positive ADR signal. Instead, if the lower-limit 95% CI of ROR value or the number of cases cannot meet the above-mentioned criteria, it suggests that there is no significant statistical association between the target drug and the occurrence of target AE, namely a negative ADR signal. In addition, for drugs with positive ADR signals, the ROR value is a quantitative index reflecting the statistical correlation between the drug of interest and AE of interest, so the ROR value can represent the ADR signal strength of the target drug and can be used to compare the risk differences of different drugs quantitatively.19,22
Data acquisition, processing, and analysis
In this study, we obtained the data through the openFDA platform and conducted a follow-up analysis. The detailed steps are as follows. First, refer to the API construction instructions issued by the openFDA (https://open.fda.gov/apis/drug/, accessed on September 15, 2023), we used the PTs in Table 1 to retrieve and download all the TLS-related AE reports in the FAERS database from January 1, 2004 to December 31, 2022. Second, we extracted the generic name of drugs recorded in the “patient.drug.openfda.generic_name” field of all the TLS reports, and the drugs were coded and classified by the ATC system. Third, according to the ATC code of drugs, TLS reports that do not involve antineoplastic agents were excluded, and the basic information of the retained reports was extracted and summarized. Fourth, according to the list of antineoplastic agents that may trigger TLS, the ADR signals were detected at the SMQ level and PT level, respectively, yielding one ADR signal at the SMQ level and three ADR signals at the PT level. Fifth, the ADR signal detection results were integrated to show the risk characteristics of different antineoplastic agents triggering TLS.
In this study, R version 4.1.0 (R Foundation for Statistical Computing, Vienna, Austria) was used for data acquisition, processing, and analysis.
Results
The basic information and patient characteristics of antineoplastic agent-related TLS reports
Using PTs in Table 1 to retrieve AE reports in the FAERS database, a total of 7269 TLS reports were returned, which involved 50,878 drugs. After removing drugs with missing generic names, removing duplicate drugs, excluding non-antineoplastic agents, and integrating synonymous drugs, a total of 164 antineoplastic agents were identified to be related to TLS. Meanwhile, the list of antineoplastic agents was used to match TLS reports further, and finally, 5819 antineoplastic agent-related TLS reports were screened for subsequent analysis (Figure 1).

Flowchart of determining TLS reports related to antineoplastic agents.
The basic information and patient characteristics of 5819 TLS reports are shown in Figure 2. In recent years, the number of TLS reports related to antineoplastic agents has been increasing, and 2022 is the year with the largest number of reports (Figure 2(a)). In terms of reporting sources, most reports (49.49%) are submitted by physicians (Figure 2(b)), and the United States is the country with the most reports (Figure 2(c)). With regard to the demographic characteristics of patients, male patients (52.74%) account for the majority of TLS patients (Figure 2(d)), and most patients are in the age group of 60–70 years old (Figure 2(e)). In addition, TLS usually results in serious adverse outcomes, and it is even directly related to the death of 1142 (19.63%) patients to some extent (Figure 2(f)).

The basic information and patient characteristics of antineoplastic agent-related TLS reports. (a) Annual distribution of reported quantity; (b) occupational distribution of the reporters; (c) top 15 countries with the largest number of reports; (d) sex distribution of patients; (e) age distribution of patients; and (f) distribution of severe adverse outcomes suffered by patients.
ADR signal detection results for 164 antineoplastic agents
In the FAERS database, 164 different antineoplastic agents were reported to trigger TLS. To investigate the association between TLS occurrence and the use of antineoplastic agents, we conducted ADR signal detection at SMQ level and PT level for 164 different antineoplastic agents, respectively. For each antineoplastic agent, there were four ADR signal detection results, including one at the SMQ level and three at the PT level. The ADR signal detection results at the SMQ level are shown in Supplemental Table S1, and the ADR signal detection results at the PT level can be found in Supplemental Tables S2–S4. On the whole (at SMQ level), the results showed that rituximab (a = 1036) was the most reported antineoplastic agent in TLS reports, followed by cyclophosphamide (a = 981), venetoclax (a = 727), doxorubicin (a = 706), etoposide (a = 604), while tagraxofusp (ROR = 141.89, 95% CI: 85.54–235.34) was the antineoplastic agent with the highest ADR signal strength followed by floxuridine (ROR = 140.75, 95% CI: 61.64–321.38), pentostatin (ROR = 73.82, 95% CI: 51.27–156.29), tebentafusp (ROR = 67.47, 95% CI: 21.39–212.89), and venetoclax (ROR = 53.08, 95% CI: 49.13–57.35).
