Abstract
Background:
The aim of this study was (1) to apply the current United Kingdom (UK) National Institute for Health and Care Excellence (NICE) clinical practice guidelines to a hypothetical older patient with multimorbidity and life-limiting illness; (2) consider how treatment choices could be influenced by NICE guidance specifically related to multimorbidity; and, (3) ascertain if such clinical practice guidelines describe how and when medication should be reviewed, reduced and stopped.
Methods:
Based upon common long-term conditions in older people, a hypothetical older patient was constructed. Relevant NICE guidelines were applied to the hypothetical patient to determine what medication should be initiated in three treatment models: a new patient model, a treatment-resistant model, and a last-line model. Medication complexity for each model was assessed according to the medication regimen complexity index (MRCI).
Results:
The majority of the guidelines recommended the initiation of medication in the hypothetical patient; if the initial treatment approach was unsuccessful, each guideline advocated the use of more medication, with the regimen becoming increasingly complex. In the new patient model, 4 separate medications (9 dosage units) would be initiated per day; for the treatment-resistant model, 6 separate medications (15 dosage units); and, for the last-line model, 11 separate medications (20 dosage units). None of the guidelines used for the hypothetical patient discussed approaches to stopping medication.
Conclusions:
In a UK context, disease-specific clinical practice guidelines routinely advocate the initiation of medication to manage long-term conditions, with medication regimens becoming increasingly complex through the different steps of care. There is often a lack of information regarding specific treatment recommendations for older people with life-limiting illness and multimorbidity. While guidelines frequently explain how and when a medication should be initiated, there is often no information concerning when and how the medications should be reduced or stopped.
Background
Clinical practice guidelines are described as ‘systematically developed statements to assist professional decisions about appropriate healthcare for specific clinical circumstances’. 1 They are used to provide support and advice to practitioners regarding a wide range of clinical situations, including patient monitoring, lifestyle advice, treatment choice through stepped care, and therapeutic drug monitoring. The use of clinical practice guidelines has become a common feature in modern day medicine, with many clinical decisions informed by them on a daily basis. Using clinical practice guidelines to inform treatment decisions has the advantage of improving the consistency of care in an attempt to improve overall health outcomes for patients. 2 This is critical, as studies have shown that treatment decisions for patients can vary dramatically depending on their clinician, hospital, or geographical region. 3
In view of this, the number of clinical practice guidelines used in medicine is exponentially increasing: in 1990 there were around 70 guideline entries listed on PubMed and, by 2012, this number grew to over 7500. 4 Within England (and for some guidelines, Wales, Scotland, and Northern Ireland), these guidelines are provided by the United Kingdom (UK) National Institute for Health and Care Excellence (NICE), which has the overarching aim of providing ‘gold standard’ cost effective guidance to optimize healthcare within the UK National Health Service (NHS). There are many stages involved in the process of NICE guideline development: topic choice, scope, evidence review, and consultation are all key steps. Critically, the recommendations from each NICE guideline are put together by subject specialists, practitioners, commissioners, and service users. NICE was first established in 1999, as the National Institute of Clinical Excellence; as more focus was placed on guideline utilization, the scope of NICE was later broadened to include guidance around public health and social care. 5
While the benefits of clinical practice guidelines have been well documented, 6 their utility has also been questioned, with some authors stating we should move away from a guideline-driven approach and embrace shared decision-making to ensure patients are cared for and considered as individuals. 7 Indeed, since the modern age of guidelines first began in the early 1990s the world has changed: people are now living longer, and the number of people living with multiple coexisting long-term conditions is increasing.8–10 In response to these challenges, in addition to disease-specific guidelines, NICE has recently developed a guideline specifically for multimorbidity. 11
In terms of prescribing medication, which many guidelines are centred upon, there are a number of challenges for older people: physiological changes brought about through the normal ageing process, as well as diseases associated with ageing (e.g. reduced hepatic blood flow, or an altered lean body mass) can affect the absorption, distribution, metabolism and excretion properties of a drug. 12 These challenges are further compounded in multimorbidity, and life-limiting illness situations, where older people are frequently prescribed multiple medications, resulting in polypharmacy.13,14 Older people are therefore more likely to experience drug–drug interactions and adverse drug reactions, which could lead to potential harms.15,16 Additionally, in people with a life-limiting disease (typically with a prognosis of less than 12 months), patient goals of care can change from extending life and preventing disease to treating symptoms and optimizing quality of life. As such, the risk–benefit ratio of medications may often change for an individual patient. It is critical that any clinical practice guideline advocating the prescribing of medication in older people reflects these challenges, and also explain how, and when, such medication should be reduced or stopped when it is no longer needed. At present, despite the large number of clinical practice guidelines used to inform daily medical practice, it is unclear how such guidelines account for the challenges of ageing, multimorbidity and life-limiting illness in their treatment recommendations.17,18
This work, therefore, aimed to (1) apply disease-specific NICE clinical practice guidelines to a hypothetical older patient with multimorbidity and life-limiting illness; (2) consider how treatment choices could be influenced by NICE guidance specifically related to multimorbidity; and, (3) ascertain if such clinical practice guidelines describe how and when medication should be reviewed, reduced and stopped.
