Abstract
Nafcillin, a beta-lactam semisynthetic penicillin, is highly resistant to penicillinase and is similar to other penicillins except that it is primarily metabolized in the liver. It is believed that nafcillin causes CYP3A4 enzyme induction which decreases warfarin’s half-life. The onset of CYP3A4 induction by nafcillin occurs within the first 7 days, but maximal effects may take up to 2 weeks. Once nafcillin is discontinued, the effects persist for several weeks. A 79-year-old male with a history of atrial fibrillation and a 53-year-old male with a history of recurrent venous thromboembolism required significantly higher weekly warfarin doses during courses of nafcillin therapy. Both patients required a 2.5–3.5-fold increase from their baseline weekly warfarin dose to achieve therapeutic international normalized ratios (INRs) while on nafcillin. Traditional protocol-driven warfarin management can result in suboptimal anticoagulation in patients on warfarin and nafcillin.
Introduction
Published literature identifies the drug–drug interaction (DDI) between warfarin and nafcillin; however, the severity and complexity of the interaction is often overlooked or mismanaged by healthcare providers. Nafcillin, a penicillinase-resistant semisynthetic penicillin, is used to treat infections such as endocarditis caused by susceptible strains of staphylococcus species. Nafcillin is highly protein bound and is similar to other penicillins except for the fact that it is metabolized by the liver. It has a half-life of 0.5–1 h, and is primarily excreted in the feces, with approximately 30% excreted in the urine as unchanged drug. 1
The proposed mechanism for the DDI with warfarin is the induction of CYP3A4 by nafcillin which then induces warfarin’s metabolism. 2 Warfarin is a racemic mixture of R-warfarin enantiomer and the three-to-five times more potent S-warfarin enantiomer. R-warfarin is metabolized by CYP3A4 and CYP1A2, while S-warfarin is predominately metabolized through CYP2C9 with minor metabolism through CYP3A4; therefore, DDI between warfarin and CYP3A4 inducers tend to be less significant. 3
A literature search in PubMed utilizing the search criteria ‘warfarin and nafcillin’ OR ‘warfarin and dicloxacillin’ without date restrictions resulted in 20 articles published between 1984 and 2015. Two articles discussed DDI between nafcillin or dicloxacillin and drugs other than warfarin, and two articles did not discuss any DDI. The 16 remaining publications included nine case reports, three retrospective studies, two prospective trials, and two summaries of possible DDI with warfarin. The studies were focused on determining if a DDI existed or the mechanism of the DDI. Within the 9 published case reports, a total of 11 patient cases were described including: 4 warfarin-naïve patients at the time nafcillin was initiated (1 of which was a pediatric patient), 4 stable warfarin patients prior to initiation of nafcillin, 2 stable warfarin patients prior to initiation of dicloxacillin, and 1 stable warfarin patient at the time of nafcillin initiation that was changed to dicloxacillin.4–19 In the warfarin-naïve patients, higher initial warfarin doses (5–12 mg/day) were needed initially during nafcillin treatment. The weekly warfarin doses were typically two- to fourfold higher than the doses after discontinuation of nafcillin.11–13,16 For those patients stable on warfarin prior to initiation of nafcillin, prothrombin times (PTs) were subtherapeutic within 7 days of starting nafcillin and required approximately a threefold increase in their weekly warfarin doses. The increased dose was needed until 2–4 weeks after nafcillin discontinuation. Two of the four patients also required bridging with unfractionated heparin during this time due to subtherapeutic PT.15–17 In the patients taking warfarin and dicloxacillin, it appears that the dicloxacillin interaction is less severe than that with nafcillin as the warfarin doses did not even reach a twofold increase.14,18–19
Despite the current published literature related to the DDI between warfarin and nafcillin or dicloxacillin, the management of this DDI remains suboptimal. One reason for the suboptimal management may be the difficultly in interpreting most of the case reports, as the results are reported in a PT instead of an international normalized ratio (INR). Other possible reasons are that many of the case reports include confounding variables such as changes in antibiotic therapy, other concomitant DDI, holding warfarin for procedures, and new initiation of warfarin therapy; all of which affect the generalizability to clinical practice. Finally, based on the very limited published data, it does not seem preferred that the DDI with warfarin be treated equivalently for nafcillin and dicloxacillin.
