Abstract
Background:
Connective tissue diseases (CTD), including systemic lupus erythematosus and rheumatoid arthritis (RA), have long been treated with hydroxychloroquine (HCQ). However, prolonged HCQ use poses a risk of adverse effects, particularly retinopathy.
Objective:
To detect early retinal changes assessed by optical coherence tomography angiography (OCTA) in CTD patients with long-term HCQ treatment and to explore the relationship between OCTA parameters and the concentrations of HCQ and its metabolites.
Design:
A cross-sectional study conducted from March 2020 to October 2021 at the First Affiliated Hospital of Anhui Medical University.
Methods:
The area and perimeter of the foveal avascular zone (FAZ), the thickness of the fovea and parafovea, and the vascular density of the superficial capillary plexus (SCP) and deep capillary plexus (DCP) in each area of the macula were measured by OCTA in 43 CTD patients treated with HCQ for over 6 months. Meantime, blood concentrations of HCQ and its metabolites were determined by high-performance liquid chromatography-tandem mass spectrometry, and the clinical documents of all 43 involved patients were collected.
Results:
There is no significant correlation between OCTA outcomes and the patient’s age, disease duration, and weight-dependent dose. HCQ cumulative duration positively correlated with FAZ area and perimeter (r = 0.419, p = 0.005 and r = 0.407, p = 0.007, respectively) and negatively correlated with the foveal vessel density in DCP (r = −0.378, p = 0.012). HCQ cumulative dose had a positive correlation with FAZ area and perimeter (r = 0.445, p = 0.003 and r = 0.434, p = 0.004, respectively) and had a negative correlation with foveal vessel density in SCP and DCP (r = −0.383, p = 0.011 and r = −0.424, p = 0.005, respectively). OCTA outcomes did not correlate with HCQ and its metabolite concentrations.
Conclusion:
OCTA could be used to detect microvascular changes in the macula of CTD patients with long-term HCQ therapy. It was not found the concentrations of HCQ and its metabolites were associated with retinal vascular changes.
Plain language summary
Introduction:
Long-term use of hydroxychloroquine (HCQ) may affect the eyes. In this study, we aimed to identify early eye changes in people with connective tissue diseases (CTD) who have been using HCQ for an extended period. Additionally, we wanted to explore the relationship between these changes and the amount of HCQ and its byproducts in their bodies.
Methods:
Between March 2020 and October 2021, we enrolled 43 patients with CTD who had been taking HCQ for more than 6 months. We collected various clinical information, including gender, age, diagnosis, disease duration, HCQ dosage, and treatment duration. We used specialized imaging called optical coherence tomography angiography (OCTA) to detect early microvascular changes in the macula and employed high-performance liquid chromatography-tandem mass spectrometry to measure HCQ and its metabolites in the blood. We also analyzed the relationships between these outcomes.
Results:
Our study did not reveal significant associations between OCTA results and patients’ age, disease duration, or HCQ dosage based on their weight. However, we found that as the duration of HCQ use increased, the size (area and perimeter) of the foveal avascular zone (FAZ) in the eye grew larger. Additionally, in individuals who had used HCQ for an extended period, the density of blood vessels in the back of the eye was lower in the foveal area. Importantly, OCTA parameters did not correlate with the blood concentrations of HCQ and its metabolites.
Conclusions:
Our use of OCTA detected subtle changes in the blood vessels at the center of the eye in people with CTD who had been using HCQ for an extended period. Notably, these OCTA parameters did not show any connection with the blood concentrations of HCQ and its metabolites.
