Abstract
Background:
The use of anti-cytotoxic T-lymphocyte antigen 4 (anti-CTLA4) therapy (ipilimumab) and anti-programmed cell-death 1 (anti-PD1) agents (nivolumab and pembrolizumab) in advanced melanoma have been associated with immune-related adverse events (irAEs) including colitis. We aimed to estimate the incidence and the risk of colitis in elderly patients with advanced melanoma treated with anti-CTLA4 and anti-PD1 in the real-world setting.
Methods:
Elderly patients (age ⩾ 65 years) diagnosed with advanced melanoma between 2011 and 2015 and treated with anti-CTLA4 or anti-PD1 agents were identified from the Surveillance, Epidemiology, and End Results (SEER)–Medicare data. We estimated the risk of colitis from start of treatment up to 90 days from the last dose of therapy. We used the log-rank test and logistic regression with adjustment for potential confounders using the inverse probability of treatment weighting method. We conducted several sensitivity analyses.
Results:
A total of 274 elderly patients with advanced melanoma were included in our cohort. The risk of colitis was similar between anti-PD1 users and anti-CTLA4 users based on log-rank test (p = 0.17) and logistic regression [odds ratio (OR) = 0.35, 95% confidence interval (95%CI) 0.04–2.79]. Sensitivity analyses for patients with all-stage melanoma showed a significantly lower risk of colitis in anti-PD1 compared with anti-CTLA4 treated patients based on log-rank test (p = 0.017) and logistic regression (OR = 0.21, 95%CI 0.09–0.53).
Conclusion:
Elderly with advanced melanoma treated with anti-CTLA4 or anti-PD1 had a similar risk of developing colitis. However, there was a statistically significant difference in the risk of colitis between anti-CTLA4 or anti-PD1 users among all-stage-melanoma patients.
Plain Language Summary
While the anti-cancer agents known as immune-checkpoint inhibitors have had a great impact on the treatment of melanoma, they may also have side effects. This study estimated the risk of colitis, a chronic inflammation of the colon, in elderly patients with melanoma treated with anti-cytotoxic T-lymphocyte antigen 4 (anti-CTLA4) or anti-programmed cell-death 1 (anti-PD1) agents, using data from the Surveillance, Epidemiology, and End Results (SEER)–Medicare linked database. Overall, we found that the risk of colitis was not different between anti-PD1 users and anti-CTLA4 users with advanced-stage melanoma. However, after including patients across all stages of melanoma, we found a significantly lower risk of colitis with anti-PD1 compared with anti-CTLA4.
Keywords
Introduction
In the United States, melanoma is the fifth most common cancer in both men and women, with an estimated 100,350 new cases and 6850 deaths in 2020. 1 Since the approval of ipilimumab, the first immune-checkpoint inhibitor (ICI), in 2011, and the subsequent approval of nivolumab and pembrolizumab in 2014, treatment options for melanoma have expanded, and treatment outcomes have improved significantly. 2 ICIs work by binding to receptors on the T cell, leading to activation of the immune system against the tumor cells. 3
Two types of ICIs have been approved by the US Food and Drug Administration (FDA). The first type targets the inhibitory path of cytotoxic T-lymphocyte antigen 4 (CTLA4); among these, ipilimumab is the only anti-CTLA4 agent approved for treating advanced melanoma. The second type targets the programmed cell-death 1 (PD1) inhibitory pathway; among these, nivolumab and pembrolizumab are approved for the treatment of advanced melanoma.2,3 ICIs have been shown to prolong survival and increasing the objective response rate in comparison with traditional chemotherapy.2,3 However, the dysregulation of the immune system by ICIs could lead to loss of tolerance and autoimmune reactions involving different organs in the body such as the skin, gastrointestinal tract, endocrine, respiratory, nervous, and cardiovascular systems. Those undesired reactions are known as immune-related adverse events (irAEs). 4
Gastrointestinal irAEs are common among ICI users and may range from mild diarrhea to severe colitis.4,5 In melanoma trials, rates of colitis of 9.1% for anti-CTLA4 users and 2.1% for anti-PD1 users (1.45% for nivolumab, and 2.1% for pembrolizumab) have been reported.6,7 However, evidence on the incidence in real-world settings is limited to case or single-center reports.8–14 A recent retrospective study based on claims from US electronic medical record data included a melanoma cohort (age ⩾18 years) of 607 patients treated with ipilimumab, 14 with nivolumab, and 157 with combination therapy of ipilimumab and nivolumab. The risk of enterocolitis or diarrhea was not statistically different between ipilimumab- and nivolumab-treated patients [hazard ratio (HR) 1.07; 95% confidence interval (95%CI) 0.68−1.70]. 15
The risk of colitis among elderly melanoma patients treated with anti-CTLA4 or anti-PD1 in real-world settings has not yet been estimated. The aim of this study was to estimate the risk of colitis in anti-CTLA4- and anti-PD1-treated elderly patients with advanced melanoma in a retrospective cohort from the SEER–Medicare linked data.
