Abstract
The Women’s Health Initiative (WHI) has conducted two randomized, placebo-controlled clinical trials to evaluate the influence of menopausal hormone therapy on chronic disease risk. Estrogen plus progestin was evaluated in 16,608 postmenopausal women without prior hysterectomy during 5.6 years’ intervention. In that setting, combined hormone therapy use significantly increased breast cancer incidence and interfered with breast cancer detection. The breast cancers were not limited to estrogen receptor positive, favorable prognosis cancers and were identified at more advanced stage. As a result, deaths from breast cancer were significantly increased by estrogen plus progestin use. While the absolute breast cancer risk for relatively short term (2–4 years) use of combined hormone therapy is small, on a population basis a therapy which nearly doubles deaths from breast cancer requires cautious use. Estrogen alone was evaluated in 10,739 postmenopausal women with prior hysterectomy during 7.1 years’ intervention. There was an overall reduction of breast cancer incidence seen with estrogen alone use and a suggestion that the effect on risk was more pronounced in women initiating hormone therapy further from menopause. Nonetheless, women with prior hysterectomy can be assured that short duration estrogen alone use for climacteric symptom management is relatively safe. Neither estrogen plus progestin nor estrogen alone should be used for chronic disease risk reduction. The safety of duration of use on chronic disease risk longer than in the WHI clinical trials is not defined.
Introduction
There has been interest in the relationships between menopausal hormone therapy and breast cancer following initial observations that hormone therapy users were at increased risk of breast cancer [Hoover et al. 1976; Ross et al. 1980]. Over several decades, reports from a series of observational studies, both case-control and prospective cohort, led to a consensus that estrogen plus progestin as well as estrogen alone (for women with prior hysterectomy) increase breast cancer risk with longer exposure required for estrogen alone influence [Colditz et al. 1995; Nelson et al. 2002; Salagame et al. 2011; Million Women Study Collaborators, 2003]. With few exceptions [Million Women Study Collaborators, 2003; Antoine et al. 2004], the preponderance of observational studies report estrogen plus progestin use mainly associated with favorable prognosis breast cancers with either no increase [Norman et al. 2010; Pocobelli et al. 2014] or decrease [Schuetz et al. 2007; Newcomb et al. 2008; Reding et al. 2011; Chen et al. 2005] in deaths from breast cancer in women on menopausal hormone therapy. Because findings were largely congruent for breast cancer outcomes when estrogen plus progestin and estrogen alone were evaluated, observational studies commonly combined results of these two interventions.
Other observational studies during the same period suggested that hormone therapy use was associated with chronic disease risk reduction benefits including reduced coronary heart disease, fracture risk, and perhaps improved cognition [Nelson et al. 2002]. The observational study findings, in conjunction with the well-established favorable influence of hormone therapy on climacteric symptoms, resulted in increasing hormone therapy usage such that in 2002 there were approximately 15 million menopausal hormone therapy users in the US [Hersh et al. 2004].
The observational study reports from the US were largely based on findings with conjugated equine estrogen (Premarin®, Pfizer), used alone for women with prior hysterectomy, or with conjugated equine estrogen plus medroxyprogesterone acetate (Prempro®, Pfizer), used in women with a uterus as these preparations had over 90% market share in the US at that time [Hersh et al. 2004]. In addition, results from European studies for combined hormone therapy, where a wider range of hormones were used, largely paralleled US findings for influence on breast cancer [Million Women Study Collaborators, 2003; Li et al. 2003]. Thus, menopausal hormone therapy was one of the most common prescriptions for postmenopausal women in the US. At the time the Women’s Health Initiative (WHI) started, randomized trials to evaluate menopausal hormone therapy influence on endometrial histology [Judd et al. 1996] and in women with cardiovascular disease [Hulley et al. 1998] were underway but the influence of estrogen plus progestin and estrogen alone on overall risk and benefits on chronic disease risk had not been prospectively evaluated in full-scale, randomized clinical trials (Box 1).
Box 1.
Menopausal hormone therapy: historical scope of the question.
Conjugated equine estrogen (Premarin®) was introduced in US in 1942 for climacteric symptom management.
Observational and surrogate endpoint studies suggested hormone role in disease prevention (osteoporosis, cardiovascular disease, dementia).
Through 2001, the prevailing concept was that, while estrogen plus progestin and estrogen alone both increased breast cancers, the cancers were mainly hormone receptor positive, low stage, with favorable prognosis.
Over 38% of postmenopausal women in US were using menopausal hormone therapy through 2001.
Two Women’s Health Initiative hormone therapy trials were begun in 1993 with outcomes reported beginning in 2002.