Method
Hypothetical patient
A hypothetical older female patient with stage IIIA non-small cell lung cancer was developed with the following long-term conditions: type 2 diabetes, hypertension, depression, and osteoarthritis. The hypothetical patient was aged 80 years, on the basis of the average female life expectancy in the UK being 82 years. 19 In addition, the 5-year survival rate for stage IIIA non-small cell lung cancer is <10% to represent a patient with a clear limited life expectancy. 20 Lung cancer was chosen as the life-limiting illness in our hypothetical patient because, in the developed world, it is the most common cause of cancer death for males and females. 21 The rationale for the choice of chronic conditions was based upon common conditions reported in the General Lifestyle Survey, and those included in the 2016 Quality Outcomes Framework. 22 A total of four long-term conditions were chosen, based upon the average number of long-term conditions reported in older people.23–25
The patient’s initial parameters were set to the following to represent typical readings in an older person:
Blood pressure: 160/90 mmHg
Blood glucose: 14.9 mmol/l
HbA1c: 97 mmol/mol
QRISK2score: 42.2%
The following disease-specific NICE guidelines were used to inform treatment recommendations for the hypothetical patient in view of the long-term conditions:
Hypertension in adults: diagnosis and management 26
Cardiovascular disease: risk assessment and reduction, including lipid modification 27
Type 2 diabetes in adults: management 28
Depression in adults: recognition and management 29
Osteoarthritis: care and management 30
These specific guidelines were identified by searching the NICE guideline registry (https://www.nice.org.uk/guidance) for the four long-term conditions in the hypothetical patient and selecting the most recent, and appropriate ones.
In addition to the disease-specific treatment guidelines, the treatment choices were also considered according to the Lung cancer: diagnosis and managementNICE guideline 31 to ascertain whether there was any specific advice in relation the treatment of long-term conditions in people with lung cancer. The Multimorbidity: clinical assessment and management NICE guideline 11 was also considered to ascertain how treatment choices could be influenced in our hypothetical patient. To mimic the stepped approach to patient care, as employed by clinical practice guidelines, each guideline was considered according to the following models: a new patient model (when the patient initially presents to the clinician without using any medication); a treatment-resistant model (when the first-line therapy fails to control symptoms), and a last-line model (the last step of therapy, where previous medications have failed to control symptoms). It was assumed that the hypothetical patient was adherent to treatment when moving between models. We only considered pharmacological intervention and did not consider additional monitoring requirements or lifestyle changes in our analysis (e.g. blood glucose testing or diet control).
Quality appraisal and data extraction
Each guideline was applied to the hypothetical patient to determine what medication, if any, should be initiated in view of the patient’s long-term conditions. The application of each guideline was done independently (by DO and AP) and, in the case of disagreement, consensus was reached with AT (senior author). Each guideline was independently data extracted, and appraised (DO and AP) according to the criteria previously proposed by Boyd and colleagues, as outlined below; 32 any disagreements in the appraisal were again reached by consensus (AT).
