Abstract
Background:
Modern oral antineoplastic and immune-modulating drugs offer an array of therapeutic advantages, and yet pose challenges in daily use for patients, physicians and pharmacists. In contrast to intravenous administration, these drugs are not subject to direct medical control. Recently, we have seen a huge rise in sales of non-prescription over-the-counter (OTC) medicines via the internet without any advice from a healthcare professional.
Objectives:
The aim of this study was to investigate whether the risk of known potential drug-drug interactions between modern oral antineoplastic and immune-modulating drugs and OTC drugs differs between sales in traditional community pharmacies versus online pharmacies.
Design:
Real-life sales data from community and online pharmacies were used as basis for the analysis.
Methods:
We determined the most frequently purchased antineoplastic and immune-modulating drug-substances in 14 local community pharmacies within the Munich area, Germany and identified the OTC substance groups that could potentially cause interactions with oncological therapies. Using sales data from 11 local community pharmacies and three online pharmacies, we investigated whether OTC purchases differed between the two sales channels.
Results:
We identified 10 relevant OTC substance classes and detected significant variations in patients’ preferred sales channels between the drug classes. Certain OTC drugs, which seem to be bought more often over the internet, pose risks during antineoplastic and immune-modulating therapy.
Conclusion:
Patients should therefore be proactively made aware of the corresponding risks in order not to jeopardize the activity of the antineoplastic and immune-modulating drugs and thus the success of their therapy.
Plain language summary
Modern anticancer and immune-modulating drugs have the advantage of often being taken orally, but they present other challenges in daily use. Unlike intravenously administered drugs, these are usually not administered by a physician but taken by the patient at home. In these cases, patients may be more likely to buy and take self-medicating drugs over-the-counter (OTC) without consulting a healthcare professional.
This study aimed to investigate whether there is a different risk of drug interactions between cancer or immune-modulating drugs and OTC drugs when bought in a community pharmacy versus an online pharmacy. Therefore, we looked at the most common cancer and immune-modulating drugs purchased in 14 local community pharmacies in Munich and identified which OTC drugs could cause problems when used simultaneously. Additionally, we analyzed the sales data from 11 local and 3 online pharmacies to determine if people were more likely to buy different OTC drugs from the two types of pharmacies.
As a result, this study showed 10 relevant OTC drug types that potentially cause problems and influence effectiveness when used with cancer or immune-modulating drugs. Furthermore, we observed that some of these OTC drugs were purchased more often online than in community pharmacies and thus are more distant from the control of a physician or pharmacist.
It is therefore essential for patients to be aware of the risks associated with easily accessible OTC drugs in combination with their cancer or immune-modulating medication, as serious side effects or decreased efficacy may develop. Patients should remember to consult their doctor or pharmacist if there is any uncertainty about potential drug interactions. At the same time, healthcare professionals should proactively draw their patients’ attention to these potential risks, especially when purchasing online.
Keywords
Introduction
The number of patients requiring antineoplastic and immune-modulating therapy has increased significantly in recent years. While sales of this group of drugs were as high as 6.8 million in Germany in 2018, the number rose even further to 7.7 million in 2020 1 all at the expense of the statutory health insurance funds. These therapy regimens require intensive patient care by different occupational groups, since they can be harmful despite their partly oral application. This high level of monitoring is particularly important for multiple reasons. As opposed to when administered intravenously, an increasing number of patients take an oral medication at home, leading to a lack of direct and frequent control by healthcare professionals. The number of home-based oncological therapies is on the rise 2 presumably in part due to the increasing number of oral anti-cancer drugs that have been approved over the last 20 years. 3 Common examples include the adjuvant use of long-term hormone deprivation medications in endocrine sensitive breast cancer, breakthroughs like Cyclin-Dependent Kinases (CDK4)/6 inhibitors for patients with hormone-receptor-positive metastatic breast cancer4,5, poly(Adenosine Diphosphate (ADP)-ribose) polymerase inhibitors for patients with BReast CAncer Gene 1 (BRCA-1) or BRCA-2 mutations.6,7 To address the special requirements that come with oral anti-cancer medications (OAMs), a multidisciplinary approach should be pursued, involving physicians, pharmacists, and other healthcare workers. 8 The responsibility shifts to the patients and, in particular, special heed must be taken regarding therapy adherence. It is of vital importance to inform the patient about specifics regarding drug use, as this is often essential for optimal efficacy of antineoplastic and immune-modulating therapy. A recent study with a group of vulnerable patients found that the understanding of how OAMs should be taken and handled was strikingly low. 9 By means of targeted communication, ambiguities can be identified, and questions clarified. It is essential to inform the patient about side effects and to sensitize the patient so that they can be recognized early. A subsequent change of preparation, dose adjustment and even when and how medication is taken at mealtimes can be decisive for a successful outcome of the therapy. Furthermore, it is imperative to recognize possible drug-drug interactions (DDIs) in order to avoid them having a detrimental effect.