Method
Study design
We conducted a retrospective cohort study of melanoma patients who were de novo users of anti-CTLA4 or anti-PD1 to estimate the risk of colitis. The University of Arizona institutional review board exempted this study because the data in the SEER–Medicare database are de-identified data.
Data source
Our study is based on a database that links cancer cases reported to the SEER program of cancer registries with corresponding data from Medicare claims. The SEER–Medicare linked data cover around 93% of SEER’s patients aged 65 years or older who were Medicare beneficiaries. 16 The SEER program is a national cancer registry that is used by the National Cancer Institute (NCI) for surveilling cancer in the United States. SEER collects information about the demographic characteristics of patients diagnosed with cancer and clinical characteristics of cancer, such as histological features, organs affected, number and type of cancers, cancer stage, tumor grade, and date of diagnosis. Also, the SEER program provides information about the first course of cancer treatment and vital data such as date of death, survival time, and cause of death, if available. The NCI implemented rigorous quality control measures, including routinely audited for data accuracy to ensure the integrity of the database. 17 Medicare is a federal program that provides health insurance for (mainly) the elderly. Around 97% of the elderly population (aged 65 years or older) are covered by the Medicare program. The coverage may involve health services provided in inpatient and outpatient settings, as well as medication coverage such as therapeutic medications for cancers. 16
Study cohort
We identified patients aged 65 years or older with a primary diagnosis of melanoma (stage 3 and 4) between 2011 and 2015 from SEER–Medicare linked data. We restricted our cohort to patients whose eligibility for Medicare was due to aging only and had continuous enrollment in Medicare part A and part B for at least 6 months prior to the diagnosis of melanoma. We excluded patients with end-stage renal disease or acquired immunodeficiency syndrome. We further restricted our sample to patients whose prescribed ICIs and diagnosis date, as specified in the Medicare claims database [International Classification of Diseases, 9th/10th Revision Clinical Modification (ICD-9-CM, ICD-10-CM)] matched the diagnosis month and year in the SEER database. Also, we excluded patients who used a combination therapy of anti-CTLA4 and anti-PD1, and those who experienced colitis prior to starting treatment with anti-CTLA4 or anti-PD1 therapies.
Exposure, outcome, and covariates
We identified patients newly exposed to anti-CTLA4 or anti-PD1 therapies from 2011 to 2016 using the Healthcare Common Procedure Coding System (HCPS) codes (Table S1). The index date was the date of the first exposure to ICIs. The outcome of interest was the development of colitis following the use of ICIs and was identified from one inpatient claim or two outpatient claims that were 30 days apart using the ICD-9-CM and ICD-10-CM codes (Table S1). Patients were followed from the start of their ICI therapy until the earliest occurrence of outcome (colitis), death, switching between ICI therapies, loss of Medicare part A and B coverage, and/or end of the available follow-up data by December 2016. The primary outcome was the risk of colitis in anti-CTLA4 and anti-PD1 analyzed at the drug class level, as well as at the specific medication level (ipilimumab, nivolumab, and pembrolizumab) during the period of initial treatment and up to 90 days from the last dose used. The baseline covariates included age at diagnosis of melanoma, sex, race, ethnicity, marital status, geographic region, urban residency, receiving public assistance to Medicare enrollee by the state Medicaid agency, melanoma stage based on the American Joint Committee on Cancer (AJCC) 7th edition, tumor grade, histopathological type, use of chemotherapy, immunotherapy, surgery, and radiation, as well as ecological surrogate for the patients’ level of education, median income, and poverty level, based on the county level obtained from the census data.