Do the guidelines address treatment for the type of patient? (Older person, person with multimorbidity)
Is the quality of evidence discussed for older adults/adults with multimorbidity?
Is time needed to treat to benefit from treatment in the context of life expectancy discussed?
In addition, to the above questions, a further three questions were posed to determine if the guidelines considered deprescribing in their recommendations; we defined deprescribing as ‘the process of withdrawal of an inappropriate medication, supervised by a health professional with the goal of managing polypharmacy and improving outcomes’, as proposed by Reeve and colleagues. 33
Do the guidelines address deprescribing/stopping treatment?
Do the guidelines address when to deprescribe?
Do the guidelines address the process of deprescribing?
Assessment of medication complexity
The medication regimen complexity index (MRCI) was used to quantify the complexity of each medication regimen, based on the dosing requirements during a typical day. The MRCI is a validated 65-instrument tool, and has three components: 34
Dosage form, accounts for dosage form, and routes of administration.
Dosage frequency, accounts for how often the medication is to be administered during a typical day.
Additional instructions, accounts for specific instructions a patient must follow when taking the medication (e.g. taking the medication with food).
The sum of the three scores form an overall score of medication complexity. Higher scores on the MRCI have been associated with reduced medication adherence levels, 35 unplanned hospital admissions, 36 and increased mortality. 37
Results
Medication regimen in the hypothetical patient according to disease-specific guidelines
Overall, five of the disease-specific guidelines recommended the initiation of medication in the hypothetical patient; if the initial treatment approach was unsuccessful, each guideline advocated the use of more medication. One guideline, the Cardiovascular Disease guideline, 27 specifically stated that the decision to start statin therapy should be made after an informed discussion between the clinician and the patient about the risks and benefits of treatment, taking into account additional factors including informed patient preference, comorbidities, polypharmacy and life expectancy. Given the limited life expectancy of our hypothetical patient, we interpreted the guideline that it would not be appropriate to initiate statin therapy. The lung cancer guidance 31 only contained information on approaches, in terms of chemotherapy selection, to manage the lung cancer, no consideration or reference was made to how long-term conditions should be managed in a patient with lung cancer.
In the new patient model, a total of 4 separate medications, totalling 9 dosage units per day, would be initiated to manage the four long-term conditions; this increased to 7 separate medications, totalling 15 dosage units per day in the treatment-resistant model; in the last-line model, this further increased to 12 separate medications, totalling 20 dosage units per day. The majority of the medications were taken to treat the symptoms associated with the long-term condition (e.g. a selective serotonin reuptake inhibitor to treat the symptoms of depression), or to prevent complications occurring in the future (e.g. using amlodipine to reduce blood pressure to reduce the risk of a myocardial infarction or ischaemic stroke). One medication, omeprazole, was initiated to reduce the gastrointestinal (GI) adverse effects associated with the ibuprofen, this was considered an important intervention, especially in view of the coadministration of sertraline, and the increased risk of bleeding. The initiation of gastroprotection was specifically recommended in the depression guideline, 29 and also in the osteoarthritis guideline 29 when systemic nonsteroidal anti-inflammatory drugs (NSAIDs) are indicated. For the different models of treatment, the MRCI scores increased alongside the different phases of treatment, showing that as treatment is stepped up, the complexity of the medication regimen also increases (Table 1).
Medication summary for the three treatment models in the hypothetical patient, based upon disease-specific NICE guidelines.
