Abstract
Background:
Retrospective analyses of large databases of treated patients can provide useful links to the presence of drug misuse or rare and infrequent adverse effects, such as agranulocytosis, diabetic ketoacidosis or neuroleptic malignant syndrome. The aim of this study is to describe the adverse effects to antipsychotics reported in the Australian Database of Adverse Event Notifications (DAEN).
Methods:
Data were collected from the DAEN – a spontaneous reporting database. The database, which covered the period from January 2004 to December 2017, was obtained from the Therapeutic Goods Administration (TGA) website (www.TGA.gov). The drugs selected for this investigation are the following: aripiprazole, clozapine, olanzapine, paliperidone, risperidone, ziprasidone, quetiapine, haloperidol and pimozide. All data were analysed descriptively. Comparison of reporting and management of adverse events between adults (older than 20 years) and children (5–19 years) was undertaken using chi squared test, where p < 0.05 is significant.
Results:
A total of 7122 adverse events associated with the antipsychotics aripiprazole, clozapine, haloperidol, olanzapine, paliperidone, pimozide, quetiapine and risperidone were reported to the TGA between January 2004 and December 2017. On average, there were 2.6 adverse events reported for each case. The most common adverse event reported for antipsychotics was neuroleptic malignant syndrome. There were no significant differences in the number of co-medications, formulations, indications, therapeutic dose, hospital admission and overdose among the antipsychotics between paediatric and adult populations. However, there were significant differences between causality, death and the management of adverse events between adult and paediatric populations (5–19 years) (p < 0.05, chi squared test).
Conclusion:
The antipsychotic drug associated with the highest adverse events in adults was clozapine, followed by olanzapine. The most common adverse event in adults, and reported with a number of antipsychotic drugs, was neuroleptic malignant syndrome. In children, the highest numbers of adverse events reported in the database were associated with risperidone, clozapine and olanzapine.
Plain language summary
Adverse events reported of antipsychotics
Background
There is growing evidence that antipsychotics are being misused for their anxiolytic, hedonic and sedative effects.1–4 Atypical antipsychotics are now being used to enhance the effects of other drugs, or as a way to counter the adverse effects of illicit substances.5–8 Moreover, antipsychotics have been associated with deliberate self-poisoning and are increasingly responsible for emergency department presentations and mortality.6–8 Malekshashi et al. found that, among 429 addiction treatment inpatients screened, 73 (17.0%) reported misuse of antipsychotics with alcohol, opioids and other illicit drugs. 9 Reasons for misuse included to “recover” from other substances (66.7%), “enhance” the effects of other substances (25.0%) and “experiment” (20.8%).
Moreover, a recent study investigated the trends of abuse of antipsychotics 10 . Klein et al. conducted a retrospective 10-year analysis of the National Poison Data System (NPDS) database and found that quetiapine abuse was more common than the abuse of other second-generation antipsychotics, comprising 60.6% of all abuse cases during the study period. This was followed by risperidone. The authors recommended that emergency physicians need to be aware of the clinical adverse events following these abuse cases.
Data extracted from spontaneous reporting systems (SRS) can provide valuable information that may not be available elsewhere and provides real-time data for use in timely analysis.11,12 Information extracted from pharmacovigilance databases such as EudraVigilance is used to identify adverse events associated with antipsychotics. 11 The United States Food and Drug Administration (FDA) Adverse Event Reporting System (FAERS) data has been used in studies to characterise these effects. 12 SRS data has been used to report on worldwide patterns of rare adverse events, associated health risks and evidence for drug withdrawals.12–14 The information gained from SRS data analysis has an important clinical practice application to assist in the appropriate use of antipsychotics.
