Abstract
Aims:
The gene polymorphism of voriconazole metabolism–related liver enzyme is notable in East Asia population. It casts a significant influence on the rational use of voriconazole. We conducted this study to investigate the relationship between steady-state voriconazole trough concentration (Ctrough) and adverse effects (AEs), especially hepatotoxicity.
Methods:
We conducted a real-world study in the Jinling Hospital from January 2015 to June 2020. A total of 140 patients receiving voriconazole were enrolled in this study. The determination and scoring of voriconazole-associated hepatotoxicity were performed according to the Roussel Uclaf Causality Assessment Method scoring scale and the severity of hepatotoxicity was graded according to the Common Terminology Criteria for Adverse Events (CTCAE).
Results:
Elevated steady-state voriconazole Ctrough with concomitant AEs are the most common reason for dose adjustments during treatment. Compared with the group without any AEs, voriconazole Ctrough was significantly higher in the hepatotoxicity and neurotoxicity groups, and the incidence of both events showed an overall increasing trend with increasing voriconazole Ctrough. Hepatotoxicity occurred in 66.7% of patients within 7 days of the first dose of voriconazole and 94.4% within 15 days of the dose. Steady-state voriconazole Ctrough >3.61 mg/l was associated with an increased incidence of hepatotoxicity (area under the curve = 0.645, p = 0.047). Logistic regression analysis showed that timely voriconazole dose adjustment was a predictor of attenuated hepatotoxicity after adjustment for confounders, but hepatotoxicity was not associated with voriconazole Ctrough measured at a single time point.
Conclusion:
Hepatotoxicity and neurotoxicity correlate with voriconazole Ctrough, and dose reduction in patients with elevated steady-state voriconazole Ctrough may prevent hepatotoxicity. In patients with early occurrence of hepatotoxicity, initial therapeutic drug monitoring (TDM) might predict the risk of hepatotoxicity. Follow-up TDM may be necessary to predict late onset hepatotoxicity.
Plain Language Summary
Introduction
Therapeutic drug monitoring (TDM) has an important role in optimizing antifungal therapy and is routinely recommended for voriconazole in the Chinese Pharmacological Society guidelines, the British Society for Medical Mycology guidelines, and the Infectious Diseases Society of America guidelines.1–4 Different guidelines recommend different voriconazole trough concentration (Ctrough). This is most likely due to the different sources of evidence. Furthermore, the proportion of slow metabolic genotypes varies across countries. Asians have a higher proportion of slow metabolic genotypes compare with Caucasian or black populations, which suggests that Asian populations are at greater risk of exposure to high drug concentrations.
In adult clinical trials with voriconazole, the rates of AEs reported in the instructions differed from those in clinical practice. A study published by the French Network of Pharmacovigilance Centers analyzed the AEs associated with voriconazole during first 4 years of marketing. They found that abnormal liver function was noticeable as the most common adverse effect (AE). 5 Other studies also showed that hepatotoxicity was the most common cause of voriconazole dose reduction or discontinuation.6–9 The results of a multicenter study by Hamada et al. 7 showed that the rate of dose reduction or discontinuation due to hepatotoxicity was significantly higher than that of visual disturbances. Therefore, monitoring on hepatotoxicity is warranted compared with other AEs of limited duration or lower frequency. 10
Clinical trials of voriconazole have reported a varying incidence of hepatotoxicity, with abnormal liver enzyme elevations reported in the range of 1–69%.11–15 A large number of studies8,9,12,16–20 utilized the Common Terminology Criteria for Adverse Events (CTCAE) to assess hepatotoxic events. 21 Meanwhile, there was a number of studies22–25 using different definitions. There is a risk that different definitions of AEs may cause underestimation or overestimation of the incidence of voriconazole toxic events and lead to errors in other conclusions related to them.
Controversy still exists regarding the correlation between voriconazole concentrations and hepatotoxicity. Several authors have reported a correlation between voriconazole Ctrough above 4.0 or 6.0 mg/l and hepatotoxicity.12,16,18,24,26–28 In determining drug-induced liver injury (DILI), 8 several potential confounding factors need to be taken into account, such as patients’ previous liver function, the concomitant use of potentially hepatotoxic drugs, ethnic groups, and so on. Many studies failed to assess these factors.