Propensity model
We used a propensity score weighting method using the inverse probability of treatment weighting (IPTW) to account for potential bias arising from observational studies due to the absence of randomized allocation. The propensity score is the conditional probability that a patient receives a treatment based on a certain list of covariates. The IPTW is a weighting method that assigns a weight of 1/(propensity score) to the treatment arm and 1/(1 − propensity score) for the control arm. The application of IPTW yields in a weighted ‘pseudo population’ in which treatment and the covariates are independent. 18 The variables included in the propensity weighting model were age at diagnosis of melanoma, sex, race, ethnicity, marital status, geographic region, urban residency, receiving public assistance for Medicare enrollee by the state Medicaid agency, melanoma stage, tumor grade, histopathological type, prior use of chemotherapy, immunotherapy, surgery, radiation, percentage of patients with high-school education, natural logarithm of median income percentage of patients below poverty level, and the baseline Charlson comorbidity index excluding cancer diagnoses. 19 We utilized generalized boosted models to estimate the propensity score and IPTW.20,21 We used the standardized mean difference (SMD) <0.2 as indicative of a good balance. 22
Statistical analysis
For our primary analysis, we intended using Cox proportional regression modeling to estimate the risk of colitis between anti-CTLA4 and anti-PD1 therapies. However, the Cox proportionality assumption for the interaction between time and treatment was violated (pinteraction < 0.05). Therefore, we utilized a non-parametric approach using the log-rank test by incorporating the IPTW and adjusted Kaplan–Meier estimate to compare the difference in time with colitis between the ICI therapies. 23 Also, we utilized logistic regression to calculate the odds ratio (OR) and 95%CI for the risk of colitis between the ICIs. We further performed sensitivity analyses to assess the robustness of the finding. First, we evaluated the risk of colitis from start of ICI treatment up to 120 days and up to 360 days from the last dose. Second, we evaluated the risk of colitis during the first 6 months and 12 months of ICI treatment. We conducted additional sensitivity analyses by including patients with all-stage melanoma to evaluate the robustness of the finding across all-stage melanoma. In both the primary and sensitivity analyses, the cumulative incidence of colitis during the first 6 months and 12 months of initiation ICI therapy was estimated. We performed all analyses using SAS, version 9.4 (SAS Institute, Cary, NC, USA) with a two-sided significance level of p < 0.05.
Results
Cohort characteristics
A total of 274 melanoma patients met the inclusion and exclusion criteria, including 205 patients treated with anti-CTLA4, and 69 treated with anti-PD1 agents (Figure 1, Table 1). The descriptive statistics of anti-CTLA4 or anti-PD1 de novo users are presented in Table 1. Among the 69 anti-PD1 users, 19 patients were treated with nivolumab, and 50 with pembrolizumab. The mean age for the anti-CTLA4 users was 74.10 years compared with 75.72 years in the anti-PD1 users. The majority of patients were White. Among the anti-CTLA4 treated patients, 7.3% had previously been treated with chemotherapy. The SMD after the IPTW was <0.2 for all variables indicating a good balance between the two groups (Table 1).

Cohort development from SEER–Medicare data.
Baseline characteristics of included patients with standardized mean difference (SMD) before (unweighted) and after (weighted) using the inverse probability of treatment weighting for advanced melanoma patients.