To date, little is known about the frequency of reporting of adverse events of antipsychotics in children, adults and the elderly. A recent clinical update by Stroup and Gray 2018, detailed the management of adverse events of antipsychotic medications in the adult populations stating that adolescents are more likely to experience weight gain and sedation than adult patients, whereas the elderly are more susceptible to falls as a result of orthostatic hypotension and cognitive impairment due to anticholinergic effects. However, the authors did not report on prevalence data or frequency of these adverse events and which events get reported by prescribers and patients. 15 Another recent observational cohort study by Ray et al., showed that increased unexpected death was observed in children and youth using antipsychotics. 16 However, the results of this study were from hospitalised patients and lacked any information about the general population.
Moreover, a systematic review assessing and reporting adverse events of antipsychotic medications found that most included studies reporting on adverse events lacked a detailed description of reporting some of the adverse events. The authors suggested the use of standardised assessment instruments to be used when collecting data on adverse events and a more thorough data collection on patients’ well-being and management need to be collated in future studies. 17 This proposed study reports on adverse events using a standardised reporting form devised by the Australian Database of Adverse Event notifications.
Therefore, the aim of this study is to characterise the reporting of adverse events associated with antipsychotics in terms of causality, co-medications, formulations, off label uses and their management among adult and paediatric populations in the Australian Database of Adverse Event Notifications using the standardised assessment form supplied by the database.18,19
Methods
The study was conducted in accordance with the Declaration of Helsinki, and the protocol was approved by the Ethics Committee of Monash University (Project ID numbers-17558). Data were collected from the Database of Adverse Event Notifications (DAEN). The database, which covered the period from January 2004 to December 2017 was obtained from the Therapeutic Goods Administration (TGA) website (www.TGA.gov). The TGA structure complies with the international safety reporting guidelines, ICH E2B. The adverse events are coded according to the Medical Dictionary for Regulatory Activities (MedDRA) preferred terminology. Pharmaceutical companies are required to report adverse events and healthcare professionals and consumers are encouraged to report adverse events. The DAEN contains more than 260,000 spontaneously reported adverse events since 1971. All adverse events are indexed in DAEN using MedRDA: International Federation of Pharmaceutical Manufacturers and Associations (Geneva, Switzerland). TGA-approved indications reviewed in this study are based on the defined statements that describe the specific therapeutic use for a medicine. They describe a medicine’s claimed purpose or health benefit according to the ‘list of permitted indications’ that is contained in the Therapeutic Goods (Permissible Indications) Determination. This database was used previously by other researchers. 19 The drugs selected for this investigation are the following: aripiprazole, clozapine, olanzapine, paliperidone, risperidone, ziprasidone, quetiapine, haloperidol and pimozide. The TGA reported no data was available for ziprasidone.
Data collection
The public case report for each adverse event in which any of the above antipsychotics was considered a potential cause was obtained. Cases defined as individual public reports collected detailed information on each patient’s age, gender, dose, indication, reporter type, causality, other suspected drugs, consequences and management received. Other data such as duration of the medication was not available.
Data analysis
All data were analysed descriptively using mean and percentages. Excel (Version 14.00,2010, Microsoft, Redmond, WA, USA) and SPSS (23.0, 2015, IBM, Armonk, NY, USA) were used to analyse the data. Data were presented as frequencies and percentages where the denominator is the number of cases and the numerator was reporter type, adverse events (total number and events reported by MedDRA preferred term), causality, suspected other medications, formulation type, TGA approved indication, therapeutic dose, frequency of reporting death, suicide, hospital admission or overdose.
We selected the following for comparison between adult and paediatric (5–19 years) populations: causality, co-medications, formulations, off label uses, doses, consequences and management of adverse events using chi squared test. We did not perform any statistical analysis on the unspecified group. Statistical significance was determined as p < 0.05.
Results
The results were presented in two parts: the first part is a summary of adverse events reported for all antipsychotics and reporter type among adult and paediatric populations as reported in the database; the second part is a characterisation of the reporting of adverse events in terms of causality, co-medications, formulations, off-label uses, doses, consequences and their management among adult and paediatric populations as reported in the database.