To further summarize the risk characteristics of different antineoplastic agents triggering TLS, we integrated the ADR signal detection results of 164 antineoplastic agents (Figure 3). In this study, the ADR signal detection results can be classified into three types, namely negative ADR signal, positive ADR signal, and no related reported cases of the combination of antineoplastic agent and SMQ (or PT). Of the 164 antineoplastic agents, 129 showed at least one positive ADR signal, while the remaining 35 did not show any positive ADR signal at the SMQ level or PT level. Among the 129 antineoplastic agents with at least one positive ADR signal, it is noteworthy that six antineoplastic agents, namely bevacizumab, carboplatin, cisplatin, fluorouracil, lenvatinib, and paclitaxel, have four positive ADR signals.

Pharmacovigilance signal distribution at the SMQ level and PT level for 164 antineoplastic agents.
TLS risk evaluation for different antineoplastic agent subclasses
Based on the ATC classification system, the 164 antineoplastic agents can be further classified into 46 chemical subgroups (fourth ATC level). To explore the whole TLS risk difference between different antineoplastic agent subclasses, we conducted ADR signal detection at the SMQ level for those 46 chemical subgroups (Table 3). Among the 46 chemical subgroups, 39 showed positive ADR signals except for ethylene imines, Janus-associated kinase inhibitors, epidermal growth factor receptor tyrosine kinase inhibitors, cyclin-dependent kinase inhibitors, hedgehog pathway inhibitors, methylhydrazines, and poly (ADP-ribose) polymerase inhibitors. In terms of reporting quantity, nitrogen mustard analogs (a = 1421) were the most reported antineoplastic agent subclass followed by clusters of differentiation 20 (CD20) inhibitors (a = 1253) and pyrimidine analogs (a = 1154). With regard to ADR signal strength, CD22 inhibitors (ROR = 33.78, 95% CI: 23.24–49.10) were the antineoplastic agent subclass with the highest ADR signal strength followed by podophyllotoxin derivatives (ROR = 28.68, 95% CI: 26.38–31.19), and actinomycines (ROR = 25.75, 95% CI: 17.48–37.94).
ADR signal detection results for 46 antineoplastic agent subclasses at the SMQ level.
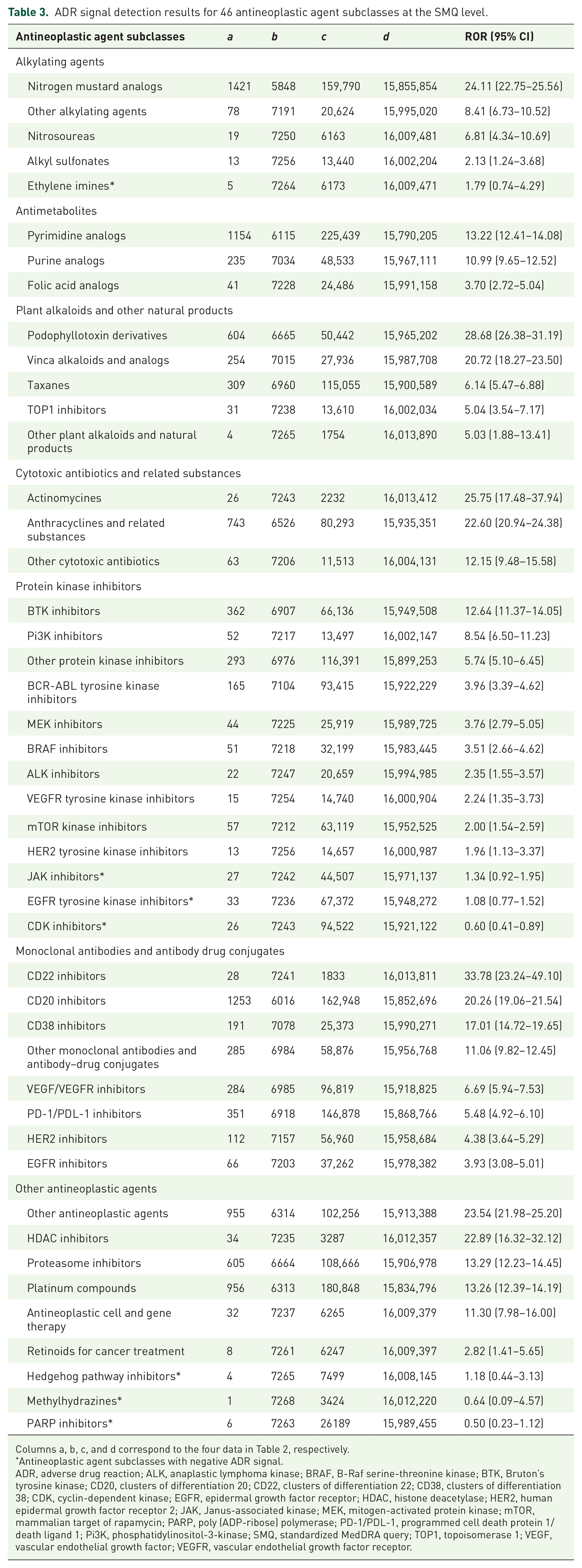
Columns a, b, c, and d correspond to the four data in Table 2, respectively.
Antineoplastic agent subclasses with negative ADR signal.
