Abstract
The introduction of long-acting beta agonists (LABAs) was considered a major advance in bronchodilator therapy for adult, as well as pediatric, patients with asthma. However, the use of LABAs has raised safety concerns, especially the potential for severe asthma exacerbations (SAEs) resulting in hospitalizations or even death. Meanwhile, the use of inhaled corticosteroids (ICSs), a cornerstone in the treatment of mild-to-severe persistent asthma, has been associated with growth suppression in children. The purpose of this review was to identify and discuss the major published safety studies surrounding LABA, ICS, and combined LABA/ICS usage in children. By way of a critical search for influential published clinical trials, meta-analyses, and observational studies, six studies relevant to the safety of LABA monotherapy, seven studies relevant to ICS monotherapy, and four studies on the subject of LABA/ICS combination usage were identified and reviewed. Based on the reviewed literature, the controversy surrounding these anti-asthma medications was clearly exposed. On the one hand, there is some evidence that LABA monotherapy may be associated with SAEs and asthma-related death, while ICS monotherapy may be associated with a higher risk of growth suppression. On the other hand, the concurrent use of a LABA with an ICS has been associated with positive outcomes including symptom reduction and reduced rate and severity of exacerbations. Further clinical research is warranted and has been called for by the US Food and Drug Administration.
Keywords
Introduction
Asthma, a chronic inflammatory disorder associated with variable airflow obstruction and bronchial hyperresponsiveness, is the most common chronic lower respiratory disease in children worldwide. It presents with recurrent episodes of wheeze, cough, shortness of breath, and chest tightness [Papadopoulos et al. 2012]. In the United States in 2011, there were an estimated 7.1 million pediatric patients suffering from asthma (representing a 9.5% prevalence rate) who accounted for 27.3% of the total asthma-patient population in the US [Centers for Disease Control and Prevention, 2011].
Children with asthma are limited in their day-to-day activities such as sports, school, and playing with pets, significantly reducing quality of life. In addition, children with asthma can be distressed by asthma symptoms and can become frightened by asthma attacks or exacerbations [Juniper, 1997]. Meanwhile, parents or caregivers of children with asthma are affected by both absenteeism from and lost productivity at work [Wang et al. 2005]. Epidemiologic studies tend to show that children do not grow out of asthma. Even if the symptoms disappear, asthma tends to reappear in adulthood [Braman, 2006]. Moreover, some children with mild asthma as children will have severe disease as adults [Braman, 2006]. Pediatric asthma is estimated to be responsible for approximately 13 million missed school days annually in the US [Akinbami et al. 2010]. In 2004, pediatric asthma was responsible for 7 million ambulatory visits (to either physician offices or hospital outpatient departments), 750,000 visits to emergency departments, 198,000 hospitalizations, and 186 deaths [Akinbami, 2006]. The annual cost of pediatric asthma in the US has been estimated at US$3.2 billion [Weiss et al. 2000]. The lifetime cost of pediatric asthma for a cohort of people born in the year 2000 in the US is estimated to be US$7.2 billion, including US$3.2 billion in medical costs and US$4 billion in work/productivity loss experienced by both the patients and their parents [Corso and Fertig, 2009]. Asthma disproportionately impacts low-income children in the US relative to the pediatric population in general, partly because of the substantial exposure poor children have to asthma triggers, such as dust and dust reservoirs, dust mites, roaches, rodents, molds, and tobacco smoke, that could exacerbate their condition [Krieger et al. 2000].