Reported adverse events
It was noted that, between 2004 and 2017, there was an overall increase in the number of adverse events reported for all antipsychotics as shown in Figure 1. Aripiprazole showed the least increase in reported adverse drug reactions (ADRs), whereas clozapine showed the highest increase.

Number of adverse events reported over the years 2004–2017.
Adverse events reported by MedDRA term for antipsychotics
A total of 7122 adverse events associated with the antipsychotics; aripiprazole, clozapine, haloperidol, olanzapine, paliperidone, pimozide, quetiapine and risperidone were reported to the TGA between January 2004 and December 2017. These were obtained from 2699 cases analysed as each case reported multiple adverse events. On average, there were 2.6 adverse events reported for each case.
There were 359 adverse events reported that were associated with the age group 5–19 years (5.0%, n = 359), 6090 associated with the age group 20+ years (86.0%, n = 6090) and 673 where the age was not specified in the report (9.0%, n = 673).
A breakdown of all the adverse events reported for each drug is shown in Figure 2. The highest number of adverse events reported was for clozapine (42.4%), followed by olanzapine (18.9%), quetiapine (13.3%) and risperidone (13.2%). The lowest adverse event reported was for pimozide (0.1%).

Breakdown of all adverse events reported for each drug.
For adults, the highest number of adverse events reported was for clozapine followed by olanzapine. In the adult population, the most common adverse event reported for antipsychotics was neuroleptic malignant syndrome (NMS), except for clozapine where neutropenia was reported to be the highest, as shown in Table 1. Antipsychotics that were associated with NMS included aripiprazole, clozapine, haloperidol, olanzapine, pimozide and risperidone. The reported adverse event ranged from 6.9% (risperidone) to 10.6% (aripiprazole) and 10.1% (olanzapine). Clozapine-associated myocarditis was reported in 28.0% of cases, and blood creatinine phosphokinase increase in 20.0%. Other adverse events include myocarditis, hyperprolactinaemia, diabetes, galactorrhea, tachycardia and extrapyramidal disorder.