ADR, adverse drug reaction; ALK, anaplastic lymphoma kinase; BRAF, B-Raf serine-threonine kinase; BTK, Bruton’s tyrosine kinase; CD20, clusters of differentiation 20; CD22, clusters of differentiation 22; CD38, clusters of differentiation 38; CDK, cyclin-dependent kinase; EGFR, epidermal growth factor receptor; HDAC, histone deacetylase; HER2, human epidermal growth factor receptor 2; JAK, Janus-associated kinase; MEK, mitogen-activated protein kinase; mTOR, mammalian target of rapamycin; PARP, poly (ADP-ribose) polymerase; PD-1/PDL-1, programmed cell death protein 1/death ligand 1; Pi3K, phosphatidylinositol-3-kinase; SMQ, standardized MedDRA query; TOP1, topoisomerase 1; VEGF, vascular endothelial growth factor; VEGFR, vascular endothelial growth factor receptor.
Discussion
The pharmacovigilance database provides an unprecedented opportunity to fully understand the potential ADR risk information of antineoplastic agents, and such information is of great significance for identifying toxic side effects of antineoplastic agents in time and providing early intervention for cancer patients. 25 In this study, we reviewed all the TLS reports recorded in the FAERS database from 2004 to 2022 and presented a comprehensive overview of TLS risk characteristics related to antineoplastic agents.
Our study provided a multi-dimensional evaluation perspective. First, we summarized 164 kinds of antineoplastic agents reported to be related to the occurrence of TLS from the FAERS database. To our knowledge, this is the most comprehensive list of antineoplastic agents used to evaluate the TLS risk signals so far. Although previous studies have used the real-world AE data in the FAERS database to investigate the risk relationship between antineoplastic agent use and the occurrence of TLS,26–30 these studies usually focus on limited anti-tumor drugs, such as lurbinectedin, 26 immune checkpoint inhibitors, 28 and monoclonal antibodies, 29 which makes their results insufficient to show the TLS risks related to antineoplastic agents comprehensively. Meanwhile, due to the difference in research purpose and research design, it is also difficult to summarize a comprehensive drug list based on the above research to integrally present the TLS risk of antineoplastic agents. In this study, the scope of research objects has been greatly expanded, covering 164 potential culprit anti-tumor drugs of TLS reported in the FAERS database from 2004 to 2022, which is helpful for medical personnel, patients, and others concerned about drug safety to comprehensively understand the antineoplastic agents that may trigger TLS.
