Abstract
Peripheral nerve injuries significantly impair quality of life due to limited regenerative capacity, which is affected by factors such as neuroma formation, injury severity, scarring, and comorbidities. The Muscle-in-Vein (MIV) repair technique, consisting of a vein filled with skeletal muscle fibers, has emerged as a promising alternative to nerve autografts. This approach supports regeneration by providing growth factors, guiding axonal growth, enhancing Schwann cell migration, and limiting scar and neuroma formation. However, its clinical use is currently restricted mainly to short gaps in sensory digital nerves, and the biological mechanisms underlying its effectiveness remain incompletely understood. In this study, we investigated the role of muscle fibers in the early phases of nerve regeneration, with a particular focus on vascularization. An 8 mm gap in rat median nerves was repaired using the MIV technique and analyzed at 3, 7, 14, and 21 days post-injury. Immunofluorescence analysis demonstrated complete macrophage infiltration and well-organized vascularization throughout the entire graft as early as 7 days post-injury. Consistently, RNA sequencing at early time points revealed significant enrichment of pathways associated with vascular development and identified key angiogenesis-related genes. Notably, our findings indicate partial anastomosis between vessels originating from the nerve stumps and those within the muscle component of the graft. These results suggest that the success of muscle-in-vein nerve repair strategy may be due to an early vascularization process mediated by the synergistic contribution of both muscle and vein.
Keywords
Introduction
Every year, thousands of medical cases report a reduction in patients’ life quality due to peripheral nerve damage, resulting in higher healthcare costs.1,2 In case of an improper nerve repair process, neuroma formation might occur. Neuromas are benign growths of scar tissue nerve fibers that form as the nerve attempts to heal after damage. The diagnosis of neuroma is usually characterized by pain sensation, while the treatment options depend on the severity and location and may include a conservative approach, such as anti-inflammatory medications or, in severe cases, surgery to remove the neuroma. 3
One of the most effective non-neural grafting approaches to facilitate injured human nerve regeneration is the Muscle-in-Vein (MIV) technique, which utilizes a vein enriched with fresh skeletal muscle fibers. This technique consists of harvesting a superficial vein with a diameter comparable to that of the nerve requiring reconstruction and a small strip of striated muscle fibers inserted within the vein to prevent the vein from collapsing over distances greater than 1–2 cm. 4
In vitro and in vivo studies have demonstrated that skeletal muscle fibers can release supporting factors essential for the survival of neuronal cells, including Glial cell-derived neurotrophic factor (GDNF), and Brain-derived neurotrophic factor (BDNF),5,6 but also Vascular endothelial growth factor (VEGF), 7 an angiogenesis stimulator, and Neuregulin 1 (NRG1),8,9 a trophic factor supporting Schwann cell proliferation, migration, and survival.
The MIV technique has been used in both preclinical10–14 and clinical studies, yielding promising results in humans. It has demonstrated impressive outcomes in repairing gaps in sensitive digital nerves,15–19 emerging as a good alternative to autograft. 20 The MIV technique offers several advantages, including the presence of the basal laminae of muscle fibers acting as natural guidance channels for regenerating axons 21 and migrating Schwann cells. 22 The vein walls also act as barriers against scar tissue ingrowth while avoiding axon sprouting. 4 Finally, MIV is a valuable tool for the surgical treatment of painful post-traumatic neuroma-in-continuity of sensory digital nerves.16–23
This promising outcome encourages further research into this model, which remains undervalued due to the limited number of preclinical and clinical investigations. 24 The positive results of the MIV technique have prompted our research group to evaluate fresh skeletal muscle as a possible enrichment of chitosan conduits in a preclinical study 25 demonstrating that the muscle-in-tube (MIT) technique supports long-term regeneration with effectiveness comparable to the autograft repairing method.
Recent research has emphasized the critical role of vascularization in the regenerating nerve process. The formation of blood vessels is essential not only for supplying oxygen and nutrients to regenerating tissues, but also for supporting the migration of Schwann cells colonizing the gap between the proximal and the distal nerve stumps.26–28 Importantly, hypoxia within the nerve gap is sensed by infiltrating macrophages, which respond by releasing VEGF-A, thereby inducing a polarized vascular network that guides Schwann cell migration. Accordingly, macrophage-specific inactivation of Vegf-a severely impairs nerve gap vascularization and, consequently, Schwann cell migration. 26 Additionally, recent studies showed that these newly formed vessels can guide mesenchymal cells in remodeling the initial fibrotic scar into protective stromal tubes. 27
The present study aimed to evaluate the processes that characterize the early stages of nerve regeneration following injury and repair using the MIV technique in a preclinical model. Through molecular and immunofluorescence analyses, we sought to clarify how the presence of muscle enhances the regenerative process, with a particular focus on vascularization.
