Abstract
Mirogabalin is a selective calcium channel α2δ subunit ligand being developed to treat neuropathic pain. In accordance with US Food and Drug Administration (FDA) guidance, the human abuse potential of mirogabalin (15–105 mg) was examined, relative to placebo, diazepam (15 or 30 mg), and pregabalin (200 or 450 mg), in two single-dose, randomized, double-blind, placebo- and active-controlled crossover studies in recreational polydrug users who could discern between positive comparator and placebo. The primary endpoint was maximum observed effect (
Introduction
Current guidelines for the management of neuropathic pain from the International Association for the Study of Pain include a range of options for first-line pharmacological therapy, including calcium channel α2δ subunit ligands, such as gabapentin and pregabalin. 1 High-quality evidence supports the use of these agents;2–4 however, both gabapentin and pregabalin are associated with a high incidence of adverse events, particularly dizziness and somnolence. 5 In addition, concerns have been expressed about the potential for misuse, abuse, or diversion of gabapentin and pregabalin, especially among individuals with a history of opioid or benzodiazepine use. 6 There is, therefore, an unmet need for a well-tolerated calcium channel α2δ ligand with a low potential for abuse.
Mirogabalin monobenzenesulfonate (mirogabalin; Daiichi Sankyo Co., Ltd., Tokyo, Japan) is a preferentially selective ligand of α2δ calcium channels. 7 A phase II proof-of-concept trial demonstrated analgesic efficacy in patients with diabetic peripheral neuropathic pain (DPNP).8,9 Results will soon be available from phase 3 studies in patients with DPNP (ClinicalTrials.gov identifier: NCT02318706) and postherpetic neuralgia (ClinicalTrials.gov identifier: NCT02318719).
The US Food and Drug Administration (FDA) recommends that all agents with central nervous system (CNS) activity be assessed for their abuse potential, so that this information can be included in the regulatory assessment. 10 Based on preclinical data and the similarity to gabapentin and pregabalin, which are both controlled substances, a human abuse potential study was required for mirogabalin. In accordance with FDA guidance, two independent studies were conducted to examine the human abuse potential of mirogabalin in adults with a history of recreational drug use. One study compared mirogabalin with placebo and diazepam (the diazepam study) and one study compared mirogabalin with placebo and pregabalin (the pregabalin study).
Methods
Study design
The diazepam and pregabalin studies both were approved by the Institutional Review Boards and were performed in accordance with the Declaration of Helsinki and International Conference on Harmonisation guidelines.
11
All participants provided written informed consent prior to study entry. In both studies, the primary objective was to compare the abuse potential of mirogabalin relative to placebo, diazepam, and pregabalin in recreational polydrug users. The primary endpoint was the maximal effect (
Secondary objectives were to evaluate the safety and tolerability and the pharmacokinetics (PK) of mirogabalin, diazepam, and pregabalin. The PK parameters were calculated from plasma concentrations using a noncompartmental approach and included maximum serum concentration (
Diazepam study
This was a single-center, randomized, balanced, placebo- and active-controlled, crossover study with two phases (Figure 1A); a double-blind qualification phase to ensure that eligible participants could discriminate the effects of the active control (diazepam) compared with placebo and an assessment phase (see Supplemental Materials). Participants reported to the clinic in the afternoon before period 1 dosing and remained in the clinic until checkout (day 3 of period 5). Qualifying participants were randomly assigned to a predetermined treatment sequence according to a two 5 × 5 Williams square design (10 sequences in total) and received a single dose of each of the following in a double-blind, double-dummy, crossover manner: mirogabalin 15 mg, mirogabalin 45 mg, diazepam 15 mg, diazepam 30 mg, and placebo (Figure 1A). Each participant was followed by serial assessments for 48 hours postdose. Treatment periods were separated by ⩾5 days between dose administrations to eliminate carryover effects.

Study design: (A) diazepam study; (B) pregabalin study.