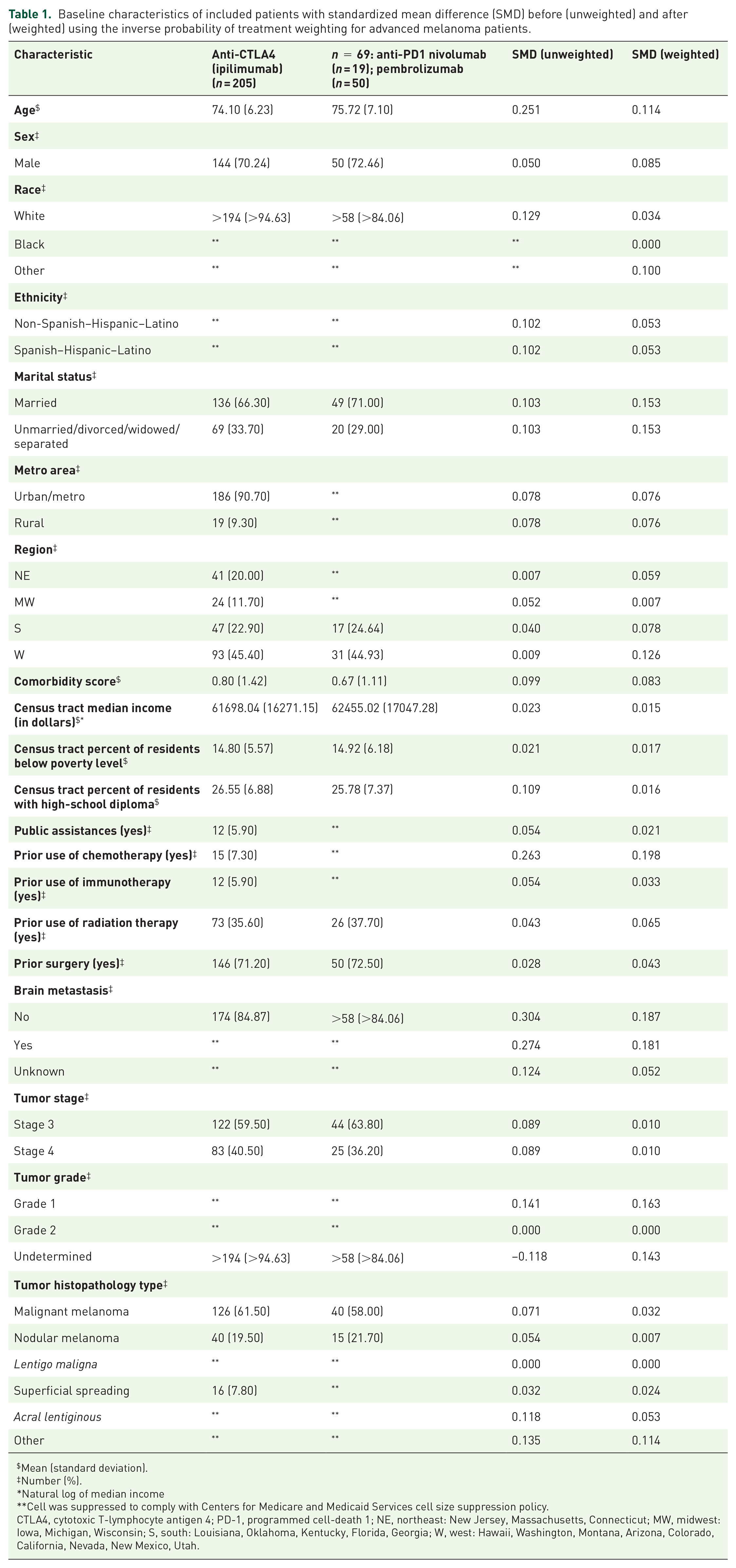
Mean (standard deviation).
Number (%).
Natural log of median income
Cell was suppressed to comply with Centers for Medicare and Medicaid Services cell size suppression policy.
CTLA4, cytotoxic T-lymphocyte antigen 4; PD-1, programmed cell-death 1; NE, northeast: New Jersey, Massachusetts, Connecticut; MW, midwest: Iowa, Michigan, Wisconsin; S, south: Louisiana, Oklahoma, Kentucky, Florida, Georgia; W, west: Hawaii, Washington, Montana, Arizona, Colorado, California, Nevada, New Mexico, Utah.
Risk of colitis
Per the log-rank test, the risk of colitis was similar in the anti-PD1 and anti-CTLA4 groups (p = 0.17; Figure 2), confirmed in the logistic regression model (OR 0.35, 95%CI 0.04–2.79). The 6- and 12-month cumulative incidence rates of colitis in the anti-CTLA4 group were 6.77% and 9.27%, respectively. In the anti-PD1 group, the corresponding values were 2.88% and 4.64%, respectively.