In addition to prescription drugs, DDIs can also occur with prescription-free medication, also known as over-the-counter (OTC) drugs such as the common proton pump inhibitors (PPIs) or non-steroidal anti-inflammatory drugs (NSAIDs). Besides endangering therapy success, severe consequences are possible in the case of undesired DDIs, a common type of drug-related problems (DRPs). 10 In Germany, this comprehensive pharmaceutical care is performed both by the physician and by the pharmacist. Beside dispensing drugs, the pharmacist increasingly fulfils the important role of providing pharmaceutical counseling services. 11 Especially during additional use of OTC drugs alongside an ongoing therapy, often no physician is involved. In most cases, however, the patient’s OTC drug use is specifically checked by the dispensing pharmacy in order to proactively avoid relevant DRPs as the pharmacist is ideally placed to guide self-medication or recommend medical advice when needed. 12
In recent years, there has been a sharp shift in the sale of self-medication with customers increasingly using online pharmacies instead of community pharmacies. In the first half of 2019, 5% more OTC drugs were purchased by mail order in Germany than in the same period of the previous year. 13 The free sale of such drugs through online pharmacies on platforms such as ‘Amazon’ and ‘ebay’ shows that there is no direct contact between the patient and the pharmacist during the sales process. 14 In addition, unlike community pharmacies, online pharmacies often do not receive information about the patient’s state of health and current medical therapy regimen. In some events, they may not store or process this data at all, 14 indicating that the quality of advice provided by online pharmacies is often not as good as is legally mandated for a german pharmacy. 15
Prescription drugs have fixed prices in Germany which are identical in every pharmacy. These costs are usually covered by the patient’s health insurance. For OTC drugs, pharmacies are free to set the prices themselves. Studies have shown that similar to other branches, 16 the selection of a particular online pharmacy by the customer is often driven by a cheaper price for the drugs, 17 which may also indicate a lack of pharmacy loyalty. Consequently, drugs that are not prescribed by a doctor, and therefore inevitably not fixed in price, are more likely to be purchased via online pharmacies. Therefore, consultation by the pharmacist is particularly important in cases where the consequences of adverse drug combinations are likely.
Whether and in what way such OTC sales via online pharmacies endanger an ongoing oncological drug therapy regimen is not known. For this purpose, a precise analysis of the drugs sold via the sales channel of the community pharmacy must be compared with that of an online pharmacy. There is sufficient data, for example, documenting the proportion of prescription and non-prescription drugs sold through these different channels. 18 However, a detailed breakdown and comparison of the drugs by indication groups is missing, although this would be an important aspect for deriving implications for pharmaceutical care, especially regarding concomitant OTC medicines. A detailed differentiation would only be possible through extensive data acquisition from many national and international pharmacies, as drugs can easily be obtained throughout Europe via online pharmacies. As a result, comprehensive data are difficult to obtain and evaluate.
Therefore, the aim of this study was to investigate whether the risk of endangering oncological drug therapy through non-prescription sales differs from purchases via online and community pharmacies. A practical approach was applied in this analysis, using real-life sales as data basis. We examined which OTC drugs frequently sold in community pharmacies have the potential to cause clinically relevant interactions with antineoplastic and immune-modulating drugs. Subsequently, investigations were made to identify whether the sales of these OTC drugs differed between online and community pharmacies in 2018 and 2019.
Materials and methods
In this market research study, sales were recorded ex-post by pharmacy inventory management systems of the pharmacies surveyed without reference to patients. The sold units were summed by month, selling pharmacy and the German central pharmaceutical registration number ‘Pharmazentralnummer’ (PZN). This number is issued by the Informationsstelle für Arzneispezialitäten – IFA GmbH for the unambiguous identification of a drug with clear indication of specific dosage and package size of a single manufacturer or importer.
To derive the active ingredient from these packages delimited by PZN, the ABDA (German Association of Pharmacists)-database was used. The ABDA-database, published by a subsidiary of the ABDA, contains a directory of data coded via the PZN on pharmaceutical and economic data such as ingredients, dosage, and indication. All pharmacies and many other German health service providers have access to the database via their computer systems. It includes other sub-modules such as the ABDA-interaction database, which were used for the analysis of possible DDIs.
For calculations and statistical analysis IBM SPSS-Statistics Version 24 was used.
Identification of relevant substance groups interacting with antineoplastic and immune-modulating drugs
To be able to analyze relevant interactions of antineoplastic and immune-modulating drugs in practice, the sales figures of corresponding prescription medications in 2018 and 2019 were collected in 14 community pharmacies in the Munich area, Germany. To identify the drugs with the indication ‘antineoplastic and immune-modulating medication’, the ABDA-database indication classification was used. All substances with a code starting with ‘AL’, coding for antineoplastic and immune-modulating medication, were selected regardless of the diagnosis for which the drug was used in the patient’s individual cases. In the German healthcare system, pharmacies routinely do not have insight into patients’ diagnoses. Drugs, for which – according to expert information – no systemic intake and thus no interactions are to be expected, for example, creams containing the active substance fluorouracil, were excluded. The sales figures of all pharmacies were summarized and grouped by active ingredient using the ABDA-database. The resulting selection of active substances was limited to the 20 most common active substances.