WHI hormone therapy trials
Against that background, two WHI menopausal hormone therapy trials were initiated in 1993 [Rossouw et al. 2002; Anderson et al. 2004]. In randomized, prospective, placebo-controlled clinical trials, estrogen plus progestin was evaluated in 16,608 postmenopausal women and estrogen alone was evaluated in 10,739 postmenopausal women with prior hysterectomy. Women between the ages of 50 and 79 years were eligible. Additional eligibility requirements included mammogram not suspicious for malignancy, no prior breast cancer or any invasive cancer within 10 years and providing informed consent. By protocol design, a proportion of women (about 25%) were entered between the ages of 50 and 59 [Women’s Health Initiative Study Group, 1998]. The primary outcome monitored for benefit was coronary heart disease (myocardial infarction or death from heart disease) and the primary outcome monitored for harm was invasive breast cancer. In addition, a global index, incorporating a series of other potentially life threatening medical conditions likely to be under hormone therapy influence (both positive and negative) including stroke, pulmonary embolism, colorectal cancer and endometrial cancer was monitored for overall risk/benefit. The study designs are outlined in Figure 1. When the study findings from the two trials were published in a series of manuscripts beginning in 2002, results differed from concepts emerging from observational studies in several aspects and differences between estrogen plus progestin and estrogen alone use on some clinical outcomes were seen.

(a) Women’s Health Initiative (WHI) estrogen plus progestin E+P trial: findings during intervention [Writing Group for the Women’s Health Initiative Investigators, 2002; Chlebowski et al. 2003; Rapp et al. 2003]. (b) WHI estrogen-alone clinical trial: findings during intervention [Anderson et al. 2004; Stefanick et al. 2006].
WHI: the estrogen plus progestin trial
The estrogen plus progestin trial was stopped early after 5.6 years’ mean follow up when the breast cancer comparison crossed the prespecified boundary for safety and analysis of the global index identified significantly more harms than benefits for estrogen plus progestin use. In the initial report, as expected, invasive breast cancer incidence was significantly and relatively modestly increased in estrogen plus progestin users (Figure 1) [Rossouw et al. 2002]. When details of the breast cancers diagnosed in the estrogen plus progestin trials became available beginning in 2003, differences from most observational studies emerged [Chlebowski et al. 2003, 2009, 2010a]. While invasive breast cancers were more commonly diagnosed in the estrogen plus progestin group [hazard ratio (HR) 1.24; 95% confidence interval (CI):1.01–1.54, p = 0.003], the breast cancers were larger and were at more advanced stage (regional/metastatic 25.4% versus 16.0%, respectively; p = 0.04) compared with those in the placebo group [Chlebowski et al. 2003]. In addition, after only 1 year, women in the estrogen plus progestin group more commonly had abnormal mammograms compared with those in the placebo group (9.4% versus 5.4%; p < 0.001) and higher frequency of annual mammograms with abnormalities persisted throughout the study duration [Chlebowski et al. 2003, 2008].
When mammogram diagnostic performance was more formally evaluated, estrogen plus progestin use was found to decrease sensitivity, specificity and positive predictive value of mammography [Chlebowski et al. 2008]. Although there were substantially more abnormal mammograms in women in the combined hormone therapy group, mammograms less reliably diagnosed breast cancer. In addition, although women in the combined hormone therapy group more commonly had breast biopsies, all done for medical cause, than women in the placebo group, the biopsies were statistically significantly less accurate in diagnosing breast cancer. These findings added risk of increased abnormal mammogram and risk of increased breast biopsies to risk benefit analyses considering combined hormone therapy use.