Introduction
Connective tissue disease (CTD) is a group of autoimmune diseases, including systemic lupus erythematosus (SLE), rheumatoid arthritis (RA), etc. Since the 1950s, hydroxychloroquine (HCQ) has been widely used as an effective anti-inflammatory and immunomodulatory agent to treat various CTDs.1–3 It is mainly metabolized in the liver into desethylhydroxychloroquine (DHCQ), desethylchloroquine (DCQ), and bisdesethylchloroquine (BDCQ). 4 Though derived from chloroquine (CQ), the risk of adverse drug reactions (ADRs) of HCQ is significantly reduced compared with CQ. These ADRs commonly involve eye lesions, heart block, skin toxicity, etc.5–8
Retinopathy is the most severe ADR associated with long-term HCQ use, although its exact mechanism has remained elusive. 9 In the rhesus monkey, the damage might first occur in the cytoplasm of ganglion cells and photoreceptors. 10 In vitro experiments, HCQ increased the lysosomal pH of retinal pigment epithelium (RPE) cells, inhibited lysosomal autophagy, and ultimately influenced the function of RPE to phagocytose debris on the outer region of the retina. Because of that, the retinal barrier might be broken down. On the other hand, the accumulation of lipofuscin in RPE leads to the degeneration of photoreceptors. The impairment of RPE was irreversible, and the drug continued to accumulate so that the severe retinopathy could keep progressing after HCQ withdrawal.11,12 It was reported that if HCQ was ceased before the occurrence of RPE damage, the progression of retinopathy rarely sustained for more than a year, nor does it affect the central recess.4,13 Therefore, early identification of retinopathy is essential.
Among the detection of HCQ retinopathy recommended by the American Academy of Ophthalmology (AAO), spectral-domain optical coherence tomography (SD-OCT) could show thinning of the outer retinal layer, which is the early changes in retinal structure. 12 Compared to SD-OCT, optical coherence tomography angiography (OCTA) could observe early retinal microvascular changes in patients without obvious retinopathy, which has been used to assess retinal involvement.14–16
OCTA is an effective method for assessing vascular lesions, an imaging technique that reconstructs the retinal capillary network by detecting changes in blood flow signals. Unlike fluorescence angiography, OCTA requires no dye injection and is safer and faster. 17 It has been used to study the pathogenesis of various retinal vascular diseases, such as retinal vein occlusion, diabetic retinopathy, etc.18,19 Studies showed that OCTA could detect the microvascular changes in the macula of SLE patients 15 and could also find the early retinopathy of patients taking HCQ for more than 5 years.16,20
In fact, in addition to the HCQ treatment course, other various factors, including patient age, daily and cumulative HCQ dose, etc., might affect HCQ retinopathy. 21 HCQ has a half-life of 40–50 days, and its steady-state blood level can be achieved after approximately 6 months of administration but there is individual variability. 4 Some investigators suggested 500 ng/mL should be taken as the effective blood concentration of HCQ.22–24 However, so far, there is no consensus widely accepted on the relationship between HCQ blood concentration and HCQ retinopathy, and there is no research reported on the relationship between HCQ metabolites and retinal changes.
In this study, we used OCTA, an objective and non-invasive test mean, to assess whether HCQ could cause damage to the retinal microvasculature in the macula and also evaluated the ocular safety of HCQ and its metabolite concentrations in CTD patients taking HCQ.
Patients and methods
Patients
A total of 43 CTD patients without ocular involvement in remission stages were recruited in this study, including 39 SLE patients and 4 RA patients who fulfilled the SLE classification criteria issued by the European League Against Rheumatism (EULAR) in 2019 and RA classification criteria defined by American College of Rheumatology and EULAR in 2010, respectively.25,26 All involved patients had been taking HCQ at a stable daily dose of 200–400 mg for at least 6 months. Their demographic data and medical documents are listed in Table 1, in which the HCQ dose was evaluated from three perspectives: the daily HCQ dose, the weight-dependent dose, and the cumulative dose. Patient with the following conditions was excluded from this study: refractive media opacities that may interfere with the acquisition of clear images, established history of ophthalmic disease, taking other medications with possible adverse reaction that may affect the eye, concomitant disorders that may cause retinal microvascular changes (such as cardiovascular disease, diabetes, hypertension, and chronic nephrosis), and other autoimmune diseases.
Demographic and clinical characteristics of the patients involved. a

Including 39 patients with SLE and 4 patients with RA.
BDCQ, bisdesethylchloroquine; DCP, deep capillary plexus; DCQ, desethylchloroquine; DHCQ, desethylhydroxychloroquine; FAZ, foveal avascular zone; HCQ, hydroxychloroquine; OCTA, optical coherence tomography angiography; RA, rheumatoid arthritis; SCP, superficial capillary plexus; SLE, systemic lupus erythematosus.