For the identified most frequent active substances of antineoplastic and immune-modulating drugs among the pharmacies observed, possible interactions with other OTC-drugs were determined using the official ABDA interaction module. The corresponding drugs causing the interaction were divided into substance groups identified by a product-group-code according to ABDA-database.
Identification of sales within the relevant substance groups
The sales among the identified product-group-codes in 2018 and 2019 were collected among 11 pharmacies without an online store and three online stores of pharmacies. Every online pharmacy in Germany must operate a public pharmacy. In the case of the three online pharmacies, only medicines ordered online were considered. The drug identified by PZN, and date of sale were recorded in each case and summed up quarterly and product-group-code according to ABDA-database. The sales within the previously determined relevant substance groups were identified.
Comparison of sales channels (online pharmacy versus community pharmacy)
The sum of drugs sold within the relevant substance groups was calculated for the sales-channels community pharmacy and online pharmacy. The shares of these sales in total sales were calculated both per substance group and in total for all relevant substance groups. A Kolmogorov-Smirnov test and subsequent Mann-Whitney-U-test was used for each substance group to investigate whether the proportion of sales by month and pharmacy differed between online and community pharmacy, although complete independence of sales within individual pharmacies cannot be assumed. A p-value < 0.05 was considered statistically significant.
For a more detailed analysis of the differences between the two sales channels, the ratio of shares by sales channel was calculated by dividing the share of sales of the online pharmacy by the share of sales of the community pharmacies.
Results
A total number of 8950 drugs, classified as antineoplastic and immune-modulating by the ABDA-database, were sold by the investigated pharmacies between 1 January 2018 and 31 December 2019. The 20 most sold active ingredients among the total of 114 were identified as shown in Table 1. At the time of analysis in January 2020, a total of 49 interactions between these 20 active ingredients with non-prescription drugs were detected.
Total sales of antineoplastic and immune-modulating drugs in 2018 and 2019 among 11 community pharmacies by decreasing order.

By summarizing repeatedly occurring interactions, a total of 10 relevant substance classes could be identified: azole antimycotics, activated charcoal, folic acid, loperamide, NSAIDs, orlistat, potassium, PPIs, Saccharomyces boulardii, St. John’s wort. A pharmacological analysis of the underlying mechanisms of action and their potential influence on antineoplastic and immune-modulating therapy showed in most cases clinically relevant interactions (see Table 2). A total of 39 product groups according to the ABDA database were identified for the substance classes.
Exemplary mechanisms of action, pharmacological effect and clinical relevance of interactions of OTC drugs with antineoplastic and immune-modulating drugs.

CYP, Cytochrome; MTX, Methotrexate; NSAID, non-steroidal anti-inflammatory drug; OTC, over-the-counter; PPI, proton pump inhibitor.