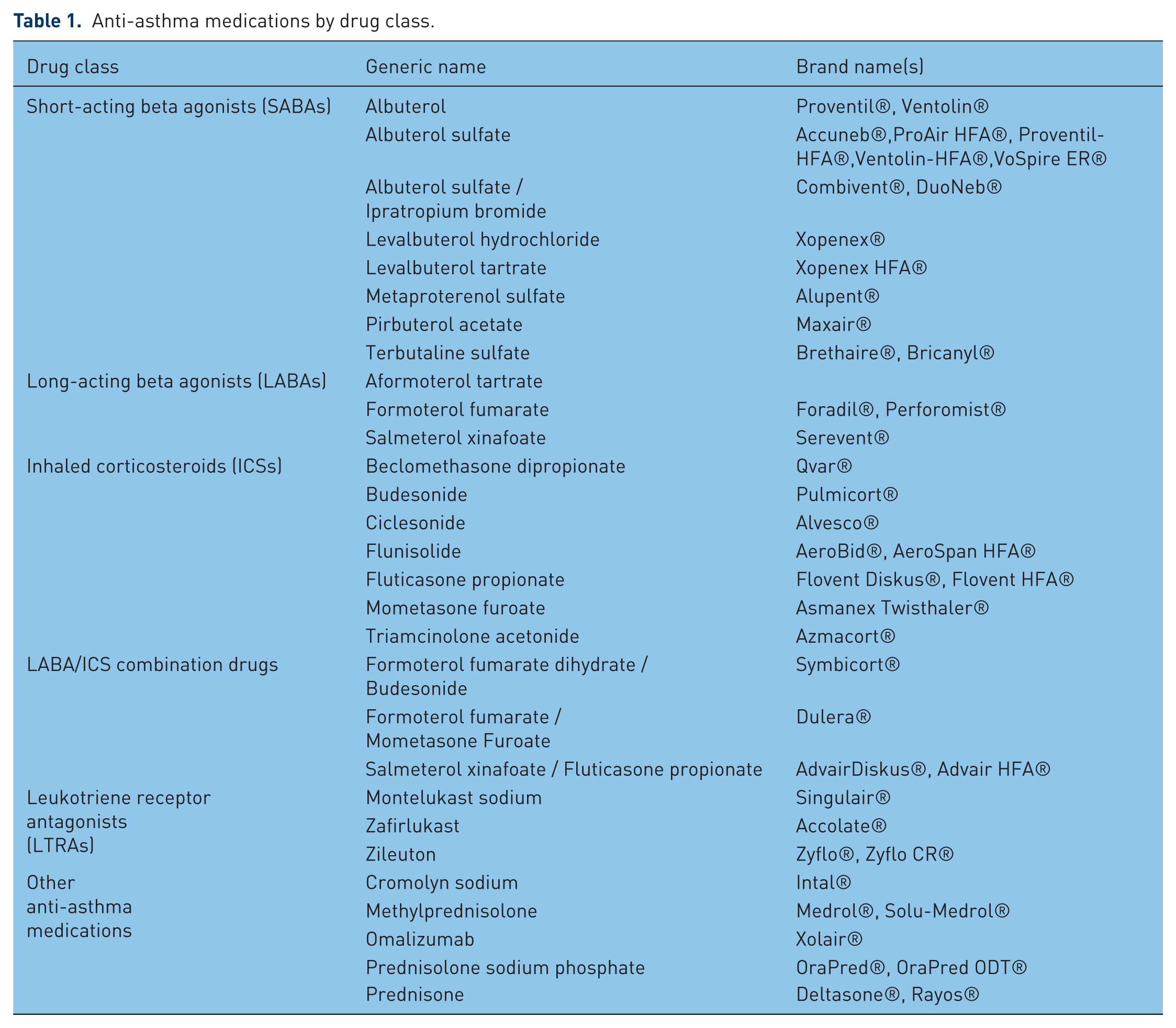
Pharmacotherapy is fundamental to the treatment of pediatric asthma. Its goal is the control of symptoms and the prevention of exacerbations with a minimum of drug-related side effects. Short-acting inhaled β2 agonists (SABAs) are the most commonly used reliever medications for pediatric patients with asthma for the treatment of intermittent and acute asthma episodes. For children with intermittent asthma, a SABA is the only asthma medication they may take. For children with persistent asthma, controller medications are used to reduce bronchial inflammation. Inhaled corticosteroids (ICSs) are used as first-line controller treatment. The patient is generally started at a low dose (200 µg BDP equivalent); if control is inadequate, the dose may be increased. Leukotriene receptor antagonists (LTRAs) are an alternative first-line therapy for pediatric patients between the ages of 5 and 11 years old with mild persistent asthma who cannot use ICSs or an add-on therapy for those children whose asthma is not adequately controlled with ICS treatment alone [Bacharier et al. 2008]. The benefit of LTRAs has been shown in children as young as 10 months, although Singulair was approved for patients 12 months and older for asthma [Straub et al. 2005]. For patients who remain uncontrolled, a long-acting β2 agonist (LABA) may be added; in step therapy, at an earlier step for children aged 5–11 years, and a later step for children 0–4 years old [U.S. Department of Health and Human Services, 2007]. LABA/ICS combination drugs are also available and are an important treatment option at least for a subpopulation of older children [Lemanske et al. 2010]. For a list of anti-asthma medications, categorized by drug class, see Table 1.