Participants were healthy male and female recreational polydrug users aged between 18 and 55 years. They were required to have used CNS depressants for nontherapeutic reasons on ⩾10 occasions in the past 5 years, including use of ⩾1 CNS depressant in the past 3 months (details in Supplemental Materials). Participants with a substance abuse disorder or those seeking treatment for addiction-related disorders were excluded. All participants were required to have a negative drug screen at baseline and to abstain from recreational drug use for the duration of the study.
Pregabalin study
The pregabalin study consisted of a dose escalation phase, followed by a qualification phase, and then an assessment phase (Figure 1B, Supplementary Materials). The dose escalation phase was included to evaluate the safety and tolerability of ascending doses of mirogabalin (60–105 mg) for use in the assessment phase. Based on data from the dose escalation phase, three doses were chosen for the assessment phase: the proposed therapeutic dose (15 mg), a low supratherapeutic dose (60 mg), and a high supratherapeutic dose (105 mg).
The assessment phase had a six-period, randomized, placebo- and active-controlled, balanced crossover design (Figure 1B, Part 2). Participants reported to the clinic on the day before period 1 dosing and remained in the clinic until check-out (day 3 of period 6). Qualifying participants were randomly assigned to a predetermined treatment sequence according to a 6 × 6 Williams square design, and received a single dose of each of the following: mirogabalin 15 mg, mirogabalin 60 mg, mirogabalin 105 mg, pregabalin 200 mg, pregabalin 450 mg, and placebo. Treatment periods were separated by ⩾3 days between dose administrations to eliminate carryover effects.
The pregabalin study included healthy men and women aged 18 to 55 years who were recreational polydrug users, using the same exclusion criteria as in the diazepam study (see Supplemental Materials for a detailed definition).
Patients were assessed for 48 h after each administration and completed 6 VAS at baseline and at predefined intervals up to 48 h postdose; these were the 4 abuse-potential VAS (Drug Liking, High, Good Drug, Bad Drug), Any Effect VAS, and Alertness/Drowsiness VAS. Participants also completed three additional VASs: one asking about similarity to other drugs (at 12 h postdose), one asking about their overall liking for the drug (at 12, 24, and 48 h postdose), and another asking about whether they would take the drug again (at 12, 24, and 48 h postdose) (see the Supplemental Materials for more details).
Blood samples were collected at baseline, 1, 2, 3, 4, 6, 12, 24, 36, and 48 h for PK assessment. Each participant was scheduled to repeat the process, receiving each possible treatment in a randomized order, with ⩾3 days of washout between each assessment. Participants remained at the research facility throughout the study, checking out on day 3 of period 6. Plasma concentrations of mirogabalin were measured using validated liquid chromatography–tandem mass spectrometry method. Detailed bioanalytic methods can be found in the Supplemental Materials.
Statistical analysis
In both studies, safety analysis was undertaken in all participants who received at least one dose of randomized medication or placebo, and had at least one relevant postdose assessment. The pharmacodynamic (PD) analysis was undertaken in all participants who were randomized into the assessment phase, received at least one dose of study drug, and had at least one postbaseline PD measurement (completer analysis set). Baseline demographic characteristics were summarized using arithmetic mean, median, range, and standard deviation (SD) for continuous variables, and frequency and percentage for categorical variables.
PD values at each time point were summarized by treatment using descriptive statistics. For each VAS, the
A mixed-effects model was used to compare the primary PD endpoint (Drug Liking VAS
PK values were summarized by study using arithmetic mean, SD, and coefficient of variation percent (CV%). AUC and
Results
Participant disposition, demographics, and baseline characteristics
For the diazepam study, 79 patients entered the qualification phase, 38 patients entered the assessment phase, and 32 completed all assessments (Supplemental Figure 1). For the pregabalin study, 126 participants entered the qualification phase, 56 participated in the assessment phase, 44 received all 6 treatments, and 41 completed the study (Supplemental Figure 1).
Most participants were male (84.2% in the diazepam study and 80.4% in the pregabalin study [assessment phase]) and Black or African American (68.4% [diazepam] and 71.4% [pregabalin]). The mean (SD) age of participants was 31.1 (6.99) years in the diazepam study and 30.4 (8.65) years in the pregabalin study. Baseline demographic characteristics for the assessment phase populations are listed in Table 1. All participants in both studies reported prior drug use (Table 1).