Materials and methods
In vivo surgical procedure
18 adult female Wistar rats (200–250 g, ENVIGO, Milan, Italy) were used for the different analyses. Animals were housed under standardized conditions (12 h light/dark cycle and ad libitum access to water and food). Animals were allocated randomly to groups/time points. The surgical procedures were performed under deep anesthesia induced by i.p. injection of ketamine (100 mg/kg) and xylazine (10 mg/kg) and conducted under a high-magnification surgical microscope. All procedures complied with the National Institutes of Health guidelines, the Italian legislation for the protection of animals used for scientific purposes (DL26/14) and the European Communities Council Directive (2010/63/EU). The experimental protocol was approved by the Bioethical Committee of the University of Torino and the Italian Ministry of Health (approval code: N692/2020_PR (E669C.64); expiry date: 21/7/2025). The MIV technique, described in 19934 begins with the preparation of the graft. An oblique skin incision is made in the anterior ventral region to expose the epigastric vein and the adductor muscle (Supplemental Figure S1). A longitudinal strip of fresh skeletal muscle tissue, 8 mm long, is dissected and inserted into a 10 mm long epigastric vein fragment to obtain an MIV graft. The rat median nerve is exposed and transected to establish an 8 mm nerve gap, immediately repaired using the fresh MIV graft, inserting 1 mm of each nerve stump inside one of the two vein ends, and fixing with stitches (Supplemental Figure S1).
The grafts, interposed between the two nerve stumps, were withdrawn at 3 and 7 days post-repair and immediately frozen in dry ice for RNA extraction and sequencing, alongside healthy veins and muscles (n = 3, for each time point and analyzed group), while samples at 7, 14, and 21 days post-repair were collected for morphological analysis (n = 3, for each time point). Each biological replicate corresponded to an independent animal. Control healthy nerves were obtained from the 8 mm long median nerves cut before the grafting repair. Animals were sacrificed using an anesthetic overdose.
The MIT technique was previously described. 25 Briefly, an 8 mm median nerve segment was removed, and 10 mm long chitosan-based tube (Reaxon® Nerve Guides; Medovent GmbH, Germany), enriched with a longitudinal strip of fresh skeletal muscle tissue (from the same animal), was used to bridge the nerve defect by inserting 1 mm of the two nerve ends inside the conduit. The grafts, interposed between the two nerve stumps, were withdrawn 7 days after the repair for morphological analysis (n = 3).
Immunolabeling
Regenerated nerves (n = 3, for each time point) were collected at 7, 14, and 21 days after the surgery. Each collected sample was fully sectioned into 50 µm-thick transverse cross-sections and analyzed by immunofluorescence as previously detailed. 28 Different combinations of primary antibodies (Supplemental Table S1) were used to label specific cellular components, followed by a 40 min incubation with 4,6-diamidino-2-phenylindole DAPI (1:1000, ThermoFisher, Waltham, MA, USA). Sections were placed on a silanate glass slide and cover-slipped with mounting medium Mowiol (Sigma-Aldrich, Merck, Darmstadt, Germany). After drying, samples were acquired using a Leica SP5 confocal microscope (Leica Microsystems, Wetzlar, Germany).
Three-dimensional reconstruction
The three-dimensional reconstruction method was previously detailed. 29 Briefly, 10 consecutive slices of 50 µm thickness each were stained and imaged through the confocal microscope (Leica SP5 Microsystems, Wetzlar, Germany) equipped with a 20× oil immersion objective (HC PL FLUOTAR 20× NA = 0.5). Images were acquired with a voxel size of 0.387 × 0.387 × 1 µm. The stacks were aligned in TrakEM2. 30 By dividing the total number of planes (263) by the 500 µm total depth, we calculated a shrinking along the z-axis of 54% and corrected the voxel size accordingly to 0.38 µm × 0.38 µm × 1.9 µm. Manual segmentation was performed to reconstruct the trajectory of the individual vessel depicted in the figure.