Adverse reactions reported by MedDRA preferred term for antipsychotics (adults and paediatric populations). Age group: Adult and Elderly (20+ years) in bold. Age group: Child and Adolescent (5–19 years) in italic. .
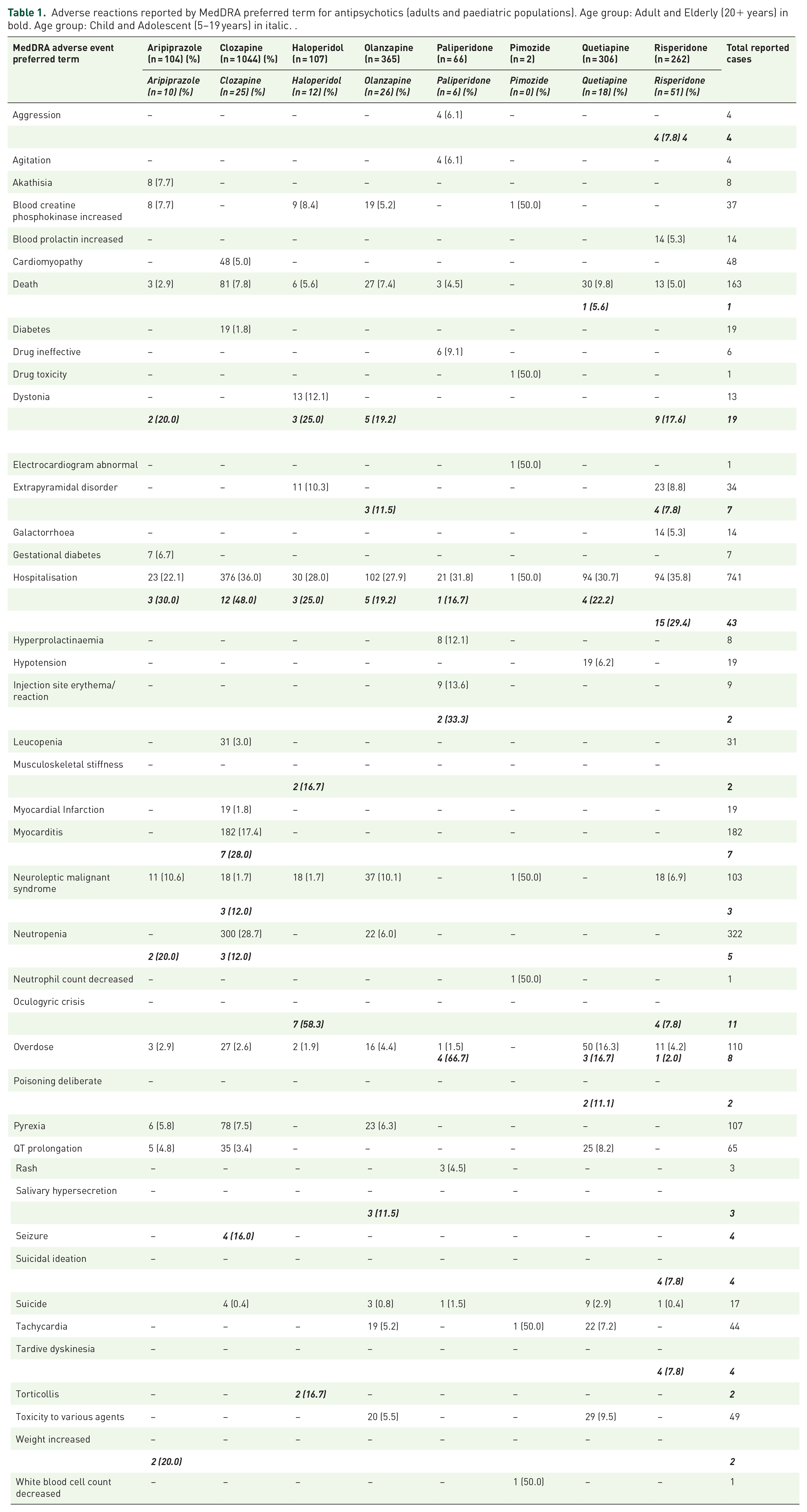
In the children’s age group (5–19 years), the highest number of adverse events was for risperidone, olanzapine and clozapine. For children, the most common adverse event reported was dystonia, which ranged from 17.6% (risperidone) to 25.0% (haloperidol). Other common adverse events included oculogyric crisis, neutropenia, weight gain and extrapyramidal disorder, as shown in Table 1.
The results for pimozide are not reliable due to the very low number of cases reported. Only three cases were reported for pimozide. Two of these cases were patients included in the adult population, and in the third case, the age was not specified.
Reporter type
Pharmaceutical companies, pharmacists, hospitals, general practitioners, specialists, nurses, members of the public, information provided from coroner reports, State and Territory Health departments and complementary health professionals reported adverse events associated with the antipsychotics aripiprazole, clozapine, haloperidol, olanzapine, paliperidone, pimozide, quetiapine and risperidone to the TGA.
Of the total 2699 cases included in this study, pharmaceutical companies reported adverse events in 1570 cases (58.0%, n = 1570), hospitals reported 659 cases (24.4%, n = 659), pharmacists were the reporter type in 173 cases (6.5%, n = 173), general practitioners reported 114 cases (4.2%, n = 114), specialists reported 58 cases (2.1%, n = 58), nurses reported 7 cases (0.3%, n = 7), members of the public were the reporter type in 85 cases (3.2%, n = 85), information from coroner reports accounted for 23 cases (0.9%, n = 23), State and Territory Health departments reported 9 cases (0.4%, n = 9) and there was 1 case reported by a complementary health professional (n = 1), as shown in Figure 3.

Reporter types by percentages.