In addition, based on the narrow-scope search of “Tumour lysis syndrome (SMQ)” in MedDRA 23.0 (International Conference on Harmonization of Technical Requirements for Human Use, Geneva, Switzerland), we introduced the ROR method to evaluate the TLS risk for 164 antineoplastic agents at the SMQ level and PT level in a unified standard (Supplemental Tables S1–S4). Our results showed that 129 (78.7%) antineoplastic agents were associated with the increased TLS risk, and even some presented a strong ADR signal combined with a large number of TLS cases. For example, venetoclax ranks fifth in the signal strength of ADR (ROR = 53.08, 95% CI: 49.13–57.35) and third in the number of TLS cases (a = 727) in our study (Supplemental Table S1), which obviously indicates its potential high TLS risk. In current literature, the TLS risk of venetoclax has been widely recognized, and prophylaxis and treatment strategies have been implemented as standard of care in patients receiving venetoclax to minimize the risk of TLS,31,32 which demonstrated that our ADR signal detection results can reliably reflect the TLS risk of antineoplastic agent to some extent, thus providing reference information in clinical practice. Meanwhile, to intuitively present the TLS risk of 164 anti-tumor drugs, we integrated the tedious ADR signal detection results into a positive-negative ADR signal distribution map (Figure 3). For each antineoplastic agent, the total number of positive ADR signals is between zero and four, which can be used as a concise tool to understand and compare the TLS risks of different antineoplastic agents conveniently. 22 For example, bevacizumab, one of six antineoplastic agents with four positive ADR signals, has theoretically higher TLS risk than an antineoplastic agent without a positive ADR signal (e.g., vismodegib). Moreover, when it is necessary for clinical practice, the positive–negative ADR signal distribution map can also be used to guide query of the ROR values and its corresponding 95% CI of the antineoplastic agent in Supplemental Tables S1–S4 to understand and (or) compare the TLS risk difference between drugs in detail, providing a reference for drug selection or drug discontinuation.
Besides paying attention to the TLS risk of a single antineoplastic agent, our study also classified 164 antineoplastic agents into 46 antineoplastic agent subclasses and explored their TLS risk (Table 3). Our results showed that 39 (84.8%) antineoplastic agent subclasses have positive ADR signals and showed potential TLS risk. Although part antineoplastic agent subclasses, such as programmed cell death protein 1/death ligand 1 (PD-1/PDL-1) inhibitors, 28 CD20 inhibitors, 30 and phosphatidylinositol-3-kinase (Pi3K) inhibitors, 33 have been reported to be associated with the occurrence of TLS, there is still a lack of clear comparison of risk differences among different antineoplastic agent subclasses. In this respect, our research has supplemented the information in this field to some extent and provided a reference for understanding the TLS risk difference of different antineoplastic agent subclasses.
Limitation
Although our study comprehensively evaluated the risk of antineoplastic agents triggering TLS from a pharmacovigilance perspective, there are still inevitable limitations. First, the FAERS database is a spontaneous reporting database; therefore, underreporting, repeated reporting, missing data, notoriety bias, and Weber effect may exit, resulting in the deviation of ADR signal detection results.22,34 Second, patient age, gender, accompanying therapeutic medications, dosage and duration of antineoplastic agents use, comorbidities, malignant disease type, and renal function influence TLS occurrence,10,22,35 but we cannot shield the influence of these factors on ADR signal detection results due to the inherent limitation of spontaneous reporting database. Hence, it is necessary to keep in mind that the results of this study can only provide a reference for the culprit-drug identification of TLS and cannot replace the professional opinion given by oncologists or clinical pharmacists after comprehensive consideration of patients’ conditions. Third, for antineoplastic agents that are not commonly used (e.g., tagraxofusp and inotuzumab ozogamicin), a small increase in reported TLS instances will significantly increase the ADR signal strength and make its feedback on TLS risk higher than the actual level, so this factor should be taken into account when explaining the TLS risk of specific antineoplastic agents. Finally, the ADR signal detection result only indicates the statistical correlation between the target drug and the target AE, but whether there is a real causality needs further verification in a well-designed clinical study.