Vessels and skeletal muscle fiber quantification
One entire single section in the middle of the graft was analyzed for each animal (n = 3, for each time point). Morphometric analysis was carried out in a blind manner. The total vessel number was manually counted using Fiji-ImageJ software for each evaluated section. The radial distribution and relative local density analyses were performed to identify three concentric areas: F1 (inner, blue), F2 (middle, orange), and F3 (outer, green; Supplemental Figure S2(A)). Once the total area ofOne entire single sectionthe cross-section approximating a circle shape was identified and quantified, it was possible to calculate the total radius (r) and, consequently, ⅓ and ⅔ of the total radius to obtain three concentric areas: F1 = Π(⅓r)2; F2 = Π (⅔r)2–F1; F3 = Π r2-F2–F1. The fluorescence quantification of myosin signal (Supplemental Figure S2(B)), labeling muscle fibers, was performed using Fiji-ImageJ software. After an optimization of brightness/contrast value, despeckle and remove outlier (2.0 pixels radius, 70 threshold) commands were applied to achieve a better noise/signal ratio. Using the Threshold tool, the myosin signal was identified, while the Analyze Particles tool was employed to quantify the selected areas, excluding particles ⩽50 μm2. This method allowed us to quantify the total amount of muscle fibers in each cross-section, while the DAPI signal, with transmitted light acquisition, was used to quantify the total section area of each sample (data not shown). GraphPad Prism 10.2.2 software (Boston, Massachusetts, USA) was used to perform statistical analysis of immunofluorescence quantification data. After confirming the normal distribution with the Shapiro-Wilk test, the statistical significance was determined by an ordinary one-way ANOVA test followed by Tukey’s multiple post-hoc comparisons. Data were presented as mean ± SEM; *p ⩽ 0.05, **p ⩽ 0.01, ***p ⩽ 0.001.
RNA extraction and sequencing
RNA was isolated using the TRIzol Reagent (Life Technologies) following the manufacturer’s instructions. TRIzol reagent was added directly to the frozen nerve samples, then mechanically dissociated with a pestle. RNA samples were quantified and assessed for quality and purity using the Agilent 5400 system. Only samples with a RIN (RNA Integrity Number) >6 were selected for sequencing.
Illumina RNA Sequencing (RNA-Seq) was performed by Novogene SRL using the 150-nucleotide paired-end reads protocol, generating approximately 20 million reads per sample. The non-directional library construction was performed as follows: mRNA was purified from 350 ng total RNA using poly-T oligo-attached magnetic beads. After fragmentation, the first-strand cDNA was synthesized using random hexamer primers, and the second-strand cDNA synthesis was performed with dTTP. The subsequent steps included end repair, A-tailing, adapter ligation, size selection, amplification, and purification. The library was checked using Qubit and real-time PCR, and size distribution was analyzed using a bioanalyzer. Libraries were pooled and sequenced on Illumina platforms, based on the effective library concentration and the required data volume. The original image data file was converted into sequenced reads using CASAVA base recognition. Sequencing data were filtered by removing reads with adapter contamination or uncertain nucleotides (N >10%) or those where low-quality nucleotides (Base Quality <5) constituted more than 50% of the reads. Alignments were performed using HISAT2 31 v2.0.5 against the Ensembl Rattus norvegicus genome (rnor_6_0_gca_000001895_4). The tool was applied with default parameters.
Bioinformatics analysis pipeline
DESeq2 v1.48 was applied with default parameters to perform the differential expression analysis. Differentially expressed genes (DEGs) were defined as those associated with a median FPKM (Fragments Per Kilobase of transcript per Million mapped reads) ⩾1, an absolute fold-change (|log2FC|) ⩾ 1 and an adjusted p-value ⩽ 0.05 (Supplemental Table S2(A)).
Metascape v3.5 32 tool was used for Gene Ontology (GO) enrichment analysis. Pathways with a false discovery rate (FDR) cut-off of 0.0001 were considered significant. The analysis was performed using the rnorvegicus_gene_ensembl_mRatBN7.2_10116 genome reference. Endothelial cell subtype signatures were obtained from single-cell RNA sequencing data. 27 Singscore v1.26 33 was applied with default settings to compute for each sample a single-sample score of each signature. The differences among scores were statistically evaluated using the Wilcoxon Rank-Sum test. To assess the mouse-to-rat conservation levels, we retrieved the level of homology and orthology confidence from Ensembl, observing that 77.1% of analyzed genes had a high orthology confidence score and a percentage of identity >80% (Supplemental Table S2(B)).
