Abstract
Background:
Our aim was to investigate whether self-administration of medication (SAM) during hospitalization affects the number of dispensing errors, perceptions regarding medication, and participant satisfaction when compared with nurse-led medication dispensing.
Methods:
A pragmatic randomized controlled trial was performed in a Danish cardiology unit. Patients aged ⩾ 18 years capable of SAM were eligible for inclusion. Patients were excluded if they did not self-administer medication at home, were not prescribed medication suitable for self-administration, or did not speak Danish.
Intervention group participants self-administered their medication. In the control group, medication was dispensed and administered by nurses.
The primary outcome was the proportion of dispensing errors collected through modified disguised observation of participants and nurses. Dispensing errors were divided into clinical and procedural errors.
Secondary outcomes were explored through telephone calls to determine participant perceptions regarding medication and satisfaction, and finally, deviations in their medication list two weeks after discharge.
Results:
Significantly fewer dispensing errors were observed in the intervention group, with 100 errors/1033 opportunities for error (9.7%), compared with 132 errors/1028 opportunities for error (12.8%) in the control group. The number of clinical errors was significantly reduced, whereas no difference in procedural errors was observed. At follow up, those who were self-administering medication had fewer concerns regarding their medication, found medication to be less harmful, were more satisfied, preferred this opportunity in the future, and had fewer deviations in their medication list after discharge compared with the control group.
Conclusion:
In conclusion, the reduced number of dispensing errors in the intervention group, indicate that SAM is safe. In addition, SAM had a positive impact on (a) perceptions regarding medication, thus suggesting increased medication adherence, (b) deviations in medication list after discharge, and (c) participant satisfaction related to medication management at the hospital.
Lay Summary
Intervention group participants self-administered their medication. In the control group, medication was dispensed and administered by nurses. Participants were allocated between groups by chance selection.
The primary result of interest was the proportion of dispensing errors collected through observation of participants and nurses. Secondary results of interest were explored through telephone calls to determine participant perceptions regarding medication, participant satisfaction, and deviations in their medication list two weeks after discharge.
Keywords
Key messages
What is already known on this subject?
(1) Self-administration of medication (SAM) has many advantages including high patient satisfaction, sense of control and independence, and increased medication knowledge.
(2) The evidence on safety of SAM during hospitalization is unclear due to the use of different outcome measures.
What this study adds
(1) Knowledge about the safety in SAM during hospitalization from a randomized controlled trial. Participants who SAM during hospitalization make fewer dispensing errors as compared with dispensing by nurse.
(2) Knowledge about participants’ perceptions regarding medication and satisfaction with SAM in a cardiology unit. In this study population, participants who self-administer medication during hospitalization have fewer concerns regarding their medication and consider medication to be less harmful than participants from the control group two weeks after discharge.
Introduction
Medication errors are frequent in both the healthcare system and in private homes.1–7 Medication errors occur for many reasons through prescribing, dispensing, and administering medication1,2 due to either a wrong or missing action. 5 Not every medication error causes harm and becomes an adverse drug event. 1 However, regardless of whether a medication error caused or had the potential to cause harm, it represents an unnecessary risk for patients. Medication errors may increase hospitalization length, healthcare costs, and mortality.8–10
In healthcare settings, medication administration errors, defined as ‘the administration of a medication dose that deviates from the prescription, hospital guidelines or written procedures,’4,11,12 occur in approximately 20% of the total opportunities for error (OEs).3,4 The medication administration process entails (a) ward-level dispensing of medication into a medicine cup/dosage box and then (b) administering it to patients, based on patient identification.4,12 Some of these errors may be avoided when patients and healthcare professionals work collaboratively through patient involvement.13–15 According to previous research, some patients prefer greater involvement in their treatment and care, 14 which may improve their knowledge, skills, and confidence in managing their condition. 13 Self-management support is an important part of practicing patient involvement. 13 Moreover, self-management programs seem to improve medication use, adherence, and clinical outcomes. 16
It has been estimated that only 50% of patients with a chronic disease adhere to the recommended treatment regimens, which may compromise treatment effectiveness and thereby increase healthcare costs. 17 Nonadherence to medication is complex and is associated with factors related to patients, doctors, and the healthcare system. 18 Patient perceptions regarding medication are related to nonadherence;19–21 and should be considered when supporting patients in their medical treatment. 21
The concept of self-administration of medication (SAM) has been known in hospitals for decades, 22 and previous studies have demonstrated that SAM is associated with numerous advantages including independence, cooperation, increased knowledge, and empowerment.22–26 Moreover, effects on clinical outcomes (e.g. pain scale score and the consumption of analgesics) have also recently been indicated. 27 SAM during hospitalization may provide patients with an opportunity to continue medication management routines from home. 25
However, concerns regarding medication safety due to risk of overdose, underdose and nonadherence have surfaced.23,25 Safety within SAM has been explored with different outcome measures (e.g. pill count, self-reported adherence, urine sampling, and disguised observation). 22 Some studies reported better adherence/fewer errors in SAM groups, whereas others reported lower adherence/more errors, or no difference between groups.22,26 Thus, evidence of SAM safety remains unclear.
The objective of this study was to investigate whether self-administration of medication during hospitalization affects the number of dispensing errors, perceptions regarding medication, and participant satisfaction when compared with nurse-led medication dispensing.
