Abstract
Clozapine, a dibenzodiazepine developed in 1961, is a multireceptorial atypical antipsychotic approved for the treatment of resistant schizophrenia. Since its introduction, it has remained the drug of choice in treatment-resistant schizophrenia, despite a wide range of adverse effects, as it is a very effective drug in everyday clinical practice. However, clozapine is not considered as a top-of-the-line treatment because it may often be difficult for some patients to tolerate as some adverse effects can be particularly bothersome (i.e. sedation, weight gain, sialorrhea etc.) and it has some other potentially dangerous and life-threatening side effects (i.e. myocarditis, seizures, agranulocytosis or granulocytopenia, gastrointestinal hypomotility etc.). As poor treatment adherence in patients with resistant schizophrenia may increase the risk of a psychotic relapse, which may further lead to impaired social and cognitive functioning, psychiatric hospitalizations and increased treatment costs, clozapine adverse effects are a common reason for discontinuing this medication. Therefore, every effort should be made to monitor and minimize these adverse effects in order to improve their early detection and management. The aim of this paper is to briefly summarize and provide an update on major clozapine adverse effects, especially focusing on those that are severe and potentially life threatening, even if most of the latter are relatively uncommon.
Introduction
Schizophrenia is a chronic and debilitating disease affecting approximately 0.5% of the population. 1 Antipsychotics are the mainstay of treatment of such a severe disorder, but it has been widely demonstrated that between 20% and 60% of patients with schizophrenia do not respond sufficiently to conventional treatments. 2
Clozapine, a dibenzodiazepine developed in 1961, is a multireceptorial atypical antipsychotic approved for the treatment of resistant schizophrenia. 3 Since the pivotal study of Kane and colleagues 4 it has remained the drug of choice in treatment-resistant schizophrenia despite a wide range of adverse effects. 5 It has been demonstrated that clozapine is more effective than any other first-generation (FGA) or second-generation antipsychotics (SGA) in the treatment of resistant schizophrenia and it is also useful in other conditions6,7 (Table 1). It has been estimated that almost two thirds of those patients who do not respond adequately to treatment with FGAs or other SGAs may respond adequately to treatment with clozapine. 8 Undoubtedly, clozapine is a very effective drug in everyday clinical practice and many patients who tolerate it will experience remarkable symptom relief that is often protracted in time and gives them the opportunity to achieve a satisfactory quality of life. 9
Benefits of clozapine use in clinical practice (modified from De Berardis et al. 10 )

Clozapine treatment may decrease mortality from natural or disease-related causes in treatment-resistant schizophrenia
However, clozapine is not considered as a first-line treatment because it may often be difficult for some patients to tolerate, as some adverse effects may be particularly bothersome (i.e. sedation, weight gain, sialorrhea etc.) and some others are potentially dangerous and life threatening. 11 Specifically, the risk of agranulocytosis is one of the main reasons why clozapine is not a first-line agent for schizophrenia. 12
Moreover, clozapine may have several interactions with other drugs as it is metabolized by the hepatic cytochrome P450 (CYP) system. 13 Clozapine is transformed to norclozapine by CYP3A4 and 1A2 and to clozapine N-oxide by CYP3A4. 14 Nevertheless, CYP2C19 is also significant at clozapine therapeutic concentration (24%) while the influences of CYP2C9 (12%) and 2D6 (6%) are more modest. 15 CYP1A2 is the most significant form at a therapeutic concentration (30%), while CYP3A4 plays an important role at higher concentrations (37%) than at therapeutic concentrations (22%).16,17 All considered, blood-level monitoring of clozapine may be needed both when inhibitors (such as antifungals, oral contraceptives, fluvoxamine, ciprofloxacin, caffeine, disulfiram) or inducers (such as rifampicin, omeprazole, phenytoin, phenobarbital, tobacco smoke) of CYP1A2 and both inhibitors (such as cimetidine, erythromycin, clarithromycin etc.) or inducers (such as carbamazepine and rifampicin) of CYP13A4 are being used. 18 It is important to note that tobacco smoking may affect clozapine metabolism through CYP1A2 induction, thus resulting in lower clozapine levels in smokers. This may cause adverse effects if a patient under clozapine treatment quits smoking because clozapine blood levels may rise up to 50–70%, potentially leading to toxic concentrations and development of adverse effects. 19
Therefore, it is important to conduct careful therapeutic drug monitoring (TDM) for any patient on clozapine. As reported by Bastiampillai and colleagues, 20 clozapine blood levels should usually be evaluated at least a week after a stable clozapine dose has been reached as a trough level. There is evidence that clozapine levels above 600 μg/ml may increase the risk of seizures, and prophylactic antiepileptics should be considered. 21 Clozapine serum-level monitoring may also be useful to evaluate clinical efficacy, in addition to checking for side effects and toxicity, with levels between 350 and 400 μg/ml significantly associated with improved clinical response rates, but also with a significantly increased side-effect burden.21–23
As poor treatment adherence in patients with resistant schizophrenia may increase the risk of psychotic relapse, 24 which may further lead to impaired social and cognitive functioning, psychiatric hospitalizations, suicide risks and increased treatment costs,25,26 clozapine adverse effects are a common reason for discontinuing this medication and this is a cause of concern.27,28 Therefore, all efforts should be made to monitor and minimize adverse effects, improve their early detection and conduct proper management, also to reduce the risk of people being harmed. 29 This must be done considering that, when clozapine fails, there are no effective therapeutic strategies that can be put in place. 30
The aim of this narrative review is to briefly summarize and provide an update on major clozapine adverse effects, especially focusing on those that are severe and potentially life threatening, even if most of the latter are relatively uncommon. Adverse reactions were divided by frequency regarding patients and treatment: common (⩾1/100), uncommon (⩾1/1000) and rare (⩾1/10,000) (see Table 2).