Raw and processed RNA-Seq data were deposited in Gene Expression Omnibus with the identifier GSE298178.
Results
Blood vessels are detectable throughout the entire MIV graft as early as 7 days post-repair
To investigate the early regeneration phenomena occurring within the graft, 8 mm gap injuries were repaired through the MIV technique. The regenerating nerves were harvested at 7, 14, and 21 days post-injury. Samples were collected and fully sectioned into 50 µm-thick transverse cross-sections for immunofluorescence analysis. For each regeneration time point, five tissue sections, spaced 2 mm apart and distributed from the proximal to the distal nerve stumps, were selected to assess the presence of vascular structures, evaluated using the Reca1 marker. Several vessels are detectable throughout the entire graft as early as 7 days post-repair, with positive signals here in red (Figure 1). Vessel architecture remains observable at 14 and 21 days post-injury. To determine whether there was an intact vascular structure connecting the internal part of the graft with the external environment, a 3D reconstruction was performed at the proximal side of the MIV graft 7 days post-injury. The 3D reconstruction of the vascular architecture, labeled with the Reca1 marker (red), reveals a continuity of vessels linking the proximal nerve stump (upstream region) and the grafted region containing muscle fibers (downstream region), as shown in Figure 2, where a selected vessel spanning both regions is highlighted in green, and muscle fibers are clearly evident in the downstream region only. Furthermore, vessels appear arranged in a parallel orientation to each other.

Vascular distribution within the MIV grafts. Representative serial cross-sections of the graft stained for Reca1 (red, marking vasculature), collected every 2 mm at 7, 14, and 21 days post-injury and repair within the MIV graft; scale bar: 200 μm.
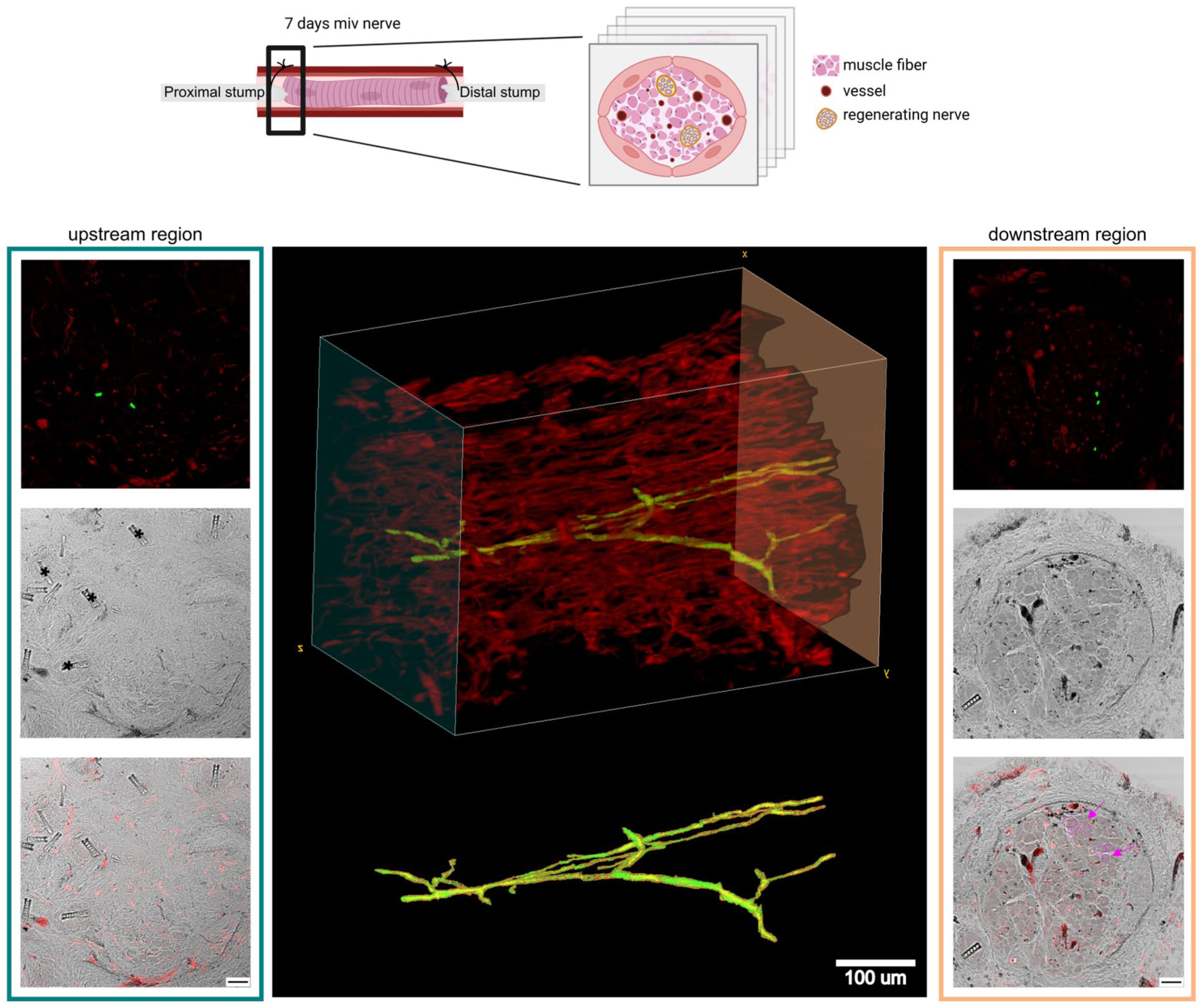