In contrast, several prospective studies found no relationship between steady-state voriconazole Ctrough and hepatotoxicity. Pascual et al. 29 showed that although an increased incidence of hepatotoxic events were observed when voriconazole was >5.5 mg/l (8% versus 19%), the difference was not statistically significant; Park et al. 6 suggested regular TDM with voriconazole reduced the incidence of discontinuation for AEs, but not the overall rate of AEs associated with voriconazole treatment.
There are no uniform criteria for determining DILI, and several methods have been developed to assess the causality of DILI. 30 The Roussel Uclaf Causality Assessment Method (RUCAM) is a scale that reflects the likelihood that hepatotoxicity is induced by a drug by assigning different scores to the clinical, biochemical, serological, and radiological characteristics of hepatotoxicity based on the aggregated calculated scores.
In this study, we evaluated voriconazole dose adjustment regimens and target drug concentration attainment rates. RUCAM and CTCAE were used to measure voriconazole-induced hepatotoxicity. In order to support voriconazole’s rational clinical use, a correlation between toxic events (especially hepatotoxicity) and voriconazole steady-state Ctrough was investigated.
Methods
Patients
The study included patients with a diagnosis of pulmonary fungal disease who visited Jinling Hospital and received intravenous or oral voriconazole (Pfizer) between 1 January 2015 and 30 June 2020. Inclusion criteria were (1) age ⩾18 years; (2) clinical diagnosis of patients with pulmonary fungal disease; (3) hospitalization for ⩾7 days; and (4) at least one steady-state voriconazole Ctrough data obtained. Exclusion criteria were (1) combined definite hepatobiliary disease, such as all types of viral hepatitis, alcoholic liver disease, autoimmune liver disease, and primary biliary or sclerosing cholangitis; (2) hepatoprotective and enzyme-lowering therapy during the study period; (3) other drugs with definite hepatotoxicity (e.g. isoniazid, rifampicin, and docetaxel) were used in combination with voriconazole during voriconazole administration.
Definition of research
Definition of voriconazole-induced hepatotoxicity
The determination and scoring of voriconazole-associated hepatotoxicity were performed according to the RUCAM scoring scale modified by Danan and Teschke 31 in 2016 (Table 3). First, the liver enzymes that were first measured to indicate the occurrence of DILI were included in the evaluation, and the R value was obtained by dividing the number of times the measured alanine aminotransferase (ALT) value was elevated compared with the upper limit of normal (ULN) by the number of times the measured alkaline phosphatase (ALP) value was elevated compared with the ULN of ALP: an R value ⩾5 indicated hepatocellular injury; an R value ⩽2 indicated cholestatic injury; and between 2 and 5 indicates mixed injury. Then, according to the R value corresponding to the type, the scale was used for the overall score: a score of 0 or below indicates that the drug is ‘excluded’ as the cause of hepatotoxicity; a score of 1–2 indicates ‘unlikely’; a score of 3–5 indicates ‘possible’; 6–8 points mean ‘probable’; and more than 8 points means ‘highly probable’. Based on the above definition, an RUCAM score of ⩾6 was considered in this study as probable or highly probable hepatotoxicity induced by voriconazole.
Criteria for determining the severity of hepatotoxicity
In this study, the severity of hepatotoxicity was graded according to CTCAE v.5.0. 21 The general grading principles of CTCAE can be divided into five categories. The standard ULN values for total bilirubin (TBil), ALP, gamma-glutamyl transpeptidase (GGT), aspartate aminotransferase (AST), and ALT in the laboratory of the Jinling Hospital Laboratory were 17.1 µmol/l, 150 U/l, 50 U/l, 40 U/l, and 40 U/l, respectively.
Dose adjustment and therapeutic drug monitoring
Therapeutic drug monitoring in this study was usually performed on day 4 or day 5 after the first dose of voriconazole administration. Clinicians typically used the following strategies for voriconazole dose adjustment (compliance rate = 93.2%): in patients with high steady-state voriconazole Ctrough over 5.5 mg/l with or without AE, 50% dose reduction was performed, followed by a further 50% dose reduction if the concentration remained high. If necessary, a direct discontinuation was adopted. On the contrary, 50% dose increase was adopted when steady-state voriconazole Ctrough were lower than 1 mg/l.