Adverse effects of clozapine (in brackets potentially life-threatening adverse effects if not recognized early, LT). Adverse reactions were divided by frequency regarding patients and treatment: common (⩾1/100), uncommon (⩾1/1000) and rare (⩾1/10,000).
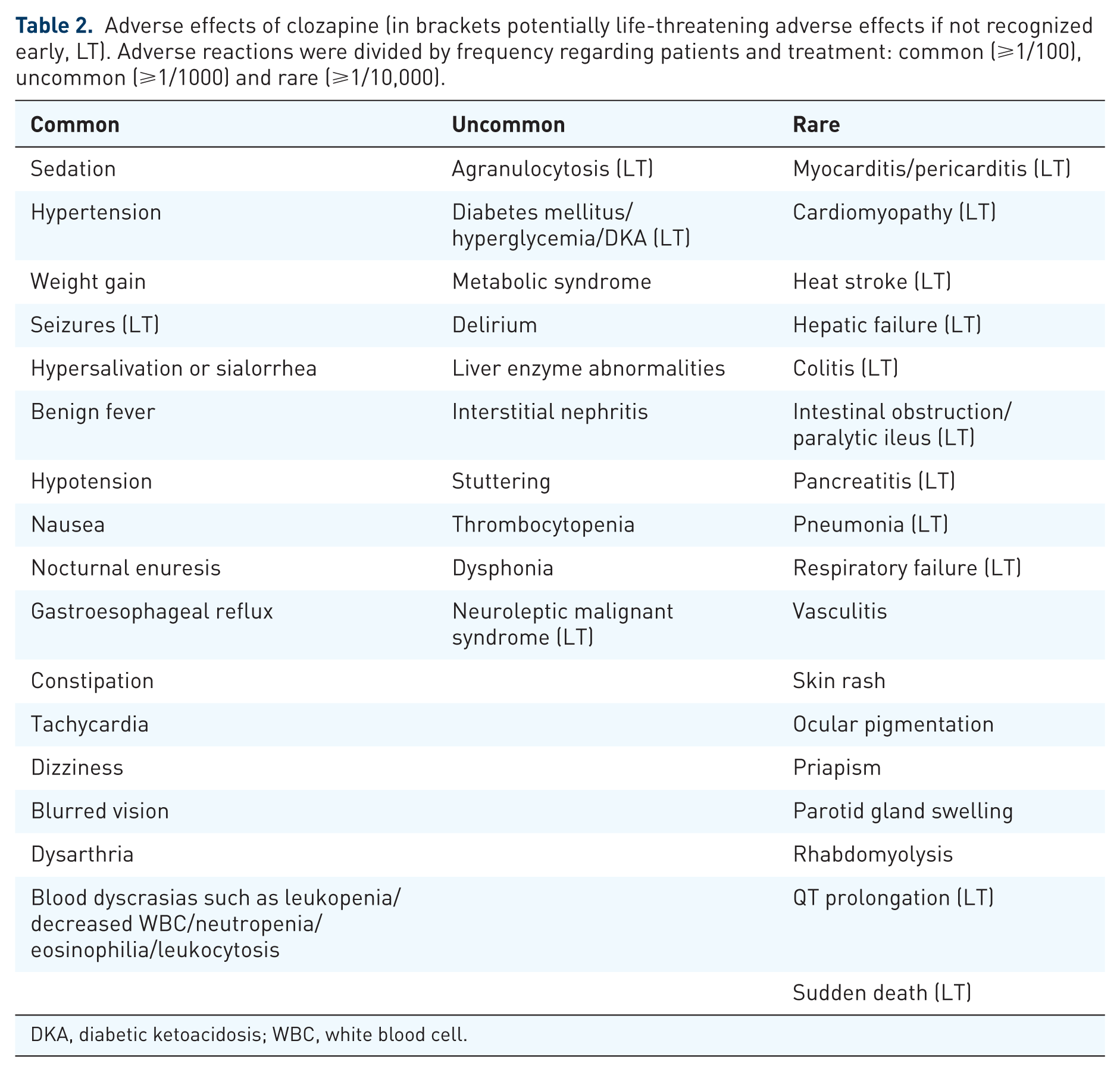
DKA, diabetic ketoacidosis; WBC, white blood cell.