3D reconstruction of the proximal region of the 7-days MIV graft. Reca1-positive vasculature is shown in red, while a selected vessel exhibiting extensive branching and spanning the entire graft is highlighted in green. Transmitted light images of the first and last sections are shown in the blue and orange boxes, respectively. The presence of muscle tissue in the last section is highlighted in purple, while absent in the first section; scale bar: 20 μm. Upper panel created with BioRender.com.
To confirm the regeneration process, the axonal elongation within the graft was assessed at 14 and 21 days, when it is well established. Qualitative observations indicate that Schwann cells migrate within the graft, following both the blood vessels and the muscle fibers. However, the substrate type on which Schwann cells migrate does not appear to influence axonal elongation. As shown in Supplemental Figure S3, the elongating axons (neurofilament/NF, white labeled) are similarly organized (purple arrow) around the muscle fibers (myosin, red labeled) and surrounding (orange arrow) blood vessels (Reca1, green labeled).
Vascular density is maintained over time after muscle degradation, and correlates with macrophage presence
To evaluate the behavior of vessels in relation to muscle presence, muscular tissue and vessel amount were quantified on two adjacent cross-sections in the center of the graft at each time point (Figure 3). Qualitative observation revealed a decrease in the absolute amount of muscle fibers and vessels over time; however, no statistical difference was detected (data not shown), while a significant reduction in the ratio between muscle and cross-section area over time was observed (p < 0.05, Figure 3(A)). However, this did not correspond to a decrease in the ratio between the number of vessels and cross-section area (Figure 3(B)), which is maintained approximately at 0.06 vessels/100 µm2.
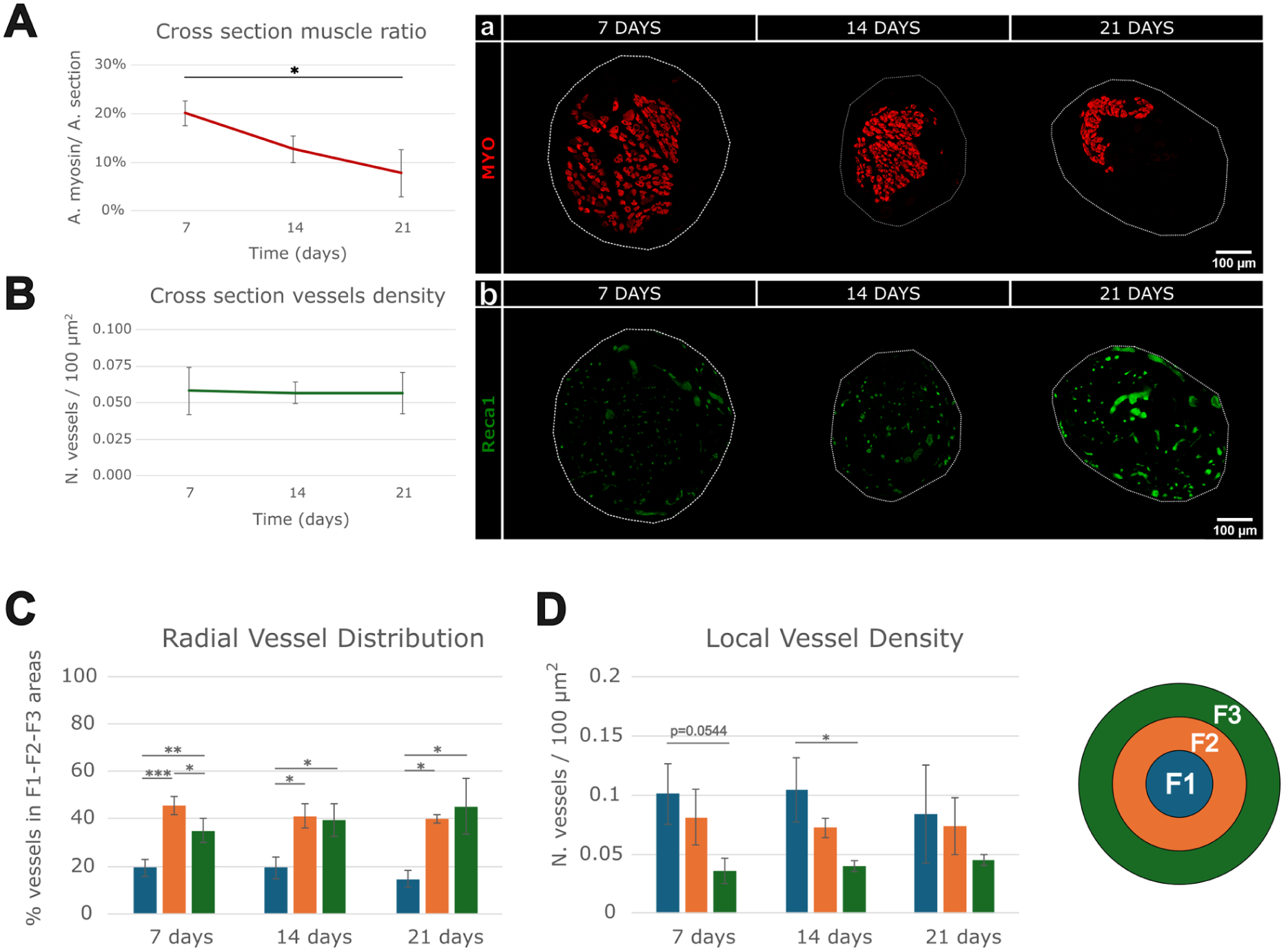