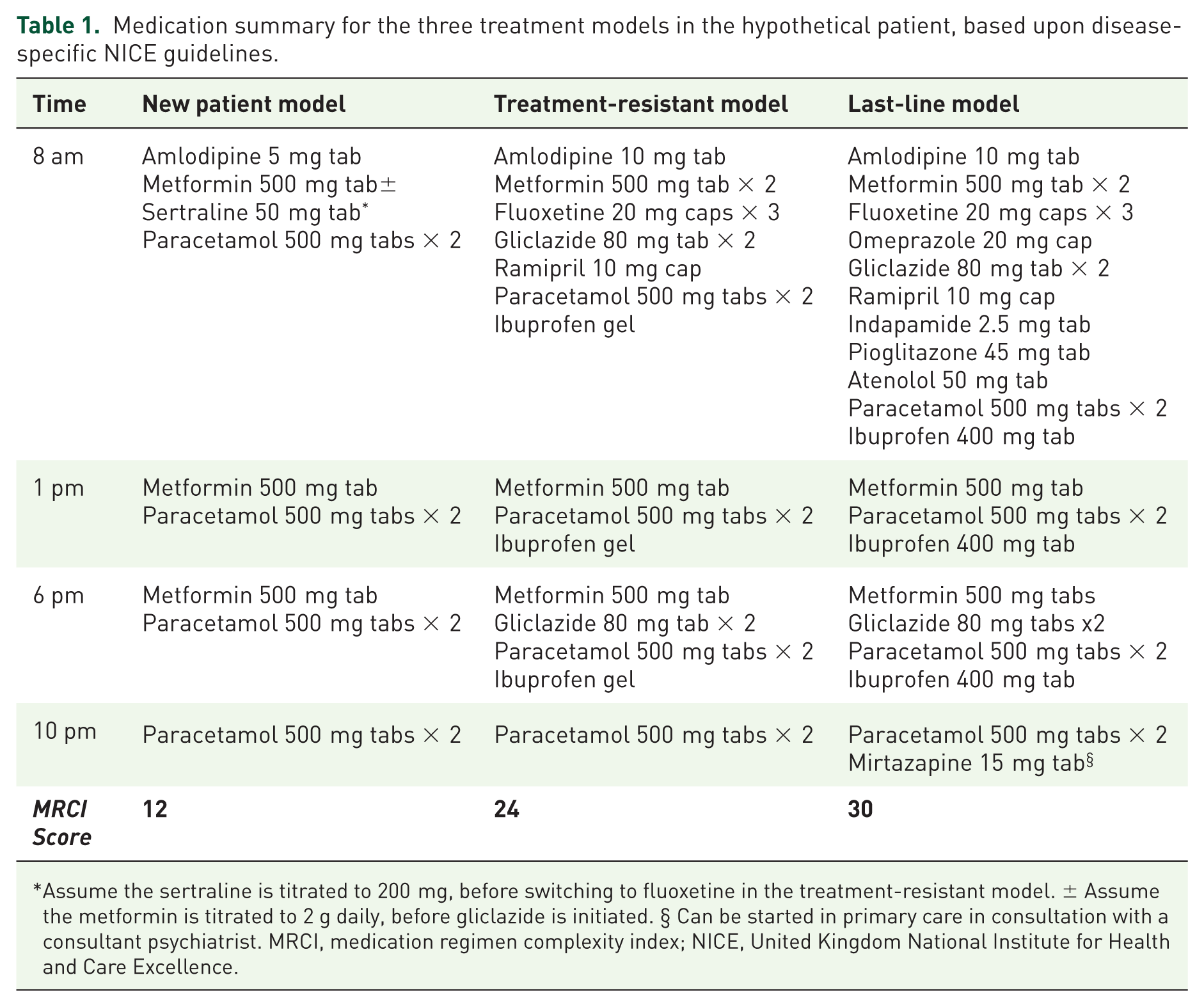
Assume the sertraline is titrated to 200 mg, before switching to fluoxetine in the treatment-resistant model. ± Assume the metformin is titrated to 2 g daily, before gliclazide is initiated. § Can be started in primary care in consultation with a consultant psychiatrist. MRCI, medication regimen complexity index; NICE, United Kingdom National Institute for Health and Care Excellence.