Anti-asthma medications by drug class.
In terms of pediatric asthma management, physicians face a delicate balance between drug safety and efficacy to achieve optimal therapeutic benefits for individual patients. Although there are side effects, such as tachycardia, dizziness, and jitteriness, associated with SABAs, these drugs tend to have generally favorable safety profiles, even in very young patients [Bacharier et al. 2008]. LTRAs are also generally safe and well-tolerated with an overall incidence of adverse events similar to placebo [Knorr et al. 2001]. The most commonly encountered side effects with LTRAs are headache and gastrointestinal upset [Bacharier et al. 2008].The importance of LTRAs in the management of pediatric asthma as well as safety issues surrounding their use are discussed in a number of recent studies [Montuschi et al. 2007; Montuschi, 2008, 2011; Montuschi and Peters-Golden, 2010; Montuschi and Barnes, 2011].
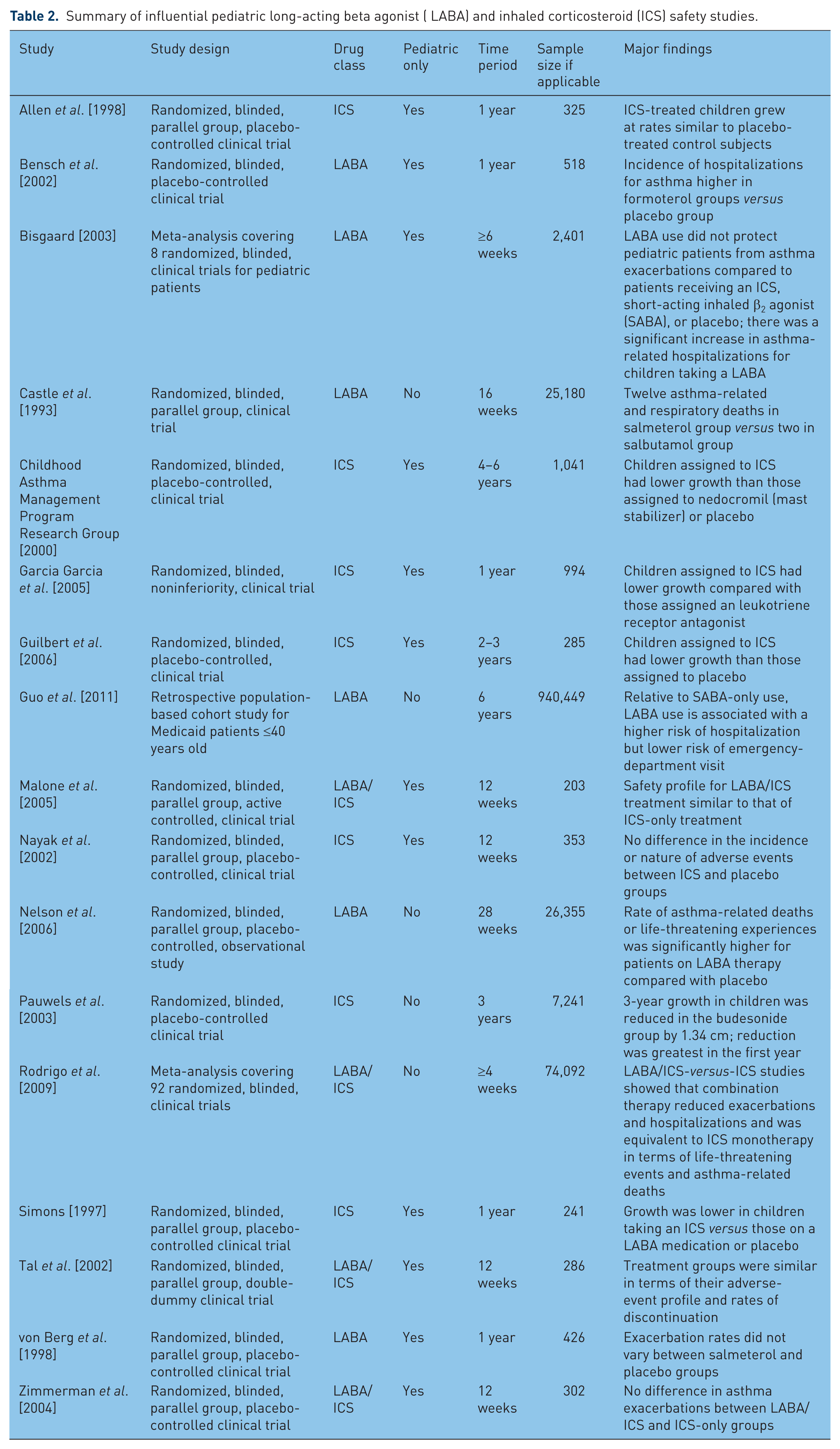
Since their introduction, however, LABAs have been associated with very serious safety issues. Several studies have presented evidence of significant risk of exacerbations and asthma-related death associated with LABA use. Meanwhile, although serious side effects are unusual, ICSs are associated with at least temporary growth suppression [Bacharier et al. 2008]. This study describes and discusses the current state of the evidence regarding LABA and ICS safety in the pediatric patient population, by reviewing some of the most impactful clinical trials, meta-analyses, and observational studies. As will become clear from this review, some of the important studies included both adults and children, while some were pediatric-specific. None of the studies involved children younger than 4 years old, hence, safety evidence has been extrapolated to that very young patient population in various treatment guidelines. For a list of studies reviewed, see Table 2.
Summary of influential pediatric long-acting beta agonist ( LABA) and inhaled corticosteroid (ICS) safety studies.