Definition of steady-state voriconazole Ctrough
For patients receiving a loading dose of intravenous or oral voriconazole (defined as voriconazole administered intravenously at 6 mg/kg q12 h twice within first 24 h, followed by voriconazole administered intravenously at 4 mg/kg q12 h or orally at 200 mg twice daily), Ctrough measured at or after 24 h of dosing was considered steady-state Ctrough. 32 For patients who did not receive a loading dose, the values measured on or after day 6 of the treatment dose were considered steady-state Ctrough; for patients who reached steady-state Ctrough during treatment but had subsequent voriconazole dose adjustments for various reasons, the values measured on or after day 4 of the dose adjustment were considered steady-state Ctrough. 33
Genotyping and genotype classification
Genotyping of three single nucleotide polymorphisms (SNPs) from stored DNA using the TaqMan analysis to identify major CYP2C19 alleles. rs4244285, rs4986893, and rs12248560 all had >98% call rates, defining the *2, *3, and *17 alleles, respectively. 34
Patients were classified into metabolizer phenotypic categories using the established common consensus star allele nomenclature. 35 Patients without a *2, *3, or *17 allele (i.e. *1/*1) were classified as ‘extensive metabolizers’, those with one *17 allele (i.e. *1/*17) and *17 homozygotes (i.e. *17/*17) were classified as ‘ultrametabolizers’. Patients with one *2 or *3 allele (i.e. *1/*2 or *1/*3) were classified as ‘intermediate metabolizers’, while patients with two *2 or *3 alleles (i.e. *2/*2, *2/*3, or *3/*3) were classified as ‘poor metabolizers’.
Statistical analysis
Normally distributed continuous data are expressed as mean value (±standard deviation, SD), non-normally distributed continuous data are expressed as median (the interquartile range, IQR), and categorical variables are expressed as counts (%). Owing to the non-normality of voriconazole concentrations, the Mann–Whitney U test was used to compare the mean values between the two sample groups. The Chi-square 2 test or Fisher’s exact test was used to compare the differences in frequency distribution between groups. Binary logistic regression analysis was used for multifactor analysis. Spearman’s method was used to study the correlation between variables. All the above analyses were performed using SPSS (version 25.0), and values were considered statistically significant when p value less than 0.05.
Results
Baseline patient characteristics
Of the 216 patients screened for this study, 76 patients were excluded. The reasons including age less than 18 years (n = 5), unmeasured steady-state blood concentration data (n = 48), and combined definite hepatobiliary disease prior to voriconazole treatment (n = 23). Finally, a total of 140 patients were enrolled in the study analysis. According to the CYP2C19 genotype classification, there were 14 (32.6%) extensive metabolizers, 25 (58.1%) intermediate metabolizers, and 4 (9.3%) poor metabolizers with voriconazole Ctrough of 2.5 mg/l (1.0–7.5 mg/l), 6.7 mg/l (1.4–8.9 mg/l), and 4.4 mg/l (2.5–6.1 mg/l). The demographic and clinical characteristics of the study patients are shown in Table 1.
Clinical characteristics of the study population.

Data are n (%), median (IQR), and mean value (SD).
Including sinusitis (n = 1), allergic rhinitis (n = 1), chronic pulmonary heart disease (n = 2), lobectomy (n = 2), gallbladder stones (n = 3), postcholecystectomy (n = 2), duodenal ulcer (n = 1), splenectomy (n = 1), ulcerative colitis (n = 1), postoperative appendicitis (n = 1), hypothyroidism (n = 2), thyroid nodule (n = 1), anemia (n = 5), prostate enlargement (n = 7), bone and joint injury surgery (n = 6), brain atrophy (n = 1), postcataract surgery (n = 1), depression (n = 1), post-tonsillectomy (n = 1), posthysterectomy (n = 1), postoperative left lower extremity varicose veins (n = 1), rheumatoid arthritis (n = 3), ankylosing spondylitis (n = 3), systemic lupus erythematosus (n = 3), mechanized pneumonia (n = 3), interstitial pneumonia (n = 3), ANCA vasculitis (n = 1), mixed connective tissue disease (n = 1), nephrotic syndrome (n = 1), dry syndrome (n = 1), gout (n = 1), and hyperthyroidism (n = 1).