Summary of demographics (assessment phase).
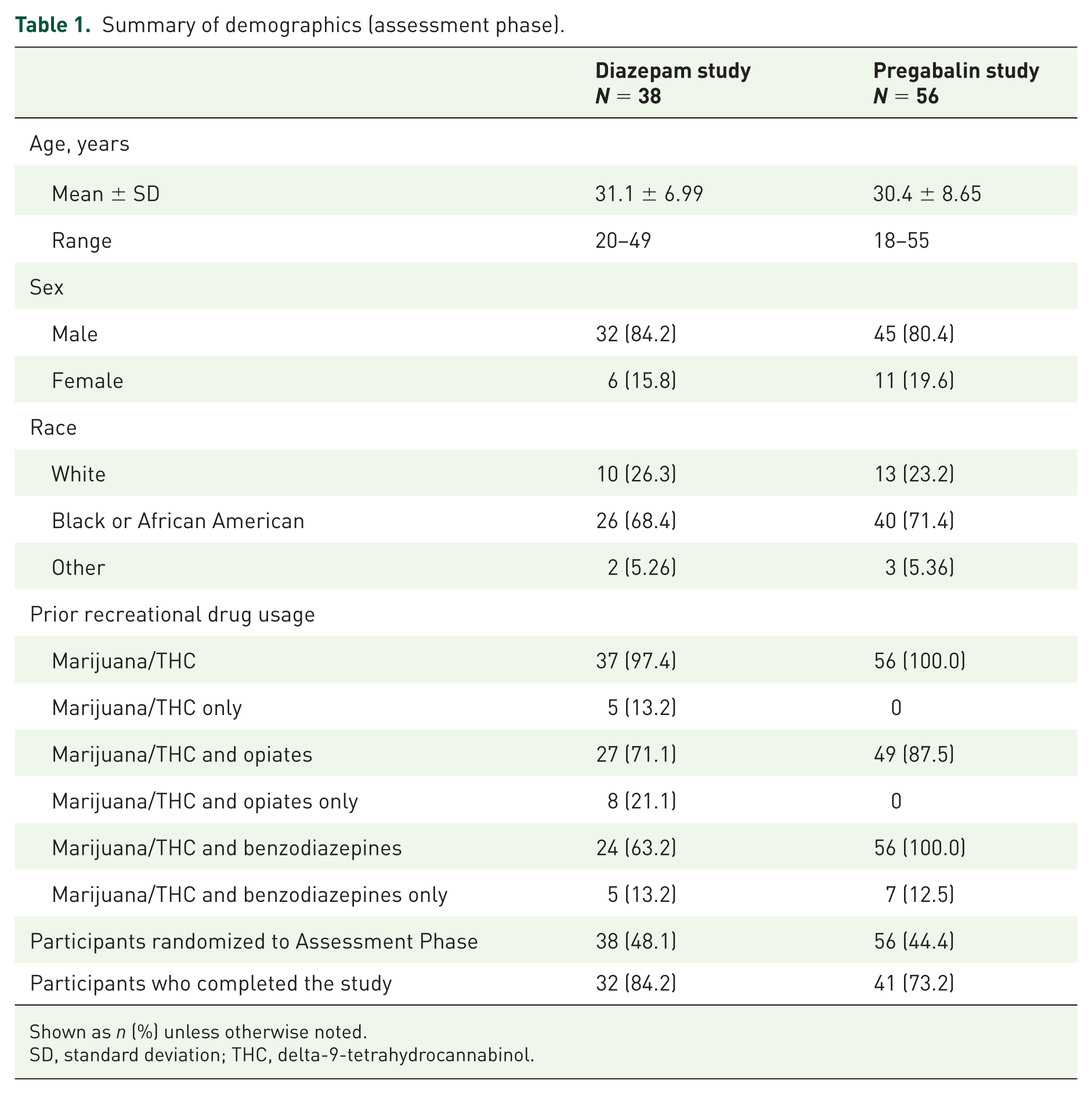
Shown as
SD, standard deviation; THC, delta-9-tetrahydrocannabinol.