Materials and methods of the review
Searches of the Medline database from 1980 to December 2017 and the PsycInfo/Embase database from 1980 to December 2017 were conducted and restricted to the English language. The search term ‘clozapine’ was combined with ‘adverse effects’, ‘side effects’ and ‘toxicity’ to identify relevant original research and review articles. All citations were screened and the full texts of peer-reviewed journal articles that were considered relevant for the purposes of the review were obtained. Bibliographies were scanned to locate additional relevant publications, even those prior to 1980. However, this paper is a narrative review. This type of review is useful when the aggregation of data is difficult because different studies or fields are being analyzed, but the methodology is less strict than in a systematic review.
Clozapine mechanism of action and relationships with adverse effects
Clozapine is a multireceptorial atypical antipsychotic approved for the treatment of resistant schizophrenia. 31 Concerning DA receptors, clozapine has a higher affinity for D1 and D4 than D2 receptors. 32 Moreover, clozapine is characterized by a rapid dissociation from D2 receptors (the ‘fast off’ phenomenon), which accounts for a more robust antipsychotic effect and a lower propensity to cause extrapyramidal symptoms and hyperprolactinemia.33,34
Clozapine has an affinity for several other receptors, including serotonergic 5-HT1A, 5-HT1C, 5-HT2A, 5-HT2C,5-HT3, 5-HT6, 5-HT7 receptors, adrenergic α1 and α2 receptors, histaminergic H1, H3, H4 receptors, and muscarinic M1 and M5 receptors. 35 These affinities, especially for adrenergic, histaminergic and muscarinic receptors, may explain some adverse effects commonly seen during clozapine administration, whereas some other adverse effects do not necessarily depend on clozapine-related receptor agonism or antagonism. 20
Furthermore, some populations, that is the elderly or adolescents, may be particularly vulnerable to clozapine adverse effects despite its proven efficacy.36–38
Besides clozapine activity on brain neurotransmitters and their receptors, a potential immune-mediated mechanism of action has been suggested.39,40 Clozapine may influence the cytokine system, especially during the first months of treatment, by increasing proinflammatory cytokines and C-reactive protein (CRP), and acting as an immunomodulatory drug in some circumstances.41–44 In fact, both in vitro and in vivo studies confirmed the immunomodulatory actions of clozapine.45–49 These effects of clozapine on the immune system may account for its efficacy in the treatment of schizophrenia in accordance with an immunological hypothesis of disorder etiology,50,51 but also for some adverse effects (i.e. eosinophilia, benign hyperthermia, cardiovascular and hematological adverse effects etc.).39,52
Common adverse effects
Orthostatic hypotension, often seen when initiating treatment, especially in the case of a rapid titration, and sexual dysfunction are mostly related to adrenergic α blockade,53–55 whereas sedation and constipation (see below for the latter) may be mainly due to H1 blockade.56,57 Clozapine-induced orthostatic hypotension and sedation may increase the risk of falls, especially in the elderly.36,58 Clozapine antagonism on muscarinic M1 receptors is responsible for anticholinergic effects such as constipation, tachycardia, blurred vision and urinary retention. 59 Other common adverse effects include dizziness, transient eosinophilia, sialorrhea, benign hyperthermia, leukocytosis and nausea. 60 Among the latter, sialorrhea may be particularly bothersome, although it is benign, dose related and often transient. However, there are some cases of aspiration pneumonia in patients treated with clozapine who have sialorrhea, so such an effect must be carefully evaluated.61,62 The management of clozapine-related sialorrhea is often problematic, but some strategies have been tried with good results, such as a low-dose amisulpiride or amytriptiline, bupropion, sublingual atropine, scopolamine, trihexyphenydil and glycopyrrolate, with the latter shown to be the most effective.63–67
Among the common adverse effects, the most dangerous and potentially life threatening are seizures and weight gain or metabolic syndrome (MetS).