Sample size of 43 people.
Including β-lactams (cephalosporins, imipenem, biapenem, piperacillin), quinolones (levofloxacin, moxifloxacin), tigecycline, glycopeptides (teicoplanin, vancomycin), linezolid, macrolides (clarithromycin, azithromycin), ornidazole, tetracyclines (minocycline), aminoglycosides (etimesine), and compound sulfamethoxazole.
Including caspofungin, amphotericin B, and polymyxin.
Including oseltamivir and ganciclovir.
Including methylprednisolone and prednisone.
Voriconazole dose adjustment regimen, reasons, and target therapeutic concentration attainment rate during treatment
Forty-four (31.4%) patients required dose adjustment during voriconazole treatment (Table 2), of which 65.9% (29/44) patients adjusted dose within 1 week after the first dose, 72.7% (32/44) required a reduction of voriconazole dose, 18.2% (8/44) required an increase of voriconazole dose, and 9.1% (4/44) voriconazole discontinued due to adverse event. Voriconazole Ctrough over the upper threshold (63.3%, 28/44) was the most common cause of dose adjustments during treatment (Table 3). Of which, the dose was reduced in 11 patients to prevent AEs simply because of high voriconazole Ctrough. Up to 60.7% (17/28) of patients had elevated voriconazole Ctrough with AEs, the combination was judged strongly correlated by clinicians, and the dose was subsequently reduced. The most common AE was elevated liver enzymes (42.9%, 12/28) followed by central neurotoxicity (10.7%, 3/28), gastrointestinal symptom (3.6%, 1/28), and rash (3.6%, 1/28). 84.1% (37/44) of patients underwent a single-dose adjustment to achieve the target Ctrough range (i.e. 1.5–5.5 mg/l). Only a small percentage of patients required more than twice dose adjustments (Table 4).
Time and program of voriconazole dose adjustment during treatment.
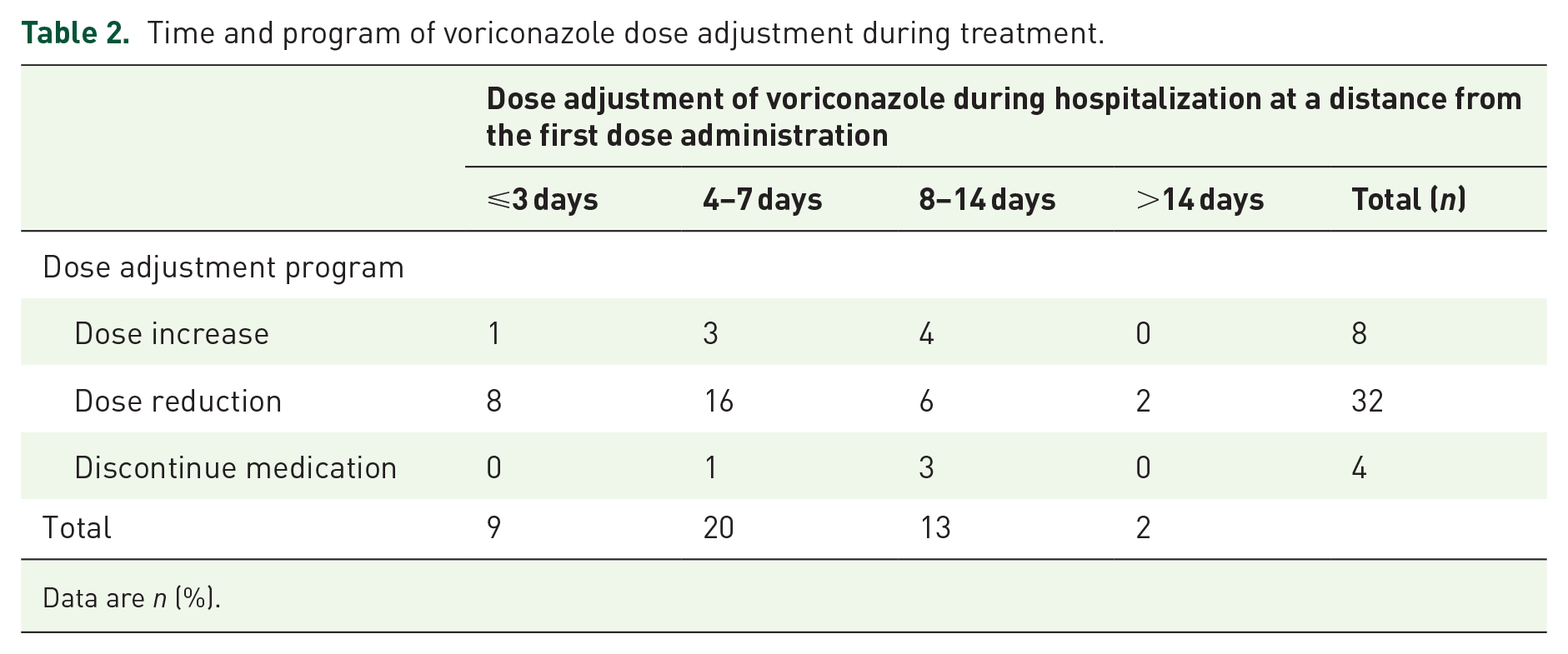
Data are n (%).