Assessment of disease-specific NICE guidelines
All of the disease-specific NICE guidelines used for our hypothetical patient (hypertension, depression, lipid modification, osteoarthritis and type 2 diabetes) contained treatment recommendations for older people, as outlined in Table 2. The guidelines concerning lipid modification and type 2 diabetes considered limited life expectancy of the patient in their treatment recommendations, although the time until benefit of treatment was not considered in depth: for the lipid modification guideline, this consideration impacted on the decision to start the statin, while for the type 2 diabetes guideline, it increased the threshold to start or add in additional medication, by creating more conservative HbA1c targets. Comorbidity was mentioned briefly in the guidelines for hypertension 26 and lipid modification, 27 while the term multimorbidity was outlined within the type 2 diabetes guideline, as part of an individualized care approach: 28 unfortunately, there were no cases where specific treatment recommendations were considered according to these terms. In our hypothetical patient, we did initiate medication (and also move between the different treatment stages by initiating additional medication) to manage the diabetes, although by creating more conservative HbA1c targets, it is possible that medication would be started in a lower proportion of older people; we also accept that depending on our Hb1AC targets, and the patients preferences, it may have been appropriate to not initiate therapy, or add in additional medications. The other NICE guidelines26,29–31 did not consider treatment recommendations in the context of (limited) life expectancy, this included guidelines for the management of chronic disease (depression, hypertension), and the specific guidance for the treatment of lung cancer. Furthermore, none of the disease-specific guidelines addressed the deprescribing of medication in their recommendations; this included when medication should be reviewed, reduced or stopped, or the process for how this could be achieved.
Examples of specific treatment recommendations from disease-specific NICE guidelines in relation to older people.
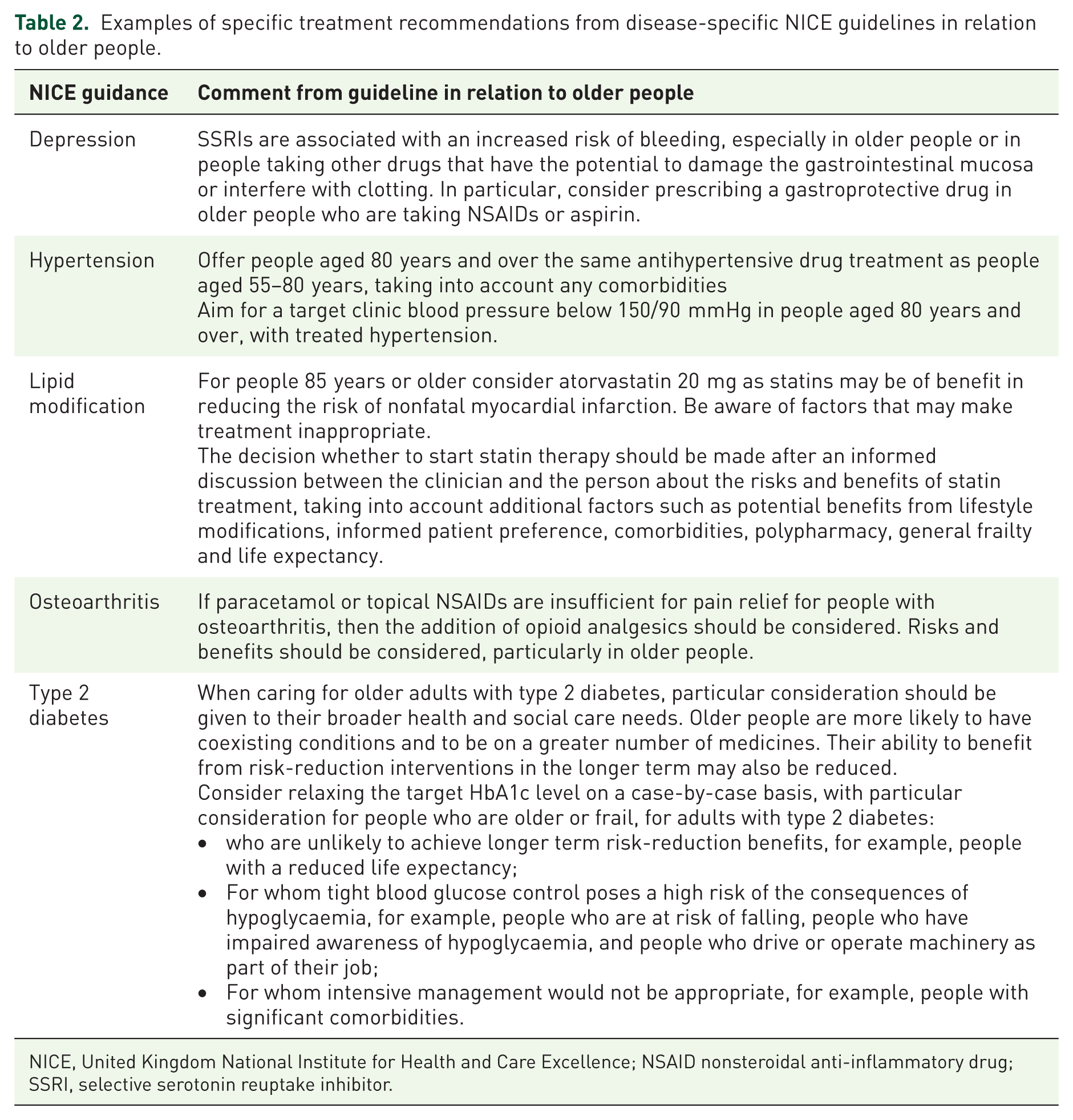
NICE, United Kingdom National Institute for Health and Care Excellence; NSAID nonsteroidal anti-inflammatory drug; SSRI, selective serotonin reuptake inhibitor.
Assessment of multimorbidity NICE guideline
The Multimorbidity: clinical assessment and management NICE guideline focused on how to optimize care for people with multimorbidity, and in stark contrast with the disease-specific NICE guidelines, only outlined one medication-specific recommendation, which related to bisphosphonate use (recommendation 1.6.16, page 12 11 ). The remaining recommendations were overarching principles of care, and included promoting shared decisions, and the importance of considering patients’ goals, values and priorities in treatment decisions. As the recommendations were overarching in nature, rather than medication specific, the quality of evidence for people with multimorbidity was not discussed or outlined. Similarly, although there was reference to people with life-limiting illness in these overarching principles of care, there was no discussion of the quality of evidence in this regard. Several of the overarching principles were pertinent to treatment considerations in our hypothetical patient, as outlined in Table 3. If our hypothetical patient’s priorities of care were focused on acute symptom management, in view of the principles of