Overall, 1,600,220 sales of OTC-drugs through 11 community pharmacies and 1,458,170 sales through three online pharmacies within these product groups were analyzed. There was a linear increase in the total number of drugs sold via the online pharmacies (p < 0.01) whereas the number of drugs sold by the community pharmacies (p < 0.01) showed a smaller increase respectively was relatively steady as shown in Figure 1.
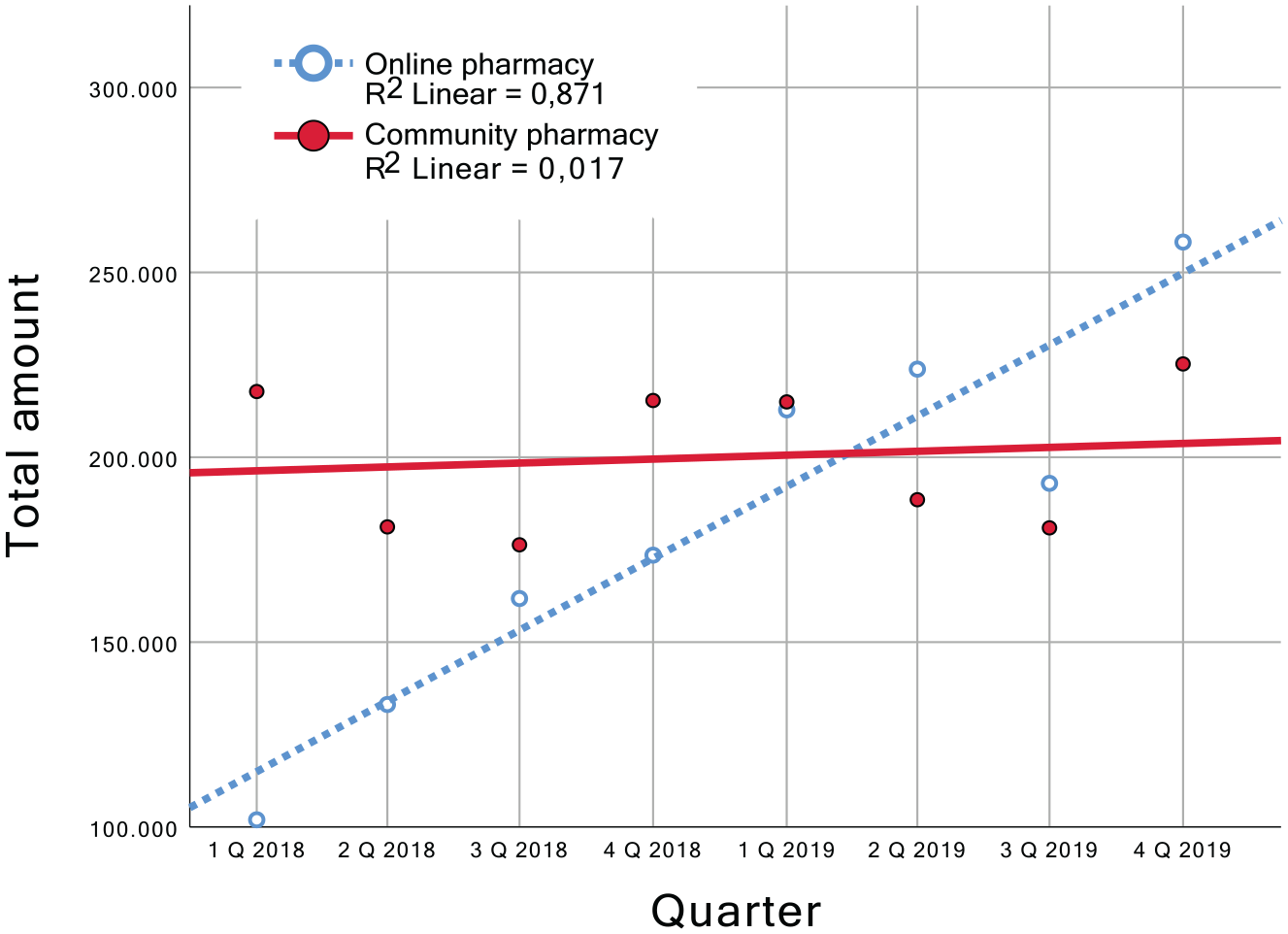
Total amount of packages sold by channel over time.
Within the identified relevant OTC product groups, 201,640 sales (12.60% of total sales) in community pharmacies and 80,071 sales (5.49% of total sales) via online pharmacies were evaluated. The relative share of drugs within the relevant product groups increased in a linear way for the online pharmacies (p = 0.048), a significant linear change within the community pharmacies could not be detected (see Figure 2).

Ratio total amount of packages sold: online pharmacy / community pharmacy by subtance group over time. This multi-plot graph illustrates the changes in sales ratios for various product groups between online and community pharmacies over a two-year period, with data presented quarterly. Ratios are calculated by dividing the percentage of sales within a product group as part of the total online sales by the corresponding percentage for community pharmacy sales. The Y-axis displays the resulting ratios on a logarithmic scale. Values greater than 1 indicate a higher proportion of sales in the online pharmacy, while values smaller than 1 signify a larger proportion of sales in the community pharmacy for each product group.
Total sales within the relevant groups ranged from 37 among orlistat drugs to 156,723 among NSAIDs as shown in Table 3. This corresponds to a share of 0.002% to 9.7% of sales within the respective sales channel.
The 10 most relevant OTC drug categories potentially causing interactions including sales and share between the two sales channels.

NSAID, non-steroidal anti-inflammatory drug; OTC, over-the-counter; PPI, proton pump inhibitor.
The monthly relative sales, determined by dividing the absolute number of sales for each substance group by the total number of sales for the respective sales channel, showed significant differences between online and community pharmacies. For azole antimycotics, NSAIDs, Saccharomyces-drugs, St. John’s wort, and PPIs, statistically highly significant differences (p < 0.001) were observed in each case. Statistically significant differences were detected for folic acid (p = 0.005), activated charcoal (p = 0.008), and potassium (p = 0.048).
An analysis of the total ratio of online versus community pharmacy sales shows differences between the individual product groups.
The ratio, determined by dividing the proportion of relevant sales in online pharmacies by the proportion of relevant sales in community pharmacies, varied from a minimum factor of 0.23 for NSAIDs to a maximum factor of 4.51 for orlistat medications, as shown in Figure 3. In relative terms, drugs from six substance groups had a ratio above one and thus were purchased more frequently in an online pharmacy, while drugs from four substance groups were purchased more frequently in a community pharmacy. Other drugs had a ratio of 1.08.
