Abstract
With the increasing prevalence of type 2 diabetes mellitus (T2DM) in women of childbearing age, prescribing antidiabetic medications in first-trimester pregnancy is becoming more common. Metformin treatment during this time is usually avoided in countries with well-resourced healthcare. This is based on historical concerns about safety to the foetus and the widespread availability of insulin. However, there is now increasing interest in the potential benefits of metformin in pregnant women with T2DM. In this commentary, the main evidence supporting metformin safety in pregnancy is summarized, with an emphasis on the first trimester. Based on a structured literature search, the recent randomized controlled trials comparing metformin and insulin are reviewed. We then show that prescribing advice for metformin in pregnancy is inconsistent and product information/package inserts (PI) are universally out of date. This causes confusion and pushes some women and their clinicians to change from metformin to insulin. The potential advantages of metformin in pregnant women with T2DM are then discussed, including oral dosing and improved acceptability, lower resource utilization and cost, decreased insulin requirements, less maternal weight gain and less risk of maternal and neonatal hypoglycaemia. The conclusion is that metformin is a cheap and efficacious antidiabetic medication for many pregnant women with T2DM, with reasonable evidence for safety. Drug information resources should be updated so that metformin can be considered more broadly in women with T2DM who present for antenatal care.
Background
The increasing prevalence of type 2 diabetes mellitus (T2DM) in women of childbearing age has led to greater use of antidiabetic medications in the first trimester of pregnancy.
1
T2DM in pregnancy includes women with pre-existing T2DM and women diagnosed in the first trimester with ‘diabetes mellitus in pregnancy’ or ‘overt diabetes’, in which glucose intolerance does not resolve in the postpartum period [based on the International Association of Diabetes and Pregnancy Study Groups diagnostic criteria endorsed by the World Health Organization; note that ‘gestational diabetes mellitus’ (GDM) is diagnosed in the second or third trimester and resolves after birth].2,3 Importantly, the first trimester is the most sensitive time in pregnancy for drug exposure because organogenesis is taking place. Ideally, all xenobiotics (from the Greek ‘xenos’ = foreigner and ‘bios’ = life), including recreational drugs, prescription drugs and complementary and alternative medicines, should be avoided. However, the risks to the foetus and mother of untreated disease are often greater than the risks of drug treatment.
4
This is true for T2DM, in which uncontrolled hyperglycaemia
Metformin is widely regarded as the first-line treatment of T2DM outside of pregnancy. In contrast, the most used treatment of T2DM in pregnancy is insulin, largely because clinical experience is so extensive, its risks are well known (e.g. maternal weight gain and maternal and neonatal hypoglycaemia), and because doses can be titrated rapidly for ongoing glycaemic control. A conservative approach is usually taken with metformin in countries with well-resourced healthcare – it is either avoided in women attempting to become pregnant or ceased and replaced by insulin when pregnancy is confirmed. This stems from historical uncertainty about the safety of metformin to the foetus and the widespread use of insulin for GDM. However, there is increasing clinical interest in the potential benefits of metformin in pregnant women with T2DM. In this commentary, we review the historical safety concerns with metformin and the main evidence regarding safety and efficacy in these women, including the recent randomized controlled trials (RCTs) comparing metformin with insulin (gold standard). Finally, the potential benefits of metformin in women with T2DM who present for antenatal care are considered.