Evaluation of the origin and maintenance of vessels related to muscle: (A) quantitative analysis of the ratio between the total muscle area (A. myosin) and cross-sectional area (A. section) at 7, 14, and 21 days post-injury and repair; (a) representative images of myosin signal (red) labeling muscular fibers, (B) quantitative analysis of vessel density over time; (b) representative images of Reca1 signal (green) labeling blood vessels, (C) radial vessel distribution: vessel percentage in three concentric areas: F1, F2, F3, and (D) local vessel density: vessel number/100 μm2 in three concentric areas: F1, F2, F3. The three areas are labeled F1 (blue), F2 (orange), and F3 (green); mean ± SEM are reported; *p ⩽ 0.05. **p ⩽ 0.01. ***p ⩽ 0.001.
A quantitative evaluation of vessel distribution in relation to the distance from the origin point of the cross-section was performed, and the local vessel density was calculated. Assuming a circular shape, the cross-section area was divided into three concentric regions: F1, F2, and F3 (Supplemental Figure S2) and the percentage of total vessels within each concentric region was quantified (Figure 3(C)), showing a radial distribution pattern of vessels: approximately 20% of the total vessels were concentrated in F1 (inner) region, while the remaining vessels were equally distributed between F2 (middle) and F3 (outer) regions. At the three time points, the vessel percentage in F2 and F3 regions was significantly higher than in the F1 region, while at 7 days, the vessel percentage in the F2 region was significantly higher than in the other regions.
Additionally, the vessel density in each region was calculated (Figure 3(D)): at 7 days, a weak significance (p-value = 0.054) was observed between F1 and F3 regions; at 14 days post-repair, the F1 region exhibited a significantly higher local vessel density compared to the F3 region. At 21 days, this decreasing density over the regions was no longer observed.