Patient cases
Institutional Review Board approval was not required as there were less than four cases being reviewed, thus not constituting ‘research.’
Patient case 1
The patient was a 79-year-old white male taking warfarin for thrombosis prophylaxis in atrial fibrillation with a goal INR of 2–3. His CHA2DS2VASc score was 5 based on the presence of hypertension, age ⩾ 65, and previous cerebrovascular accident (CVA). His HAS-BLED score was 4 due to hypertension, age, aspirin use, and previous CVA. The patient was newly started on warfarin approximately 1½ months prior to initiation of nafcillin for endocarditis. Prior to initiation of nafcillin, the patient’s home dose of warfarin was 35 mg per week. This dose was stabilized just prior to the time nafcillin was initiated. He denied any missed doses of warfarin while taking nafcillin and warfarin concomitantly.
The patient’s INR was checked while on nafcillin therapy for the first time 2 weeks after it was started. His INR was subtherapeutic and the weekly warfarin dose was increased. Subsequently, the patient’s dose continued to be increased every 4–7 days in response to continued subtherapeutic INR values. Despite a threefold increase in warfarin dose to 100 mg per week, the patient’s INR continued to be subtherapeutic and trended downwards. He ultimately required a 3.3-fold increase in his weekly warfarin dose to achieve therapeutic INRs while on nafcillin. Nafcillin was stopped then restarted three times during this course of therapy due to hospitalizations and the infectious disease team changing his regimen while in the hospital. The patient did not have any side effects, intolerances, or adverse reactions to nafcillin, and the only notable DDI was from nafcillin and warfarin. The first time, nafcillin was stopped for 10 days; the next time, it was stopped for 28 days; and the final time, it was discontinued permanently.
At 2½ weeks after nafcillin was stopped the second time, the patient’s INR had climbed to 4.1, and the dose of warfarin was reduced to 70 mg per week. At 1 month after the nafcillin had been stopped the second time, it was restarted for 10 days. He required another dose increase after nafcillin was restarted for the final 10-day course. The nafcillin was then stopped permanently and the patient started lifelong cephalexin. Approximately 3 weeks after nafcillin was discontinued for the final time, the patient’s INR increased to 3.6 and the warfarin dose was decreased to 62.5 mg per week. His warfarin dose required additional gradual reductions over the next 8 weeks. See Figure 1 for a visual representation of the dose adjustments during the courses of nafcillin.