Conclusion
In this study, we focused on the issue of TLS risk in the process of using antineoplastic agents. As an exploratory pharmacovigilance study, the real-world AE data in the FAERS database were used to summarize the TLS risk of different antineoplastic agents, and we obtained meaningful results. We found that 164 antineoplastic agents, involving 64 antineoplastic agent subclasses, were reported as the potential culprit drugs of TLS in the FAERS database. The ADR signal detection results showed that 129 antineoplastic agents and 39 antineoplastic agent subclasses may be closely related to the occurrence of TLS, suggesting that we need to attach importance to the TLS risk of these antineoplastic agents and antineoplastic agent subclasses in the process of medication. In addition, we integrated the TLS risk characteristics of 164 antineoplastic agents based on the ADR signal detection results at the SMQ level and PT level, which can be used as a handy tool to quickly understand and compare the TLS risks of different antineoplastic agents, providing reference information for regulators, health professionals, and others concerned about the safety of antineoplastic agents. However, it is particularly noteworthy that the ADR signals data given in this study only represent statistical correlation rather than a real causal relationship, so our results cannot replace the professional opinions of oncologists or pharmacists in clinical practice. Besides, due to the uncertainty of causality, it is still necessary to further assess or validate the real TLS risks of some antineoplastic agents in well-designed clinical studies.
Supplemental Material
sj-docx-1-taw-10.1177_20420986241274909 – Supplemental material for Assessing the risk of tumor lysis syndrome associated with the use of antineoplastic agents: a real-world pharmacovigilance study based on the FDA Adverse Event Reporting System database
Supplemental material, sj-docx-1-taw-10.1177_20420986241274909 for Assessing the risk of tumor lysis syndrome associated with the use of antineoplastic agents: a real-world pharmacovigilance study based on the FDA Adverse Event Reporting System database by Dongxuan Li, Chunmeng Qin, Hongli Wang, Dan Du, Yalan Wang, Qian Du and Songqing Liu in Therapeutic Advances in Drug Safety
Supplemental Material
sj-docx-2-taw-10.1177_20420986241274909 – Supplemental material for Assessing the risk of tumor lysis syndrome associated with the use of antineoplastic agents: a real-world pharmacovigilance study based on the FDA Adverse Event Reporting System database
Supplemental material, sj-docx-2-taw-10.1177_20420986241274909 for Assessing the risk of tumor lysis syndrome associated with the use of antineoplastic agents: a real-world pharmacovigilance study based on the FDA Adverse Event Reporting System database by Dongxuan Li, Chunmeng Qin, Hongli Wang, Dan Du, Yalan Wang, Qian Du and Songqing Liu in Therapeutic Advances in Drug Safety
Supplemental Material
sj-docx-3-taw-10.1177_20420986241274909 – Supplemental material for Assessing the risk of tumor lysis syndrome associated with the use of antineoplastic agents: a real-world pharmacovigilance study based on the FDA Adverse Event Reporting System database
Supplemental material, sj-docx-3-taw-10.1177_20420986241274909 for Assessing the risk of tumor lysis syndrome associated with the use of antineoplastic agents: a real-world pharmacovigilance study based on the FDA Adverse Event Reporting System database by Dongxuan Li, Chunmeng Qin, Hongli Wang, Dan Du, Yalan Wang, Qian Du and Songqing Liu in Therapeutic Advances in Drug Safety
Supplemental Material
sj-docx-4-taw-10.1177_20420986241274909 – Supplemental material for Assessing the risk of tumor lysis syndrome associated with the use of antineoplastic agents: a real-world pharmacovigilance study based on the FDA Adverse Event Reporting System database
Supplemental material, sj-docx-4-taw-10.1177_20420986241274909 for Assessing the risk of tumor lysis syndrome associated with the use of antineoplastic agents: a real-world pharmacovigilance study based on the FDA Adverse Event Reporting System database by Dongxuan Li, Chunmeng Qin, Hongli Wang, Dan Du, Yalan Wang, Qian Du and Songqing Liu in Therapeutic Advances in Drug Safety
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