Because of the critical role that immune response plays in maintaining vascular structures and remodeling, 34 the behavior of vessels in the central cross-sections of MIV grafts was assessed in relation to macrophage presence. To achieve this, a comparison was made at 7 days post-injury and repair between MIV and MIT grafts, where chitosan tubes were enriched with muscle fibers. 9 While the vein undergoes permeability during its degeneration, allowing an early crossing of cellular components, the chitosan tube applied in this study was characterized by lasting stability and non-adhesive properties 35 and, due to its manufacturing method, does not form porous structures,36,37 preventing cellular infiltration. A clear qualitative association was observed between the lack of macrophages and the absence of vascular structures in the MIT graft. Specifically, only a small number of Iba1-positive macrophage (purple) were detected, which corresponded to sparse Reca1 staining (green), suggesting a markedly reduced vascular network at 7 days post-injury and repair (Figure 4). In contrast, the MIV graft exhibited a widespread distribution of macrophages, often located near the widely distributed blood vessels.

Macrophage infiltration of MIT and MIV 7-day grafts. Two representative areas from a middle cross-section of each graft are labeled with Iba1 (magenta-macrophages), Reca1 (green-vessel) and DAPI (blue-nuclei). Different amount of Iba1 signal is shown correlating with the Reca1 signal presence in MIT graft cross-section; scale bar: 20 μm.
Transcriptomic analysis of MIV grafts revealed differential expression of vascular and angiogenesis-related genes
To investigate the biological processes occurring within the MIV graft at 3 (MIV_3d) and 7 (MIV_7d) days post-injury and repair, RNA-Seq was performed on biological triplicates. Because muscle fibers and veins were predominant in the early stages, RNA-Seq was also conducted on freshly collected healthy muscle, vein tissues, and healthy nerve.
A Principal Component Analysis (PCA) was performed to assess the variability between the MIV_3d and MIV_7d groups. While the MIV_3d samples were more tightly grouped compared to the MIV_7d group, no overlap was observed along the highest variance axis, PC1 (34.82%), confirming a distinct divergence between the two experimental groups (Figure 5(A)). Additionally, a PCA comparison was conducted among the MIV groups and healthy muscle, vein and nerve tissues (Figure 5(B)), to provide a global overview of transcriptional heterogeneity across tissue types. The rationale for including these tissues in the sequencing analysis was specifically to compare markers of cell populations present in healthy nerve, muscle and vein with those present in MIV samples.

RNA-Seq analysis: (A) PCA of MIV-treated samples, (B) PCA of all analyzed samples, (C) volcano plot showing the genes differentially expressed in MIV-7d compared with the MIV-3d group. The dashed lines represent the threshold of 0.05 on the adjusted p-value and of 1 on the absolute log2FC, (D) top 20 most significant terms enriched for the DEGs by the Metascape analysis, (E) top 10 most up-regulated and down-regulated genes in each pathway related to vascular development and related log2FC, (F) selected genes associated with vessel formation and vessel development analyzed in MIV-7d compared with MIV-3d. Color intensity reflects the magnitude and direction of fold change (red, up-regulated, blue down-regulated genes). Genes not significantly differentially expressed (NO-DEG) are also included to provide a comprehensive overview of the analyzed pathways (more details in Supplemental Table S3).