LABA studies
The Serevent Nationwide Surveillance (SNS) study was a randomized, double-blind, clinical trial with parallel groups over 16 weeks, which was designed to compare the safety of salmeterol (LABA) and salbutamol (SABA) in asthma treatment. There were 25,180 subjects, with approximately 6% between the ages of 12 and 18, who participated in treatment. The SNS study found no statistically significant differences between the two groups in terms of risk of hospital admission, risk of life-threatening event, or risk of other serious event. However, there were 12 asthma-related deaths in the salmeterol group versus 2 in the salbutamol group [Castle et al. 1993].
The Salmeterol Multicenter Asthma Research Trial (SMART) was a randomized, double-blind, parallel-group, placebo-controlled, observational study over 28 weeks involving 26,355 asthmatic patients at least 12 years old. The trial found that the rate of respiratory-related deaths or life-threatening experiences was not significantly different (relative risk [RR] = 1.40, 95% confidence interval [CI] 0.91–2.14) for salmeterol versus placebo patients. However, there was a statistically significant difference between salmeterol and placebo for asthma-related deaths or life-threatening experiences (RR = 1.71, 95% CI 1.01–2.89) [Nelson et al. 2006].
In a much smaller, but pediatric-focused, multicenter, randomized, double-blind, and parallel study by von Berg and colleagues, a total of 426 children aged 5–15 years were randomly assigned to receive salmeterol 50 µg or placebo twice daily. The study design included a 2-week baseline period, a 12-month treatment period incorporating a 2-week period ‘off treatment’ after 6 months, and a 2-week follow-up period at the end of the study. The study found that 109 (53%) patients assigned a placebo experienced an asthma exacerbation compared with 137 (62%) patients in the salmeterol group; this difference was not statistically significant (p = 0.2) [von Berg et al. 1998].