Methods
The study is reported according to the CONSORT 2010 statements for randomized controlled trials (RCTs). 28
Trial design and study setting
The study was designed as a pragmatic RCT of patients admitted to the Medical Department (Cardiology Unit, 28 beds), Randers Regional Hospital, Denmark, from August 2017 to September 2018. The hospital catchment area has approximately 225,000 inhabitants. The unit provides basic cardiology services, with approximately 2000 patients being referred to the unit annually.
The Danish healthcare system provides free, tax-funded access to healthcare for the entire population. 29 Medication is usually provided by hospitals; however, it is permitted to ask patients to administer their own medication at hospital. According to Danish law, doctors are responsible for prescriptions and medication reconciliation. 30 Electronic Medication Administration Record (eMAR) updating is performed at, for example, the Emergency Department, Randers Regional Hospital, prior to referral to the Cardiology Unit. 30 Nurses are responsible for ward-level medication dispensing and administration. 30 The medicine room is supplied with medicine by pharmaconomists, while pharmacists are not regularly involved.
Participants
Patient records were retrieved to determine fulfillment of the exclusion/inclusion criteria. Patients were eligible for inclusion if they were capable of SAM during hospitalization, were prescribed at least one medication suitable for self-administration, practiced SAM at home, spoke Danish, and were aged ⩾ 18 years old.
SAM capability assessment was delegated from doctors to nurses, and included an evaluation of the patients’ current cognitive, emotional, and health status. 31
Patients’ knowledge regarding their medication and drug/alcohol abuse history were also part of the evaluation (Appendix 1).
Consecutive participants were recruited from Monday to Thursday by the primary investigator (PI) or a research assistant. Written informed consent was obtained prior to participation; thus, the patients had the opportunity to decline. The reasons for declining were registered.
Intervention
Medications brought to the hospital by participants were checked by a nurse for the quality (e.g. expiration) and quantity (amount of tablets) 32 and were compared to the eMAR prescription data. In case of uncertainties, a doctor was consulted. Medication was provided by the hospital if the participant had not brought the medication or if new medication was prescribed. A complete medication package was delivered as small blister packages (⩽20 pieces), if available, or as loose tablets in a container. If the medication was only available in large blister packages, it was delivered as a blister card. The medication was labeled with the participant’s name and Civil Registration Number. The medication was then placed in plastic bags; a green bag for medication to be used during hospitalization and a red bag for medication not in use. The medication was stored in participants’ wardrobes and the keys were kept by participants. Prescriptions in the eMAR were marked with ‘Own medication’ and ‘Self-administration.’ An updated medication list and information regarding new medication 33 were printed and provided to participants. A nurse subsequently instructed participants on their medication, and the PI observed participants when they dispensed their medication for the first time. The participants were informed about any medication changes, and participants’ SAM capabilities were reviewed at least once daily by a nurse.
Participants in the intervention group self-administered their medication during hospitalization, with the exception of medication unsuitable for self-administration. Prior to the study, the project group (nurses, a senior doctor, a pharmaconomist, a pharmacist, and the PI) decided that injections and infusions, inhalations through nebulization, one-time prescriptions, medication stored in the refrigerator (except insulin) and variable high doses of digoxin were considered unsuitable for self-administration. The decision was made based on criteria of what was considered safe and possible in clinical practice (including special storage conditions, certain administration forms and dosages).
Control group
In the control group, medication was dispensed and administered by nurses during hospitalization. Participants were allowed to self-administer nonstocked medication and inhalations brought to the hospital. Information on prescription changes were largely provided to participants verbally, at discharge.
Data collection
Baseline
Baseline data included age, sex, education, residential situation, comorbidity,34,35 medical specialty, prescriptions, brought medication, and length of stay. Baseline data were obtained from patient records and participants.
Primary outcome
The primary outcome was the proportion of dispensing errors in relation to the number of OEs observed. A SAM intervention at hospital will, by default, remove the majority of administration errors by nurses (e.g. lacking patient identification, wrong patient). Therefore, to measure errors during dispensing is considered the best way to compare safety of nurse- and participant-led medication processes.
Dispensing errors were defined as ‘the dispensing of a dose of medication that deviates from the prescription, from hospital guidelines or written procedures’ and were divided into clinical and procedural errors as described in the work of Westbrook and colleagues 36 and Risør and colleagues. 12 A clinical error occurred if participants did not receive their medication as prescribed in the eMAR.12,36 A procedural error occurred if nurses deviated from written procedures or guidelines for the dispensing process.12,30,36 Common types of dispensing errors are described in Table 1.11,12,37
Dispensing error types.

These error types were used in the feasibility and pilot study as well (unpublished).
eMAR, Electronic Medication Administration Record.
An OE was defined as any dose dispensed plus any dose prescribed but omitted.11,12 The proportion of errors was calculated by dividing the total number of dispensing errors by the total number of observed OEs and multiplying by 100.
A modified disguised observation technique was used to observe the dispensing of medication.11,38–40 The nurses being observed were aware of time measurements and the study’s overall purpose, but unaware of the real purpose of the observation. The participants in the intervention group were asked to fill their dosage box while the PI was around; however, they were not informed about the observation or the purpose.