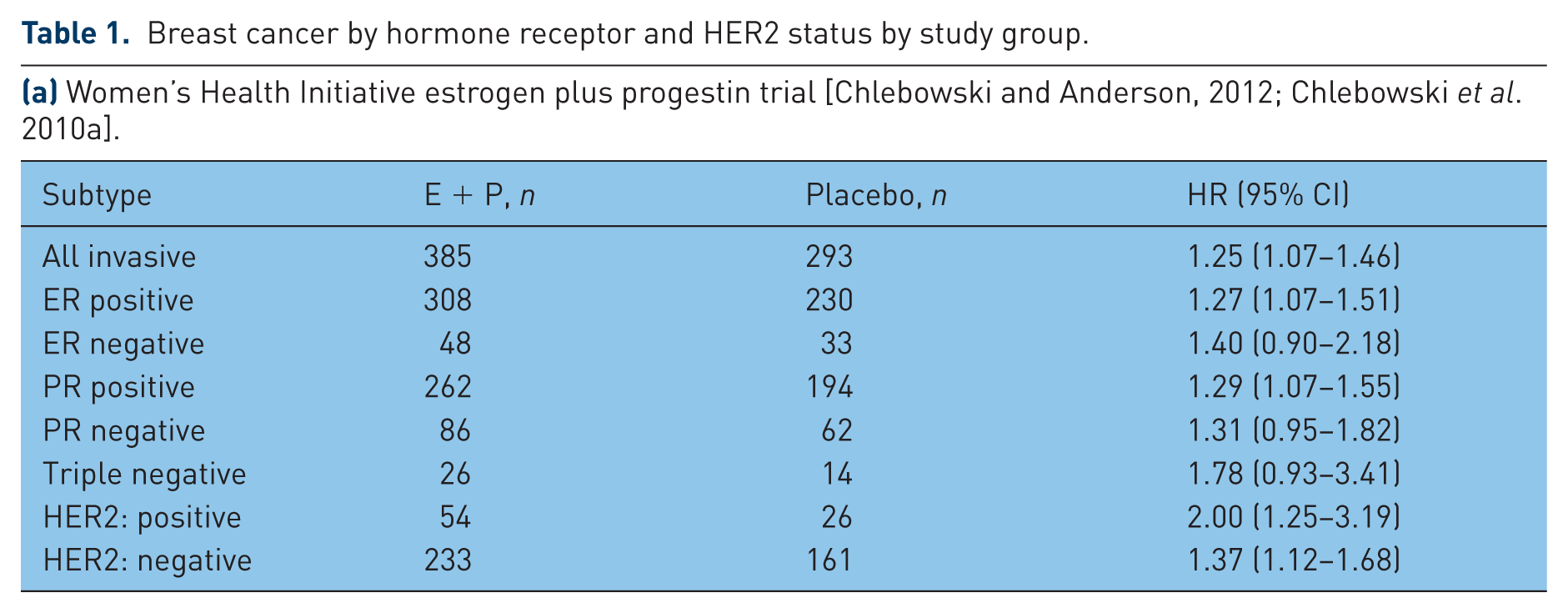
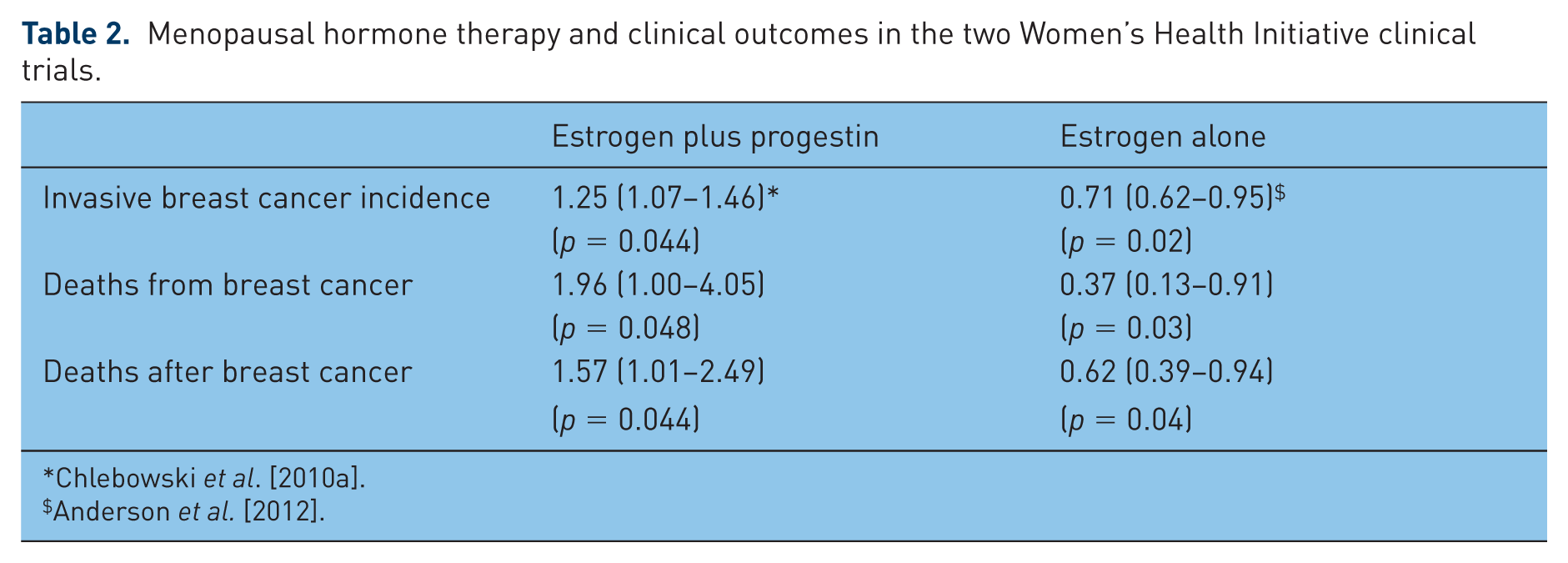
Taken together, these results suggested that estrogen plus progestin use stimulated breast cancer growth and hindered breast cancer diagnosis. Examination of the Kaplan–Meier curves finds an apparent lower breast cancer incidence in the initial years for estrogen plus progestin users (Figure 2), but the more advanced stage at diagnosis was suggestive of diagnostic delay rather than a true lower incidence. The adverse influence of estrogen plus progestin was not limited to hormone receptor positive cancers, rather all categories of breast cancer appeared to be increased although numbers in some subgroups were small (Table 1a) [Chlebowski et al. 2010a]. In addition, there was a suggestion of a greater adverse effect of combined hormone therapy on breast cancer in women initiating hormone therapy closer to menopause [Prentice et al. 2008, 2009; Chlebowski and Anderson, 2011; Chlebowski et al. 2013, Fournier et al. 2009]. Finally, with additional follow up, deaths from breast cancer, those directly attributed to the cancer, were found to be significantly increased (HR 1.96; 95% CI 1.00–4.04; p = 0.049) as were deaths after a breast cancer diagnosis [Chlebowski et al. 2010a] (Table 2).