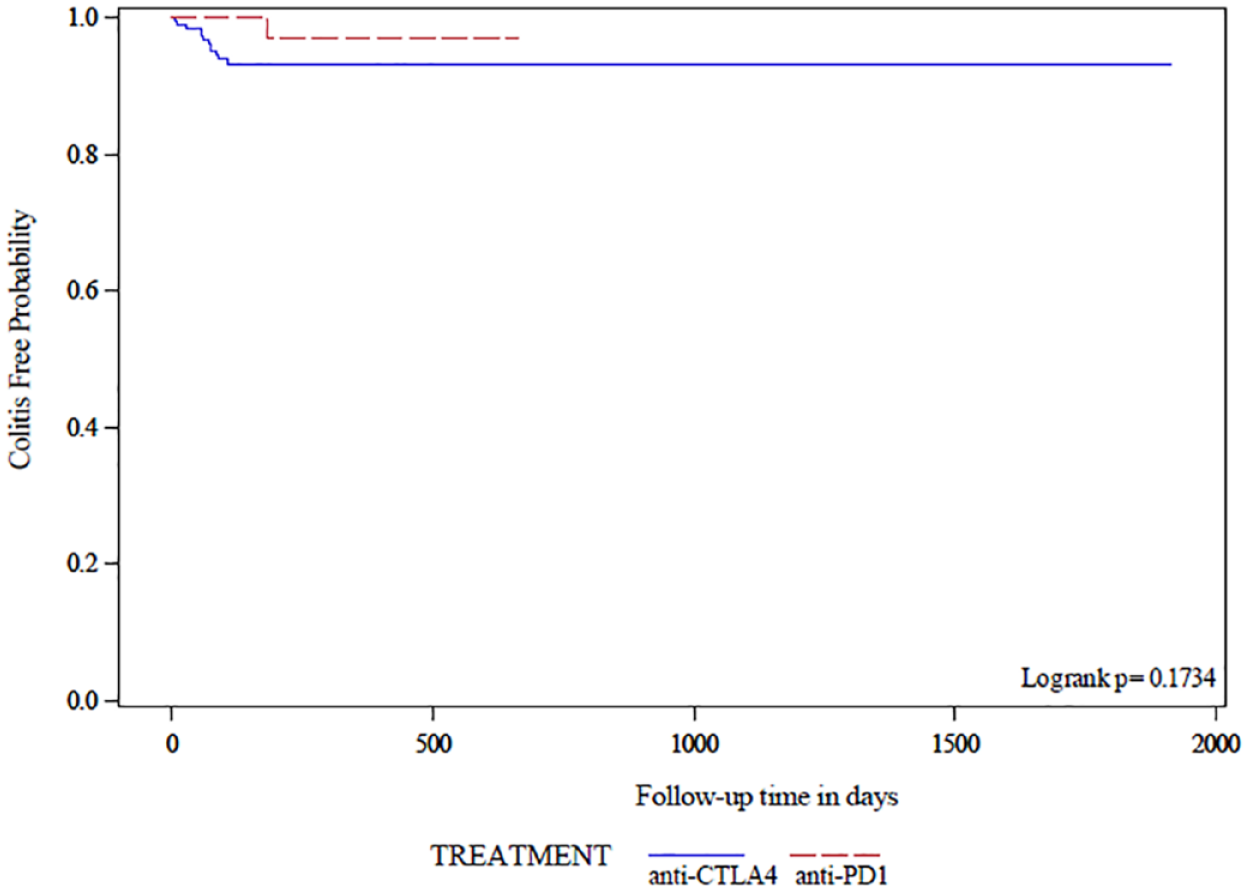
Risk of colitis between anti-cytotoxic T-lymphocyte antigen 4 (anti-CTLA4) and anti-programmed cell-death 1 (anti-PD1) from start of treatment up to 90 days of the last dose of treatment.
Cohort characteristics (ipilimumab versus nivolumab versus pembrolizumab)
Comparisons by type of medication (ipilimumab, nivolumab, and pembrolizumab) and including the SMD prior to and after the IPTW are presented in Tables 2 and 3. Prior use of immunotherapy was statistically different in the patients treated with either of the three agents (p = 0.02; Table 2). The average SMD after IPTW indicated acceptable balance though with median income, percentage of county residents with high-school diploma, prior use of chemotherapy or immunotherapy, and tumor grade showing SMD >0.2 (Table 3).