Historical safety concerns
The limited use of metformin in early pregnancy is due to historical safety concerns. These are based on uncertainty about potential risks to the foetus from transfer via the placenta,
6
broader concerns about oral antidiabetic medicines causing birth defects and neonatal hypoglycaemia,
7
questionable efficacy in maintaining an adequate postprandial glucose response
8
and unknown long-term effects on offspring. Added to this are observational data suggesting adverse pregnancy outcomes, in particular increased rates of perinatal mortality and preeclampsia, and decreased mean birth rate.9–11 However, these concerns and the validity of the observational data have been questioned.12–17 A major limitation of these observational studies is poor matching between groups of women on metformin and those not, with the metformin group in one study
9
mostly treated in the third trimester and having increased risk factors for preeclampsia, such as obesity and nonadherence to treatment. In the study by Piacquadio and colleagues
7
showing an association between oral antidiabetic medications and birth defects, biguanides (two phenformin and one metformin) were analysed together with sulphonylureas (
Evidence supporting safety
Several reports by Coetzee and colleagues from the Peninsula Maternal and Neonatal Service in Cape Town, South Africa describe metformin use in pregnancy over a period of >40 years.17–22 In one retrospective analysis of first-trimester exposure, after accounting for poor glycaemic control in later pregnancy, there was no greater liability to birth defects or perinatal mortality in women on metformin compared with those who received insulin or no medication.
20
In a larger retrospective analysis of 379 pregnancies over 10 years (1991–2000), the overall rate of perinatal mortality, but not birth defects, was higher in women treated with oral antidiabetic medications (metformin, glibenclamide, and metformin + glibenclamide analysed together as one group) compared with those converted early to insulin or those treated with insulin only. The perinatal mortality rates per 100 births were 125, 28 and 33, respectively (
During the 1990s interest grew in using metformin for insulin-resistant states other than T2DM. Metformin was shown to treat or prevent clinical problems associated with polycystic ovarian syndrome (PCOS), including oligomenorrhea, hirsutism and obesity, and to induce ovulation in infertile women. 23 In short, metformin does not increase the risks of major birth defects or abortion in women affected by PCOS and treated during the first trimester.24–26 Furthermore, maternal and neonatal outcomes in the primary studies have been examined in systematic reviews and meta-analyses, and these support the safety of metformin exposure in the first trimester.24–28
Similarly, for third trimester metformin exposure, the MiG trial showed that metformin can be used safely to treat GDM 29 without adverse effects on children to at least two years of age, including blood pressure 30 and neurodevelopmental outcomes 31 (safety data beyond this age are starting to be prepared in abstract form at the time of this commentary). 32 This is consistent with several systematic reviews and meta-analyses that found metformin treatment of GDM does not increase adverse maternal or neonatal outcomes,27,28 although other similar analyses have detected a small but statistically significant increase in the rate of pre-term birth.33–35
Taken together with the South African experience, these studies vindicate the expert commentaries in the mid-2000s re-evaluating metformin use in pregnancy and calling into question the putative link with adverse outcomes described in the previous section.12–17 There are also newer safety data in the RCTs described in the next section. The lack of adverse effects in these studies is reassuring, recognizing that most were not powered to detect significant differences between treatments, so the outcomes represent weaker evidence of no harm with metformin.
Recent studies of metformin for T2DM in pregnancy
Despite years of experience with metformin in poorly resourced countries, 17 only recently have RCTs been published that compare metformin with insulin for T2DM in pregnancy. To identify these, a structured search of the electronic databases MEDLINE, EMBASE, CENTRAL (Cochrane Central Registry of Controlled Trials) and Google Scholar was conducted using, in combination with ‘metformin’, the search terms ‘pregnancy’, ‘type 2 diabetes mellitus’, ‘type 2 diabetes in pregnancy’, ‘congenital abnormalities’, ‘birth defects’, ‘pregnancy outcomes’ and ‘HbA1c’. Reference lists from relevant papers were also used to identify studies suitable for review. Five RCTs and two prospective cohort studies were found, as described below.36–42
Waheed and colleagues 40
This study was conducted in Pakistan and enrolled 64 pregnant women with diabetes who were beyond 14 weeks’ gestation. 40 A combined efficacy outcome was defined as fasting glucose concentration between 3.5 and 5.5 mmol/L after 1 month of treatment and at term, random glucose concentration below 7.8 mmol/L after 1 month of treatment and at term, and haemoglobin A1c (HbA1c) below 6.1% ‘post-treatment’. Overall, there was no difference between metformin and insulin in controlling diabetes, with exactly 27/34 (79.4%) in each group meeting the combined efficacy endpoint. This paper failed to explain how patients with poor glycaemic control were managed and did not show baseline demographics, including prior diabetes treatment, gestational week at enrolment and classification of diabetes (T2DM in pregnancy or GDM). Pregnancy outcomes were also not reported.