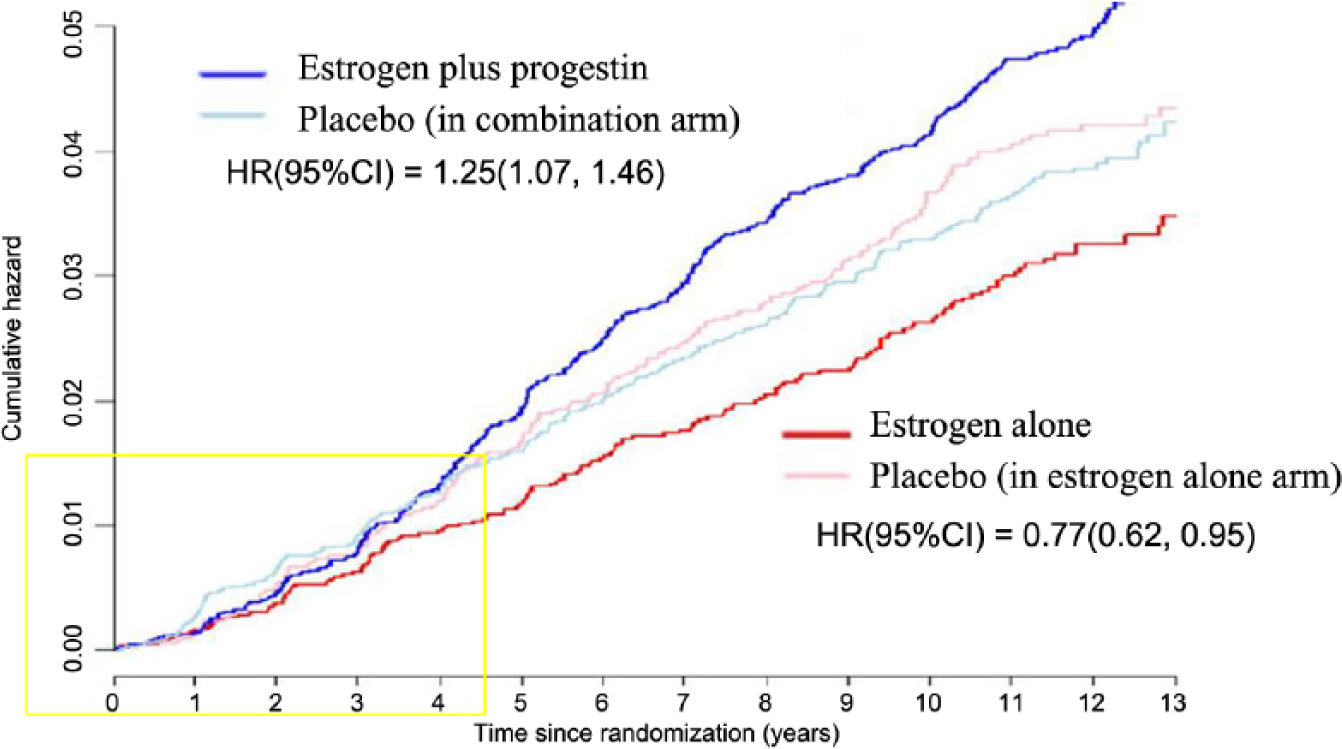
Estrogen plus progestin and estrogen alone, and breast cancer incidence in the Women’s Health Initiative trials [Anderson et al. 2012].
Breast cancer by hormone receptor and HER2 status by study group.
(a) Women’s Health Initiative estrogen plus progestin trial [Chlebowski and Anderson, 2012; Chlebowski et al. 2010a].