Seizures
Seizures may be a relatively common and potentially life-threatening adverse effect of clozapine. 68 In fact, clozapine may lower the seizure threshold not only in individuals at risk of epilepsy, but also in apparently healthy subjects. 69 The seizure risk during clozapine treatment has been estimated at roughly 1–6%, is usually dose dependent and may appear more frequently in younger patients.70,71 It has been reported that rapid titration and higher dosages of clozapine (⩾600 mg/day) were associated with greater risk than medium (300–600 mg/day) or low dosages (<300 mg/day). 72
Moreover, rapid clozapine titration may further increase seizure onset. 70 Even if a higher risk of seizures during clozapine titration has been reported, this adverse effect may appear during all phases of treatment, even after several years of therapy. 73 Patients who develop seizures during clozapine titration or maintenance treatment may continue clozapine with dose reduction, or better still, the addition of an antiepileptic medication. Valproate may be the best choice, even if this combination must be carefully monitored.74,75 Electroencephalography and clozapine plasma blood-level monitoring are recommended to avoid or minimize seizure risk.76,77
Weight gain and the risk of MetS
Second-generation antipsychotics are generally associated with weight gain, particularly olanzapine and clozapine, and with related risks of metabolic disturbances such as insulin resistance, type 2 diabetes, dyslipidemia and diabetic ketoacidosis (DKA). 78
Weight gain is a common adverse effect of clozapine that, per se, may be not potentially life threatening if properly managed and treated. 77 If not, the consequent development of MetS may be a major health concern which highly increases the risk of developing type 2 diabetes mellitus and cardiovascular diseases.79,80 A relationship between weight gain and clozapine doses has been proposed, but this point remains controversial. 81
Clozapine metabolic abnormalities are mostly linked to 5-HT2A and 5-HT2C receptor blockade, but the effects on histaminergic and adrenergic receptors are also thought to play a significant role.82–84
Even if the prevalence of MetS in patients with schizophrenia is higher compared with the general population, going up to 40%, the effect of clozapine is remarkable with a prevalence rate varying from 11% to 64% among treated patients.85,86 Allison and colleagues reported that clozapine treatment was associated with an average gain of 9.8 lb over 10 weeks and this was the highest figure of all antipsychotics. 87 The incidence of clozapine-related weight gain is considered to be the most significant at 5–35%, and almost half of patients may gain 20% or more of their initial body weight, especially if they are on polypharmacy, have a sedentary lifestyle or are not overweight at baseline. 88 Several studies have also associated clozapine treatment with the development of dyslipidemia and hypertension,77,89–91 all factors that may affect MetS or make it worse. The metabolic risk associated with clozapine treatment may be increased in the short term after treatment initiation. In fact, it has been demonstrated that the increased short-term metabolic risk associated with clozapine may be diminished over time because multiple other variables are also likely to affect the metabolic risk during the lifespan. 92
Clozapine treatment may also be associated with diabetes and DKA.93,94 Regarding DKA, Cohen and colleagues 95 found a 1-year incidence rate of 1.2–3.1% and a case fatality rate of 20–31%, making DKA one of the riskiest side effects, with higher mortality than agranulocytosis with an incidence of 3.8–8.0% and case fatality rate of 2.2–4.2%. Therefore, the authors proposed that the screening guidelines should be modified with an early detection of treatment-emergent hyperglycemia through an obligatory monthly measurement of fasting plasma glucose. It has also been demonstrated that clozapine-related DKA typically occurs early in the course of treatment, when clozapine treatment duration is short and doses are low. 96
Especially with combined behavioral interventions consisting of both nutritional counseling and diet modification), aripiprazole, topiramate, orlistat and metformin additions have been shown to be beneficial in reducing clozapine-induced weight loss, even if the evidence is based on a limited number of randomized controlled trials and further studies are needed.88,97,98 Counseling for patients needing proper diet and exercise for weight and glucose control should always be recommended. 99
Gastrointestinal hypomotility and constipation
The anticholinergic peripheral properties of clozapine as well as its H1 antagonism properties were related to several grades of impairment of intestinal peristalsis ranging from transient to obstinate constipation and paralytic ileus up to intestinal obstruction.100,101 It is known that even low or therapeutic doses of clozapine may prolong colonic transit time (CTT). 102
These effects are mainly mediated by the clozapine antagonism on cholinergic and serotoninergic receptors in the gut wall. 103 It has been demonstrated that clozapine, but not norclozapine, has potent effects on the motility of the colon of a rabbit, thus inhibiting neurogenic contractions at lower concentrations and myogenic contractions at higher concentrations. 104 These effects may be mediated by cholinergic and serotonergic receptors, presumably via M1–M3 and 5HT-3 and 5HT-7 receptors. Moreover, de Alvarenga and colleagues 105 showed that clozapine significantly reduced the frequency of cycles of contractions of zebrafish larvae gastrointestinal tracts through anticholinergic action.
These adverse effects are bothersome for several patients who develop mild constipation, which is often a treatment discontinuation cause, but it may become severe and potentially life threatening in some others who develop moderate to severe untreated constipation or even an adynamic ileus. 103 In fact, constipation is very frequent in patients treated with clozapine with an estimate range varying from 15% to 60% in accordance with several studies.106,107 Moreover, Every-Palmer and Ellis 108 reviewed all reports of serious clozapine-induced gastrointestinal hypomotility (CIGH, also known as clozapine-related ‘slow gut’) submitted to the Australian Therapeutic Goods Administration and New Zealand Pharmacovigilance Centre between 1992 and 2013 and found that the reported prevalence of serious CIGH was 37/10,000, likely an underestimation of the true prevalence. CIGH is relatively common as between 50% and 80% of patients treated with clozapine have unambiguous objective evidence of CIGH in colonic transit studies, 102 with mean transit times that are four times longer than normal, affecting all regions of the colon. 106 It is worth noting that studies which have relied on reporting of constipation are not very sensitive in diagnosing slow gut.