Baseline characteristics of included patients with advanced melanoma.

NE, northeast: New Jersey, Massachusetts, Connecticut; MW, midwest: Iowa, Michigan, Wisconsin; S, south: Louisiana, Oklahoma, Kentucky, Florida, Georgia; W, west: Hawaii, Washington, Montana, Arizona, Colorado, California, Nevada, New Mexico, Utah.
Mean (standard deviation).
Number (%).
Natural log of median income
Cell was suppressed to comply with Centers for Medicare and Medicaid Services cell size suppression policy.
Standardized mean difference (SMD) in pre-inverse probability of treatment weighting (IPTW) (unweighted) and post-IPTW (weighted) for patients with advanced melanoma.

NE, northeast: New Jersey, Massachusetts, Connecticut; MW, midwest: Iowa, Michigan, Wisconsin; S, south: Louisiana, Oklahoma, Kentucky, Florida, Georgia; W, west: Hawaii, Washington, Montana, Arizona, Colorado, California, Nevada, New Mexico, Utah.
Mean (standard deviation).
Number (%).
Natural log of median income
Risk of colitis
The overall log-rank test indicated no significant difference between the agents of interest (p = 0.40; Figure 3). Results from logistic regression showed a similar risk of colitis for nivolumab versus ipilimumab (OR 0.13, 95%CI 0.01–2.04), pembrolizumab versus ipilimumab (OR 0.50, 95%CI 0.19–1.32), and pembrolizumab versus nivolumab (OR 3.90, 95%CI 0.23–66.88). The 6-month cumulative incidence of colitis was 6.76% in ipilimumab, 0% in nivolumab, and 3.76% in pembrolizumab while the 12-month cumulative colitis incidence was 9.48% in ipilimumab, 0% in nivolumab, and 8.36% in pembrolizumab.

Risk of colitis between ipilimumab, nivolumab, and pembrolizumab from start of treatment up to 90 days of the last dose of treatment.
Sensitivity analyses
Sensitivity analyses for comparing anti-PD1 versus anti-CTLA4 treated patients using different follow-up times did not show a difference in colitis risk. Similarly, comparing colitis risk between ipilimumab, nivolumab, and pembrolizumab over the first 6 or 12 months from start of treatment or up to 120 or 360 days from the last dose of treatment showed no differences in colitis risk between these agents (Table S2).
The characteristics of 324 anti-CLA4 and 145 anti-PD1 (36 nivolumab,109 pembrolizumab) patients at all stages of melanoma stratified by anti-CTLA4 versus anti-PD1 or stratified by agents (ipilimumab, nivolumab, and pembrolizumab) are presented in Tables S3 and S4, respectively. The SMD after IPTW was <0.2 for all variables indicating a good balance between the two groups (Table S3). The average SMD after IPTW at the medications level (ipilimumab, nivolumab, and pembrolizumab) showed acceptable balance in the covariates with some variables showing SMD exceeding 0.2 (age at diagnosis, percentage of county residents with high-school diploma, prior use of chemotherapy or immunotherapy, and tumor stage; Table S5). Per the log-rank test, the risk of colitis was statistically lower in the anti-PD1 versus anti-CTLA4 group across different follow-up periods (p < 0.05; Table S6). This was confirmed in logistic regression analyses (Table S6). The cumulative incidence of colitis in the first 6 months and 12 months among anti-CTLA4 was 8.19% and 9.49%, respectively, while among anti-PD1 was 2.48% and 3.67%, respectively.
Comparing the individual medications with each other using the overall log-rank test indicated a significant difference in colitis risk following the initiation of the treatment and up to 90 days of last dose (p = 0.03) and during the first 6 months of treatment with ICIs (p = 0.03). Post hoc analyses showed a lower colitis risk among pembrolizumab- versus ipilimumab-treated patients for follow-up periods up to 90 or 360 days from last dose or for the first 6 months and 12 months of treatment (p < 0.05; Table S6). However, logistic regression analyses revealed a statistically lower odds of colitis risk among nivolumab or pembrolizumab users versus ipilimumab users across the various follow-up periods (Table S6). The risk of colitis was similar between nivolumab- and pembrolizumab-treated patients (Table S6).