Reasons for dose adjustment of voriconazole during treatment.
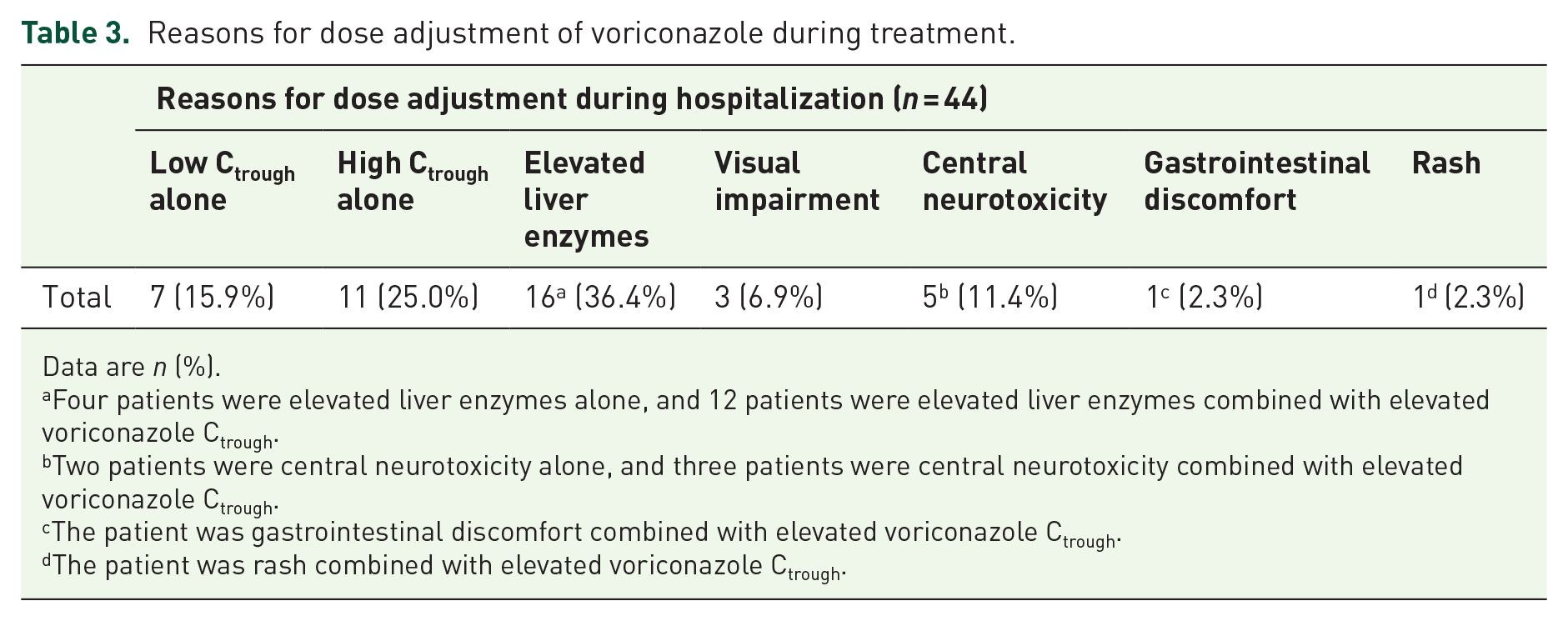
Data are n (%).