All observations were performed following the same protocol from Monday to Friday by the PI or a research assistant on the day of participants’ inclusion to the study, or the following days, with a maximum of two observations per participant to minimize dependency between data. Observation of nurses was performed in the medicine room each morning during the day shift, when most medication was dispensed. Observation of participants was performed in their rooms when they filled medicine cups with their morning medication (or a dosage box with medication for the next 24 h after the ward round). Only medication for intake during the day shift was registered in observations. For both groups, medication was dispensed from original medication packages or blister cards; thus, the name of the medication was visible on the container or blister card. Observations were registered and subsequently compared with prescriptions in the eMAR and written procedures. 30 Deviations were recorded and categorized by error type (Table 1). The observer only intervened when observing a severe error, to prevent an error from reaching the patient.
Secondary outcome
Secondary outcomes were collected either at inclusion or through telephone calls two weeks after discharge by the PI, using a structured interview guide. Questions to explore participant satisfaction and deviations in medication list after discharge were tested for face validity among 10 SAM candidates prior to the study by the PI in order to ensure user friendliness in this particular population (Appendix 2).41,42
Perceptions regarding medication. Participants’ perceptions regarding medication were evaluated using a Danish translation 43 of the Beliefs about Medicines Questionnaire (BMQ) at inclusion and two weeks after discharge.19,20 The BMQ consists of 18 items divided into two sections; BMQ-Specific and BMQ-General. BMQ-Specific comprises two five-item factors assessing beliefs about the necessity (Specific-Necessity) and concerns (Specific-Concerns) regarding medication for personal use. BMQ-General comprises two four-item factors assessing general beliefs about the harmfulness of medication (General-Harm) and overused by doctors (General-Overuse). Responses were provided for each item on a five-point Likert scale (1 = Strongly disagree to 5 = Strongly agree). Total scores ranged from 5 to 25 for BMQ-Specific factors and from 4 to 20 for BMQ-General factors. 19
Participant satisfaction. Participants were asked about their satisfaction with how they received medication during hospitalization; a five-point Likert scale was used (1 = Very unsatisfactory to 5 = Very satisfactory). Additionally, participants were asked if they would prefer SAM in a future hospitalization.
Deviations in medication list after discharge. Participants were asked to list the medication they consumed on the day of the interview. This self-reported medication list was compared with the medication list in their discharge letter. Participants were asked to explain any deviations. Deviations in prescribed medication for regular intake that were not in accordance with their general practitioner (GP)’s or the hospital’s prescription were registered.
Sample size
The calculation of sample size was based on a power of 90% and a statistical significance level of 5%. A proportion of dispensing errors of 16% was used based on results from the feasibility and pilot study (unpublished). A 30% reduction in error proportion was considered clinically relevant. 12 Based on these values, we calculated a sample size of 1020 OEs to be observed in each study group. From the number of medications and observations in the feasibility and pilot study, it was estimated that 1020 OEs per group would correspond to approximately 150 participants per group. Recruitment proceeded until at least 1020 OEs were observed in each study group.
Randomization
Randomization was performed by the Hospital Pharmacy’s Department of Quality Assurance using http://www.randomization.com and random block sizes between 8 and 20 to avoid possible distribution prediction.
For each participant, the Department of Quality Assurance wrote the group allocation down and placed it in a sealed opaque envelope. These envelopes were opened by the PI or the research assistant upon participant recruitment.
Blinding was not possible due to the intervention type.
Data analysis
Data were analyzed per protocol using Stata® v.15 (StataCorp, 4905 Lakeway Drive, TX, USA).
Continuous outcomes were compared by Student’s t test or Wilcoxon rank-sum test. As the distribution of length of stay by nature is positively skewed, the data were log transformed before analysis.
Binary outcomes were primarily compared in a chi-squared test. Fisher’s exact test was used in the test of small samples with expected values less than 5. 44
Total scores of the four factors of BMQs were calculated for each participant and as a mean for each group at point of inclusion, follow up, and as the change between the two time points.
Ethics
Study procedures were in accordance with the ethical standards of the Helsinki declaration and its later amendments, 45 as well as Danish legislation. The intervention did not include a biomedical intervention; thus, the Regional Committee of Health Care Ethics waived the need for approval. The study was registered with ClinicalTrials.gov and the Danish Data Pro-tection Agency (1-16-02-106-17) [ClinicalTrials.gov identifier: NCT03541421].
Results
Participant flow
A total of 2245 patients were admitted to the unit during the study period (21 August 2017 to 26 September 2018; with 174 inclusion days). A total of 1666 patients were assessed according to the inclusion/exclusion criteria. Of these, n = 632 (37.9%) were eligible; however, a large proportion n = 250 (39.6%) was discharged before inclusion was possible and some were readmissions (Figure 1). In total, n = 354 (56.0%) patients were invited to participate, of which n = 104 (29.4%) declined. Of these, n = 36 (34.5%) wanted self-administration and refused to be randomized (i.e. possibly allocated to the control group).