In fact, rare but potentially fatal clozapine-related adverse effects are intestinal obstructions and a paralytic ileus that occur in almost 2% of treated patients. 109 In severe cases, constipation progresses to ileus and bowel ischemia, with multiple fatalities related to aspiration of feculent vomit, sepsis and perforation, as described in the literature. 110 Prolonged gastrointestinal hypomotility and constipation may promote ischemia, increasing intraluminal pressure and compression of mucosal vessels. As a consequence, bacterial translocation, inflammation and ischemia of the segment of bowel proximal to the obstruction will develop.111–113 One case of clozapine-induced severe constipation leading to a silent presentation of pneumonia with a subsequent respiratory arrest 114 has also been reported.
Clozapine-related ileus has been reported to be dose dependent and may occur several days after initiation of treatment as well as at any time during treatment, with the latter often triggered by concomitant medications known to affect the gastrointestinal system, especially those with anticholinergic properties. 115 Therefore, early detection and treatment of constipation and ileus are mandatory to avoid fatalities.
The recommendation of an adequate intake of fluids, especially in the presence of hypersalivation, and regular physical activity is mandatory in such patients, as well as dose reduction or laxative prescriptions to avoid the progression of severe gastrointestinal adverse effects.107,116,117 Recently, Every-Palmer and colleagues 118 demonstrated that patients with clozapine-related constipation treated with docusate and senna augmented by macrogol had reduced CTTs and this was beneficial in treating constipation. Also, orlistat was found to be helpful, 119 but further studies are needed.
Uncommon adverse effects
Less common adverse effects associated with clozapine treatment may include delirium, liver enzymes abnormalities, interstitial nephritis, stuttering, thrombocytopenia and dysphonia. 120
Moreover, several cases of clozapine-related neuroleptic malignant syndrome have been reported, often characterized by atypical features, that is, with less intense extrapyramidal symptoms or high fever.121,122 However, it should be noted that the majority of published cases were reported in subjects with comorbid conditions 123 or under polypharmacy with different antipsychotics, especially those with higher affinity on dopamine D2 receptors, such as risperidone, amisulpride and olanzapine or other drugs.124–126
Blood dyscrasias and agranulocytosis/granulocytopenia
Blood dyscrasias, such as mild to moderate leukopenia decreased white blood cell count, mild neutropenia, anemia, eosinophilia and leukocytosis, are often seen with clozapine but, if cautiously monitored, in the majority of the cases this may be transient or benign.127,128
The most severe and potentially life-threatening clozapine-related blood dyscrasias is neutropenia, which may eventually develop into clozapine-induced agranulocytosis or granulocytopenia. This occurs in roughly 0.8–2% of patients and requires mandatory hematological monitoring.20,129 The occurrence of agranulocytosis or granulocytopenia even led to the partial withdrawal of clozapine from the market and strong restrictions to its use in the mid 1970s. 130
Neutropenia was defined in the 2005 clozapine guidelines as an absolute neutrophil count (ANC) below 1500 and occurs in approximately 3% of treated patients, 131 whereas agranulocytosis was defined as an ANC below 500/mm3.132,133 A transient neutropenia (1 week) or weekly variations of ANC are relatively common during clozapine treatment and do not necessarily require treatment discontinuation, but it is not known why some patients develop transient neutropenia whereas others progress to having agranulocytosis.134,135
Although the highest risk of agranulocytosis has been reported to occur during the first 12 weeks from clozapine titration and during the first 6 months of treatment, patients may develop severe blood dyscrasia anytime during the treatment, even if the risk decreases exponentially over time after the first 6 months of treatment. 135 However, some cases of late-onset clozapine-induced agranulocytosis have been reported.136,137 The development of agranulocytosis may be independent of dosage and risk factors include older age, female sex, human leukocyte antigen haplotypes and concomitant treatment with other drugs known to cause agranulocytosis.52,138–142 Interestingly, in cases with severe clozapine-induced agranulocytosis, the occurrence did not depend on the clozapine dosage. 143
To explain this dangerous adverse effect, several studies have hypothesized a potential interaction between clozapine and the immune system, suggesting an immune-mediated mechanism to be involved in its adverse effects or even in its clinical efficacy.144,145 It is also possible that some subjects may be genetically predisposed; oxidative clozapine degradation may cause the development of some metabolites with reactive nitrenium ions, which may covalently bind to human leukocytes, resulting in direct toxicity or leading to the development of aptenic formation of an antigenic structure that will stimulate a targeted immune response.146–148
The 2015 US Food and Drugs Administration guidelines for hematologic monitoring during clozapine therapy149–151 recommend that treatment is not discontinued in the case of mild neutropenia (⩾1000–1499/mm3), with ANC monitored three times weekly until it is at least 1500/mm3. However, in the case of moderate neutropenia (⩾500–999/mm3), they recommend that treatment is interrupted when clozapine-induced neutropenia is suspected, with daily monitoring performed until ANC is at least 1000/mm3 (when treatment may resume), and consequently monitoring carried out three times weekly until ANC is at least 1500/mm3, then proceeding with weekly checks for 4 weeks, and finally returning to the patient’s last normal range ANC monitoring plan.