In a randomized, double-blind, placebo-controlled, multinational, multicenter study, Bensch and colleagues examined the efficacy and safety of formoterol dry powder in pediatric asthma patients. A total of 518 children aged 5–12 years were randomly assigned to receive 12 or 24 µg of formoterol dry powder or placebo twice daily for 12 months. Results showed 68 (40%) patients in the formoterol 24 µg group, 67 (39%) patients in the formoterol 12 µg group, and 71 (41%) patients in the placebo group experienced asthma exacerbations. Again, there was no statistically significant difference in the number of asthma exacerbations across the treatment groups [Bensch et al. 2002].
In a pediatric-specific meta-analysis, Bisgaard examined the asthma-related exacerbation rates of LABA drugs in pediatric asthma patients. Eight studies (including those by Bensch and colleagues and von Berg and colleagues mentioned above) were included, with a total of 2,401 patients 4–17 years old with mild-to-severe persistent asthma [Bisgaard, 2003]. The conclusion was that LABA treatment with either salmeterol or formoterol, among children taking an ICS, provided no additional protection from asthma exacerbation compared to children receiving a SABA or placebo. A significant increase in asthma-related hospitalizations (the RR ranged from 3.3 to 21.6 across the three studies that included hospitalizations as study endpoints) was, however, observed for children taking a LABA.
In a large, retrospective, cohort, database study with close to 1 million Medicaid patients (out of which there were 767,715 children less than 18 years old) in 10 states over 6 years, Guo and colleagues came to a similar conclusion regarding hospitalizations. Compared with patients taking a SABA only, LABA use in patients with pre-existing asthma was found to be positively associated with hospitalizations and intubations. On the other hand, the estimated hazard ratio for emergency-department visits was 0.68 (95% CI 0.63–0.73) for those same patients with pre-existing asthma. After controlling to the greatest extent possible for patient severity, patients taking a LABA were less likely to go to the emergency room than those on a SABA only [Guo et al. 2011].
ICS studies
A number of studies have reported a direct association between ICS use and short-term growth suppression. Simons and colleagues conducted a randomized, double-blind, placebo-controlled, parallel-group, 1-year study on the use of ICSs by 241 pediatric asthma patients. During the study period, height on average increased by 3.96 cm in the beclomethasone group (200 µg twice a day), which was significantly (at least statistically) less than both the average growth in the placebo group (5.04 cm, p = 0.018) and the average growth in the salmeterol group (5.40 cm, p = 0.004) [Simons et al. 1997].
The Childhood Asthma Management Program Research Group found similar results through a double-blind, placebo-controlled study. A total of 1,041 children, aged 5–12 years old, with mild-to-moderate persistent asthma, were randomly assigned to receive budesonide (200 µg), nedocromil (8 µg, a mast cell stabilizer), or placebo twice daily. The study participants were treated for 4–6 years. The children taking budesonide grew on average 22.7 cm, statistically significantly (p = 0.005) less than the average growth of 23.8 cm among the children assigned the placebo [Childhood Asthma Management Program Research Group, 2000].
Pauwels and colleagues again found lower average growth among asthmatic children taking an ICS during a randomized, double-blind clinical trial. A total of 7,241 asthma patients with mild persistent asthma in 32 countries were enrolled in the study. Among them were 1,974 children aged 5–10 years old and 1,221 children and adolescents aged 11–17 years old. They were randomly assigned to receive budesonide or placebo once daily for 3 years. Results showed that the mean growth difference was 0.43 cm (p < 0.0001) less per year in the budesonide group than in the placebo group [Pauwels et al. 2003].
Garcia Garcia and colleagues examined the efficacy and safety of fluticasone versus montelukast in a 12-month, multicenter, randomized, double-blind, clinical trial. Out of a total of 994 patients with mild persistent asthma who were 6 to 14 years old, 499 were assigned fluticasone (100 µg, twice daily), and 495 received montelukast (5 µg, once daily). The results showed that the average growth was statistically significantly lower (p = 0.018) in the fluticasone group (5.81 cm) than in the montelukast group (6.18 cm) over a year’s time [Garcia Garcia et al. 2005].