Four patients were elevated liver enzymes alone, and 12 patients were elevated liver enzymes combined with elevated voriconazole Ctrough.
Two patients were central neurotoxicity alone, and three patients were central neurotoxicity combined with elevated voriconazole Ctrough.
The patient was gastrointestinal discomfort combined with elevated voriconazole Ctrough.
The patient was rash combined with elevated voriconazole Ctrough.
Clinical data of patients who received ⩾2 voriconazole dose adjustments during treatment.

iv, intravenous drip; M, male; qd, administered once daily; q8 h, administered every 8 h; q12 h, administered every 12 h; qod, administered every other day.
Voriconazole-associated AEs and their correlation with steady-state Ctrough
Of the 140 patients, 24.3% (34/140) had at least one or more AEs during voriconazole treatment. The most common AE was hepatotoxicity [12.9%; the median steady-state voriconazole Ctrough was 6.57 mg/l (IQR = 4.28–8.22 mg/l)], followed by the neurotoxicity group [7.1%; 8.00 mg/l (IQR = 4.73–10.54 mg/l)]. As shown in Figure 1, compared with the steady-state voriconazole Ctrough in 52 patients without any AEs during treatment, the visual impairment group (6.89 versus 4.17 mg/l; p = 0.543), the gastrointestinal AEs group (4.49 versus 4.17 mg/l; p = 0.886), cardiac-related AEs group (8.28 versus 4.17 mg/l; p = 0.279), and skin-related AEs group (3.89 versus 4.17 mg/l; p = 0.538) were not statistically significant differences. Steady-state Ctrough, however, were significantly higher in the CTCAE (Δ ⩾2) group (8.20 versus 4.17 mg/l; p = 0.016), hepatotoxicity group (6.57 versus 4.17 mg/l; p = 0.021), and neurotoxicity group (8.00 versus 4.17 mg/l; p = 0.031) (Figure 1).

AEs and voriconazole Ctrough of patients (n = 140) during voriconazole treatment.
Voriconazole-induced hepatotoxicity
Voriconazole-induced hepatotoxicity staging and CTCAE grading
Based on RUCAM, a total of 18 patients (12.9%) were diagnosed as probable or highly probable to have DILI due to voriconazole, with a mean RUCAM score of 7.78 (±1.2). By R value type, five patients (3.8%, 5/140) were hepatocellular injury type, five patients (3.8%, 5/140) were cholestasis type, and eight patients (5.7%, 8/140) were mixed type. According to the CTCAE grading, 27.8% (5/18) patients with hepatotoxicity were evaluated as grade 1, 38.9% (7/18) patients with hepatotoxicity were evaluated as grade 2, and 33.3% (6/18) patients with hepatotoxicity were evaluated as grade 3, and no patients with grade 4 were identified (Table 5).
Hepatotoxicity classification and CTCAE classification.

CTCAE, Common Terminology Criteria for Adverse Events; RUCAM, Roussel Uclaf Causality Assessment Method.
Data are n (%).
Incidence and timing of voriconazole-induced hepatotoxicity
The incidence of hepatotoxicity and neurotoxicity increased with the steady-state voriconazole Ctrough. Among the four groups, the incidence of hepatotoxicity and neurotoxicity ranged between 22.2% and 25% at steady-state voriconazole Ctrough of >4.0 mg/l. This was higher than the incidence at 4.0 mg/l (5–13.3%; Figure 2). There were 66.7% of patients who experienced hepatic toxicity within 7 days of the first voriconazole dose, and 94.4% within 15 days (Figure 3).

Correlation between the incidence of hepatotoxicity and central nervous system toxicity and voriconazole Ctrough.

The time interval between the onset of hepatotoxicity and the first dose of voriconazole.
Predictive thresholds for Ctrough in voriconazole-induced hepatotoxicity
The analysis of receiver operating characteristic (ROC) showed that steady-state voriconazole Ctrough >3.61 mg/l were associated with an increased incidence of hepatotoxic events, with an area under the ROC curve of 0.645 (95% confidence interval (CI) = 0.534–0.757, p = 0.047; Figure 4).