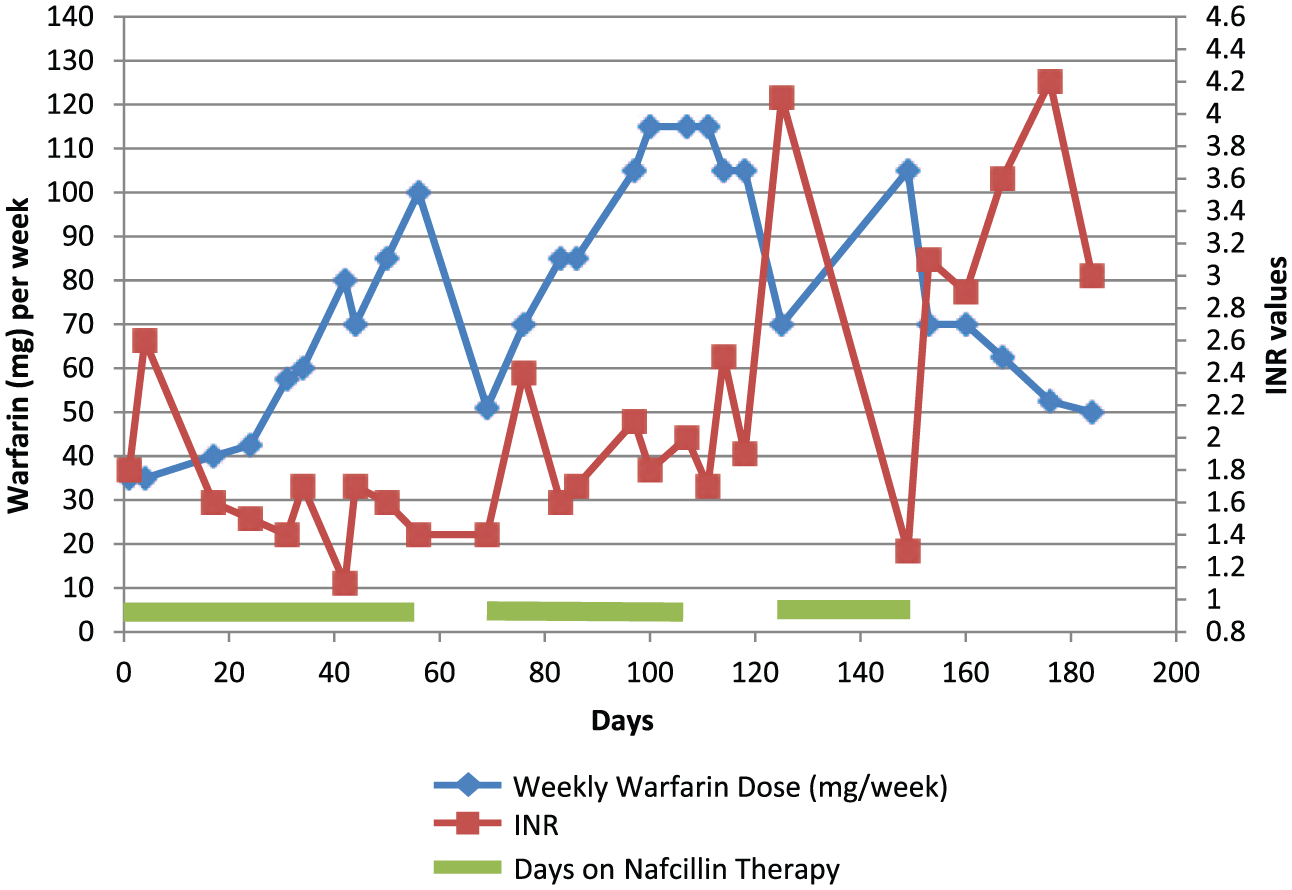
Patient case 1: warfarin dosing over time with the impact of nafcillin.
Patient case 2
The patient was a 53-year-old white male taking warfarin for thrombosis prophylaxis, with a history of recurrent, unprovoked pulmonary embolism (PE) and deep vein thrombosis (DVT). His goal INR was 2–3, and his HAS-BLED score was 3, due to labile INRs, aspirin use, and a history of a retroperitoneal bleed. The patient had been on warfarin for approximately 1½ years when he had an L3–L5 posterior laminectomy performed. At 3 weeks following surgery, he developed an incision site infection that ultimately required nafcillin therapy. Prior to initiation of nafcillin, the patient’s home dose of warfarin was 24 mg per week. He had been stable on a dose of 20–26 mg per week for approximately 5 months.
Within 8 days of starting nafcillin, the patient’s INR dipped to 1.7, and his dose was adjusted to 30 mg for 1 week due to a one-time dose adjustment, and then told to continue 28 mg per week. The patient’s dose was adjusted over the following 6 weeks of nafcillin therapy and he ultimately required 56 mg per week, a 2.5-fold increase from his baseline weekly dose. The patient’s nafcillin was discontinued at the end of those 6 weeks. He was continued on warfarin 56 mg per week until his INR rose to 5.7, 4 weeks after stopping nafcillin. The patient was instructed to hold a dose, and then decrease to 28 mg per week, and the patient’s INRs remained therapeutic over the following month on this dose. See Figure 2 for a visual representation of the dose adjustments during the course of nafcillin.