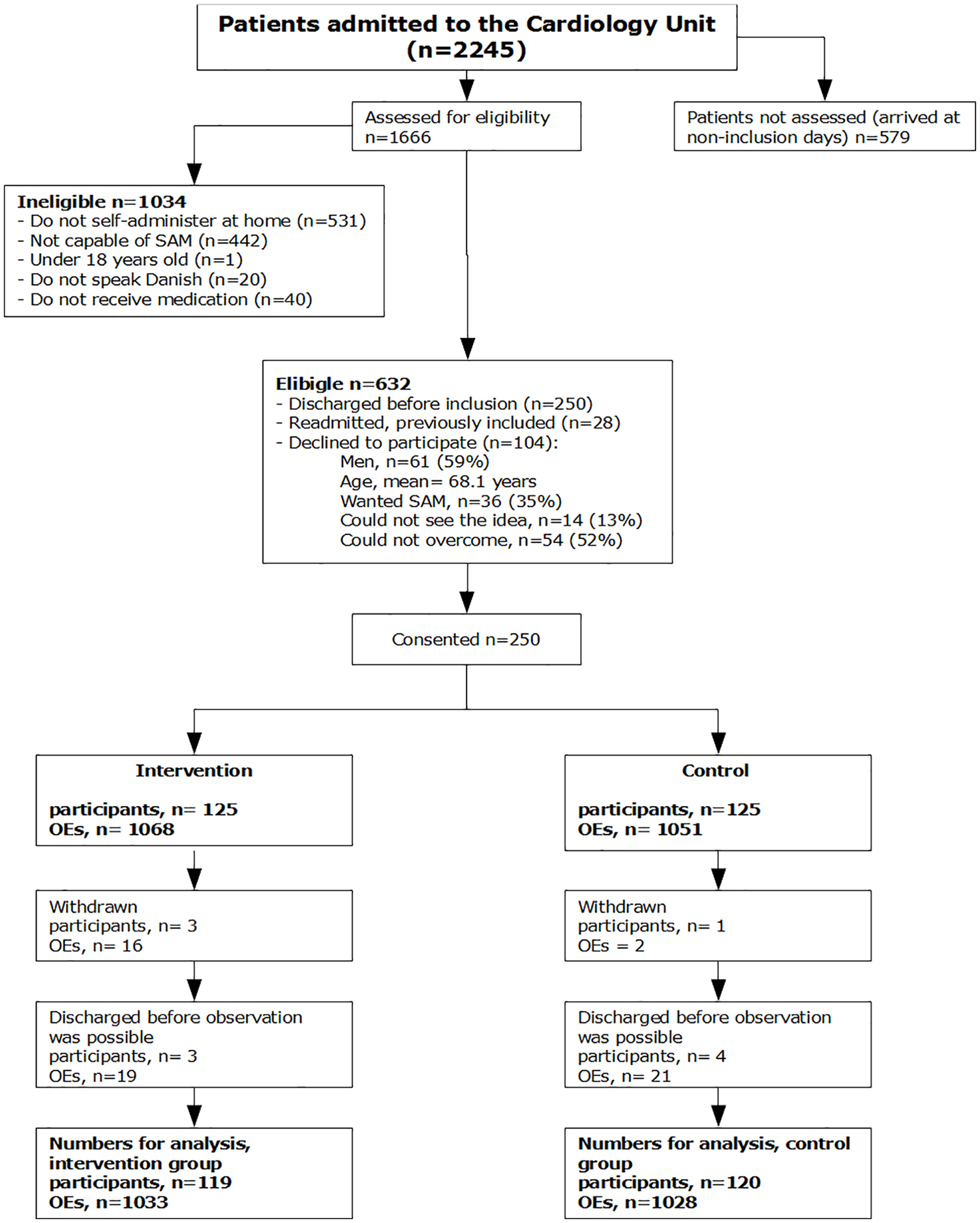
Patient flow (CONSORT).
Recruitment
A total of 250 participants were recruited and randomized (125 participants per group), corresponding to 1068 OEs in the intervention group and 1051 OEs in the control group. Three participants (16 OEs) from the intervention group withdrew due to a decline in their capability of SAM. One participant (two OEs) withdrew due to a refusal to be in the control group. Seven participants (40 OEs) were discharged earlier than expected and were thus not observed.
Baseline data
Baseline characteristics of participants in the groups were similar (Table 2). The majority of participants were cardiological patients. At admission, participants in both groups were taking a mean of four different medications, and seven at discharge, indicating similar medication regimens.
Baseline characteristics.

Charlson comorbidity index: 0 = no comorbidity and 6 = severe disease, e.g. malignant tumor.
Student’s t test.
Chi2 test.
IQR, interquartile range; SD, standard deviation.
Primary outcome: dispensing errors
A statistical significant difference in the total number of dispensing errors was observed (p = 0.02), with 100 errors in 1033 OEs (9.7%) in the intervention group (41 participants) and 132 errors in 1028 OEs (12.8%) in the control group (68 participants; Table 3).
Primary outcome: dispensing errors.