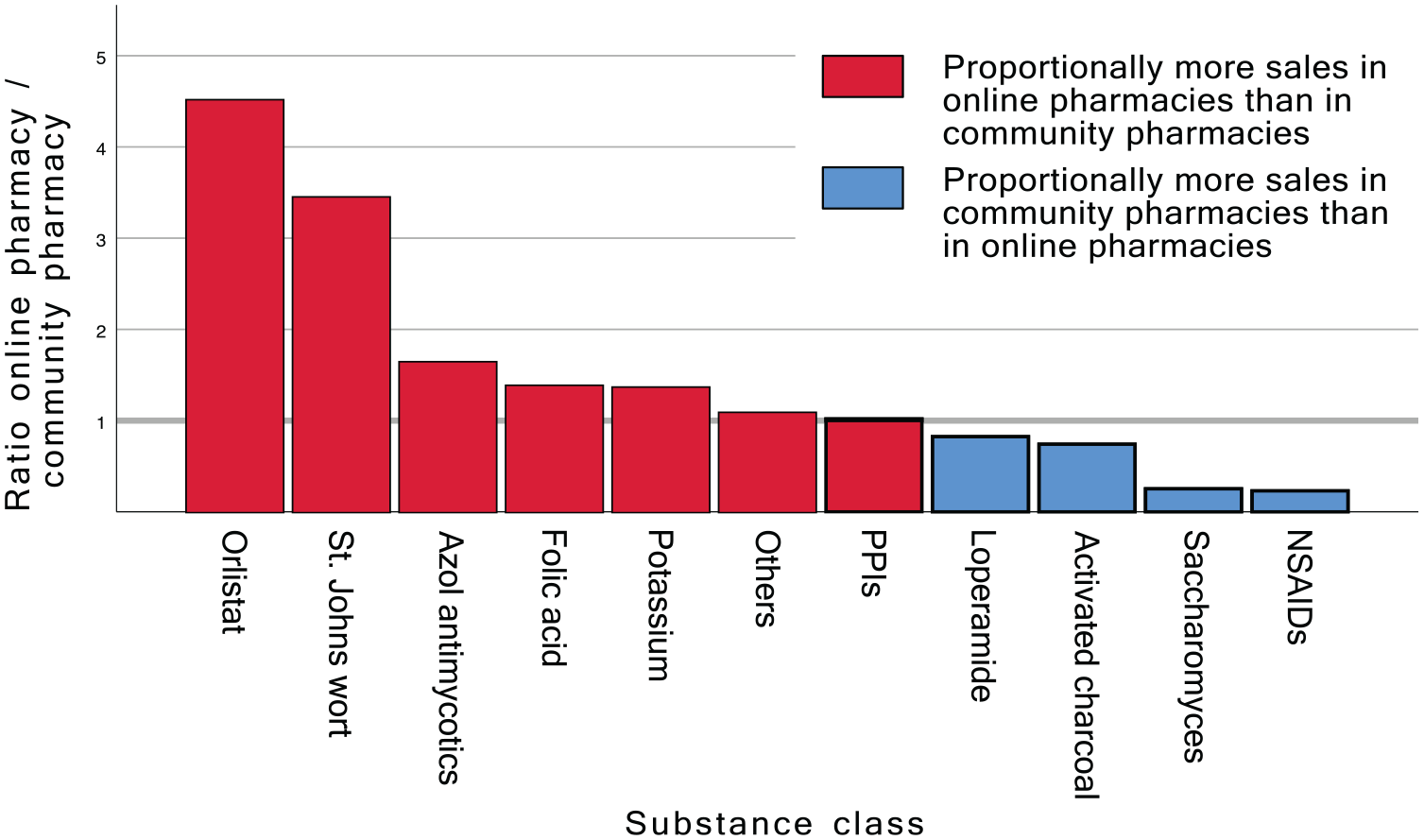
Comparison of sales: online pharmacy - community pharmacy. This graph compares sales ratios for various product groups between online and community pharmacies. Ratios are calculated by dividing the percentage of sales within a product group as part of the total online sales by the corresponding percentage for community pharmacy sales. The Y-axis displays the resulting ratios on a logarithmic scale. Values greater than 1 indicate a higher proportion of sales in the online pharmacy, while values smaller than 1 signify a larger proportion of sales in the community pharmacy for the respective product group.
Discussion
The aim of this study was to compare possible risks from OTC-drugs, purchased via online versus community pharmacies, interacting with high-potency drugs, based on real sales data. Through the usage of the ABDA-database, only established standards and data available to every German pharmacy were used. Most of the described interactions with antineoplastic and immune-modulating drugs which are listed in the interaction module of the ABDA-database, exist with other prescription drugs. To recognize and thus avoid potentially relevant and dangerous interactions, the prescribing physician should have a comprehensive understanding of the patient’s full medication regimen. This includes prescription and non-prescription drugs, dietary supplements and multivitamin preparations. However, several potential interactions with OTC drugs have been identified, particularly with 10 distinct groups of active substances. During the period covered by the study, we found that within the participating community pharmacies, about every eighth purchase of a non-prescription item was from one of these 10 groups, whereas within the participating online pharmacies, only every eighteen purchase of a non-prescription item was from these groups. Thus, the drugs obtained in a community pharmacy have more than twice the risk of a potential interaction. Eickhoff et al. 40 showed, that in about one of five encounters a direct pharmacist–patient interaction during a self-medication purchase revealed at least one relevant DRP. Hence, it can be assumed that a head-to-head consultation with a staff member in a community pharmacy will make it easier to identify corresponding interactions. However, the lower total rate of potential interactions might suggest that there is no increased risk of relevant DRPs with purchases in online pharmacies.
A closer look at the individual substance groups reveals that eight of the ten product groups showed a significant difference between the relative sales among online and community pharmacies. While NSAIDs were bought about four times more often in a community pharmacy, there were other drugs that were clearly more frequently bought via an online pharmacy like St. John’s wort (about 3.5 times as often).