Finally, in a randomized, double-blind clinical trial, a total of 285 pediatric asthma patients were randomly assigned to receive either fluticasone (88 µg, twice daily) or placebo for 2 years, then were followed for an additional year without medication. Compared with the placebo group, patients in the fluticasone group grew an average of 1.1 cm less (p < 0.001) over the first 24 months. However, the height increase was 0.7 cm less (p = 0.008) by the end of study period [Guilbert et al. 2006].
Conversely, some studies have shown no statistically significant association between ICS use and growth suppression. Allen and colleagues conducted a double-blind, placebo-controlled study with 325 pediatric asthma patients, age range of 4 to 11 years old, who were randomly assigned to receive a low dose of fluticasone (50 µg), a higher dose of fluticasone (100 µg), or a placebo twice daily for 1 year. The results showed that, by the end of study period, mean height increased 5.94 ± 0.16 cm in the 50 µg fluticasone group, 5.73 ± 0.13 cm in the 100 µg fluticasone group, and 6.15 ± 0.17 cm in the placebo group (p = 0.308, overall) [Allen et al. 1998].
Other research has studied the association between ICSs and other adverse events. Nayak and colleagues examined the efficacy and safety of beclomethasone dipropionate, formulated as an extra fine aerosol (hydrofluoroalkane-134a [HFA]-BDP), in children with moderate persistent asthma. A 12-week, randomized, multicenter, double-blind, placebo-controlled, parallel-group study was conducted, with 353 children aged 5–12 years with persistent asthma enrolled. After a 2-week run-in period, children were randomized to receive low-dose HFA-BDP (80 µg), high-dose HFA-BDP (160 µg), or HFA-placebo twice daily. No statistically significant difference in the rate of adverse events was found across the groups [Nayak et al. 2002].
LABA/ICS combination studies
Although SMART was not designed to assess the effect of ICS use on asthma-related death or life-threatening experiences, post hoc analyses of the study results revealed that the higher risk of asthma-related deaths or life-threatening experiences held only for those not using an ICS at baseline. For those using an ICS, there was no statistically significant difference between the salmeterol and placebo groups [Nelson et al. 2006].
The meta-analysis by Rodrigo and colleagues also compared LABA/ICS combination therapy with ICS-only therapy. Their results showed that combination therapy could reduce exacerbations (RR = 0.73; 95% CI 0.67–0.79) compared with the ICS-only group. The results also showed there was no statistically significant difference between the LABA/ICS combination group and the ICS-only group (RR = 2.96; 95% CI 0.50–17.57) in asthma-related death [Rodrigo et al. 2009].
Zimmerman and colleagues conducted a double-blind, placebo-controlled, randomized, parallel-group, multicenter study. A total of 302 Canadian asthmatic children, aged 6–11 years, already taking an ICS medication, were enrolled in a 12-week study. The children were randomly assigned to receive either formoterol 4.5 µg, formoterol 9 µg, or placebo twice daily. A total of 19 asthma exacerbations occurred during the study, with no statistically significant difference across the groups [Zimmerman et al. 2004].
Malone and colleagues conducted a pediatric-specific randomized, double-blind, active-controlled, parallel-group study to compare the safety of inhaled fluticasone propionate with or without inhaled salmeterol. A total of 203 children aged 4–11 years were randomly assigned to receive salmeterol/fluticasone propionate (50/100 µg) or fluticasone propionate (100 µg) only twice daily for 12 weeks. The results showed that three (3%) patients in the salmeterol/fluticasone propionate group experienced asthma exacerbations, compared with eight (8%) patients in the fluticasone-propionate-only group [Malone et al. 2005].
Tal and colleagues conducted a double-blind, randomized, parallel-group, multicenter study, with 286 enrolled children, age 4-17 years old, with asthma. The asthmatic children were randomized to receive formoterol/budesonide (4.5/80 µg, twice daily) or budesonide (100 µg, twice daily). There were eight (5.4%) children in the formoterol/budesonide group who had asthma exacerbations, compared with four (2.9%) patients in the budesonide-only group [Tal et al. 2002], essentially the opposite of the result from the study by Malone and colleagues.