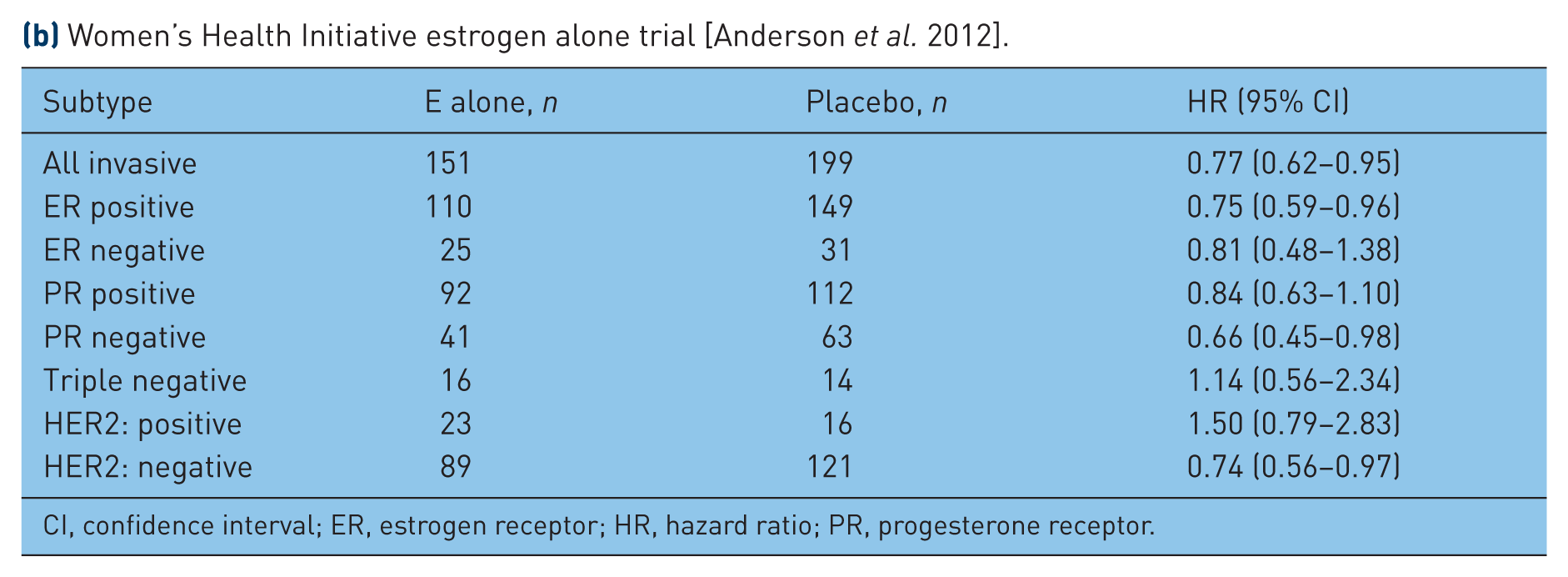
(b) Women’s Health Initiative estrogen alone trial [Anderson et al. 2012].
CI, confidence interval; ER, estrogen receptor; HR, hazard ratio; PR, progesterone receptor.
Menopausal hormone therapy and clinical outcomes in the two Women’s Health Initiative clinical trials.
WHI: estrogen alone trial
In the WHI randomized, placebo-controlled clinical trial evaluating estrogen alone in postmenopausal women with prior hysterectomy, intervention was ended after 7.1 years (mean) because of an increased stroke risk and an absence of overall clinical benefit (Figure 1) [Anderson et al. 2004]. The breast cancer findings related to estrogen alone use differed substantially from most of observational study reports where increased breast cancer risk was associated with longer duration estrogen alone exposure. At the end of intervention, there were fewer invasive breast cancers in the estrogen alone group (HR 0.80; 95% CI 0.62–1.04) but the difference was not statistically significant (p = 0.09) [Stefanick et al. 2006]. While more women in the estrogen alone group had mammograms with abnormalities (9.2% versus 5.5%; p < 0.001), the difference was mainly in reports suggesting short interval follow up [Stefanick et al. 2006; Chlebowski et al. 2010b]. After longer follow up (median 11.8 years), a statistically significant, lower incidence of invasive breast cancer emerged in the estrogen alone group (151 cases versus 199 cases; HR 0.77; 95% CI 0.62–0.95; p = 0.02) [Anderson et al. 2012], an effect seen both during and after intervention [Manson et al. 2013]. In addition, fewer women died from breast cancer in the estrogen alone group (HR 0.37; 95% CI 0.13–0.91; p = 0.03) (Table 2) [Anderson et al. 2012], providing support for the finding of a reduced breast cancer incidence.