This study was conducted at the First Affiliated Hospital of Anhui Medical University between March 2020 and October 2021, obtained the approval of the Ethics Committee of the First Affiliated Hospital of Anhui Medical University, and followed the Tenets of the Declaration of Helsinki. This design adheres to the Strengthening the Reporting of Observational studies in Epidemiology (STROBE) guidelines (Supplemental Table S1). 27
OCTA measurement
The AngioVue OCTA device (Optovue, Inc., Fremont, CA, USA) was used to detect and evaluate the angiography images. After dilating the pupil with tropicamide drops, a macular area of 6 mm × 6 mm centered on the foveal avascular zone (FAZ) was scanned by the built-in software. A series of parameters were obtained for the area and perimeter of the fovea and the density of blood vessels surrounding the FAZ including superficial capillary plexus (SCP) and deep capillary plexus (DCP) by the built-in angio analytics software and its PAR algorithm. This software also filters out noise and artifacts. The FAZ area is defined as the area of the central 1 mm diameter ring; the parafovea is defined as the area of the middle ring with a diameter of 3 mm. All parameters were detected by the same ophthalmologist between 11:00 a.m. and 12:00 p.m.
High-performance liquid chromatography-tandem mass spectrometry detection of HCQ, DHCQ, DCQ, and BDCQ concentration in blood
On the same day as the ophthalmic examination, 1.5 mL of peripheral venous blood was collected from every patient in a fasting state before taking the HCQ. Referring to the method reported in previous studies, the concentrations of HCQ and its metabolites (DHCQ, DCQ, and BDCQ) in the blood were measured by high-performance liquid chromatography-tandem mass spectrometry. 28 By Poroshell 120 EC-C18 column (2.1 × 100 mm, 2.7 μm) (Agilent, CA, USA), Shimadzu Nexera X2 HPLC system (Shimadzu, Kyoto, Japan), and API 3200DX mass spectrometer (AB SCIEX, MA, USA), the peak areas of HCQ and its metabolites were calculated from the serial standard concentrations with quinine as the internal standard. The concentrations of HCQ, DHCQ, DCQ, and BDCQ in each peripheral blood sample were obtained from the standard curve.
Statistical analysis
We obtained all outcome-related data from participants. All statistical analysis was performed by SPSS version 26.0 (SPSS, Inc., Armonk, IL, USA). Shapiro–Wilk test was used to assess for characteristics of the data distribution. Data normally and non-normally distributed were presented as mean ± standard deviation (SD) and median with 25th–75th percentile range (P25, P75), respectively, while categorical data were expressed as number (frequency). Comparisons between the two groups were drawn by the Student’s t-test or Mann–Whitney U test based on the data distribution. Pearson and Spearman’s approaches were employed to analyze the associations between the normally and non-normally distributed data, respectively. p < 0.05 was considered statistically significant.
Results
Measurement results of OCTA, HCQ, and its metabolites
The OCTA data of all 43 patients are shown in Table 1. The perimeter and area of FAZ were 2.19 ± 0.42 mm and 0.34 (0.27, 0.37) mm2, respectively, while the foveal and parafoveal thickness of macula were 243.26 ± 19.90 μm and 321 (313, 328) μm, respectively. When the density of superficial capillaries and deep capillaries was assessed from three different regions (Figure 1), the foveal and parafoveal vessel density in SCP was (16.54 ± 6.22)% and 49.30 (46.10, 50.90)%, respectively, and the density of the three vessels in DCP was (31.80 ± 8.21)% and (51.20 ± 4.80)%, respectively.

Graphic expressions of FAZ, macular thickness, SCP, and DCP detected by OCTA. (A) The perimeter and area of FAZ. (B) The foveal and parafoveal thickness (macular thickness). (C) The density of SCP is indicated by the colored map on the right. (D) The density of the DCP is represented by a colored map on its right. a, FAZ; b, the foveal regions; c, the parafoveal regions.
In addition, the median concentrations of HCQ, DHCQ, DCQ, and BDCQ detected from the 43 patients were 563.31 (376.39, 794.22), 355.24 (262.27, 482.30), 44.73 (36.55, 80.63), and 27.22 (14.79, 36.66) ng/mL, respectively, as listed in Table 1.