Hickman and colleagues 38
In this North American study of 28 women, 15 of whom had T2DM prior to conception, the primary outcome was mean fasting blood glucose concentration at 18–20, 28–30 and 36–38 weeks’ gestation.
38
There was no statistical difference in fasting glucose concentrations at all time-points. Haemoglobin A1c values were also similar between the treatment groups in each trimester. In total, 43% of women randomized to metformin required insulin supplementation to achieve glycaemic control. Importantly, all women who required supplementary insulin remained on maximum doses of metformin until delivery and had fewer subjective (0
Refuerzo and colleagues 39
Another small North American study randomized 8 women to receive metformin and 11 women to receive insulin.
39
All women had T2DM prior to conception and the primary outcome was HbA1c <7% at delivery. This was achieved in all the women randomized to metformin and 9 of 11 women in the insulin group [RR 0.79; 95% confidence interval (CI): 0.64–1.10]. There was no statistical difference (defined as
Beyuo and colleagues 37
This study of 104 pregnant Ghanaian women randomized those with T2DM (called ‘pre-gestational diabetes’ in the paper) or GDM to either metformin or insulin between 20 and 30 weeks’ gestation.
37
The proportions of women with T2DM in each group were 25.6% and 42.5%, respectively. Two-hour postprandial blood glucose was the primary outcome. This was significantly lower in the metformin group and only 2/52 (4%) required supplementary insulin. There were no differences in the rates of miscarriage, stillbirth, caesarean section or early neonatal death, whereas the number of babies with macrosomia (defined as >4.0 kg) was greater in mothers treated with metformin ± insulin (
Ainuddin and colleagues 36
The final RCT from the structured literature search was another study from Pakistan.
36
This was a randomized, open-label, parallel-group study comparing metformin with insulin in 206 women with T2DM in pregnancy, of which about two-thirds had pre-existing T2DM controlled by diet alone or by a combination of diet and oral antidiabetic medication. In contrast to the other RCTs, the primary outcomes were adverse events: perinatal mortality, neonatal hypoglycaemia requiring intravenous dextrose therapy, and neonatal intensive care unit (NICU) admissions >24 h. The mean gestational time at enrolment was 10 weeks, so this is the only RCT to commence predominantly in the first trimester. Overall, 84% of patients randomized to metformin required add-on insulin. Thus, three groups were compared in the final analyses: metformin alone, metformin + insulin and insulin alone. There were no neonatal deaths and glycaemic profiles were similar in all groups. Neonatal hypoglycaemia, NICU admissions >24 h and pregnancy-induced hypertension were significantly less in the metformin groups, although small-for-date babies were more common (
Other relevant studies
Rai and colleagues conducted a prospective cohort study in Indian women with GDM or T2DM, with metformin use in one obstetric unit compared with insulin use in another. 42 Glycaemic control was better in the metformin group over the duration of gestation and there were no differences between groups in birth weight or mean gestational age. However, of the 60 women enrolled, only eight had T2DM – two received metformin and six received insulin. In another prospective study, this time in Egypt, one group of pregnant women with diabetes who showed insulin resistance had metformin added, while a parallel group of similar women had their insulin dose increased. 41 In the group to which metformin was added, approximately one-third achieved maternal glycaemic control with a daily metformin dose of 1500 mg, another one-third required 2000 mg metformin per day, and the remaining one-third remained on 2000 mg of metformin per day but required an increase in insulin dose.