The optimal voriconazole Ctrough threshold for predicting the occurrence of hepatotoxicity.
Multifactorial analysis of voriconazole-induced hepatotoxicity
Reverse stepwise binary logistic regression models identified timely voriconazole dose adjustment as a protective factor in reducing hepatotoxicity [odds ratio (OR) = 0.189, 95% CI = 0.054–0.652, p = 0.008; adjusted OR = 0.190, 95% CI = 0.065–0.554, p = 0.002; Figure 5]. After adjusting for confounders, patients with elevated voriconazole Ctrough showed an increased risk of hepatotoxicity (OR = 1.088, p = 0.390). The model had good goodness of fit [Hosmer and Lemeshow test Chi-square = 9.96, degree of freedom (df) = 1, p = 0.268; Nagelkerke R2 = 0.187], and its overall prediction power was 82.1%.

Logistic regression analysis of voriconazole-induced hepatotoxicity.
Correlation between voriconazole steady-state Ctrough and liver enzymes
Spearman’s correlation analysis showed a positive correlation between steady-state voriconazole Ctrough and TBil (r = 0.246), ALT (r = 0.270), AST (r = 0.405), ALP (r = 0.276), and GGT (r = 0.185) levels (p value < 0.05; Figure 6).

Spearman’s rank-order correlation coefficient between liver enzymes and voriconazole Ctrough (n = 140).
Change in CTCAE classification of liver enzymes before and after voriconazole treatment
Among 140 patients, TBil, ALT, AST, ALP, and GGT CTC ⩾3 points were 1 (1%), 4 (3%), 3 (2%), 0 (0%), and 19 (17%) patients, respectively. TBil, ALT, AST, ALP, and GGT were elevated ⩾2 points in 2 (1%), 11 (8%), 6 (4%), 2 (2%), and 32 (29%) patients, respectively. Considering that GGT was significantly higher than other liver enzymes after treatment, we further performed a subgroup analysis of the correlation between GGT and voriconazole, and the results showed that steady-state voriconazole Ctrough (p = 0.002) and voriconazole treatment duration (p = 0.026) were independent positive predictors of ΔCTC (GGT) ⩾2 (Table 6).
Logistic regression analysis of the increase of GGT after voriconazole treatment.

△CTC, CTCAE grade difference of liver enzyme before and after treatment with voriconazole; CI, confidence interval; GGT, gamma-glutamyl transpeptidase; OR, odd ratio.
Discussion
There are no guideline recommendations for dose adjustment based on TDM results. Park et al. 6 used a threshold of 1.0, 5.5, and 10.0 mg/l for adjusting dose based on the exposure data and the severity of the AEs. By following this strategy, the percentage of patients achieving their target therapeutic range increased from 49% to 77%. In this study, the majority of patients (84.1%, 37/44) only required one-time dose adjustment to reach the target concentrations (1.5–5.5 mg/l) with this regimen.
The Dutch Pharmacogenetics Working Group has provided more specific dose adjustment options for voriconazole based on pharmacogenomic treatment recommendations. Working group members suggested determining the initial dose of voriconazole in patients according to their CYP2C19 genotypes. For slow metabolizers, the dose should be reduced by 50% and for fast metabolizers, increased by 50%. Similarly, Zubiaur et al. 36 used a physiology-based pharmacokinetic model to analyze the dose adjustment of voriconazole in a study and suggested that the standard dosing regimen in the current guidelines may only be applicable to the normometabolic phenotype.
Studies have shown that CYP2C19 nonwild (mutant) phenotypes are prevalent in Asian populations (60–70%), whereas this proportion is only about 30% in Caucasian and African. 24 In this study, the intermediate metabolizers were also predominant in patients with CYP2C19 genotype, and the mean steady-state voriconazole Ctrough was higher than those of the extensive metabolizers. This may indicate that the dosing regimen of voriconazole needs to be adjusted for Chinese patients.