Discussion
In this retrospective study from a nationally representative SEER–Medicare dataset of elderly patients with advanced melanoma treated with anti-CTLA4 or anti-PD1, we found a comparable incidence of colitis between both cohorts at both the class and the individual agent levels. However, colitis risk was significantly lower among all-stage melanoma patients treated with anti-PD1 versus those treated with anti-CTLA4. In the all-stage melanoma cohort, nivolumab- and pembrolizumab-treated patients were more likely to have lower odds of colitis compared with those treated with ipilimumab as presented in the logistic regression analyses.
The incidence of colitis among ICI users in clinical trials was estimated in several meta-analyses.6,7,24 In a recent review of ICI-induced colitis, the incidence across different types of cancers in anti-CTLA4 and anti-PD1 users was reported to be 7–11.6% and 1.3–2.9%, respectively. 24 A meta-analysis of 34 trials of ICIs in patients with melanoma, non-small-cell lung cancer, or renal-cell carcinoma showed an overall colitis incidence of 9.1% in anti-CTLA4-treated patients compared with 1.4% in those treated with anti-PD1 agents. When stratifying by tumor type, melanoma patients treated with anti-CTLA4 therapy had an incidence of colitis of 9.1% versus 1.8% among patients treated with anti-PD1 agents. 6 Another meta-analysis of 46 anti-PD1 studies across different cancer types reported the overall incidence of colitis to be 2.24% (2.5% in nivolumab users, 2.02% in pembrolizumab). However, in the melanoma patients treated with anti-PD1, the rate of colitis was 2.06% (1.45% in nivolumab, and 2.11% in pembrolizumab). 7 In our analysis, the cumulative incidence of colitis was 6.77–9.27%, in advanced melanoma patients treated with anti-CTLA4 agent, which is comparable with the incidence reported in prior meta-analyses.6,24 In contrast, the cumulative incidence of colitis in our anti-PD1 cohort was 2.88–4.64% in patients with advanced melanoma, which is slightly higher than the incidence rate of colitis (2.06%) in a prior meta-analysis. 7 This may be attributed to different dosing of anti-PD1 in the clinical trials, as well as our relatively small sample size of anti-PD1 users considering the relative recency of these agents’ regulatory approval, and therefore, their inclusion in the SEER–Medicare dataset.6,7
Nivolumab users in our study did not experience any colitis, a result also observed in two prior trials that included small samples size similar to our nivolumab sample size.25,26 The incidence of colitis associated with a nivolumab 3 mg/kg every 2 weeks regimen from a pooled analysis of clinical trials involving 576 patients was around 1%. 27
The incidence of colitis in the real-world setting is limited to case reports, single-center experiences, or surveillance studies.8,28–33 In a review of 234 published case reports of irAEs in patients treated with ipilimumab, 80 case reports were about colitis. 28 However, another review of 139 case reports of irAEs following the use of anti-PD1 revealed that 14 reports were about colitis. 8 The incidence of colitis in melanoma patients in single-center reports ranged from 6.5 to 22.3% in ipilimumab, 2.7–6.6% in pembrolizumab, and 1–2.4% in nivolumab.29–33 This variation in the incidence of colitis in the real-world setting may be due to the differences in sample size, follow-up time, definition of colitis, and type of cancer.