Limitations of recent studies
A few limitations of these studies warrant consideration. Except for Ainuddin and colleagues, the RCTs were not designed to investigate differences in adverse effects between treatments. Second, the studies were open-label, which is almost unavoidable given their locations. Third, the North American studies were underpowered to detect safety signals due to recruitment difficulties (Hickman and colleagues had 15 participants with T2DM leading into pregnancy and Refuerzo and colleagues had 21).38,39 Fourth, the proportion of women treated with metformin prior to enrolment was not clear and the gestational age at enrolment ranged between 4 and 30 weeks’ gestation. Thus, the degree to which early pregnancies were exposed to metformin is not easily deduced. Despite these limitations, the trend towards favourable outcomes is consistent with the evidence in PCOS and GDM.
Further studies of metformin for T2DM in pregnancy are currently recruiting [ClinicalTrials.gov identifier: NCT00678080; NCT01353391]. This includes the MiTy trial, a multicentre, randomized, double-masked study that aims to enrol 500 participants (gestation 6–22 weeks) already on insulin to either metformin 1000 mg twice daily or placebo (1:1). 44 Pharmacoepidemiological studies are still required to investigate the long-term effects of metformin on offspring, such as the follow up of neurodevelopmental and metabolic function as children grow into adolescence and become adults. Using routinely collected health data is an attractive approach. 45
Prescribing advice
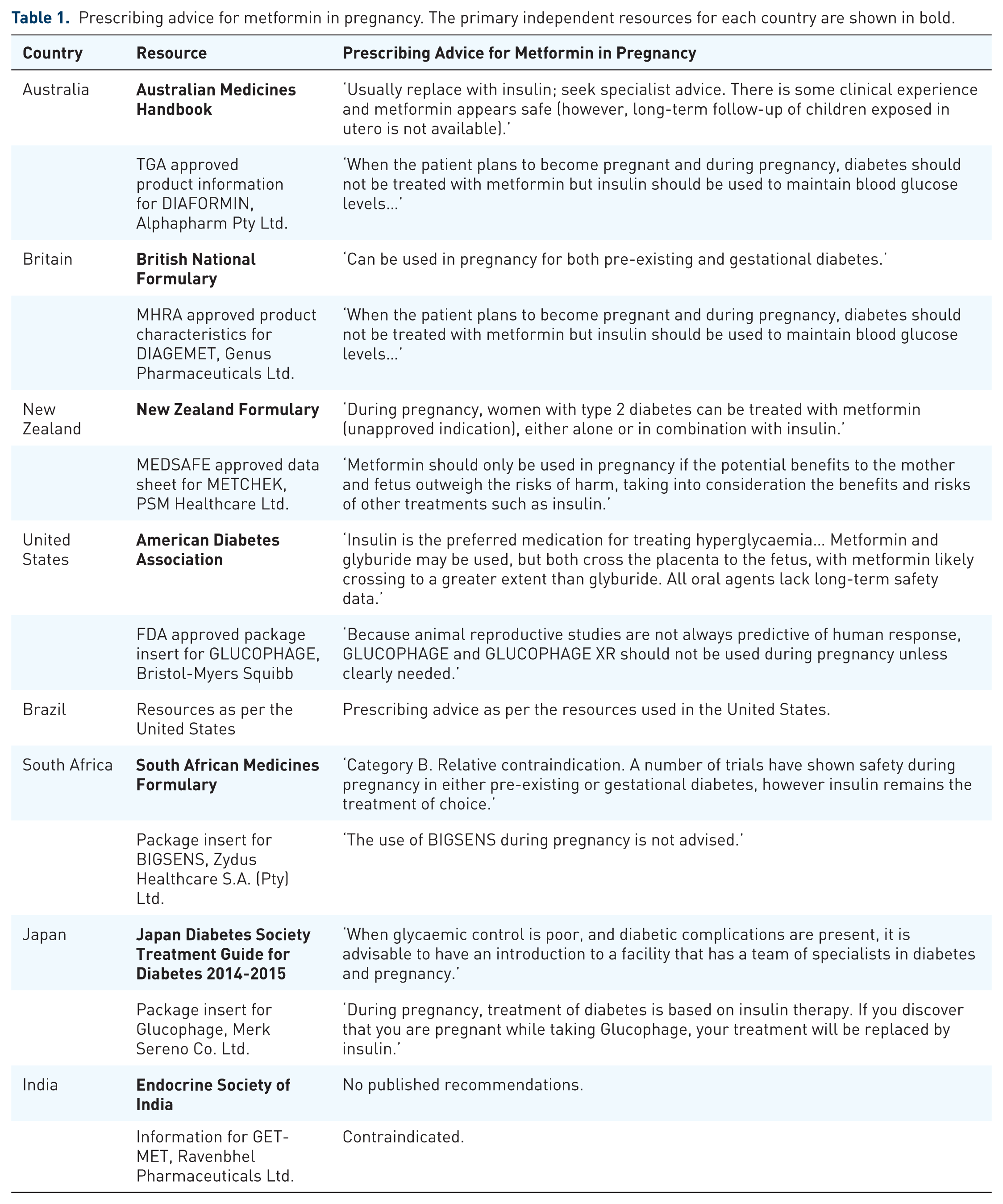
There are many drug information resources that provide clinicians with prescribing advice in pregnancy. These include independent national drug formularies and therapeutics information services, regulator-approved product information/package inserts (PI) from manufacturers, and guidelines from specialist societies and interest groups. Table 1 shows prescribing advice for metformin in pregnancy from different resources in selected countries. In recent years, a ‘softening’ of wording in some independent resources has allowed for greater prescriber interpretation and autonomy – for example, the national formularies of Britain and New Zealand now indicate that metformin can be used in T2DM in pregnancy (Table 1). This is not endorsed by specialist societies, such as the Australasian Diabetes in Pregnancy Society. 46 Importantly, all PI for metformin, independent of manufacturer or regulatory jurisdiction, appear to reflect the historical safety concerns, advising prescribers to avoid metformin and use insulin. This has important implications, since PI populates the commercial electronic prescribing software packages widely used in clinical practice, such as eMIMs. We previously showed that PI for off-patent drugs such as metformin are often at odds with current evidence and guidelines, probably because incentives for updating are absent and no single manufacturer retains ‘ownership’. 