The estrogen alone influence on reducing breast cancer incidence was less in women with time from menopause to hormone initiation (gap time) <5 years compared with women with longer gap times [HR 0.89 (95% CI 0.66–1.20) compared with HR 0.65 (95% CI 0.48–0.89)], but the interaction was not statistically significant (p interaction 0.13) [Anderson et al. 2012]. Similar results were seen in the Million Women Study cohort [Beral et al. 2011]. It appeared the effect of estrogen on reducing invasive breast cancer was mainly on infiltrating ductal carcinomas and estrogen receptor positive tumors (Table 1b) but interaction terms were also not significant for the subgroups examined.
The effect of estrogen alone in reducing breast cancer is unlikely to be a result of detection bias as annual mammography was protocol defined and the frequency of mammography was similar between randomization groups throughout [Stefanick et al. 2006; Chlebowski et al. 2010b]. Given the suggestion of the gap time effect, it is unclear whether women beginning estrogen alone use close to menopause would see a beneficial effect on breast cancer incidence. However, the randomized clinical trial findings provide no suggestion of an increased breast cancer risk even for women beginning estrogen alone with short gap times.
WHI observational study
To address the differences between the two WHI randomized hormone therapy trials and most observational studies regarding some breast cancer finding, analyses were conducted in the WHI observational study cohort of over 93,000 postmenopausal women. In this cohort, a population with similar eligibility to that in the randomized clinical trial was identified, namely, women with no prior hysterectomy and mammography within 2 years of entry.
In the WHI observational cohort, the incidence of invasive breast cancer was statistically significantly higher in estrogen plus progestin users compared with nonusers (0.60% versus 0.42%, annual rate, respectively; HR 1.55; 95% CI 1.41–1.70; p < 0.001) [Chlebowski et al. 2013]. Following up on findings from an earlier time from menopause analysis [Prentice et al. 2008, 2009], the highest breast cancer risk was seen for women initiating estrogen plus progestin at menopause (0 gap time; HR 1.73) with subsequent linear decrease in risk through gap times of 15 years (p < 0.001) [Chlebowski et al. 2013]. In addition, compared with non-users, estrogen plus progestin users had more deaths from [HR 1.32; 95% CI 0.90–1.93; not significant (NS)] and after breast cancer (HR 1.65; 95% CI 1.29–2.12), findings consistent with those in the WHI randomized trial.
In a series of analyses in the WHI observational study cohort, several factors likely influencing much of the differences seen between the randomized clinical trial and observational study results were identified [Prentice et al. 2008, 2009; Chlebowski et al. 2013]. One obvious issue is that observational studies still not uncommonly report breast cancer findings combining results from estrogen alone and estrogen plus progestin [Harlid et al. 2012; Hou et al. 2013; Cui et al. 2014]. Additional potential confounding factors often overlooked in cohort analyses include absence of mammogram clearance before cohort entry, limited information on mammogram frequency while on hormones and consideration of hysterectomy, bilateral oophorectomy and prior hormone therapy use.
In many older observational studies, mammography frequency was not determined. However, women undergoing screening mammography have more breast cancers identified and the cancers are more likely to be hormone receptor positive and be diagnosed at earlier stage compared with those identified in a population without screening [Kerlikowske et al. 2003; Hofvind et al. 2006]. Women using menopausal hormone therapy are more likely to be under medical supervision and consequently more likely to have screening mammography compared with women not using hormone therapy. For example, in the Nurse’s Health Study, mammography was not assessed during the first 12 years of follow up (1976–1988). When mammography frequency was reported, substantial differences between hormone therapy users and nonusers was seen. For women who reported no screening in the prior 2-year period, 31% of hormone users reported no screening in the subsequent 2 years, while 58% of hormone nonusers reported no screening in the subsequent 2-year period, a nearly 30% difference [Joffe et al. 2001].
Analyses to control for mammography frequency differences are complex [Gordon et al. 1993] and several approaches to adjust for this variable have been proposed [Weiss, 2003]. The Nurses’ Health Study investigators identified factors associated with screening mammography frequency to compute weights for integration in a breast cancer prediction model [Cook et al. 2009]. The approach, which used in a population of nurses with high screening frequency, led to attenuation of estimated combined hormone therapy influence on breast cancer risk, bringing findings in closer agreement with those from the WHI randomized clinical trial. Whether this innovative approach can be formally validated and/or provide reliable results in other settings remains to be determined.
Age at first use and the associated concept of time from menopause (gap time) initiation is another factor explaining the discrepancies between the randomized trials and observational studies [Prentice et al. 2008, 2009; Chlebowski et al. 2013; Fournier et al. 2009; Beral et al. 2011]. Women in the WHI hormone therapy trials tended to be older at the time of initial hormone therapy use than occurs in clinical practice. The observation that initiation of combined hormone therapy further from menopause is associated with lesser breast cancer risk supports the hypothesis that gene expression profile changes associated with the menopause-induced involution process is an effect modifier for hormone therapy [Jordan and Ford, 2011] and thus could explain some of the differences in magnitude between the observational studies and the WHI hormone therapy trials.
