Abstract

Increasing evidence indicates that proton pump inhibitors (PPIs) are being overprescribed and are associated with numerous adverse effects [Forgacs and Loganayagam, 2008; McCarthy, 2010].Studies over the past decade have linked PPI use to increased rates of Clostridium difficile infection, pneumonia, fractures, hypomagnesemia and both acute and chronic kidney disease [Schoenfeld and Grady, 2016]. Two studies, one involving institutionalized older patients, and another involving patients in acute geriatric wards and nursing homes, have demonstrated a link between PPI use and increased mortality [Bell et al. 2010; Teramura-Gronblad et al. 2012]. PPI use in older patients discharged from acute care hospitals was also associated with increased mortality [Maggio et al. 2013]. We sought to investigate whether the use of PPIs, specifically in older hospital patients discharged to residential aged care facilities (RACFs), had any impact on hospital readmission rates and mortality.
Patients ⩾75 years, originally from home or a RACF, admitted to general medicine wards between 30 October 2014 and 1 May 2015 and discharged to a RACF, were recruited into the study. Palliative care patients were excluded. A comprehensive list of all preadmission medications, completed by a clinical pharmacist, was utilized as the list of medications on admission. A discharge medication chart for each RACF, completed by a medical officer and countersigned by a clinical pharmacist, was utilized as the list of medications on discharge. Only regular medications were included in data analysis. Readmission data was obtained via the electronic hospital clinical information system. Mortality data was collected via Births, Deaths and Marriages South Australia, who require mandatory reporting for all RACF deaths. Each patient was followed up for six months in relation to readmission and mortality. Ethics approval was obtained via the Southern Adelaide Clinical Human Research Ethics Committee (457.14). The Charlson Comorbidity Index (CCI) scores were calculated via the electronic hospital clinical information system. Statistical analyses were performed using IBM® SPSS® Statistics Version 21. Cox proportional hazards regression (hazard ratios) and negative binomial regression with log link (incidence rate ratios) both with 95% confidence intervals were used to determine the association between PPIs and mortality and hospital readmissions, respectively. The possible indication for each PPI was identified based on the medical discharge summary, taking into account the patients’ concurrent medical conditions and medications.
The mean (SD) age of participants (N = 102) was 87 (6) years, and 62 (60.8%) were female. Of the total population, 51 (50%) were taking a PPI on discharge. Of the population taking a PPI on discharge, 45 (88%) were already taking a PPI prior to their admission, 4 (8%) were initiated onto a PPI during their admission, and 2 (4%) had their PPI continued at a decreased dose. Of the 51 patients taking a PPI, 37 patients received pantoprazole; 9, esomeprazole; 4, omeprazole; and 1, rabeprazole. In adjusted analyses, PPI use was associated with an 82% increased rate of hospital readmission (Table 1), and 124% increased rate of mortality within six months from initial discharge from hospital (Table 2). The majority of patients (n = 30/51; 59%) had an identifiable indication for a PPI based on their medical discharge summary, the most common of which was gastroesophageal reflux disease (n = 16/30; 53%).
Relationship between use of PPIs and readmissions.

PPI, proton pump inhibitors; CI, confidence interval; IRR, incidence rate ratio.
Indicates negative binomial regression with log link IRRs adjusted for age, gender, initial place of residence, CCI, number of days alive following discharge and number of regular medications taken on discharge.
Relationship between use of PPIs and mortality.
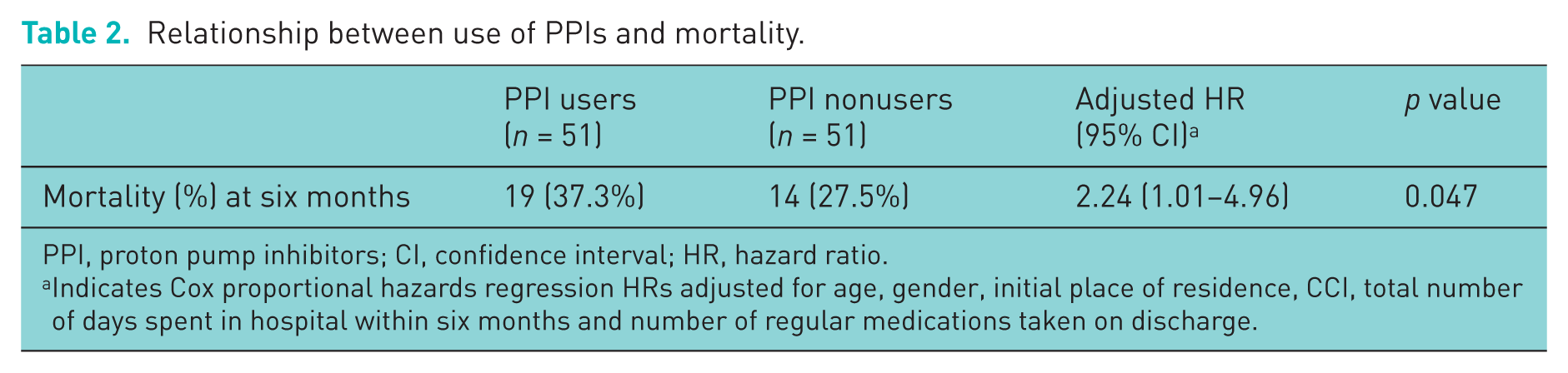
PPI, proton pump inhibitors; CI, confidence interval; HR, hazard ratio.
Indicates Cox proportional hazards regression HRs adjusted for age, gender, initial place of residence, CCI, total number of days spent in hospital within six months and number of regular medications taken on discharge.
PPI use in our patient population was associated with an increased rate of hospital readmission and mortality within six months. Our mortality data is consistent with previous studies. However, we also observed a significant increase in hospital readmission rates, as further evidence of potential PPI-related adverse events. These results may be explained by PPIs increasing the likelihood of infection, fractures and other adverse effects mentioned in literature. Increasing hospital readmissions places strain on the health care system, and a portion may be avoidable due to 41% of patients not having an identifiable indication for a PPI based on their medical discharge summary. A possibility of residual confounding cannot be excluded, the indication for each PPI was limited to that which was identifiable from medical discharge summaries, and we lacked data relating to how long each patient had already been on a PPI and for how long they remained on a PPI post-discharge. This frail patient population are already at a high risk of readmission and mortality, and PPIs may further increase this risk unnecessarily. Our study further highlights the need for physicians and pharmacists to continually review PPI use, especially in the older inpatient population.
Footnotes
Acknowledgements
Matthew A. Pegoli had full access to all the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis. Study concept and design, acquisition of data, analysis and interpretation of data, drafting of the manuscript, critical revision of the manuscript for important intellectual content, statistical analysis: Pegoli, Dedigama, Mangoni, Grzeskowiak, Russell, Thynne. Gregory W Roberts (BPharm, FSHP, CRP) and Cameron Phillips (BPharm, MClinPharm) provided assistance with study guidance and concept. Justin P. Turner (PhD, BPharm, MClinPharm) and Sepehr Shakib (MBBS, FRACP, PhD) provided assistance with data management. No compensation was received for all of the above contributions. This study was approved by the Southern Adelaide Clinical Human Research Ethics Committee (457.14).
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of interest statement
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