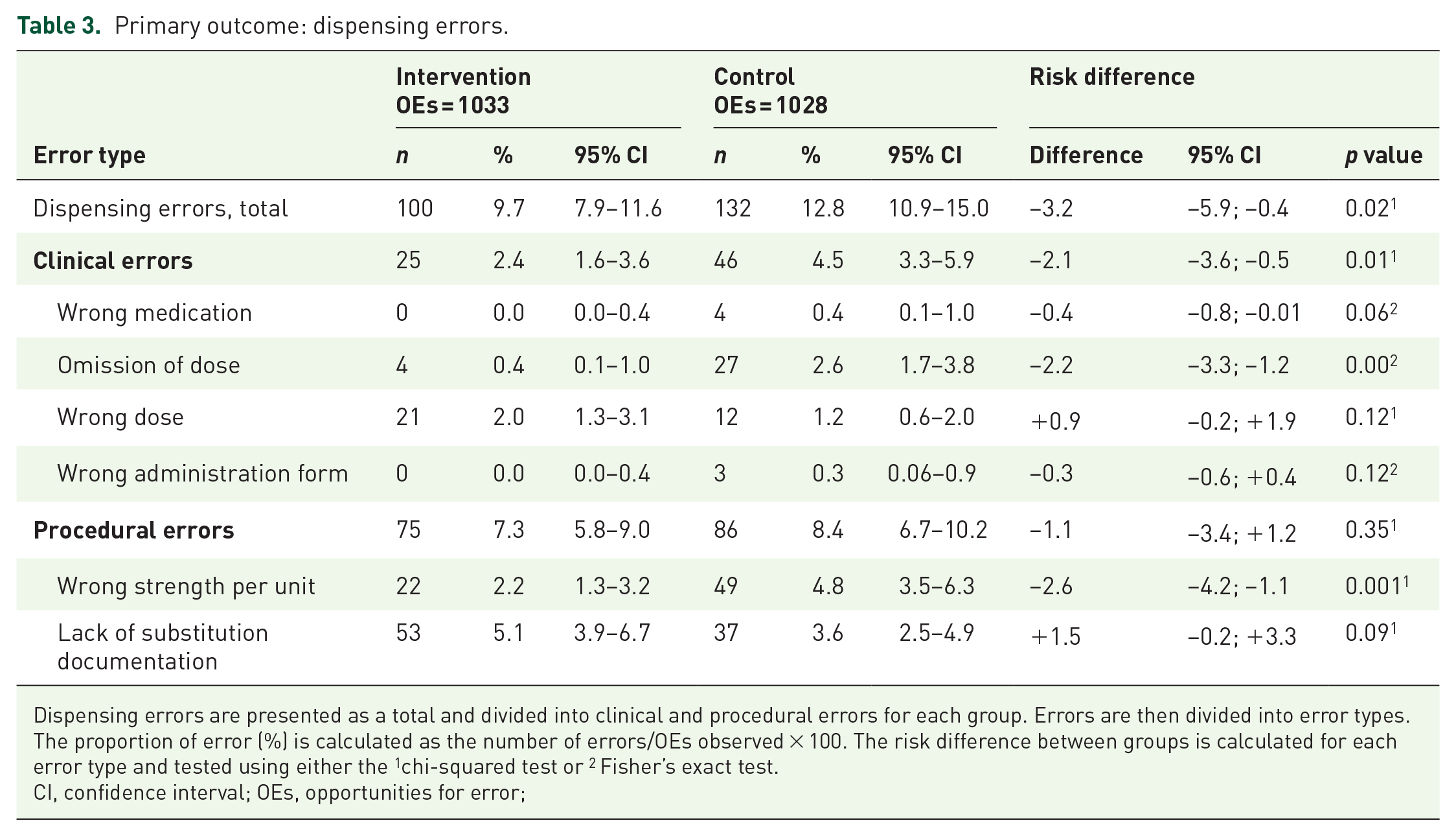
Dispensing errors are presented as a total and divided into clinical and procedural errors for each group. Errors are then divided into error types. The proportion of error (%) is calculated as the number of errors/OEs observed × 100. The risk difference between groups is calculated for each error type and tested using either the 1chi-squared test or 2 Fisher’s exact test.
CI, confidence interval; OEs, opportunities for error;
The number of clinical errors in the intervention group was 25, compared with 46 in the control group. The difference between groups was statistically significant (p = 0.01) and corresponded to a relative risk reduction of 46.7%. The number of procedural errors was 75 in the intervention group, compared with 86 in the control group (not statistically significant; p = 0.35).
In the intervention group, n = 21 (84%) of the clinical errors were due to wrong dose (e.g. dispensing 50 mg losartan when 100 mg was prescribed) whereas n = 27 (59%) of the clinical errors in the control group were omissions (e.g. omitting nonstocked candesartan; Table 3). More examples of observed errors are presented in Appendix 3.
In the intervention group, n = 53 (71%) of procedural errors involved a lack of substitution documentation, whereas n = 49 (57%) of procedural errors in the control group were ‘wrong strength per unit’ (nurse did not document a change in strength per unit).
There was no statistically significant dependency between data on error proportion; neither in relation to the number of observations per participant (p = 0.23), nor to which nurse was involved (p = 0.07).
Secondary outcomes
Perceptions regarding medication
A total of n = 224 (93.7%) participants were reached by telephone. Three participants did not answer the BMQ at follow up, leaving n = 221 (92.5%) participants for analysis. Compared with baseline characteristics, the n = 18 (7.5%) nonresponders were slightly younger (mean age 59.7 years), healthier at inclusion (Charlson comorbidity index; mean = 0.5), and fewer were men (55.5%).
No statistically significant difference was observed for perceptions regarding medication at the time of inclusion (Table 4).
Secondary outcomes.