With respect to breast cancer characteristics, in analyses in the overall WHI cohort, similar to most observational studies [Antoine et al. 2004], more estrogen receptor positive cancers were seen in combined hormone therapy users compared with nonusers. However, in the WHI (as in most cohorts), women who were already on hormone therapy were allowed to enter the cohort. When women who initiated estrogen plus progestin use only after entering the cohort were examined, more aggressive breast cancers were seen in the estrogen plus progestin users [Chlebowski et al. 2013], a finding closely in line with that seen in the randomized trial. Thus, a woman on estrogen plus progestin for 5 or 6 years prior to entering the cohort had a ‘guarantee time’ that more aggressive estrogen receptor (ER) negative and HER2 positive breast cancer could not occur. If they did, the woman would not be eligible for participation in the cohort. Thus, observational studies which enter individuals who have been on combined hormone therapy for some time could not be expected to identify early aggressive breast cancers stimulated by estrogen plus progestin use.
Some observational studies report relatively low excess breast cancer risk associated with combined hormone therapy use in obese [Collaborative Group on Hormonal Factors in Breast Cancer, 1997; Rosenberg et al. 2008; Schairer et al. 2000] and in African American women [Hou et al. 2013; Hall et al. 2005], suggesting that the greatest risk of combined hormone therapy is in lean, non-black women. However, both obese women [Segars et al. 2014; Lethaby and Vollenhoven, 2011; Ritte et al. 2012] and black women [Segars et al. 2014] are substantially more like to have had a hysterectomy and thus be candidates for estrogen alone use. In addition, nearly half of the women in the US with hysterectomy have had a bilateral oophorectomy as well [Jacoby et al. 2009; Vorwergk et al. 2014], a procedure known to reduce breast cancer risk [Nichols et al. 2013]. These considerations could potentially explain some of the observational study findings regarding relative breast cancer safety for hormone therapy use in obese and black women, especially when estrogen alone and estrogen plus progestin results are combined. While no interaction was seen for breast cancer risk and estrogen plus progestin use in African–American and obese women in the WHI randomized trial during the 5.6 years’ active intervention, power was limited by small sample size within subgroups [Chlebowski et al. 2003]. Further analyses will address this issue after longer post-intervention follow up. At present, breast cancer safety for estrogen plus progestin use in black or obese women cannot be assumed.
Clinical implications
Following the initial presentation of the WHI estrogen plus progestin trial findings in 2002 [Rossouw et al. 2002], subsequent reports which reinforced the findings of increased breast cancer risk [Million Women Study Collaborators, 2003; Chlebowski et al. 2003] and no overall health benefit with combined hormone therapy use, clinical practice rapidly changed in the US and around the world. Using the US National Prescription Audit database, Hersh and colleagues reported that there were about 15 million hormone therapy users in the year prior to the WHI report, but in 2003, prescription had declined by 66% for estrogen plus progestin and by 33% for estrogen alone [Hersh et al. 2004]. During the same period, a substantial decrease in breast cancer incidence was seen in US women between the ages of 50 and 79 years and the decrease was attributed to the decline in hormone therapy use [Clarke et al. 2006; Ravdin et al. 2007].
A recent review summarized results from 40 reports examining relationships among menopausal hormone therapy use, mammography and breast cancer incidence. Although some variability in time lag and magnitude of effect was seen, the data were found to ‘convincingly support a direct association between decreasing hormone therapy use and declining breast cancer incidence’ [Zbuk and Anand, 2012]. However, others have found limitations of such studies as they were seldom based on individual data and reports frequently lacked breast cancer details [Antoine et al. 2014].
Analyses were conducted in the WHI estrogen plus progestin trial to address these issues. In the WHI trial evaluating estrogen plus progestin, all women currently on study medication were instructed to immediately stop their study pills on the same day in 2002 when the study results were published [Rossouw et al. 2002] and about 98% did do so [Ockene et al. 2005]. A subsequent analysis found breast cancer incidence to rapidly decline in the combined hormone therapy group while mammography frequency remained high and closely comparable between groups [Chlebowski et al. 2003]. The initial rapid decline likely represents the effect of a hormone therapy on already established but preclinical breast cancers (estrogen removal similar to oophorectomy). Questions remain regarding how the excess breast cancer risk related to estrogen plus progestin evolves over time after therapy is ended. In both the WHI randomized trial [Anderson et al. 2012] and the E3N cohort [Fournier et al. 2014], a significantly elevated risk was seen post stopping and further study of this issue is needed.