Pharmacodynamic results
Pharmacodynamic balance of effects measures: Drug Liking VAS
Mean Drug Liking VAS scores over 24 h postdose are presented in Figure 2.

Mean profile of Drug Liking visual analog scale scores by treatment: (A) diazepam study; (B) pregabalin study.
The therapeutic dose of mirogabalin 15 mg was not significantly different from placebo on the primary endpoint of

Statistical analysis of maximum observed effect for Drug Liking: (A) diazepam study; (B) pregabalin study.
Pharmacodynamic parameters for Drug Liking VAS assessment during the assessment phase in patients who received study drug in all treatment periods a .

Shown as arithmetic mean (standard deviation), unless otherwise noted.
The visual analog scale used was a 100-point bipolar scale where 50 was neutral.
DZP, diazepam;
Pharmacodynamic secondary measures
The therapeutic dose of mirogabalin 15 mg was not significantly different from placebo on any of the secondary endpoints (Table 3). In contrast, the low and high supratherapeutic doses of mirogabalin (60 and 105 mg) demonstrated significantly greater mean
Secondary PD outcome measures (assessment phase) a .

Shown as arithmetic mean (standard deviation), unless otherwise noted.
The visual analog scale used was a 100-point bipolar scale where 50 was neutral.
DZP, diazepam;
In the pregabalin study, pregabalin (particularly at the 450 mg dose) was rated as being similar to benzodiazepines, and less so to opioids, on the Drug Similarity VAS. Mirogabalin 15 mg was not rated as being similar to any drugs of abuse; however, the two supratherapeutic doses were rated as being similar to benzodiazepines.
Pharmacokinetic results
The concentration-time profile of mirogabalin in the two studies is shown in Figure 4. Plasma concentrations and exposure parameters (
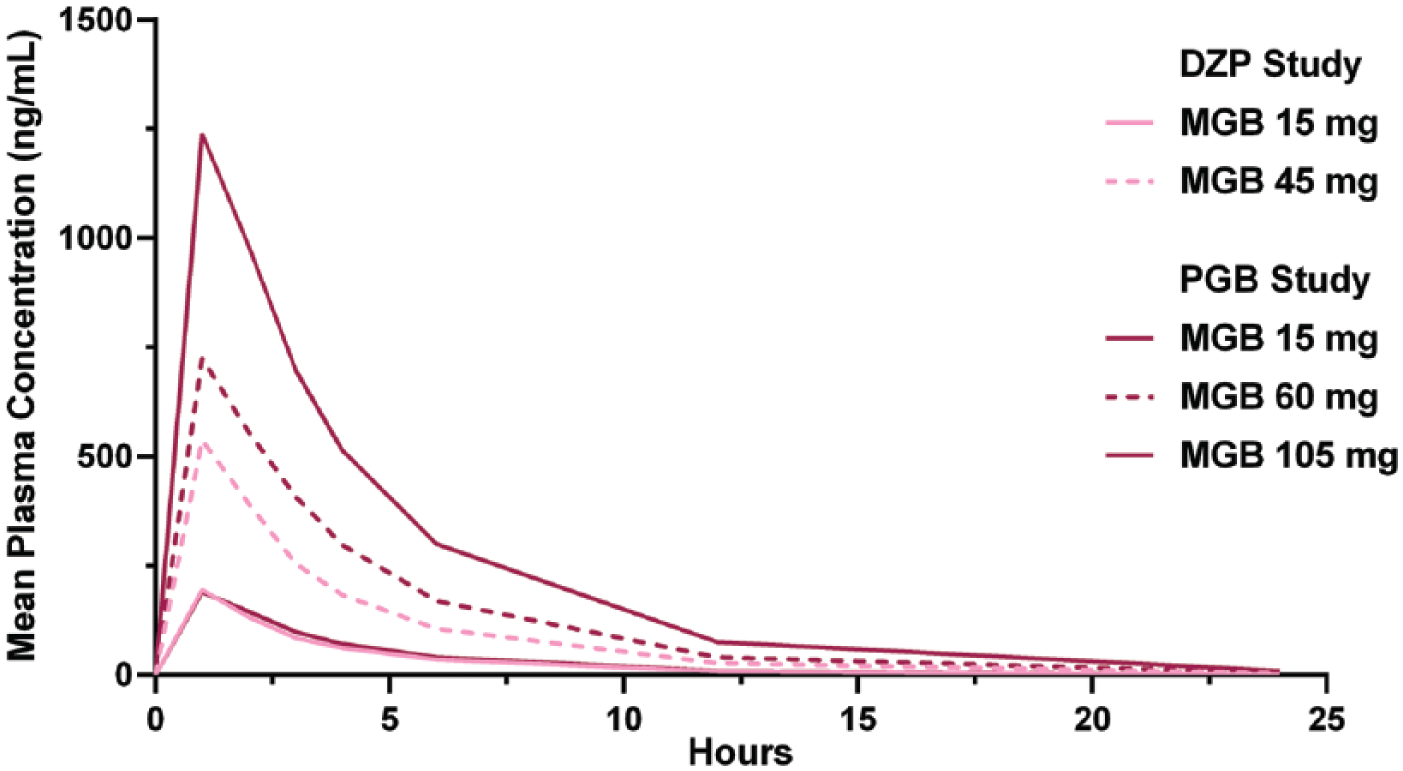
Pharmacokinetic profile of mirogabalin after a single dose in the diazepam and the pregabalin studies.
Safety results
No deaths or serious adverse events occurred in either study. With the exception of one severe adverse event that occurred with diazepam 30 mg, all adverse events were either mild or moderate in intensity. One participant withdrew from the assessment phase of the diazepam study because of cellulitis after receiving diazepam 30 mg, and one withdrew from the assessment phase of the pregabalin study due to mild postural orthostatic tachycardia that was considered to be unrelated to study drug (pregabalin 450 mg).