Characteristics of adverse events among paediatric and adult populations
In terms of causality, the numbers of cases reported for the paediatric and adult populations as probable were 6.0% and 3.5%, respectively, and the number of cases reported as possible were 92.6% and 96.0%, respectively. There was significant (p = 0.03) difference between paediatric and adult populations in the reporting of causality as possible. Reported cases in children were more likely to report the cause as possible than adults.
Suspected other medications
Overall, in 1726 cases, the antipsychotic was the only medication suspected of causing the reported adverse events (63.3%, n = 1726).
Overall, of the total 2699 cases analysed, co-administered medications that were suspected of contributing to the adverse events were reported in 982 cases (36.4%, n = 982).
In the adult age group (20+ years), the antipsychotic and at least one other medication were reported as possibly causing adverse events in 824 cases (30.5%, n = 824). There was no significant (p = 0.047) difference between paediatric and adult populations.
Suspected formulations
In both the adult and paediatric populations, oral forms of the medications were associated with more adverse events than injectable forms as shown in Table 2. However, there was no significant (p > 0.05) difference between the two populations.
Characterisation of reported adverse events among paediatric and adult populations.

ADR, adverse drug reaction; TGA, Therapeutic Goods Administration.
Adverse events and TGA approved use
The antipsychotics aripiprazole, clozapine, haloperidol, olanzapine, paliperidone, pimozide, quetiapine and risperidone were used for TGA-approved conditions in 490 (18.0%) of the total 2699 reported cases. There were 2166 reported cases where the indication was not specified by the reporter, therefore TGA approval could not be assessed (80.2%, n = 2166).
In both the adult and paediatric populations, a similar number of adverse events were associated with off-label uses (2.0% and 1.6%, respectively). There was no significant difference (p > 0.05) found between the two population groups.
Adverse events and therapeutic dose
A total of 2203 doses were reported for all cases associated with the antipsychotics aripiprazole, clozapine, haloperidol, olanzapine, paliperidone, pimozide, quetiapine and risperidone. In several cases, the reporter specified more than one route of administration (both an oral and injectable dose form) in a single report.
For all age groups, 1560 doses fell within the reasonable dose range according to therapeutic guidelines (70.8%, n = 1560) and 109 doses were above therapeutic limits (4.9%, n = 109). In 1050 cases, the dose was not reported. The age group reporting the highest number of doses that were above therapeutic guideline limits was the adult group 20+ years, with a total of 100 doses (92.0%, n = 100), where the antipsychotic olanzapine accounted for 53 of these cases, quetiapine for 27, and risperidone for 13. There was no significant (p > 0.05) difference between the two populations in the doses prescribed.
Frequency of reporting of death, suicide, hospital admission and overdose
Overall, the highest frequency of death was reported for quetiapine (9.0%), clozapine (8.0%) and olanzapine (7.2%). In adults, quetiapine (9.8%) had the highest reported frequency of death. In children, quetiapine was associated with one death (5.6%). Overall, suicide was reported to be the highest with quetiapine (2.9%). There were nine cases (2.9%) of suicide reported for quetiapine in adults and no suicide was reported in children. Overall, in adults, hospitalisation was the highest for clozapine (36.0%), pimozide (50.0%), risperidone (35.8%), paliperidone (31.9%) and quetiapine (30.7%). Hospitalisation was reported to be high for pimozide (50.0%), representing one case out of a total of two cases. In children, hospitalisation was the highest for clozapine (48.0%) reported in 12 cases, followed by aripiprazole (30.0%) reported in 3 cases and risperidone (29.4%) reported in 15 cases.
Overall, there were more reported cases of death and hospital admissions for adults compared with the paediatric population; however, there were more reported cases of overdose in the paediatric population (5.4%) compared with the adult group (4.8%).