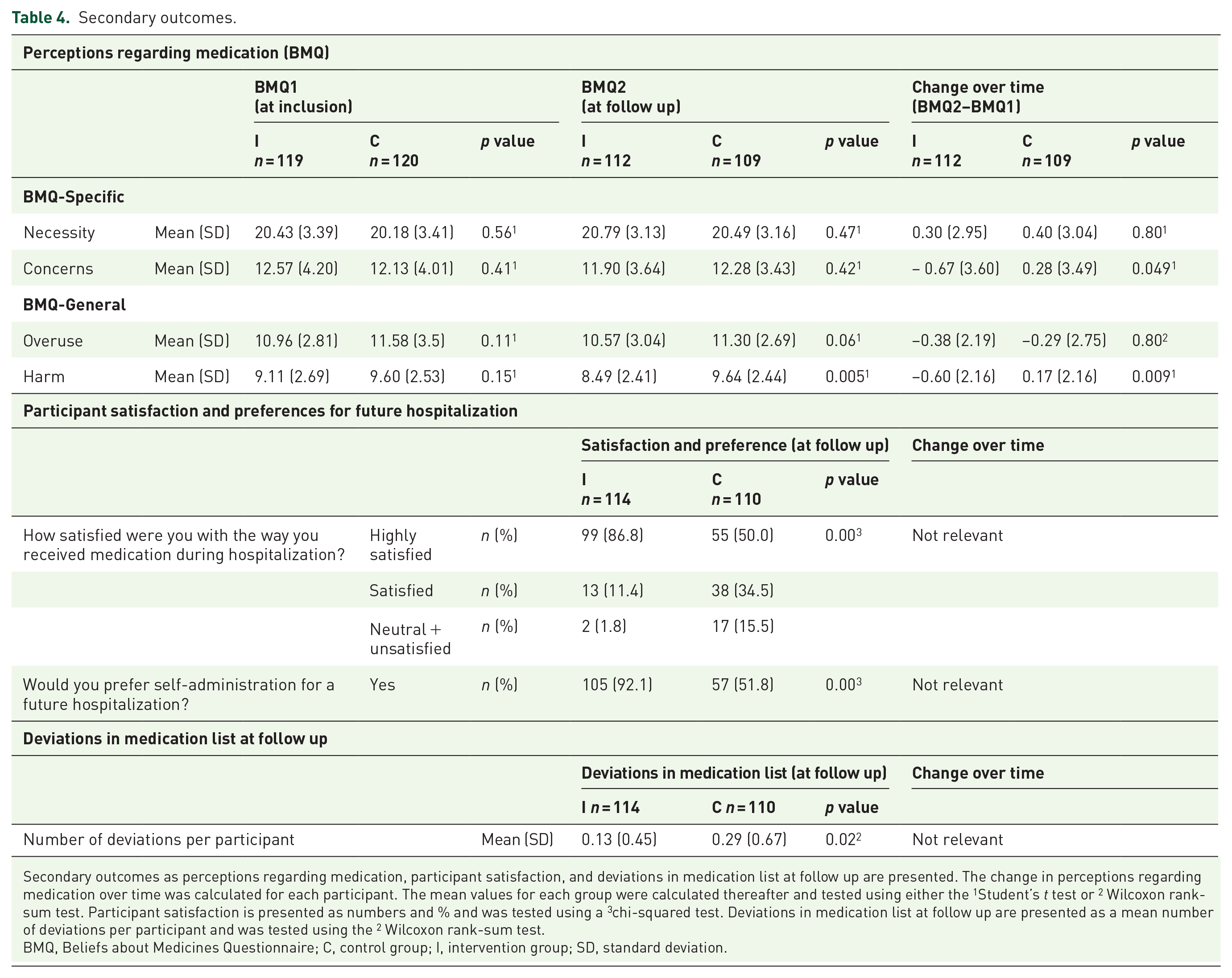
Secondary outcomes as perceptions regarding medication, participant satisfaction, and deviations in medication list at follow up are presented. The change in perceptions regarding medication over time was calculated for each participant. The mean values for each group were calculated thereafter and tested using either the 1Student’s t test or 2 Wilcoxon rank-sum test. Participant satisfaction is presented as numbers and % and was tested using a 3chi-squared test. Deviations in medication list at follow up are presented as a mean number of deviations per participant and was tested using the 2 Wilcoxon rank-sum test.
BMQ, Beliefs about Medicines Questionnaire; C, control group; I, intervention group; SD, standard deviation.
When comparing changes in the BMQ over time, significantly smaller values of the factors ‘Concerns’ (p = 0.049) and ‘Harm’ (p = 0.009) were observed in the intervention group. There was no statistically significant difference between groups concerning the factors ‘Necessity’ and ‘Overuse’ (Table 4).
Participant satisfaction
A total of n = 99 (86.8%) participants in the intervention group versus n = 55 (50.0%) in the control group expressed high satisfaction (p < 0.01) with how they received medication during hospitalization. A total of n = 105 (92.1%) participants in the intervention group and n = 57 (51.8%) participants in the control group preferred SAM for possible future hospitalization (p < 0.01; Table 4).
Deviations in medication list
There were more deviations between the self-reported medication list at follow up and prescriptions from the hospital and the GP (p = 0.02) in the control group, when compared with the intervention group (Table 4). For example, one control group participant did not wonder why the doctor had not sent prescriptions to the primary pharmacy. Hence, he was not treated with anticoagulant medication following myocardial infarction.