In the case of severe neutropenia (<500/mm3), all patients must interrupt treatment for suspected clozapine-induced neutropenia and clozapine rechallenge should not be considered unless the prescriber, along with a hematologist consultation, determines that the benefits outweigh the risks. 152 Recently, some studies have pointed out that granulocyte colony-stimulating factor may be beneficial in the treatment of clozapine-induced agranulocytosis, 153 but more evidence is needed and withdrawing clozapine is still mandatory in such cases.
Clozapine rechallenge is always a risk after an episode of agranulocytosis as several reports observed a new episode of blood dyscrasia.154,155 However, there are also some reports that observed no blood dyscrasia when clozapine was reintroduced after an episode of agranulocytosis with or without adjuvant therapies.156–158 Dunk and colleagues 159 conducted clozapine rechallenge in 53 patients who developed leucopenia or neutropenia during previous clozapine treatment and found that 38% experienced a further blood dyscrasia whereas 55% did not. More recently, Prokopez and colleagues 160 analyzed the results of a clozapine rechallenge after leucopenia or neutropenia in 19 patients and observed that almost 70% of the patients did not develop a new hematological adverse effect, whereas the remaining 30% had a faster but less serious neutropenia. In conclusion, clozapine rechallenge requires careful evaluation and should be taken into consideration on a case-by-case basis when the risks outweigh the benefits for all other strategies.
Uncommon adverse effects
Clozapine may be associated with several uncommon adverse effects such as vasculitis, skin rash, ocular pigmentation, priapism, parotid gland swelling and rhabdomyolysis that may not always be severe but require cautious monitoring. 5 However, some of the (fortunately) rare adverse effects may be severe and potentially life threatening, and these include heat stroke, hepatic failure, colitis, pancreatitis, pneumonia and respiratory failure. 7
QT prolongation
It has been reported that clozapine may be associated with QT prolongation in a dose-dependent manner.161,162 In most cases, drug-induced QT prolongation is not significant per se, whether it is symptomatic or malignant, 163 but in some individuals it may lead to the life-threatening torsades de pointes, a sudden and often fatal arrhythmia. 164
The effect on QTc is not peculiar to clozapine; it is often seen with other antipsychotics, especially FGAs, and other psychiatric drugs.165–167 Almost all of the drugs known to prolong the QT interval both directly or through one of their metabolites seem to preferentially block the rapid component of the delayed rectifier outward K+ current, IKr, of an ion channel in ventricular cardiomyocytes, whose α subunit is encoded by the human ether-a-go-go related gene (HERG). 168
Clozapine has demonstrated the capability to block HERG currents and this may account for drug-induced QT prolongation. 169 However, it is known that clozapine may induce an increased heart rate and this may influence the QTc determination (when using Bazett’s formula), leading to an overestimation of this effect. 170
All considered, clozapine seems to rarely influence QT traits. However, electrocardiogram monitoring is always useful when clozapine is commenced or when administered together with other drugs, and in elderly patients.171–175 The QT interval should be calculated using methods involving linear regression, adjusting for age and sex, as recommended by the American Heart Association, 176 and not with Bazett’s formula. Clozapine dose reductions may be useful for QT prolongation, especially in the case of polypharmacy. 177
Clozapine-induced cardiomyopathy, myocarditis and pericarditis
Clozapine may be associated with the occurrence of severe cardiovascular side effects, including dilated cardiomyopathy, myocarditis and pericarditis, which may be fatal when not timely recognized and managed.10,170