With regards to drugs purchased more frequently in a community pharmacy one possible explanation could be that OTC NSAIDs and activated charcoal are often used to treat urgent complaints such as pain, diarrhea, or as an overdose or poison treatment and can be obtained very quickly by customers. Even though a significant difference between the share of sales via the two channels for loperamide could not be found in every case when looking at individual months and pharmacies, this does in principle fit into this scheme. NSAIDs and loperamide drugs are subject to prescription in Germany. There is the exception that these drugs may be purchased in small quantities for the temporary treatment of acute complaints without a prescription. 41 Online buyers may circumvent this restriction by making multiple purchases which in turn distorts the comparison between the two groups. Saccharomyces boulardii drugs are often used as adjuvant therapy in the context of an antibiotic therapy 42 and are proactively recommended in many cases by the pharmaceutical staff of a pharmacy during consultations.
The medicines purchased more frequently online are subject to a different characteristic. The intake of folic acid over a long duration has been prophylactically recommended in order to prevent diseases, especially during pregnancy. 43 Potassium drugs could be perceived by customers as harmless dietary supplements because of their non-prescription status and are also often taken over a long period of time mostly without an easily observable effect for patients. St. John’s wort is mostly used as a mild antidepressant. As depression is still seen as a stigma, particularly by the elderly, it is often concealed by those affected who also tend to self-medicate. 44 Azole antimycotics are usually used to treat fungal infections, which are often unpleasant for patients. Orlistat is used to treat obesity, also a stigmatized condition. 45 For orlistat, the greatest absolute difference between the groups was observed. However, the difference in the groups by month and pharmacy was not significant, possibly due to the small number of cases. A supposedly anonymous purchase of a drug among the three groups over the internet could therefore be preferred by patients in an attempt to avoid discrimination.
The novel approach based on real sales figures may be particularly helpful indicating latent problems in practice. However, an increased statistical probability of an interaction does not mean that it has effectively occurred. A practical clinical relevance of interactions described in studies and subsequently in databases like the ABDA-database is also not always given. The individual patient characteristics like indication for antineoplastic or immune-modulating therapy can also influence the importance of the interaction and can both up- and downgrade its significance. Even if an interaction itself seems irrelevant in a standard case as shown in Table 2, the mechanism of interaction can be very relevant when transferred to other drugs that are not included in the most sold drugs within this study, or even to food products as the following examples show.
Azole antimycotics exhibit a very high DDI potential through cytochrome (CYP) inhibition after systemic administration. Particularly in combination with drugs with a narrow therapeutic range as ciclosporin, serious consequences of the interaction are possible, as presented in Table 2. In contrast, the clinical relevance of this interaction is usually less relevant if azole antimycotics are applied in topical or vaginal dosage forms on intact skin. Although drug absorption from these methods of application is low, a systemic effect cannot be excluded. For example, econazole, a known inhibitor of CYP3A4 and CY2C9, has been shown to exhibit clinically relevant interactions with oral anticoagulants, which like ciclosporin, are CYP3A4 substrates inhibition of cytochromes P450 by antifungal imidazole derivatives. Thus, in Germany, only topical and vaginal products with azole antimycotics are available OTCs; all other forms of administration require a prescription. However, clinically relevant interactions due to CYP enzyme inhibition with OTC products frequently occur in practice. While most other inhibitors of CYP enzymes are prescription only, there are also foods such as grapefruit and bitter orange with the same effect that can seriously jeopardize the effect of antineoplastic and immune-modulating therapy.19–21
Folic acid reduces both the efficacy as well as the folate antagonism-dependent side effects of the folic acid antagonist Methotrexate (MTX) as shown in Table 2. MTX frequently leads to mucosal, gastrointestinal, hepatic or hematologic adverse reactions which can be attenuated by supplementation with folic or folinic acid. Therefore, intentional folic acid substitution in patients receiving MTX for the treatment of rheumatoid arthritis is common; in Germany 5–10 mg once a week approximately 24 h after the weekly MTX administration are recommended. Shorter time intervals risk reduced therapeutic effect of MTX.23–25 Thus, it can be debated whether to consider the use of folic acid and MTX as a ‘real’ drug interaction as they are concomitantly prescribed with preventative intent. As in this study drug interactions were determined using the ABDA-database interaction module and this database classifies folic acid and MTX as an interaction, it is considered as an interaction in Table 2. The opposite effect – increased drug toxicity – is another possible interaction that can occur with OTC folic acid, for example, in combination with the cytostatic capecitabine. Capecitabine is a pro-drug of 5-fluorouracil that is used, for example, for the therapy of breast cancer. While co-administration with folinic acid to enhance the effect of i.v. 5-fluorouracil administration is common, the combination of capecitabine with folinic or folic acid does not bring any therapeutic advantages, but only increases its toxicity. Hence, the combination must be avoided. Remarkably, folic acid is not only contained in OTC products but also in food supplements and multivitamin preparations. Taken together, these drug interactions with folic acid demonstrate the high need for counseling of patients requiring folic acid.