Discussion
Based on the results of the studies discussed in this article, as well as on its own research, the United States Food and Drug Administration (FDA) issued a safety announcement on February 18, 2010, regarding the use of LABAs in adults and children. The FDA’s announcement included the following: ‘Pediatric and adolescent patients who require the addition of a LABA to an inhaled corticosteroid should use a combination product containing both an inhaled corticosteroid and a LABA, to ensure compliance with both medications’ [Food and Drug Administration, 2010]. At that time, the FDA also required a Risk Evaluation and Mitigation Strategy (REMS) for these products, which included a revised Medication Guide for patients and class-labeling changes. Although LABAs remain on the market, the FDA encourages them to be used at the lowest possible doses, for the shortest amount of time, and only in combination with ICSs [Food and Drug Administration, 2010].
Meanwhile, a number of studies have now been devoted to understanding the effect of ICS utilization on adult height, arguably a more important endpoint than a short-term growth rate. Doull summarized the results from a number of longitudinal studies, concluding that the vast majority of asthmatic children will attain a normal adult height [Doull, 2004]. With few side effects at recommended dosages and growth suppression a temporary phenomenon, the benefits of ICS treatment seem to outweigh the costs.
In its February 2010 safety announcement, the FDA asked that the LABA manufacturers conduct additional clinical trials to evaluate further the safety of LABAs when used in combination with ICSs. It did not feel that there was enough known about their safety in children. This decision was consistent with the conclusion reached by Cates and colleagues in their recent review of reviews: ‘Regular combination therapy is likely to be less risky than monotherapy in children with asthma, but we cannot say that combination therapy is risk free. There are probably an additional three children per 1000 who suffer a non-fatal serious adverse event on combination therapy in comparison to ICS over 3 months. This is currently our best estimate of the risk of using LABA combination therapy in children and has to be balanced against the symptomatic benefit obtained for each child.’ [Cates et al. 2012, p. 2].
Concerns over the safety of asthma medication obviously make treatment more difficult. Moreover, pediatric asthma can be hard to diagnose in the first place because a number of other childhood conditions, such as rhinitis, sinusitis, and respiratory tract infections, can have symptoms similar to those caused by asthma and can co-occur with asthma. In children aged 6 and older, physicians diagnose asthma using the same tests, including spirometry, used to diagnose the disease in adults [Mayo Clinic, 2013]. For younger children, there is a focus on the frequency and severity of symptoms including wheeze, nocturnal cough, exercise-induced symptoms, persistence of cough with colds, and exposure to environmental factors including allergens [Bacharier et al. 2008]. Because asthma is characterized by clinical variation in presentation throughout the course of the disease, there is a strong need for noninvasive assessment of airway inflammation. Progress is being made in researching the use of exhaled breath concentrate (EBC) to study concentration of leukotrienes and other biomarkers in asthma [Montuschi, 2009]. However, EBC requires additional study before being introduced to everyday clinical practice.
Although we have focused on asthma treatment in developed countries, asthma is a common disease in both high- and low-income countries, with prevalence increasing rapidly in the latter. Although prevalence may be leveling off somewhat in the former, this is a recent event after decades of rising prevalence and severity [Greenwood, 2011]. In a large, global, multicenter study, the highest prevalence was found in the United Kingdom, Australia, New Zealand, and the Republic of Ireland, followed by countries in North, Central, and South America. The lowest prevalence was found in Eastern Europe, Albania, Greece, China, Taiwan, Uzbekistan, India, Indonesia, and Ethiopia [Pearce et al. 2007]. The high cost of ICSs is a major barrier to persistent asthma management in low- and middle-income countries [International Union Against Tuberculosis and Lung Disease and the International Study of Asthma and Allergies in Childhood, 2011].
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest statement
Over the past 5 years, Dr Guo has received grant support from Bristol-Myers Squibb, Eli Lilly, Genentech, Janssen Ortho-McNeil, and Novartis. Dr Kelton has received grant support from Eli Lilly, Janssen Ortho-McNeil, and Novartis. Ms Xia, Mr Bian, Dr Xue, and Dr Wigle have no conflict of interest to report. The opinions and conclusions expressed in the article are solely those of the authors.