Dilated cardiomyopathy is an uncommon, dose-independent, but potentially life-threatening adverse effect of clozapine treatment. It may occur even after a longer drug exposure and is often confused with myocarditis. 178 The clinical manifestations of clozapine-induced cardiomyopathy range from subclinical forms to severe ones with fulminant pulmonary edema and cardiogenic shock.179,180 Often the symptoms are insidious and include persistent tachycardia at rest, palpitations, arrhythmias, dyspnea, chest pain, malaise and other signs and symptoms that may be underestimated by the clinicians until the symptoms resemble the clinical picture of heart failure or myocardial infarction. 181
Myocarditis is an uncommon, dose-independent, life-threatening adverse effect and occurs in up to 3% of treated patients.182,183 Moreover, several sudden deaths reported with clozapine use may be due to an unrecognized or fulminant myocarditis. 184 Although myocarditis has a tendency to appear at any time during treatment with clozapine, this has been reported more frequently from 4 days to 22 weeks after treatment initiation, 185 with a particular ‘danger period’ during the first 4 weeks following initiation. 186
The clinical presentation of clozapine-related myocarditis in adults may be highly variable, ranging from subclinical diseases to fulminant heart failure.187,188 Flu-like symptoms such as fatigue, fever, myalgias, dizziness, arthralgias, nasal congestion and sensations of ‘scratchy throat’ may often be the first to appear and are sometimes unrecognized or underestimated.189,190
Respiratory symptoms, such as dyspnea, cough, subjective sensations of chest discomfort and orthopnea, as well as cardiovascular symptoms, such as persistent resting tachycardia, increased heart rate, palpitations, chest pain, syncope, arrhythmias and hypotension, are the main symptoms that drive the diagnosis. However, in some cases, clozapine-related myocarditis may develop in an atypical form without accompanying symptoms and these cases are often fatal.191,192
It has been suggested that risk factors for any cardiac complications of clozapine may be the rapid dose titration, the presence of weight gain or metabolic adverse effects, the coadministration of selective serotonin reuptake inhibitors and the use of illicit substances.193–195 Moreover, concomitant treatment with medications that have been implicated as the cause of cardiovascular complications like myocarditis and heart failure, namely some antibacterials, cisapride, thyroxine, ranitidine, cyclophosphamide, lithium, phenotiazines and certain antidepressant agents like amytriptiline, imipramine and desipramine, may represent another risk factor when a patient is taking clozapine. 196 Interestingly, some cases of clozapine-related myocarditis were detected in patients taking valproate, which is often prescribed to prevent seizures or as a mood stabilizer.184,195,197 Hence, when clozapine is administered together with valproate, patients should be more carefully and strictly evaluated for an eventual myocarditis onset.
The early detection of clozapine-related myocarditis is mandatory as early recognition improves clinical outcome and reduces mortality.170,198 To date, the monitoring protocol adapted from De Berardis and colleagues 10 and Ronaldson and colleagues 185 seems to be scientifically sound and clinically useful in everyday clinical practice (see Figure 1).
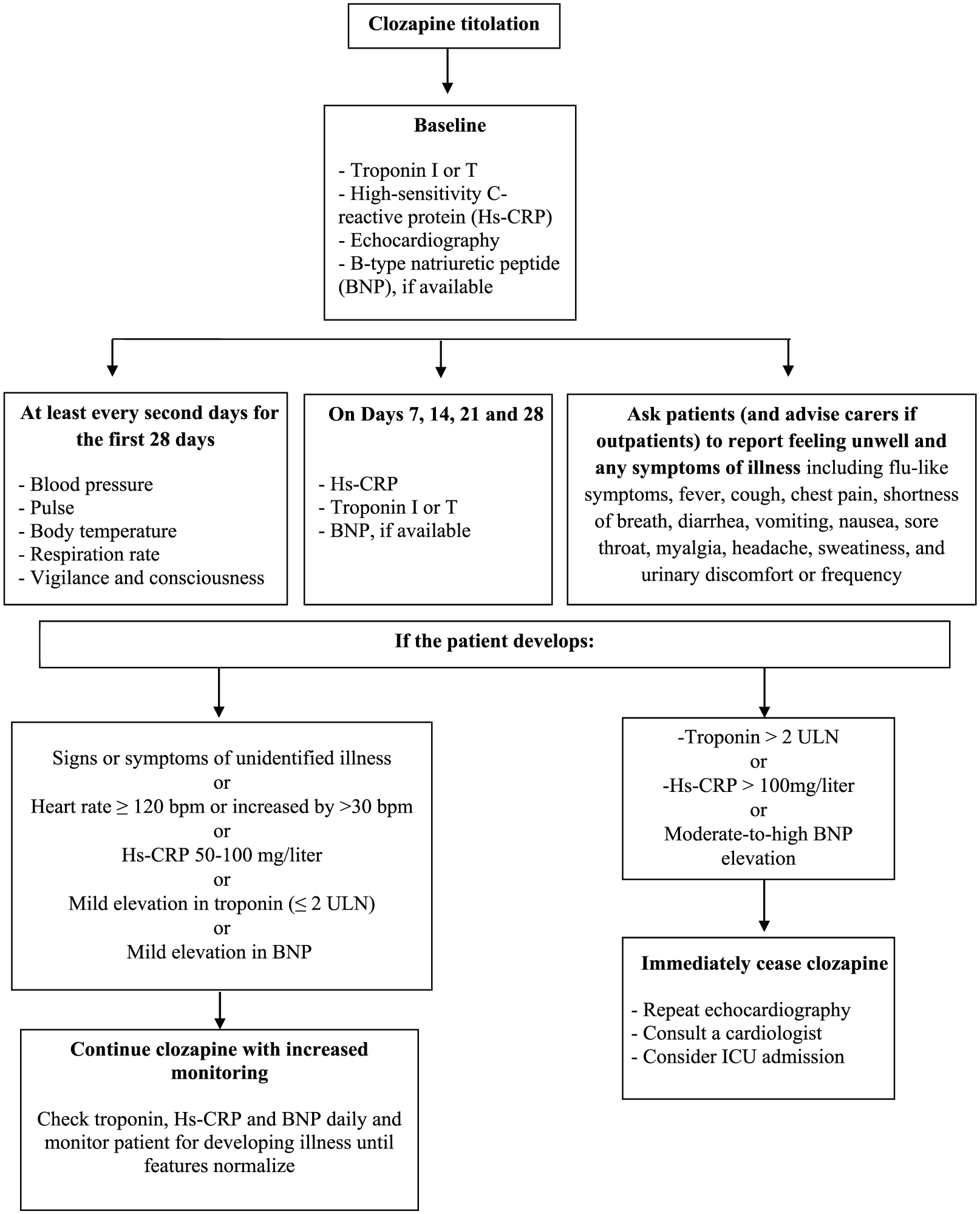
When myocarditis is suspected or diagnosed, immediate clozapine cessation is mandatory as usually this may resolve the case within a few days or weeks. 60 However, some patients may require treatment with a β blocker or angiotensin-converting enzyme inhibitor, whereas severe cases will need mechanical ventilation.199,200 Corticosteroids have been administered in some cases, but their efficacy is uncertain.201,202
As far as clozapine rechallenge after an episode of myocarditis is concerned, it should be noted that this strategy may be dangerous, is not recommended, and should be employed after careful evaluation of the pros and cons together with a consultation with a cardiologist,170,203,204 even though there are some positive reports.191,205–207