Harm
Overall, the intervention was performed without harm. However, in one case, an unintended event was reported to the Danish Patient Safety Database. An SAM participant was not informed about prescription changes and therefore he dispensed and administered a discontinued medication (isosorbide mononitrate).
Discussion
In this pragmatic RCT, we observed fewer dispensing errors among SAM participants during hospitalization compared with dispensing by a nurse. We found fewer clinical errors in the intervention group, but no difference between groups in the proportion of procedural errors. At follow up, participants from the intervention group had less concerns regarding their medication, generally considered medication to be less harmful, were more satisfied with how they received medication during hospitalization, and preferred SAM in future hospitalizations compared with the control group. Finally, we found fewer deviations in the medication list at follow up compared with the control group.
Medication safety at hospital
Only one previous study compared errors performed by nurses/technicians and patients. 46 They observed a higher proportion of errors in the self-administering group, with 154 errors (omissions) in 3548 doses (4.3%; 24 patients) compared with 27 errors (omissions or wrong dose) performed by nurses/technicians in 1135 doses (2.4%; 37 patients). 46 In contrast, we observed significantly fewer dispensing errors in the intervention group, and most clinical errors were dose errors, not omissions. These conflicting results are most likely due to the previous study not being an RCT and that errors in the self-administering group were detected through interviews and pill counts instead of direct observation.
Unfortunately, comparisons with other studies are not possible due to the use of different outcome measures, such as adherence or medication errors caused by patients using methods such as pill count, self-reported adherence, and urine sampling.22,26
We found ‘wrong dose’ to be the most frequent clinical error in the intervention group. This may be due to participants not being properly instructed about prescription changes or that the participant had misunderstood that the strength and dose of the medication were not the same. This emphasizes that staff maintain an important, albeit different, task in supporting participants in medication management, as prescription changes must be properly communicated from staff to participants. As suggested by Vanwesemael and colleagues, 47 SAM may include supporting, screening, monitoring, and empowering patients and, therefore, they suggest using the broader term ‘self-management of medication’ covering a range of aspects that need to be looked into during self-administration.
These errors could have had other reasons, and we recommend further studies to fully understand them.
The most frequently recorded clinical error in the control group was ‘omission,’ often because the prescribed medication was not stocked on the ward and, thus, caused primarily by external circumstances tied to the medication distribution system. If more participants brought own medication to hospital, this error type would certainly have been minimized.
SAM is not suitable for every patient; it depends on their current situation including their cognitive, emotional, and health status.31,48 Thus, SAM will not be suitable for many elderly living in a nursing home or for those who receive help for medication management at home. Proper objective assessment of SAM capability is therefore very important.
Participant preferences and perspectives
This study demonstrates that participants who were instructed about their medication and who self-administered medication during hospitalization were more satisfied with how they received medication compared with dispensing and administration by a nurse.
Another Danish study also reported a high level of satisfaction with self-administration among 66 surgical patients, where 94% were satisfied with being responsible for self-managing their medication. 31 However, this study did not compare with medication managed by nurses.
In our study, participants who self-managed their medication during hospitalization preferred to have this opportunity in possible future hospitalizations. This result is in accordance with a UK study in which 86% of the self-administering group and 50% in the control group preferred SAM. 49 It is worth noting that participants in a control group have not tried SAM during hospitalization; thus, they might not have the knowledge to answer the question about preference for future hospitalization.
Patient perspectives have also been explored in qualitative studies in Australia, Belgium24,25 and Denmark. 50 All studies have noted positive attitudes toward SAM, as it gave participants a sense of control, independence, and improved medication safety.24,25,50
Effects after discharge
The participants’ perceptions regarding medication were explored at the point of study inclusion and at follow up. Participants exposed to the intervention had fewer concerns regarding medication and generally considered medication to be less harmful, compared with the control group. To the best of our knowledge, no previous studies have explored the impact of SAM on participants’ beliefs about medicine.
Previous research demonstrated that higher necessity scores correlate with higher medication adherence and higher levels of concern correlate with lower medication adherence. 20 Previous studies have also shown correlations between nonadherence and higher scores for General-Harm and General-Overuse.51,52 Thus, the lower concern score and harm score found in our study can be considered a surrogate marker of higher medication adherence in the intervention group.
There were a larger number of deviations in the medication list at follow up in the control group compared with the intervention group. This could be due to a lack of knowledge regarding medication, as participants were often unaware of the deviations. According to systematic reviews, some evidence suggests that patients’ knowledge about medication increases through involvement in an SAM scheme.22,26 Further studies on the postdischarge impact are recommended.
Strengths and limitations
This study has some strengths and limitations that merit further discussion.
Patients were eligible for the study if they were capable of self-administration (nurses assessed eligibility). However, in the assessment, we did not use a validated assessment tool,48,53 which may have introduced an information bias to the study through misclassification. However, we believe the potential misclassification is nondifferential, and thus will not affect the overall estimates of the study.
Furthermore, we only observed the proportion of dispensing errors since the intervention removed the majority of administration errors in the intervention group. Thus, we did not observe actual medication consumption.