The number of deaths reported for adults was significantly higher than in the paediatric population (7.2% versus 0.7%), p < 0.05. There were no cases of suicide reported for the paediatric population. There were 18 cases of suicide identified in the adult population reports. The number of hospital admissions and reported overdoses were comparable in both paediatric and adult populations and there was no significant (p > 0.05) difference between the two populations.
Management of adverse events
The management of adverse events associated with the antipsychotics aripiprazole, clozapine, haloperidol, olanzapine, paliperidone, pimozide, quetiapine and risperidone that were reported included cessation of the medication or medications suspected of causing the adverse event and/or other treatment. In 1214 cases, the antipsychotic suspected of causing the adverse event was ceased (45.0%, n = 1214) and in 349 cases other medications were ceased (13.0%, n = 349).
The ‘other’ category included specific treatment associated with each antipsychotic. For example, the administration of benztropine, midazolam, lorazepam or other benzodiazepines; administration of intravenous fluids; intensive care unit (ICU) supportive measures and the monitoring of further treatment; and change of medications including antipsychotics, antidepressants and other medications, or a reduction in the dose of medications suspected of causing the adverse event and monitoring for further adverse events. Other treatments were reported in 773 cases (28.6%, n = 773). The management of adverse events remained unspecified in 974 cases (36.0% n = 974). There was a significant difference (p < 0.05) between the paediatric and adult populations associated with the management of adverse events.
Discussion
This study identified the characteristics of ADR associated with antipsychotics from voluntary reporting to the adverse drug event database in Australia. The analysis was undertaken based on two populations: paediatric (5–19 years) and adults (20 years and older).
Overall, this study identified that, in adults, the drug associated with the most reporting of adverse events was clozapine, followed by olanzapine. In children, the highest numbers of adverse events reported in the database were associated with risperidone, olanzapine and clozapine. The most common adverse event reported in adults was NMS. Other adverse events included myocarditis, hyperprolactinaemia, diabetes, galactorrhea, tachycardia and extrapyramidal disorder. The most common reported adverse events in children were dystonia and myocarditis related to clozapine cases. Other reported adverse events included blood creatinine phosphokinase increase, oculogyric crisis, neutropenia, weight gain and extrapyramidal disorder. The types of adverse events reported between the general and paediatric populations differ. This is consistent with recent studies investigating the differences in antipsychotic-related adverse events in adult, paediatric, and geriatric populations.20,21
The high frequency of reported adverse events presented in this study is consistent with the increase in antipsychotic use reported by Brett et al. 1 The authors proposed that some of this increase could be the result of the changes in indications approved by the TGA for some antipsychotics, such as quetiapine for generalised anxiety disorder as well as treatment-resistant depression. 22 This allowed a higher percentage of patients without psychosis or schizophrenia-related conditions to be prescribed these medications.
NMS is a serious and potentially fatal condition. The drugs associated with this ADR were aripiprazole, haloperidol, olanzapine, pimozide and risperidone. A systematic review of case reports analysis published in 2015 found that NMS is induced by both older antipsychotics as well as newer ones. The authors found that clozapine-, aripiprazole- and amisulpride-induced NMS can present with atypical features more frequently than other second-generation antipsychotics induced NMS, for example, displaying less intense extrapyramidal symptoms or high fever. This is consistent with our findings of pyrexia reported by adults taking aripiprazole, clozapine and olanzapine. NMS was also reported in children in our study to be 12.0% with clozapine. The number of paediatric cases reported in the database was relatively small compared with the number of adult cases.23,24
Another serious adverse event reported in adults was QT prolongation. Our study identified that QT prolongation was reported in adults taking aripiprazole, clozapine and quetiapine. The reporting of QT prolongation ranged between 3.4% and 8.2% in adults. Kimura et al. and Belissima et al. found that only ziprasidone and risperidone were associated with QT prolongation.22,25 A recent systematic review addressing QT prolongation with psychotropic medications found that ziprasidone appears most likely to prolong the QT interval. The authors also recommended the frequency of cardiac monitoring for patients receiving psychiatric medications to be determined individually, based on the prescribed agent(s) and additional risk factors for Torsade de Pointes.25,26 Note QT prolongation could be subjective when it comes to its interpretation as there are various methods of reporting. A recent study by Vandenberk et al. stated that tools such as Bazett equation overestimated the number of patients with potential dangerous QT prolongation, 27 whereas the Fridericia and Framingham correction formulae showed the best prediction for 30 day and 1 year mortality. Therefore, these results should be interpreted cautiously. 27