Of the 140 patients in this study, 44 (31.4%) experienced a dose adjustment during voriconazole treatment. The simultaneous elevation of Ctrough and liver enzymes (42.9%) was the most common reason for the first voriconazole dose adjustment. Among them, 94% of patients had their voriconazole dose adjusted within 14 days, with the greatest proportion occurring between 4 and 7 days (45.5%, 20/44), which is likely due to routine monitoring after voriconazole administration. In addition, we observed that the dose was not adjusted in 10 patients with elevated Ctrough and AEs, 13 patients with normal Ctrough, and 21 patients with elevated Ctrough but no AEs. Thus, in clinical practice, therapeutic decisions are influenced by expected or observed toxicity as well as disease severity and host factors. 37
In a meta-analysis of 39 randomized controlled trials by Wang et al., 9 the risk of discontinuing voriconazole due to elevated liver enzymes was high. There is still some controversy whether there is a correlation between hepatotoxicity and voriconazole Ctrough, however. 8 Some studies have reported that steady-state voriconazole Ctrough above 4.0–6.0 mg/l are associated with an increased incidence of hepatotoxicity.12,16,18,24,26–28,38 Meanwhile, several other studies have also shown no correlation between steady-state voriconazole Ctrough and hepatotoxicity.6,19,29,39
A wide variation of hepatotoxicity is observed in different studies, which is likely due to the different study population,8,10,25 CYP2C19 genotype, 15 disease severity,14,18 and dose and method of administration.17,40 The heterogeneity among studies may make it difficult to generalize the results based on population and definition criteria to all populations. A meta-analysis by Jin et al., 10 which included 21 studies, showed a large difference in the incidence of voriconazole-associated liver injury between Asian and non-Asian studies. A reasonable downward adjustment of the upper target voriconazole concentration threshold recommended by current guidelines may be warranted in the Asian population.
In this study, compared with the steady-state voriconazole Ctrough in 52 patients without any AEs during treatment, there was no statistically significant difference in other AEs such as visual disturbances, except for the hepatotoxicity group and neurotoxicity group, which is consistent with some other studies.10,19 Using subject curve analysis, we determined that a steady-state Ctrough of 3.61 mg/l could predict hepatotoxicity with 94.4% sensitivity and 41.8% specificity. In other words, if the steady-state Ctrough was considered the only predictor of hepatotoxicity, 94.4% of patients would be identified, but 58.2% of the population would be overestimated. This led us to propose a more plausible hypothesis that increased voriconazole Ctrough may lead to an increased likelihood of toxic events. But, there is no perfect positive linear correlation with toxic events (especially hepatotoxicity) and that steady-state Ctrough at a single time point may not ideally predict the risk of hepatotoxic events.
Furthermore, logistic regression analysis was used to analyze the factors influencing voriconazole-associated hepatotoxicity, and the results indicated that timely voriconazole dose adjustment was an effective protection against hepatotoxicity. In other words, clinicians would actively reduce the dose or discontinue treatment based on the assessment of liver function to prevent hepatotoxicity.
Extracellular GGT acts as a membrane-bound zinc protein with a main function in glutathione recycling. Several studies have shown that GGT overexpression is associated with melanoma,41–43 and the association of long-term voriconazole use with photosensitivity has been confirmed by several studies.44–46
We compared the CTC scores of patients before and after treatment, and the percentage of GGT in △CTC ⩾2 was significantly higher than that of the other enzymes and correlated with steady-state voriconazole Ctrough and length of voriconazole treatment during hospitalization. This is consistent with the observation that a certain proportion of patients requiring long-term oral voriconazole treatment showed isolated mild-to-moderate elevations in GGT, which may suggest the presence of high oxidative stress in this group of patients. Currently, only sporadic cases of the association between long-term voriconazole use and melanocytoma development exist, but due to the poor prognosis of melanoma complications, it is worthwhile to be alert to this indicator in clinical practice.
This study had some limitations. First, the CYP2C19 genotype polymorphism, which has been shown in several studies to affect voriconazole blood levels, was not identified in this study, which may be due to the small sample size tested for the genotype in this study. Second, this is a study of a specific Asian population and the findings may be only applicable to Asia populations.
In conclusion, this study found that around one-third of Chinese patients with pulmonary fungal disease required dose adjustment after regular doses of voriconazole. Hepatotoxicity was the most common cause of dose adjustment. In patients with early occurrence of hepatotoxicity, initial TDM might predict the risk of hepatotoxicity. Follow-up TDM is also required to predict late onset hepatotoxicity, however. GGT may be used to reflect the level of oxidative stress in patients on long-term voriconazole.