The relationship between OCTA parameters with clinical characteristics
Referring to previous studies,16,29 enrolled patients were divided into groups with duration >5 and ⩽5 years according to their duration of HCQ administration. The duration of treatment was 24.00 (12.00, 37.50) months for patients in the HCQ duration ⩽5 years group, whereas it was 109.50 (72.00, 136.25) months in the HCQ duration >5 years group. The statistical results showed that compared with the group with a duration of ⩽5 years, the perimeter and area of FAZ in the group with a duration >5 years increased significantly, the foveal vascular density in SCP and DCP decreased significantly, and the parafoveal vascular density in DCP increased significantly (Table 2). According to the HCQ daily dose >5 and ⩽5 mg/kg/day, and HCQ cumulative dose >1000 and ⩽1000 g, respectively, patients were divided into different groups. It was demonstrated that the foveal thickness was enhanced in the low daily dosing group than in the high daily dosing group [248.00 (243.00, 260.00) versus 238.50 (228.25, 249.00) μm, p = 0.018]. There were no significant differences in other OCTA parameters between these groups (Supplemental Tables S2 and S3).
Comparison of OCTA outcomes between patients treated with HCQ for ⩽5 years and for >5 years.

The results of the Mann–Whitney U test, the others were the result of the Student’s t-test.
p < 0.05.
DCP, deep capillary plexus; FAZ, foveal avascular zone; HCQ, hydroxychloroquine; OCTA, optical coherence tomography angiography; SCP, superficial capillary plexus.
Based on the results of statistical correlation analysis (Table 3), in OCTA parameters, FAZ outcomes were significantly correlated with HCQ treatment duration and cumulative dose, respectively, while the foveal vessel density in SCP had a markedly negative relationship with HCQ cumulative dose. Also, the foveal vessel density in DCP showed significant negative correlations with HCQ treatment duration and cumulative dose, respectively. No correlation was found between OCTA parameters and patients’ age, disease duration, or other variables of HCQ administration.
Correlation analysis between OCTA findings and the patient characteristics as well as their HCQ administration.
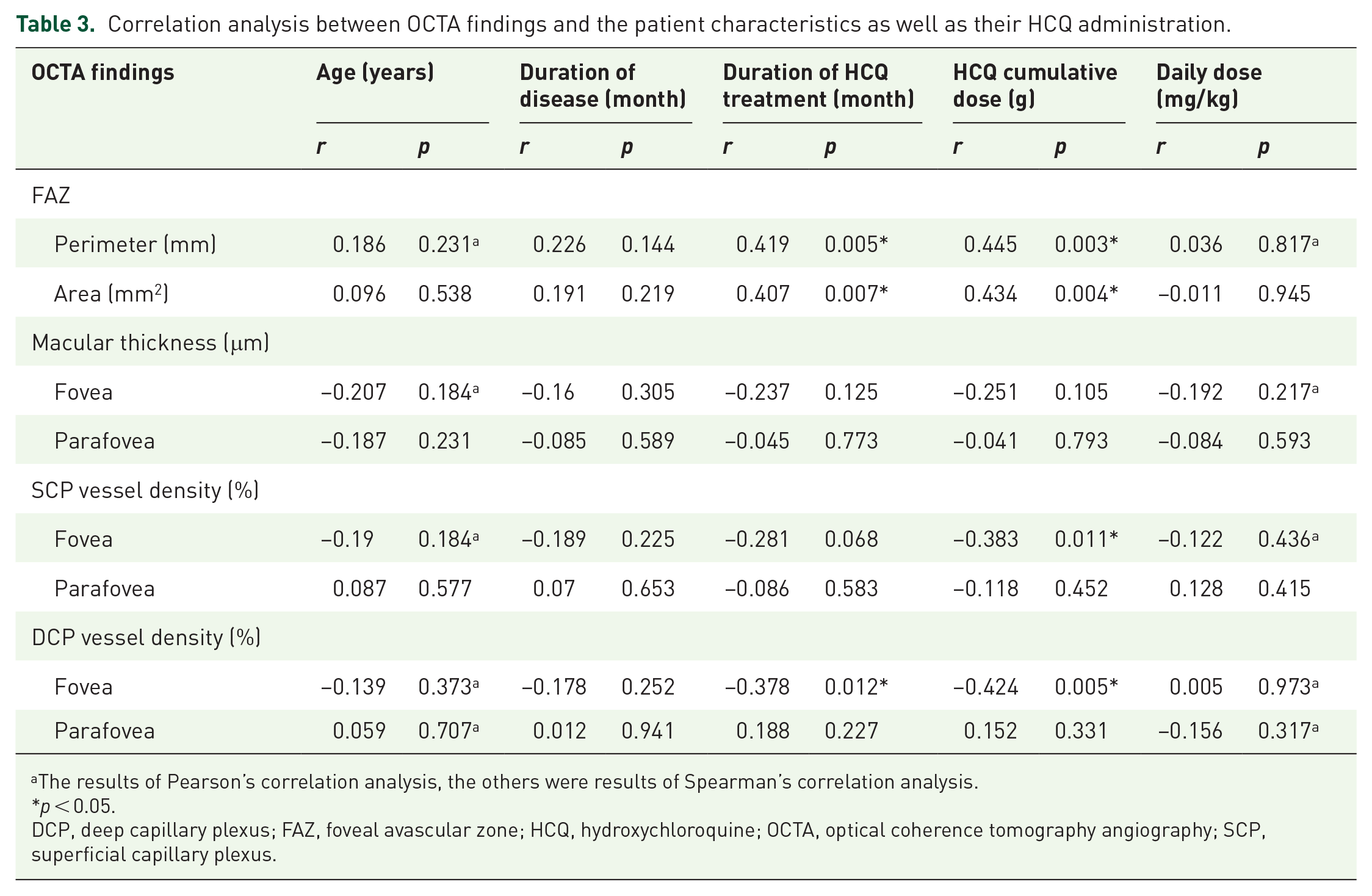
The results of Pearson’s correlation analysis, the others were results of Spearman’s correlation analysis.
p < 0.05.
DCP, deep capillary plexus; FAZ, foveal avascular zone; HCQ, hydroxychloroquine; OCTA, optical coherence tomography angiography; SCP, superficial capillary plexus.
The relationship between OCTA parameters with blood concentrations of HCQ and its metabolites
There is no uniform standard for the higher concentration of HCQ.30,31 In our study, patients were divided into the higher concentration group (the blood concentration above the median concentration) and the lower concentration group (the blood concentration below the median concentration). The median blood concentrations of HCQ, DHCQ, DCQ, and BDCQ were 563.31, 355.24, 44.73, and 27.22 ng/mL, respectively. Noteworthy, there were no statistical differences in OCTA parameters between different groups (Table 4). Furthermore, correlation analysis showed that none of these concentrations were associated with OCTA findings (Table 5).