Akathisia is commonly reported with antipsychotics, and was reported also in our study. It is distressing to the patient and has been associated with suicide in some cases. A recent systematic review addressing Akathisia assessment and treatment found that the best approach to this adverse event is a personalised treatment with a consideration of either dose reduction or cessation of the antipsychotics. Other options include switching the patient to another antipsychotic with lower tendency of developing akathisia. 28
In our report, myocarditis in adults and children was reported for clozapine. Myocarditis is often a difficult condition to diagnose and is under-reported in the literature. A recent systematic review that analysed all studies including patients taking clozapine found that the presentation of myocarditis is highly variable and hence difficult to diagnose. The authors recommended close monitoring of cardiac symptoms in patients taking clozapine.26,29
Overall, there was no significant difference between the adult and paediatric populations in most of the parameters examined, including other suspected medications, formulations, indications, therapeutic doses and frequency of hospital admissions and overdose. However, significant differences between the two populations were seen in the causality, death and management of adverse events. The significance seen in causality could be related to the fact that children, on average, are on a lower number of ongoing medications than most adults and, hence, it is easier to identify the possible cause of ADR in this population, unlike adults with chronic conditions who are more likely to be on polypharmacy.20,21
In our study, suicide was reported for quetiapine, paliperidone, olanzapine, risperidone and clozapine, whereas death was reported in all antipsychotics except for pimozide, due to the small number of cases. A 5-year follow-up study investigating patients taking antipsychotics and mortality rate found high mortality and suicide reporting in this group of patients, which required close monitoring during treatment. 26 The high rate of death reported in this study is consistent with a recent meta-analysis conducted by Ralph and Espinet. 30 They revealed that antipsychotic medication prescribing is associated with all-cause mortality and recommended avoiding prescribing these drugs for behavioural problems. 30 These results are consistent with a recent study examining the drugs associated with suicide risk, which highlighted quetiapine as one of the five drugs associated with high suicide rate. 31
Management of adverse events was found to be significantly different between the paediatric and adult populations. Various management strategies were reported for both populations and these included ceasing the drug, reduction of dosages and pharmacological treatment of other symptoms. To date, this is the first study to report on some of the management strategies undertaken by healthcare professionals to manage the reported adverse events. Cessation of the drug and other reported measures such as reduction of dose, switch to another medication and/or modified additional treatment were found to be significantly higher in the paediatric population. This could be related to the severity of adverse events reports in this population compared with adults. Clinicians should be aware of the latest guidelines to manage adverse events. The Canadian Alliance for Monitoring Effectiveness and Safety of Antipsychotics in Children (CAMESA) has detailed comprehensive management guidelines that include the most common adverse events experienced by children taking antipsychotics. Appropriate recognition and management of these adverse events by healthcare professionals is warranted. 32
It should be mentioned that the data reported in this study is from spontaneous reporting of the DAEN database, which is lacking some fields and is not very reliable. The main limitation revealed in using data from SRS was under-reporting.11,12 In addition, it was found that the quality of the information varied from very detailed to limited, and data distortions were possible in the form of duplicates and errors in coding.11,12,14 Nguyen et al. noted that doses and durations were not always recorded, and the Medical Dictionary for Regulatory Activities (MedDRA) terminology did not always fit the complex character of movement disorders. 17 Liu et al. and Minjon et al. commented that, with the exception of drug companies, SRS are limited by their passivity because they are voluntary.16,18 Montastruc et al., 2015 suggested that notoriety bias (from safety alerts) accounted for the high numbers of reports with olanzapine and clozapine. 