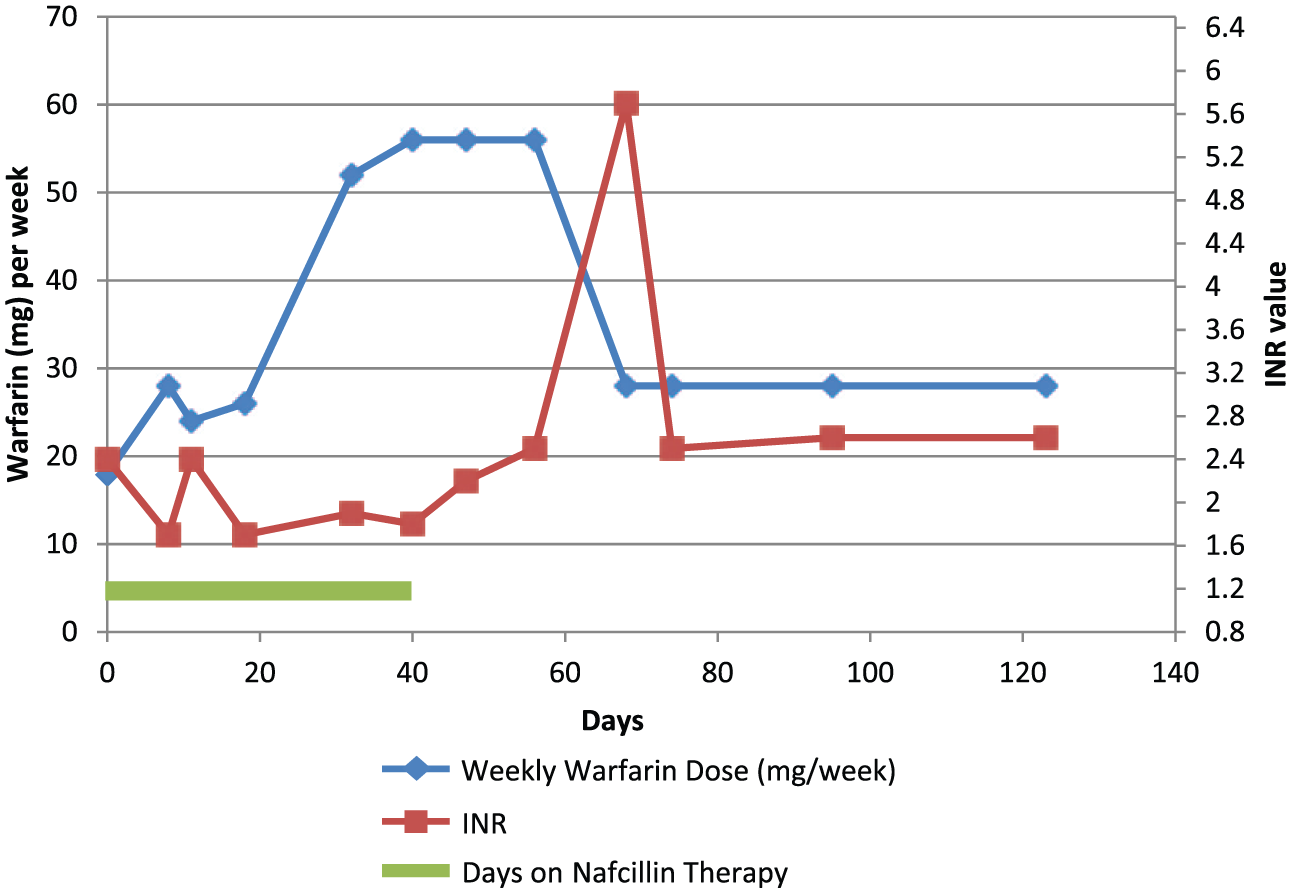
Patient case 2: warfarin dosing over time with the impact of nafcillin.