care stated in this guideline, and the relatively long time to benefit of treatment, it could be interpreted that the medication used to treat hypertension, and diabetes would not have been initiated, although the guideline did not state this specifically. The multimorbidity guideline did not specifically recommend when or how to deprescribe medications, instead only providing a recommendation to discuss whether individuals wish to continue their medications. In addition, the NICE multimorbidity guideline did not make any reference to the five disease-specific NICE guidelines that were utilized by our hypothetical patient; nor was there any reference to the multimorbidity guideline in the disease-specific NICE guidelines, although we acknowledge the multimorbidity guideline was published after some of the disease-specific guidelines used in our hypothetical patient.
Principles of care from outlined in the NICE multimorbidity guideline in relation to older people.

NICE, United Kingdom National Institute for Health and Care Excellence.
Discussion
This is the first study to explore the appropriateness of UK guidelines regarding treatment recommendations for older people with multimorbidity and limited life expectancy. We have identified four key findings that are of importance to healthcare practitioners and policy makers: (1) disease-specific guidelines routinely advocate the initiation of medication to manage chronic conditions in older people, with medication regimens becoming increasing complex through the different steps of care; (2) although there is often information regarding the initiation of treatment in older people in disease-specific guidelines, there is often a lack of detailed information regarding specific treatment recommendations for older people with life-limiting illness and multimorbidity; (3) in contrast with the disease-specific guidelines, guidance specifically related to multimorbidity contains overarching principles that seek to optimize care, although such guidelines fall short of stating specific treatment recommendations (prescribing or deprescribing) regarding medication; and, (4), current guidelines advocate the initiation of medication to manage long-term conditions, without considering when and how these medications should be reviewed and stopped (deprescribed); this causes polypharmacy, which has potential to cause adverse drug events. Overall, when managing a complex older patient, the interplay between the disease-specific guidelines and the multimorbidity guideline is analogous to driving a car: the disease-specific guidelines act as an accelerator pedal regarding treatment (by focusing on initiating medication), while the multimorbidity guideline acts as the break (by focusing on treatment optimization). When these guidelines are used in tandem, a balance may be reached in regard to appropriate medication use; however, when disease-specific guidelines are used in isolation, there is potential for the car to speed, with more focus on initiating (potentially inappropriate) medication. Given the need to keep disease-specific guidelines concise (to optimize user friendliness) it may not be appropriate to include an in-depth discussion of multimorbidity and limited life expectancy in all disease-specific guidelines, however, at a minimum they should refer to and signpost people to the multimorbidity guideline. These are potentially important and significant findings given the ageing population and the exponential increase in the use of clinical practice guidelines to support clinicians in their decision-making.