To ensure the validity of the observations, we used two trained observers following the same protocol. However, the dispensing process was largely observed each morning; this may have affected external validity, potentially making us unable to compare our results with medication routines at other times of the day. Dispensing was observed a maximum of twice per participant due to a concern that SAM participants were not informed about prescription changes. This design was considered as a strength, as we actually saw this error a couple of times. The observation was not repeated further to minimize dependency between data, and we did not see any statistically significant difference in error proportion; neither in relation to the number of observations per participant, nor to which nurse was observed. It was a concern that the observer may influence the person being observed (‘the Hawthorne effect’); however, previous research has demonstrated that this bias holds limited importance, as the observed person quickly gets accustomed to being observed and returns to normal behavior.4,5,11,38 Additionally, as both groups were observed, we believe a possible misclassification to be nondifferential.
This pragmatic RCT reflected a typical hospital ward; as such, several nurses were replaced, and new staff was regularly trained over the study period. This can be considered a limitation to the study, since only few nurses became experienced with SAM. Meanwhile, nurses who were inexperienced may have failed to perform some intervention tasks. However, we believe that a strong focus on training and working procedures is recommended for future SAM implementation.
Perceptions regarding medication were measured using BMQs. BMQ questions were read to participants at their inclusion to the study, and they also had the opportunity to read the questions on paper. However, the questions were read aloud through the telephone at follow up. This difference in data collection method could potentially have influenced the answers; though, we believe the misclassification to be nondifferential.
Participants were selected and recruited at a cardiological unit from Monday to Thursday. We acknowledge that there may be more or less patients eligible for SAM in other types of wards and on weekends. Furthermore, our findings are limited to experiences with SAM in participants with a low level of comorbidity who were generally younger than those who declined participation, which makes us unable to generalize our results for older patients with more comorbidity. We consider it to be a strength of our study that only n = 11 (4.4%) withdrew from the study. Of those, only n = 3 (1.2%) withdrew due to a change in their capability of SAM.
The clinical consequence of errors was not investigated. The power calculation was based on the number of dispensing errors detected in the control group of the feasibility and pilot study (unpublished); however, only procedural errors were observed in this study. Clinical errors were the most important errors to avoid, since participants in these cases did not receive medication as prescribed.12,36 It was not feasible to perform an RCT with a power based on clinical errors, since it would require a huge number of OEs to be observed. Procedural errors have the potential to cause injury;12,36 however, further research on the clinical consequence of such errors is recommended.
In conclusion, the reduced number of dispensing errors in the intervention group indicate that SAM is safe. In addition, SAM had a positive impact on perceptions regarding medication, suggesting stronger medication adherence among SAM participants. Finally, we observed a positive impact on the number of deviations in medication lists following discharge, as well as on participant satisfaction related to medication management in the hospital setting.
Supplemental Material
Appendix_1_capability_assessment – Supplemental material for Self-administration of medication: a pragmatic randomized controlled trial of the impact on dispensing errors, perceptions, and satisfaction
Supplemental material, Appendix_1_capability_assessment for Self-administration of medication: a pragmatic randomized controlled trial of the impact on dispensing errors, perceptions, and satisfaction by Charlotte Arp Sørensen, Marianne Lisby, Charlotte Olesen, Ulrika Enemark, Signe Bredsgaard Sørensen and Annette de Thurah in Therapeutic Advances in Drug Safety
Supplemental Material
Appendix_2_questions – Supplemental material for Self-administration of medication: a pragmatic randomized controlled trial of the impact on dispensing errors, perceptions, and satisfaction
Supplemental material, Appendix_2_questions for Self-administration of medication: a pragmatic randomized controlled trial of the impact on dispensing errors, perceptions, and satisfaction by Charlotte Arp Sørensen, Marianne Lisby, Charlotte Olesen, Ulrika Enemark, Signe Bredsgaard Sørensen and Annette de Thurah in Therapeutic Advances in Drug Safety
Supplemental Material
Appendix_3_error_examples – Supplemental material for Self-administration of medication: a pragmatic randomized controlled trial of the impact on dispensing errors, perceptions, and satisfaction
Supplemental material, Appendix_3_error_examples for Self-administration of medication: a pragmatic randomized controlled trial of the impact on dispensing errors, perceptions, and satisfaction by Charlotte Arp Sørensen, Marianne Lisby, Charlotte Olesen, Ulrika Enemark, Signe Bredsgaard Sørensen and Annette de Thurah in Therapeutic Advances in Drug Safety
Footnotes
Acknowledgements
This study could not have been performed without the engaged work performed by the nurses in the Cardiology Unit when they delivered the intervention. Thanks to the constituted department nurse Lotte Grøn for active participation in the study phases. Thanks to the pharmaconomist Karin Bruun Kastrup who acted as a research assistant, and participated in both the recruitment of new participants and the data collection. Thanks also must be given to the steering committee for their support throughout the study. Thanks to professor Robert Horne (UK) for permission to use the “Beliefs about Medicines Questionnaire.
CAS, ML, CO, UE and AT were responsible for the planning and design of the study. CAS was responsible for data collection and analysis. KBK participated in recruitment of new participants and data collection. CAS wrote the first draft of this manuscript. ML, CO, UE, SBS, and AT provided critical revision. All authors read and approved the final manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: the study was funded by the Hospital Pharmacy Central Denmark Region, the Hospital Pharmacies and Amgros’ Research and Development Foundation and Randers Regional Hospital.
Conflict of interest statement
The authors declare that there is no conflict of interest.
Data availability statement
Data are available from the corresponding author upon reasonable request.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