Our primary analysis focusing on elderly patients with advanced melanoma did not show a significant difference in the risk of colitis between those treated with anti-CTLA4 and anti-PD1agents. However, when considering the data for all stages of melanoma, a lower risk of colitis was identified in anti-PD1 versus anti-CTLA4 treated patients. This is consistent with the results from a meta-analysis of two studies in advanced melanoma patients (age ⩾18 years) that found a relative risk of 0.2 (95%CI 0.07–0.62) 34 and a meta-analysis involving three studies that yielded a relative risk of 0.16 (95%CI 0.05–0.51). 35 A real-world claims-based study using the IBM Explorys Universe Dataset found that the incidence of enterocolitis in adult (age ⩾18 years) melanoma patients treated with ipilimumab was 13.2% versus 5.7% in those treated with nivolumab. The risk of diarrhea or enterocolitis was not significantly different between nivolumab- and ipilimumab-treated patients (HR 1.07, 95%CI 0.68−1.70). No separate results for enterocolitis risk were reported. 15
Our finding of no difference in colitis risk among elderly patients with advanced melanoma treated with anti-CTLA4 versus those treated with anti-PD1 in the real-world setting may be a function of several factors. Most clinical trials involve patients aged 18 years and older, but elderly populations tend to be under-represented. 36 One reason for their under-represention may be the general impact of usual aging on physiology and organ function. Another, more specific, factor is immunosenescence: the attenuation and alteration of the immune system due to aging. 37 Further, the elderly are more likely to develop sarcopenia, the loss of skeletal muscle mass and strength due to aging, which is believed to be associated with changes in the immune system. 38 Despite these aging-related immunological changes, the irAE profile of ICIs in the elderly is generally assumed to be comparable with that in younger populations.39–41 However, an observational study from a multi-center French registry of 615 patients treated with anti-PD1 agents for different types of cancer found a higher rate of irAEs in patients age 70 and older versus those younger than 70 years (31% versus 25%, p = 0.035). 42 A retrospective study involving 75 patients with advanced non-small-cell lung cancer treated with ICIs revealed a higher rate of colitis in patients 80 years or older compared with patients below the age of 80 (24% versus 5%, p = 0.04). 43 A retrospective study of 48 melanoma patients treated with ipilimumab found a higher colitis risk in patients with sarcopenia versus those without sarcopenia (16.7% versus 2.1%, p = 0.039). 44 Considering the context of changes in the immune system in the elderly and the irAE profile of ICIs, clinicians may need to personalize ICI treatment in elderly melanoma patients.
Our study has several strengths. We used a cohort of elderly patients with melanoma that represented real-world practice during the period of 2011–2016. We utilized the IPTW method to adjust and balance potential confounders and gain a better estimate of the risk of colitis between anti-CTLA4 and anti-PD1 users. Our analyses provide clinicians with real-world evidence about colitis risk in advanced melanoma patients age 65 and older treated with anti-CTLA4 or anti-PD1 therapies. Yet, our findings should also be addressed within the inherent limitations of observational retrospective claims analyses. The SEER–Medicare dataset does not include such potentially informative covariates as severity grading for colitis, obesity, and smoking status that could be added to the adjustments in the IPTW method. Relying on diagnosis codes such as ICD9-CM/ICD10-CM may lead to misclassification error, and therefore to an underestimation of the incidence of colitis. Similarly, by using the HCPS codes to identify ICIs, some ICI users may not have been included, though this may have been corrected by the high sensitivity and specificity of Medicare data in defining cancer treatments. 45 The SEER database only collects the tumor stage at diagnosis and does not capture changes in staging over time. Being focused on elderly patients and given the relative recency of ICI therapy, the nivolumab cohort was limited by small sample size, and this may have affected the precision of the colitis risk in nivolumab-treated patients, and the results should be interpreted with caution. Larger studies are needed to detect any significant differences between individual ICIs.
Conclusion
The incidence and risk of colitis was comparable among elderly patients with advanced melanoma treated with anti-CTLA4 versus those treated with anti-PD1. However, the use of anti-PD1 therapy is more likely to be associated with a lower risk of colitis compared with anti-CTLA4 when considering elderly melanoma patients at all stages of disease. Furthermore, treating elderly melanoma with ICIs warrants personalized care by clinicians to optimize the beneficial effects of ICIs and minimize potential irAEs such as colitis.
Supplemental Material
sj-docx-1-taw-10.1177_2042098621991279 – Supplemental material for Association of immune-checkpoint inhibitors and the risk of immune-related colitis among elderly patients with advanced melanoma: real-world evidence from the SEER–Medicare database
Supplemental material, sj-docx-1-taw-10.1177_2042098621991279 for Association of immune-checkpoint inhibitors and the risk of immune-related colitis among elderly patients with advanced melanoma: real-world evidence from the SEER–Medicare database by Abdulaali R. Almutairi, Marion Slack, Brian L. Erstad, Ali McBride and Ivo Abraham in Therapeutic Advances in Drug Safety
Footnotes
Conflict of interest statement
The authors declare that there is no conflict of interest.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