The most common TEAE reported in the diazepam study was sedation, which occurred in 25 participants overall (65.8%), but none in the mirogabalin 15-mg group (Supplemental Table 2). Other events commonly reported in this study were postural orthostatic tachycardia syndrome (23.7% overall) and a feeling of relaxation (26.3% overall). TEAEs (reported by ⩾5% of participants in any single treatment group) in the pregabalin study included euphoric mood, somnolence, postural orthostatic tachycardia syndrome, dry mouth, upper respiratory tract infection, muscle spasms, headache, and dizziness (Supplemental Table 2). A dose-dependent trend was observed in the incidence of the most common TEAEs following administration of mirogabalin and pregabalin, with the exception of dizziness, which did not occur in a dose-dependent manner following administration of pregabalin. The incidence of the most common TEAEs was generally comparable between mirogabalin 15 mg and placebo, and between mirogabalin 60 mg and pregabalin 450 mg, and higher for mirogabalin 105 mg than all of the other treatments, including pregabalin 450 mg.
No clinically relevant laboratory or ECG abnormalities were noted by the investigator in either study, and no participant experienced hepatic transaminase elevations >3× upper limit of normal.
Discussion
These two studies demonstrate that the planned therapeutic dose of mirogabalin (15 mg) shows limited potential for abuse, based on maximal Drug Liking scores that were not significantly different from placebo in recreational polydrug users. In addition, based on the primary endpoint of Drug Liking
The effects of mirogabalin on most of the tested PD parameters appeared dose-related. However, even at a supratherapeutic dose of 45 mg, the maximal effects of mirogabalin on Drug Liking, Positive Effects High, and Good Drug Effects were not significantly different from those of placebo and were less marked than those of diazepam 15 or 30 mg. Only in the pregabalin study, when mirogabalin was administered at doses four or seven times higher than the planned therapeutic dose, did the
These findings demonstrate that single doses of mirogabalin are well tolerated. In the diazepam study, the incidence of adverse events with mirogabalin 15 mg (8.6%) or 45 mg (27.0%) was similar or lower than the incidence with placebo (28.6%), and lower than after single doses of diazepam 15 (69.4%) or 30 mg (80.0%). The planned therapeutic dose of mirogabalin was similarly well tolerated in the pregabalin study. The incidence of adverse events with mirogabalin 15 mg was comparable with the incidence in the placebo group (36.0%
The PK profile of mirogabalin was consistent with previous reports in patients receiving single doses of mirogabalin.