Discussion
When utilizing the Drug Interaction Probability Scale, patient case 1 scored an 8 and patient case 2 scored a 6. Both scores resulted in a probable drug interaction. 20 Although CYP3A4 interactions with warfarin tend to be less significant, as it is not the predominant metabolizer, these cases demonstrated that the interaction between warfarin and nafcillin is significant. If this interaction is inappropriately managed, it will result in suboptimal anticoagulation management and increases the patient’s risk for thrombosis. Although it would be ideal to avoid the concomitant use of warfarin and nafcillin, unfortunately, due to increased drug-resistant organisms and indication for anticoagulation, this is not always possible. Anticoagulants that do not interact with nafcillin include heparin, low-molecular-weight heparin, and fondaparinux. Currently, it is unknown whether a significant interaction exists between nafcillin and apixaban, edoxaban, rivaroxaban or dabigatran. Further research would be needed to determine if these medications could offer a safe alternative for patients requiring anticoagulation while on nafcillin therapy. Of note, oral factor Xa inhibitors and direct thrombin inhibitors should not be used in the setting of valvular atrial fibrillation and are not approved for use in patients with mechanical valves.21–24
Although the anticoagulation management in the two cases described above is not ideal, several critical take away points from these cases can help improve anticoagulation management in patients requiring concomitant use of warfarin and nafcillin. First, even though infection can and traditionally does increase the INR, this should not be a reason for conservative dosing in patients taking nafcillin. Second, it has been demonstrated that if the patients are continued on their stable home dose of warfarin, the INR will decrease within the first 5–7 days of nafcillin therapy. Since warfarin has a delayed onset, ideally warfarin doses should start to be increased within the first 3–5 days to help to maintain therapeutic INRs. A modest dose increase would be appropriate at this point, as the INR will likely be at the low end of the therapeutic range or slightly subtherapeutic. As you can see in case 2, a 50% dose increase was given after 1 week of nafcillin therapy that resulted in a therapeutic INR at the next visit. Third, based on the continued subtherapeutic INRs since the two cases presented, despite increased weekly doses of warfarin, the full induction of warfarin metabolism due to nafcillin seems to take at least 2 weeks. During this time, the patient’s doses will need continued adjusting. Case 2 supports this, as the weekly warfarin dose was not increased further after day 11 when the INR was 2.4 and then the next several INRs were subtherapeutic. Fourth, the induction of warfarin’s metabolism is not immediately reversed with the discontinuation of nafcillin. Patients still require increased doses for at least 2 weeks and typically 4 weeks post nafcillin discontinuation. Finally, extreme caution is needed with the anticoagulation management of patients on nafcillin. Protocol-driven anticoagulation management in patients on concomitant warfarin and nafcillin therapy will result in suboptimal management. Traditional one-time dose increase with a 10–20% weekly dose increase will not result in therapeutic INRs when these cases and past literature support that at least a twofold increase and upwards of a four- to fivefold increase is often needed to achieve and sustain therapeutic INRs.
Conclusion
The DDI between warfarin and nafcillin can be significant, with the onset of induction occurring within the first 5–7 days of nafcillin therapy but may take up to 2 weeks for full effect. The effects of the induction typically last for 2–4 weeks after the discontinuation of nafcillin but may extend up to 2 months. Recommendations are to start with a 25–50% empiric dose increase, similar to other strong inducers, starting 3 days after initiation of nafcillin and continue to monitor INRs every 3–7 days initially until stable. Ultimately, at least two- to fourfold dose increases are typically required to achieve therapeutic INRs while a patient is on nafcillin.
Footnotes
Funding
This case report was completed without assistance from a pharmaceutical manufacturer and the authors did not receive any funds or grants for this research.
Conflict of interest statement
The authors declare that there is no conflict of interest.