More recent analyses indicate the use of hormone therapy has continued to decline [Tsai et al. 2011; Sprague et al. 2012; Lagro-Janssen et al. 2010] through 2009. In a Dutch study involving women who presented with menopausal symptoms for the first time to primary case physicians, hormone therapy prescriptions decreased 37% in 2002 to 14% in 2003 and to only 4% in 2004 with an increase back to 14% in 2006 [Lagro-Janssen et al. 2010]. Subsequent reports from the WHI regarding differences between estrogen plus progestin and estrogen alone use [Stefanick et al. 2006; Anderson et al. 2012; Manson et al. 2013; Chlebowski and Anderson, 2012] for relative breast cancer safety and potential overall benefit for short-term estrogen alone use when initiated dose to menopause has had apparently limited impact on prescribing practice.
Reviewing all available randomized clinical trial evidence, the US Preventative Services Task Force recently concluded that menopausal hormone therapy is ‘currently not recommended’ to prevent chronic conditions, such as cardiovascular disease and cancer. In addition, they found that, among subgroup analyses, there were ‘no consistent differences by age and comorbidities’ sufficient to guide clinical use [Nelson et al. 2012].
Full discussion of the risks and benefits of estrogen plus progestin use for women with no prior hysterectomy or estrogen alone use for women with prior hysterectomy is beyond the scope of the current discussion. Several conclusions can be put forward with the emphasis on breast cancer findings. Estrogen plus progestin increases breast cancer incidence, interferes with breast cancer detection and increases deaths from breast cancer. The absolute risk for women beginning combined hormone therapy around menopause is small especially with relatively short-term use (2-4 years). However, on a population basis, a therapy which nearly doubled deaths from breast cancer [Chlebowski et al. 2010a] requires cautious use, especially since no coronary heart disease benefit was seen with estrogen plus progestin use, even in younger women. Clearly, some women with debilitating symptoms will benefit from combined hormone therapy use but this is an individualized decision that needs to be informed by an understanding of their broad health effects and revisited at regular intervals.
For short-term use of the duration in the WHI clinical trial, the situation for estrogen alone in women with prior hysterectomy is more straightforward. While subgroup analysis suggests a potential coronary heart disease benefit in younger women, the absolute benefit is small since coronary heart disease is uncommon in this age group. There was an overall reduction in breast cancer incidence seen with estrogen alone use, but the effect may be more pronounced in women initiating such therapy further from menopause [Prentice et al. 2009; Fournier et al. 2009; Anderson et al. 2012], not the usual prescribing pattern. In any event, a women with a prior hysterectomy can be assured that estrogen alone use for short duration management of climacteric symptom management is relatively safe. The safety of longer term use of estrogen plus progestin or estrogen alone is unknown.
Footnotes
Acknowledgements
We gratefully acknowledge the dedicated efforts of the WHI participants and of key WHI investigators and staff at the clinical centers and the Clinical Coordinating Center.
Program Office: (National Heart, Lung, and Blood Institute, Bethesda, Maryland) Jacques Rossouw, Shari Ludlam, Dale Burwen, Joan McGowan, Leslie Ford, and Nancy Geller. Clinical Coordinating Center: (Fred Hutchinson Cancer Research Center, Seattle, WA) Garnet Anderson, Ross Prentice, Andrea LaCroix, and Charles Kooperberg. Investigators and academic centers: (Brigham and Women’s Hospital, Harvard Medical School, Boston, MA) JoAnn E. Manson; (MedStar Health Research Institute/Howard University, Washington, DC) Barbara V. Howard; (Stanford Prevention Research Center, Stanford, CA); Marcia L. Stefanick; (The Ohio State University, Columbus, OH) Rebecca Jackson; (University of Arizona, Tucson/Phoenix, AZ) Cynthia A. Thomson; (University at Buffalo, Buffalo, NY); Jean Wactawski-Wende; (University of Florida, Gainesville/Jacksonville, FL) Marian Limacher; (University of Iowa, Iowa City/Davenport, IA) Robert Wallace; (University of Pittsburgh, Pittsburgh, PA) Lewis Kuller; (Los Angeles Biomedical Research Institute at Harbor-UCLA Medical Center) Rowan T. Chlebowski; (Wake Forest University School of Medicine, Winston-Salem, NC) Sally Shumaker. Women’s Health Initiative Memory Study: (Wake Forest University School of Medicine, Winston-Salem, NC) Sally Shumaker.
Conflict of interest statement
R.T.C. is a consultant for Novartis, Amgen, Novo Nordisk and Genetech. He is a speaker for Novartis and carries out review activities for Pfizer. G.L.A. has no conflicts of interest in preparing this article.
Funding
Funding for the WHI program findings described in this manuscript was as follows. The WHI program is funded by the National Heart, Lung, and Blood Institute at the National Institutes of Health, US Department of Health and Human Services through contracts HHSN268201100046C, HHSN268201100001C, HHSN268201100002C, HHSN268201100003C, HHSN268201100004C and HHSN271201100004C.