12
The
Although these studies examined only a single dose of mirogabalin, both were carefully designed to identify abuse potential based on FDA guidelines. 10 Key strengths include the use of a qualification phase to identify appropriate participants; use of both placebo and active comparators; randomized, double-blind, double-dummy, crossover designs; examination of a range of doses, including supratherapeutic doses; use of specific VAS assessing potential drug effects; and conduct of the study in a controlled laboratory setting to prevent other drug use.10,13 The significant effects of the two positive controls (diazepam and pregabalin) compared with placebo on the primary endpoint and most of the secondary endpoints demonstrate the validity of the study and sensitivity of the measures for detecting abuse-related effects.
Conclusions
Overall, the results of these studies demonstrate that mirogabalin had limited abuse potential at the therapeutic dose of 15 mg and is well tolerated in participants with a history of recreational polydrug use. At supratherapeutic doses (e.g. 60 and 105 mg), mirogabalin has abuse potential greater than placebo, but similar to that of pregabalin in a highly sensitive population of recreational polydrug users.
Supplemental Material
Revised_supplemental_material_5Feb2019 – Supplemental material for Abuse potential of mirogabalin in recreational polydrug users
Supplemental material, Revised_supplemental_material_5Feb2019 for Abuse potential of mirogabalin in recreational polydrug users by Jeanne Mendell, Naama Levy-Cooperman, Ed Sellers, Bradley Vince, Debra Kelsh, James Lee, Vance Warren and Hamim Zahir in Therapeutic Advances in Drug Safety
Supplemental Material
Supplemental_figure_1A_(1) – Supplemental material for Abuse potential of mirogabalin in recreational polydrug users
Supplemental material, Supplemental_figure_1A_(1) for Abuse potential of mirogabalin in recreational polydrug users by Jeanne Mendell, Naama Levy-Cooperman, Ed Sellers, Bradley Vince, Debra Kelsh, James Lee, Vance Warren and Hamim Zahir in Therapeutic Advances in Drug Safety
Supplemental Material
Supplemental_figure_1B_(1) – Supplemental material for Abuse potential of mirogabalin in recreational polydrug users
Supplemental material, Supplemental_figure_1B_(1) for Abuse potential of mirogabalin in recreational polydrug users by Jeanne Mendell, Naama Levy-Cooperman, Ed Sellers, Bradley Vince, Debra Kelsh, James Lee, Vance Warren and Hamim Zahir in Therapeutic Advances in Drug Safety
Supplemental Material
Supplemental_tables – Supplemental material for Abuse potential of mirogabalin in recreational polydrug users
Supplemental material, Supplemental_tables for Abuse potential of mirogabalin in recreational polydrug users by Jeanne Mendell, Naama Levy-Cooperman, Ed Sellers, Bradley Vince, Debra Kelsh, James Lee, Vance Warren and Hamim Zahir in Therapeutic Advances in Drug Safety
Footnotes
Author contributions
Hamim Zahir and Jeanne Mendell were the clinical study leaders, designed the studies, interpreted the data, and wrote the manuscript. Naama Levy-Cooperman designed the research, interpreted the data, and wrote the manuscript. Bradley Vince and Debra Kelsh performed the research. All authors provided critical feedback and approved the final version.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by Daiichi Sankyo, Inc., Basking Ridge, NJ. Writing and editorial assistance was provided by Senem Kurtoglu, PhD, MBA, and Claire Daniele, PhD, of AlphaBioCom, LLC, King of Prussia, PA, and supported by Daiichi Sankyo, Inc.
Conflict of interest statement
The author(s) declared following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: JM and HZ are employees of Daiichi Sankyo, Inc. JL and VW were employees of Daiichi Sankyo, Inc. at the time the study was conducted. NLC, ES, DK, and BV received funding to conduct the study.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