Moreover, several cases of clozapine-related pericarditis have been reported in the literature in patients on stable clozapine regimens for several weeks, months or even years, or during the titration phase.208–211 In the majority of published cases, clozapine treatment was immediately discontinued, resulting in complete resolution of symptoms. 212 In some cases, clozapine rechallenge was tried, but this strategy is controversial as pericarditis may develop again, even though this is not the rule.213,214
Venous thromboembolism
There are several reports on clozapine-related venous thromboembolism (VTE), but luckily it seems to be relatively rare and dose independent.215–217 The mechanism of this dangerous adverse effect remains unknown.218–220 Axelsson and colleagues 221 compared several antipsychotics and found that clozapine was the only compound that increased platelet adhesion and aggregation, and shortened activated partial thromboplastin time, even at therapeutic concentrations. Moreover, in a case report, clozapine induced an allergic vasculitis that resulted in VTE. 222
Nevertheless, the risk of VTE seems to be an adverse effect of several antipsychotics including clozapine,223,224 and may be higher in elderly patients and in women taking high doses or receiving parenteral antipsychotics.225–228 However, it has been suggested that VTE may be more a consequence of several risk factors for VTE present in patients with psychiatric disorders, such as MetS, inactivity, low levels of water intake and other medical illnesses, than the treatment with antipsychotics itself.229–231
All considered, caution is needed to recognize VTE symptoms early, for example painful edema of the lower limbs, chest pain and dyspnea in patients taking clozapine, especially in those with known risk factors for VTE, like patients over 65, with previous VTE episodes, obesity, MetS, inactivity, lower limb trauma, estrogen treatment, among others.170,218,225
Conclusion
Despite its potentially life-threatening adverse effects which deserve special attention, clozapine remains the gold standard in the treatment of treatment resistant schizophrenia (TRS) with, to date, inimitable results compared with other available antipsychotics. However, regardless of the evidence, clozapine is still underprescribed or utilized too late and this may be due to, but not limited to, blood monitoring because it is often tedious for the patients, 129 and the psychiatrists’ fear of severe adverse effects.232,233
Some psychiatrists do not prescribe clozapine when required as they are anxious about its management and perceive a lack of knowledge and a negative attitude toward this drug.234,235 Such discomfort with clozapine is a major cause of concern that the patients will pay in terms of a lack of a therapeutic possibility and, therefore, efforts should be made to overcome this.3,236 This brief review was written not to scare prescribers, but to increase knowledge on the adverse effects of clozapine and to improve their early identification and management. Despite the fear of death of clozapine-treated patients with its possible legal consequences, it is important to emphasize that clozapine treatment may decrease mortality in treated patients by reducing self harm, risk of suicide and attempted suicide.237,238 Furthermore, Hayes and colleagues220,239 reported an association between clozapine and reduced risk of mortality from natural causes in a sample of 14,754 individuals with serious mental illness.
Last, but not least, clozapine is the only drug able to restore satisfactory functioning in patients with TRS by improving their lives and self care, even in the long term.240–243 This should always be kept in mind. Therefore improving our knowledge of clozapine is important to increase the possibility of prescribing it to those in need. Also, improving our knowledge on the adverse effects of clozapine and their management may help to reduce the understandable apprehension of physicians and help them to be more confident when prescribing clozapine.
Footnotes
Acknowledgements
The authors thank Prof. Roberta Polimanti for checking and editing the English language.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest statement
No author or immediate family member has financial relationships with commercial organizations that might appear to represent a potential conflict of interest with the material presented.