OCTA parameters between groups with different concentrations of HCQ and its metabolites.

The results of the Mann–Whitney U test, the others were the result of the Student’s t-test.
BDCQ, bisdesethylchloroquine; DCP, deep capillary plexus; DCQ, desethylchloroquine; DHCQ, desethylhydroxychloroquine; FAZ, foveal avascular zone; HCQ, hydroxychloroquine; OCTA, optical coherence tomography angiography; SCP, superficial capillary plexus.
Correlation analysis between OCTA measurements and the concentrations of HCQ as well as its metabolites.

The results of Pearson’s correlation analysis, the others were results of Spearman’s correlation analysis.
BDCQ, bisdesethylchloroquine; DCP, deep capillary plexus; DCQ, desethylchloroquine; DHCQ, desethylhydroxychloroquine; FAZ, foveal avascular zone; HCQ, hydroxychloroquine; OCTA, optical coherence tomography angiography; SCP, superficial capillary plexus.
Discussion
Acting as an anti-inflammatory and immunomodulatory agent with regulatory effects on thrombosis and blood lipids, HCQ is currently used as the first-line medication for the treatment of CTD, including SLE, to increase survival rates by reducing the risk of organ damage and disease activity.32–34 However, long-term use of HCQ might increase the risk of retinal toxicity and the patients would have to be checked regularly for changes in visual acuity and visual field. It was recommended that fundus lesions be ruled out before taking HCQ and that annual ophthalmic assessments be performed 5 years after taking it. Guidelines from the 2016 AAO suggested multiple means of screening for HCQ retinopathy, including SD-OCT, fundus autofluorescence, and multifocal electroretinogram (mfERG). 21 Still, up to now, there is no examination method as a consensus gold standard for screening HCQ-related retinopathy. In this study, we made use of OCTA as a method to assess retinal microvascular changes since the three-dimensional nature of OCTA allows for the visual analysis and quantification of retinal and choroidal capillaries without the need for any dye injection, with the advantage of being safe, fast, and accurate. 35
Our study showed that, compared to the patients taking HCQ for less than 5 years, those who took HCQ for more than 5 years had reduced vascular density in SCP and DCP and increased FAZ. The FAZ is responsible for marginal and central vision, and an increased FAZ indicates ischemic changes in the macula. 36 This was consistent with the AAO recommendation that HCQ usage for more than 5 years might be a high-risk factor for HCQ-induced retinopathy. Some studies reported that, in patients who had taken HCQ for more than 5 years, some preclinical changes in the fundus could be detected by OCTA earlier than the onset of symptoms in the retina. These preclinical changes found by OCTA included the enlargement and distortion of the FAZ and reduction in retinal thickness and blood density in the macula.16,20,29,37 Since OCTA could differentiate macular changes from the patients with different HCQ treatment courses, it should be helpful in the early detection of retinopathy.