Although the clinical relevance of interactions between loperamide in normal doses and CYP3A4- and P-glycoprotein inhibitors seem to be small (Table 2), extremely high dosages of loperamide can have serious central and peripheral impact. Euphoric effects, severe heart problems (e.g. QT prolongation and ventricular tachycardia) and even death have occurred.26–28 The FDA (U.S. Food and Drug Administration) has released several ‘Drug Safety Communication’ letters in regard to this problem. 46 Since antineoplastic and immune-modulating drugs often cause diarrhea as a side effect, this interaction should be considered by pharmacies when dispensing OTC loperamide.
Clinically high relevant interactions with orlistat are possible, as demonstrated in Table 2. Orlistat reduces the absorption of fat by the inhibition of gastrointestinal lipases. As ciclosporin absorption is fat-dependent, it is assumed that orlistat treatment leads to impaired ciclosporin absorption resulting in reduced immune-suppressive effects of ciclosporin. Therefore, in Germany, OTC orlistat is contraindicated when used with ciclosporin.32,33 Despite this relevant example of an interaction between orlistat and an immune-modulating drug, interactions with orlistat in general are not very prevalent due to its overall low number of sales. Nevertheless, although immune-modulating and antineoplastic drugs themselves often induce weight loss, it is not uncommon that affected patients are also prescribed antidepressants as part of their therapy. Antidepressants in turn often lead to severe weight gain, which could trigger the use of orlistat.
In a recent study, Hall et al. 35 sum up the frequently described interaction of low-dose MTX and PPIs respectively NSAIDs as not substantiated, see also Table 2. Much more frequently, however, relevant interactions between PPIs and oral anti-cancer drugs occur due to the PPI-induced increase in gastric acid pH leading to altered absorption of the interaction partner. This particularly frequently affects tyrosine kinase inhibitors (TKIs) such as erlotinib, 47 as TKI absorption is often dependent on gastric pH. For some drugs, the interaction can be avoided by adhering to the correct time of administration. For example, with palbociclib (see Table 1), the effect is not significant when taken during a meal. 48 Since PPIs are in widespread use and furthermore available without prescription, there is a special and extremely important need for pharmacist consultations for patients on antineoplastic and immune-modulating therapy.
Potassium intake not only represents electrolyte substitution or nutritional supplementation. Due to the additive effects of the increase in potassium, it can enhance the toxicity of various drugs. Simultaneous use with, for example, tacrolimus can lead to increased occurrence of severe hyperkalaemia and renal failure; see Table 2. This interaction is highly relevant as hyperkalaemia and renal dysfunction already occur very frequently (>10%) with tacrolimus monotherapy. 36 On the other hand, potassium substitution may be intentionally used for treatment of hypokalemia, which also constitutes a high-risk condition. Therefore, patient requests for compounds with potassium requires precise counseling as well as the monitoring of serum levels of potassium by healthcare professionals. Sometimes even serum levels of the drug interaction partner like tacrolimus have to be monitored. Specific caution is also required when eating high amounts of potassium-rich foods (e.g. bananas, apricots). This is often considered harmless but can lead to very serious consequences in practice.
St. John’s wort extracts containing hyperforin are strong CYP and P-glycoprotein inducers that leads to reduced plasma levels affecting the dose-requirement of a wide range of drugs, including ciclosporin, tacrolimus and palbociclib, as presented in Table 2. The reduction or even lack of therapeutical effect is highly relevant, as most drugs in targeted cancer therapy are CYP substrates.38,39 Moreover, this interaction is likely to occur frequently in practice, because in order to treat depression that often arises during cancer therapy, many affected patients turn to mild herbal OTC drugs rather than more potent antidepressants. This very serious example shows particularly clearly the risk that can emanate from self-medication during antineoplastic and immune-modulatory therapy.
While the strong interaction is well researched for hyperforin-containing St. John’s wort extracts and therefore already often communicated pro-actively, this is not the case for all herbal drugs due to the lack of clinical studies in this area, although in vitro analyses describe strong potential problems. 49 Protective mechanisms of the human body limit interactions at naturally occurring doses, which is why many of the herbal medicines receive little attention in practice. However, new methods to improve the systemic availability of purportedly healthful phytochemicals bear the risk that interactions with herbal drugs that were previously considered irrelevant, may now become a major problem in drug therapy. 50
There is also a fluent transition from drugs to herbal or synthetic dietary supplements and even functional food products that can cause the same effects. Such foods are also referred to by the not clearly defined name nutraceuticals, a combination of the terms nutrient and pharmaceutical. They have been the fastest growing segment of the food industry for years and are expected to grow even further.51,52 However, the exact legal classification is often unclear and the associated regulation is not sufficiently implemented. 53 Nutraceuticals and dietary supplements can be advertised and often purchased online with even less control than with OTC drugs.
Studies have shown that most patients search for information on medical topics online. 54 However, users often showed limited awareness of how to evaluate this internet-based information about medicines. 55 In addition, studies demonstrated that even in the online marketing of specific drugs and supplementary products, often not all prescribing information is provided as required.56,57 Presumably a certain number of patients after obtaining information via the internet, for example, through an online store, will also order directly there. Therefore, in addition to OTC drugs, special attention should be paid to herbal medicines and dietary supplements and nutraceuticals when analyzing self-medication of patients with antineoplastic and immune-modulatory therapy especially those purchased via the internet.