47 The drugs in pregnancy classifications for metformin also differ between countries – metformin is class C in Australia (‘Drugs which, owing to their pharmacological effects, have caused or may be suspected of causing harmful effects on the human foetus or neonate without causing malformations’) but class B in the United States (‘Animal reproduction studies have failed to demonstrate a risk to the foetus and there are no adequate and well-controlled studies in pregnant women’). 48 This highlights difficulties in the drugs in pregnancy classification system, which does not have a clear hierarchy of risk stratification. In mid-2015 the US Food and Drug Administration scrapped the letter categorizations and began implementation of the Pregnancy and Lactation Labeling Final Rule (PLLR). This system is mandatory for newly registered drugs after mid-2015, but for old drugs such as metformin it will be several years before the pregnancy letter categories are removed from PI and the PLLR classification system is applied. We believe that the inconsistent prescribing advice about metformin described in this section causes confusion and pushes some women and their clinicians to change from metformin to insulin.
Prescribing advice for metformin in pregnancy. The primary independent resources for each country are shown in bold.
Potential benefits of metformin
There are sound pharmacological and practical reasons to use metformin in T2DM in pregnancy. The potential benefits of metformin include the following.
Conclusion
Metformin is a cheap and effective antidiabetic medication for many pregnant women with T2DM. There is very little evidence for harm and reasonable evidence for tolerability, although long-term data are lacking. Recent studies, predominantly from countries with poorly resourced healthcare, provide encouraging efficacy results for metformin. Continuing or commencing metformin treatment for T2DM in pregnancy has potential advantages over switching to or starting insulin immediately. Prescribing advice for metformin in pregnancy is inconsistent and often reflects historical safety concerns based on uncertainty rather than evidence. Drug information resources should be updated so that metformin can be considered more broadly in women with T2DM who present for antenatal care.
Footnotes
Acknowledgements
Funding
This research received no specific grant from any funding agency in the public, commercial or not-for-profit sectors.
Conflict of interest statement
The authors declare that there is no conflict of interest.