In addition to the treatment course, macular area changes measured have been reported to be associated with older age (>60 years), larger daily doses (>5 mg/kg), and higher cumulative doses (>1000 g).9,25,38 Our study did not observe a correlation between OCTA parameters and age, which might be due to the relatively younger age (36.40 ± 11.15 years, Table 1) of the included patients. We found that the foveal thickness was enhanced in the group with daily doses ⩽5 mg/kg, which is consistent with the results of previous studies, whereas no correlation between OCTA outcomes and the weight-dependent dose was found. Although the patient’s weight-dependent dose of HCQ (5.66 ± 1.66 mg/kg/day, Table 1) was not less than the dose of 5 mg/kg/day recommended by the 2016 AAO guideline, 21 the mean daily dose [300.00 (200.00, 400.00) mg/day, Table 1] did not exceed 400 mg/day. Furthermore, the weight-dependent dose did not correlate with macula-related parameters, so the HCQ daily dose in our study [300.00 (200.00, 400.00) mg/day, Table 1] was considered safe while the regular close follow-up should be obligatory. Ocular involvement in patients with CTD, especially in SLE, may be caused by CTD itself, leading to retinopathy. 39 Therefore, in our study, we selected patients in their remission state with no previous ocular involvement to minimize the likelihood of CTD affecting OCTA results. The result showed that the OCTA parameters were independent of the disease duration but were related to the HCQ treatment course, suggesting that retinopathy might be the ADR from HCQ accumulation. Also, HCQ treatment duration was positively correlated with FAZ area and FAZ perimeter, respectively, and was negatively correlated with the foveal vessel density of DCP. Furthermore, the HCQ cumulative dose was positively correlated with FAZ area and perimeter, respectively, and was negatively correlated with foveal vessel density in SCP and DCP, respectively. These results were consistent with the previous research reports. After assessing retinal function by mfERG in 67 patients treated with HCQ and 62 healthy controls, Lyons et al. 40 found a significant correlation between HCQ cumulative dose and mfERG abnormalities. In a large group of 3995 HCQ-treated patients, Wolfe et al. 41 revealed that the risk of HCQ-induced retinal toxicity was low in patients with an HCQ treatment course of less than 7 years but was very common in patients treated for more than 7 years with HCQ cumulative doses exceeding 1000 g. Based on the fundus outcomes detected by the swept-source OCTA, Forte et al. 20 demonstrated that, with the increase in HCQ cumulative dose and treatment duration, the vessel density in SCP and DCP decreased, and the FAZ increased. All these investigations indicated that HCQ cumulative dose and long treatment duration potentiated substantial risks for retinopathy.