It can be summarized that an often time-consuming and precise differentiation of the seriousness and practical clinical relevance of the individual DRPs would therefore still have to be made in individual cases, for example, using systematic medication reviews as already practiced during routine care in some public pharmacies. 58 This should be carried out by specially trained personnel and not solely based on databases. Prely et al. 59 investigated the DDIs found using multiple interaction databases in patients receiving oral anti-cancer therapy. They concluded that the interactions found were not systematically significant and called for the development of more sensitive and specific interaction databases. According to a recent review, this is of particular importance, because patients who are increasingly using DDIs databases with huge differences in regard to service quality, when in doubt, will turn to their doctor or local pharmacist. 60
Limitations
This study is subject to several limitations. First, other herbal and synthetic OTC drugs, dietary supplements, and nutraceuticals with potentially high interaction potential might be commonly sold by pharmacies but are hidden in substance groups that were not investigated in this study. Their identification is difficult because antineoplastic and immune-modulatory therapy have different and partly individual aspects to consider. There is also a lack of relevant published studies related to these substances. Second, it is not easy to obtain economically relevant data, especially from online pharmacies. Therefore, a limited number of pharmacies was observed, which makes it difficult to compensate for individual fluctuations in the sales figures. Subsequent investigations should examine whether the composition of the sales was representative and thus could be transferred to other locations, pharmacies and online-shops. Hence, other drugs or active substances could also be relevant in the context of antineoplastic and immune-modulating therapy. Third, on the other side, the methodology used does not identify drugs taken concomitantly by a given patient as sales were recorded without reference to patients. Furthermore, the screening of the ABDA-indication group ‘antineoplastic and immune-modulating medication’ was carried out independently of the diagnoses, which are not available to pharmacies in Germany. Some drugs classified in this group are not only used in cancer treatment, for example, MTX is also used for rheumatoid arthritis. Fourth, there was no inclusion of drugs which cannot be purchased from the community pharmacies or drugs that are not registered via the corresponding ABDA product groups, such as drugs produced individually for a patient. Even in the context of a non-oral therapy, whether in a clinic, doctors practice or at home, analogous interactions with OTC drugs can occur in the same way and have therefore to be considered. Fifth, as described, internet purchases can depend heavily on the price offered. The recorded sales, especially by online pharmacies, could therefore also be influenced by the offer consisting inter alia of price, stock, competition, and further factors. Subsequent studies need to examine the influence of these aspects on the purchasing process both in online and community pharmacies. In addition, certain factors such as regional and national outbreaks of diseases such as COVID and influenza or severe weather conditions could influence the two sales channels differently. Sixth, due to differing legal restrictions, these findings may not be transferable to other countries. Thus, possibly different drugs are prescribed to patients as here in Germany or others available without a prescription in certain countries. The availability of drugs via online pharmacies is also often regulated differently. Other OTC drugs could therefore be of greater relevance abroad.
In view of the current developments in Germany regarding electronic prescriptions, the matter of drug interactions is becoming even more relevant. The proportion of drugs sold via the internet is currently increasing every year 18 which is consistent with the observed linear increase in the number of drugs sold by online pharmacies in this study. The nationwide introduction of an electronic prescription in Germany, which is currently being introduced, 61 would eliminate the need to send in the physical prescription in advance when purchasing a prescription drug via the internet. This is expected to further increase the sale of drugs online, especially prescription drugs. At the same time, it has been reported that electronic prescriptions have even led to an increase in prescribing errors,62,63 which was inter alia justified by a reduction in face-to-face interactions. Taken together with the results of this study, it therefore seems reasonable to explore possible further risks caused by the shift of drug purchases away from the community pharmacy to the internet and identify possible solution approaches.
Conclusion
The study shows that there are large numbers of OTC drugs that exhibit clinically relevant DDIs with antineoplastic and immune-modulating drugs. In oncological care, we identified specific substance groups within the OTC drugs that the patients take additionally to their cancer or immune-modulating medication and which have a particular high risk for DDIs. The reason why this patient group uses OTC drugs that interact with their oncological therapy may be due to patients’ reluctance to talk to their local pharmacist about conditions such as depression, fungal infections or obesity that are typically treated with these substance groups. This problem could be solved by pro-active communication initiated by the treating physician pointing out the risk of taking OTC drugs with these indications during oncological therapy. In the same way, pharmacies, both online and community, could also point out corresponding risks especially within these substance groups. Structured and maybe even automated communication could be a useful tool, especially for online pharmacies. Based on these findings, further research could investigate not only which other drugs, but also dietary supplements or foods, these findings can be applied to in addition to the statistically most frequently sold active substances.
It is likely that these measures together with the improvement of databases could help to turn this existing risk associated with potentially interacting OTC drugs, sold via community pharmacies as well as via less controlled online pharmacies, into ensured effectiveness of the antineoplastic and immune-modulating drugs and thus into therapeutic success.