15 Minjon et al. also cited selective reporting as a limitation, in addition to the inability to quantify the true risk and occurrence in absolute terms, because the actual number of children using antipsychotics is unknown. 16 Minjon et al. 2019 also recognised that causality and severity assessment of the evaluated ADRs was not performed, whilst Raschi et al. acknowledged that the analysis of spontaneous reports does not support the establishment of causality, and reflected in a further study by Raschi et al., who commented that the lack of complete patient-related risk factors and information on drug administration does not allow a full causality assessment of individual reports.12,14,16
This study has some limitations. The reporting of adverse events in the TGA database is voluntary, spontaneous and random in nature. This results in the under-reporting of many adverse events by healthcare professionals due to reasons such as lack of time and lack of knowledge about the reporting process. Therefore, the results presented are observational and may not be regarded as representative of the true frequencies. Another limitation is the lack of clarity of the adverse events reported. For example, unpleasant effects of the disorder, for example, agitation rather than drug-induced akathisia. On the other hand, a side effect may not be drug-related, but may be related to an underlying physical origin requiring further investigation of the aetiology. The low rates of reporting of metabolic adverse events could be due to the fact that the symptoms such as hyperglycaemia may have been confused with diabetes, etc. The reporters may not be trained observers and, even if they are, a comprehensive evaluation of the patient is needed to improve accuracy. Although most reports concerned listed whether the reported ADR is likely, suspected or unlikely, no causality assessment of the evaluated ADRs reports was performed, nor was the severity of the reported ADRs taken into account.
Other limitations include the lack of information regarding the proportion of pharmaceutical companies that were from clinical trials and the proportion of cases that arose from ‘real cases’ and lack of data regarding the total number of antipsychotic users in Australia during the study period. This made it difficult to report the proportion of all antipsychotic ADRs. Healthcare professionals may also have been reporting on serious events rather than expected ones, and this means that the reported adverse events of antipsychotics are under-reported in clinical practice. Members of the public reporting of ADR may be highly emotive, lacking objectivity and clinical knowledge. All these factors may have contributed to the inconsistencies and the quality of the reports. Therefore, it was unfeasible to conduct disproportionate analysis. The lack of known denominator of the total reported ADR has the potential to overestimate the numbers of ADR reported by those who are prescribed these medications. Moreover, other limitations of the study include its retrospective design and reporting that dated from 2004 until 2017. Earlier reporting had more incomplete data than more recent reports. This is in addition to other confounding factors such as other medical conditions.
To summarise, our study found serious adverse events reported with antipsychotics including NMS, prolongation of QT interval and myocarditis. Most of these adverse events are not easily diagnosed as they can manifest as pyrexia, tachycardia and other symptoms. The high reporting of myocarditis in both children and adults needs to be closely monitored and accounted for by psychiatrists. Death was found to be more frequent in adult compared with paediatric populations. Moreover, management of ADR is found to be different between paediatric and adult populations. Further consideration of prescribing practices is warranted to ensure the benefits are considered against the risks and that medicines are monitored closely and deprescribed as appropriate, to minimise the risk of ongoing side effects. The results of this study need to be considered in light of the voluntary nature of reporting the adverse events collected in the database.
Footnotes
Author contributions
HK designed the study and obtained ethics approval, wrote the first draft of the manuscript and undertook the statistical analysis. DH analysed the data from the TGA database. NA was involved in the study design and commenting on the manuscript. All authors have read and approved the manuscript.
Availability of data and materials
The data is not available to the public as per Monash Research Ethics Committee.
Conflict of interest statement
The authors declare that there is no conflict of interest.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethics approval and consent to participate
This study was approved by the Human Research Ethics Committee (Project ID numbers-17558). The data received from the Therapeutic Goods Administration was de-identified.