While this is the first study to examine the appropriateness of clinical practice guidelines in a UK context, other studies have explored the appropriateness of clinical practice guidelines in other settings. A seminal study by Boyd and colleagues evaluated the applicability of United States clinical practice guidelines to the care of older individuals with several comorbid diseases. 32 In agreement with our study, Boyd and colleagues showed that most disease-specific clinical practice guidelines do not modify or discuss the applicability of their recommendations for older people with multiple comorbidities. They also constructed a hypothetical patient with chronic obstructive pulmonary disease, type 2 diabetes, osteoporosis, hypertension and osteoarthritis, and showed that if the relevant clinical practice guidelines were followed, the patient would be prescribed 12 medications, costing over $400 per month. Several other studies reviewing disease-specific guidelines internationally have also found that there is a lack of information and guidance for managing older adults with multimorbidity, especially in relation to treatment in the presence of discordant comorbidities.38,39
The lack of appropriateness of disease-specific practice guidelines for older adults with multiple coexisting long-term conditions may not be directly attributed to guideline developers. 40 Protocols and guidance on clinical practice guideline development requires the use of high quality evidence, which in the case of treating long-term conditions is usually randomized controlled clinical trials. 41 It has been shown that older people and those with multimorbidity are often excluded from clinical trials, meaning that the results may not be applicable to the general patient population.42,43 In cases where older people are included in clinical trials, the results are often not reported or statistically interpreted according to age, although this limitation is acknowledged in some guidelines. 44 For example, the Heart Protection Study, a key study assessing the effectiveness of statin therapy in 20,536 high-risk individuals, only recruited people aged between 40 to 80 years. 45 As such, NICE specifically acknowledge in their guidance on cardiovascular disease that assessing the effectiveness of statin therapy in older people should be a research priority. 26 In contrast, there have been studies that explore the effectiveness of antihypertensive therapy in older people: 46 and as a consequence of this research, NICE specifically consider initiating antihypertensive medication according to age, with a higher threshold of treatment recommended for people aged over 80 years. 26 In keeping with this approach, the NICE multimorbidity guideline, which seeks to optimize care for adults through a series of recommendations, uses terms such as ‘taking account of multimorbidity in tailoring an approach to care’, and ‘how to identify people who may benefit’. 11 The approach of dealing with multimorbidity as a separate entity is a significant step forward since the work by Boyd and colleagues, 32 but it is clear that more could be done to integrate these overarching principles of care into more practical and specific recommendations for disease-specific treatment guidelines.
Guidelines often contain a disclaimer and may recommend shared decision-making between the healthcare professional and the patient to decide whether (or not) a medication should be initiated. We note, for example, that each of the NICE guidelines applied to our hypothetical patient stated:
When exercising their judgement, professionals and practitioners are expected to take this guideline fully into account, alongside the individual needs, preferences and values of their patients or the people using their service. It is not mandatory to apply the recommendations, and the guideline does not override the responsibility to make decisions appropriate to the circumstances of the individual, in consultation with them and their families and carers or guardian.