The median concentration in peripheral blood for HCQ, DHCQ, DCQ, and BDCQ from Eryavuz Onmaz’s investigation was 806 (range 61.7–2760), 452 (range 20.0–1700), 76.4 (range 4.0–502.0), and 291 (range 14.0–1477) ng/mL, respectively. 42 In comparison, the average concentration of HCQ and its metabolites detected in our study was relatively lower [563.31 (376.39, 794.22), 355.24 (262.27, 482.30), 44.73 (36.55, 80.63), and 27.22 (14.79, 36.66) ng/mL for HCQ, DHCQ, DCQ, and BDCQ, respectively, Table 1], which might be attributed to the different HCQ metabolism by various races or inconsistent detection methods. By employing OCT and mfERG as the assessment method for fundus oculi, Petri et al. 43 reported that HCQ blood concentration was related to retinal changes, while Munster found that high HCQ concentration in blood was associated with gastrointestinal side effects rather than retinal toxicity. 44 In our study, based on OCTA parameters, neither HCQ nor its metabolite concentrations presented any correlation with the detected parameters of the macula. However, it was reported that BDCQ blood concentration is correlated with fundus lesions found by Munster et al. 44 Different examination methods, including OCTA and mfERG, used to evaluate the fundus lesion might account for the different results in these researches. Several studies demonstrated that blood levels of HCQ and its metabolites correlated with disease activity. Blanchet measured HCQ blood concentration from 573 patients with SLE. They observed that complete blood HCQ levels in patients with low disease activity [systematic lupus erythematosus disease activity index (SLEDAI) ⩽4] were higher than the patients with high disease activity (SLEDAI >4) (940.8 ± 448 versus 765.9 ± 426 ng/mL, p = 0.001). 45 Also, Munster et al. 44 found that the therapeutic efficacy of HCQ in RA patients was positively correlated with DHCQ blood levels; therefore, maintenance of high HCQ concentration was recommended, though there is no consensus on the effective HCQ concentration for various CTD treatments by now. However, our research results revealed that the macular area detected by OTCA was not affected by changes in HCQ concentration. So, based on the follow-up results detected by non-invasive OCTA, it would be safe for the retina to maintain a higher HCQ concentration in the treatment of CTD.
Limitations
This study has several limitations. First, we did not include healthy controls and disease controls. Because fewer SLE patients were treated without HCQ, and the majority of participants in this study were SLE patients. We had difficulty finding SLE patients who were not taking HCQ as disease control. Considering that SLE could result in retinal vascular changes, the comparison between patients treated with HCQ and HCs was not exact; therefore, we did not include healthy controls as well. Second, we included a small number of study subjects, and no patients with HCQ retinopathy were observed in this study. Third, we did not analyze the density of choroidal capillaries because we used the SD-OCTA instrument, which is less sensitive to choroid angiopathy with a shorter wavelength and weaker penetration.
Conclusion
In conclusion, OCTA can assess the macula microvascular changes caused by long-term HCQ therapy. The retinal changes detected by OCTA were related to the HCQ cumulative course and cumulative dose but not to higher blood concentrations of HCQ and its metabolites. To improve HCQ efficacy in the treatment of CTD, it is recommended to maintain the concentrations of HCQ and its metabolites at relatively high levels. Even if achieving higher levels of HCQ and its metabolites, the retinal vessels have no significant changes. Noteworthy, regular follow-up examinations should be indispensable.
Supplemental Material
sj-docx-1-taw-10.1177_20420986231225851 – Supplemental material for Evaluation of early retinal changes in patients on long-term hydroxychloroquine using optical coherence tomography angiography
Supplemental material, sj-docx-1-taw-10.1177_20420986231225851 for Evaluation of early retinal changes in patients on long-term hydroxychloroquine using optical coherence tomography angiography by Huanhuan Zhao, Menglu Pan, Yaping Liu, Fangyue Cheng and Zongwen Shuai in Therapeutic Advances in Drug Safety
Supplemental Material
sj-docx-2-taw-10.1177_20420986231225851 – Supplemental material for Evaluation of early retinal changes in patients on long-term hydroxychloroquine using optical coherence tomography angiography
Supplemental material, sj-docx-2-taw-10.1177_20420986231225851 for Evaluation of early retinal changes in patients on long-term hydroxychloroquine using optical coherence tomography angiography by Huanhuan Zhao, Menglu Pan, Yaping Liu, Fangyue Cheng and Zongwen Shuai in Therapeutic Advances in Drug Safety
Supplemental Material
sj-docx-3-taw-10.1177_20420986231225851 – Supplemental material for Evaluation of early retinal changes in patients on long-term hydroxychloroquine using optical coherence tomography angiography
Supplemental material, sj-docx-3-taw-10.1177_20420986231225851 for Evaluation of early retinal changes in patients on long-term hydroxychloroquine using optical coherence tomography angiography by Huanhuan Zhao, Menglu Pan, Yaping Liu, Fangyue Cheng and Zongwen Shuai in Therapeutic Advances in Drug Safety
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