While this is encouraging, medical practitioners have reported a fear of litigation if they do not follow guidelines. 47 For example, if a guideline recommendation for treatment is not followed, and harm occurs to a patient, they (or their family) may seek claims of negligence, and litigation, causing significant stress to the prescribing clinician. 48 Additionally, some health care systems include a ‘pay for performance’ system whereby clinicians’ reimbursement for service may be influenced by their adherence to standards of care for single diseases. These standards of care are often extracted from guidelines (despite the guidelines not being developed for this purpose), and such systems disincentivize managing complex issues in people living with multiple coexisting long-term conditions. 32 The statement regarding statin therapy in the NICE Cardiovascular Disease guideline that treatment initiation should also consider additional factors, such as informed patient preference, polypharmacy and life expectancy, is helpful in this regard, and this was main reason why statin therapy was not initiated in our hypothetical patient. Recent trial evidence shows that statins can be discontinued in patients with limited life expectancy without causing negative outcomes for patients; 49 however, preventive medications, including statins, are routinely inappropriately prescribed to older patients with limited life expectancy. 50
In terms of once treatment was initiated, it was clear from our findings that none of the NICE clinical practice guidelines addressed how and when treatment should be stopped or discontinued, so-called deprescribing. This finding is timely given the findings of Markovitz and colleagues who showed that guidelines for cardiovascular disease and diabetes primarily recommend intensification, rather than deintensification, of therapy. 17 Similarly, Jansen and colleagues, who reviewed cardiovascular guidelines, found that none included any information about when or how to stop medications, with one even encouraging against discontinuation. 51 In recent years, a great deal of attention has been given to the concept ‘deprescribing’: the process of which has recently been outlined, 52 and studies suggest that deprescribing initiatives aimed at older people are feasible and safe and may improve health outcomes and reduce mortality. 53 In view of this and general practitioner (GP) reports of a lack of guidelines as a significant barrier to deprescribing, there have been a number of medication-specific deprescribing guidelines developed that consider when and how to reduce and stop medication.54–56 These guidelines have been developed following the robust standards in which clinical practice guidelines are developed. 57 Indeed, rather than separate out the process of initiating, monitoring, and deprescribing medication, we believe in order for the recent deprescribing guidelines to gain maximum utility, it is important that these deprescribing recommendations, or the principles of them, are included within disease-specific clinical practice guidelines; or as a minimum, from a UK perspective, it would be helpful if NICE were to develop a separate deprescribing guideline, in a similar approach to the multimorbidity guideline, and make reference to it in disease-specific guidelines. We acknowledge that it is challenging, especially considering the current models of care in general practice where GPs typically have 12 minutes for a patient consultation. One possible solution to dealing with complex older patients taking multiple medications is the recent introduction of clinical pharmacists working in general practice as part of the primary care team as part of a pilot by the NHS England. 58
This work has several limitations. Firstly, we only considered guidelines underpinning treatment recommendations for four long-term conditions; it is possible that guidelines for other conditions include specific treatment recommendations for deprescribing medication in older people and, as such, may not be representative of all NICE guidelines. Secondly, we only considered the brief NICE guideline; the full supporting evidence used to formulate the guideline recommendations were not considered, even though they were available on the NICE website. The rationale for this approach was that each brief guideline contains circa 50 pages, while the full guideline with all of the supporting evidence is circa 500 pages. We therefore believe it is unlikely that a clinician would have the time to read a 500-page guideline to inform their routine prescribing practice. However, it is possible that the supporting evidence of each of the guidelines contains information about prescribing medication to older people with multiple coexisting long-term conditions. We therefore recommend that our findings are interpreted in view of these limitations.
Conclusion
In a UK context, disease-specific clinical practice guidelines routinely advocate the initiation of medication to manage long-term conditions, with medication regimens becoming increasingly complex through the different steps of care. There is often a lack of information regarding specific treatment recommendations for older people with life-limiting illness and multimorbidity. While guidelines frequently explain how and when a medication should be initiated, there is often no information concerning when the medications should be reduced or stopped. Overall, this approach increases the potential for polypharmacy, the development of drug-related events, and possible harm to patients. In view of this, it is important for future guideline development for specific conditions to holistically consider all aspects of medication utilization, including how and when the medication should be reviewed, reduced, and stopped.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest statement
The authors declare that there is no conflict of interest.