The comparison between MIV_7d and MIV_3d identified 1415 Differentially Expressed Genes (DEGs), with 828 up-regulated and 587 down-regulated (Figure 5(C) and Supplemental Table S2(A)). To further explore the biological processes underlying the early stages of regeneration, an enrichment analysis was performed using the GO database via Metascape (Figure 5(D)). The top 20 most significantly enriched pathway clusters included terms associated with the cell cycle and vasculature development. Five clusters were directly or indirectly associated with vascularization. The heatmap of each vascularization-related cluster is reported in Figure 5(E). A focused evaluation was carried out manually, based on the literature,38–43 with particular attention to gene families playing key roles in vascularization: Vegfs (Vegf-A, -B, -C, Pgf), Eno1, Vegfrs (Vegfr-1, -2, -3), Pdgfs (Pdgf-A, -B, -C, -D), Pdgfr-A/B, Angpt-1/2/Tie2 axis, Angptl1, Fgf-1/2, and the Igf1/Igfr families. In the comparison between MIV_7d versus MIV_3d, critical genes involved in angiogenesis, such as VegfA, Pgf, Igfb3, and Eno1, were significantly down-regulated; nevertheless, their expression levels remained within the range (but higher) observed in healthy nerve tissue, where vascularization is physiologically maintained. In contrast, genes involved in endothelial progenitor cell differentiation and in the maintenance of newly formed blood vessels (Pdgf-D, Pdgfr-B, Tie2 and, with a lower fold change, Pdgf-C and Vegf-B), were up-regulated (Figure 5(F) and Supplemental Table S3), suggesting ongoing vascular maturation. Moreover, although angiogenic factors such as Angpt-1/2 and Fgf2 were not significantly regulated, Fgf1 (also known as aFgf) and Fgfr2 were up-regulated. Similarly, Igf1r and Igf2r were not significantly regulated, while Igf-1 was up-regulated. Furthermore, Angptl1, Igfbp-4, -5, and -6 (known inhibitors of angiogenesis) were up-regulated, while Igfbp-3 (a proangiogenic factor) was down-regulated. Finally, it has been evaluated whether markers consistent with endothelial cell differentiation toward arterial or venous phenotypes were present. Genes associated with arterial phenotypes (e.g. GJa4, Sema3g, Sox17) were up-regulated, showing patterns consistent with arterial phenotype development switch.
Comparative gene signatures of endothelial cell subtypes in MIV grafts
To examine vascular remodeling during nerve regeneration, the gene expression signature of 10 endothelial cell (EC) subtypes, based on single-cell RNA sequencing data, 27 was evaluated (Supplemental Table S2(B)). The gene signature of 9 out of 10 EC subtypes was identified, except for the Intermediate group (Figure 6(A)). The results showed different trends across the various specialized EC subtypes in the MIV_3d and MIV_7d groups (Figure 6(B)). Although not statistically significant, there was a positive increment in the prevalence of EC_CAP-PV+/– and EC_VEIN-PV+/–, while a decrease was observed in the signature of immature ECs (EC_immat), proliferating cells (EC_prolif), as well as tip cells (EC_tip) associated with new growing vessels. The same analysis was also performed on healthy tissues: muscle, vein, and nerve (Figure 6(A)). Interestingly, in the muscle tissue, which is most abundant in the early time point, the gene signatures of all different endothelial cells were lower compared to the MIV_3d group, especially in EC_immat, EC_prolif, and EC_tip.

Endothelial cell subtype signature evaluation: (A) boxplot of the single-sample scores computed for each endothelial cell subtype signature in healthy tissues (muscle, nerve, vein) and MIV groups, (B) detailed boxplot of the single-sample scores computed for each endothelial cell subtype signature in MIV groups. At the top, the pie chart represents the percentage of genes that were positively (red, up-regulated) or negatively (blue, down-regulated) differentially expressed, or not in the analysis; p-value by the Wilcoxon Rank-Sum test.
Discussion
This study investigates the early stages of regeneration within MIV-grafts. This cost-effective surgical technique is particularly compelling due to its proven success in humans,15–19,23 not only in promoting nerve regeneration, but also in preventing neuroma recurrence and in alleviating pain. 23
This study found complete vascularization of the MIV-graft by 7 days post-injury and repair. In contrast, recent studies on empty chitosan conduits28,44 used to repair the same gap length, showed only partial vascularization at 7 days, reaching completion at 14 days. At the same time, revascularization after autologous nerve graft repair demonstrated that blood vessels from the stumps of the injured nerve can form anastomoses with the donor nerve vasculature, particularly between 4 and 7 days post-repair. 44 In this study, the 3D reconstruction of 10 consecutive cross-sections, covering 0.5 mm of length connecting the proximal stump to the graft at 7 days post-repair, shows a continuous vascular network. Moreover, the evaluation of vessel distribution shows that the vessel density within the graft was approximately 0.06 vessels/100 µm2, a value consistent with that observed in rat skeletal muscle tissues. 45 The radial distribution of these vessels remains stable over time, despite the reabsorption of muscle tissue, with a homogeneous local density, suggesting a successful integration of the vessels into the newly formed neural tissue. While this uniform distribution contrasts with what is observed in the empty tube model, 28 where vessels tend to concentrate at the periphery, it is consistent with the vascular distribution described in muscle tissues. These findings, together with reported literature,46,47 support the hypothesis that anastomoses might form between the vascular structures of the stump and the endogenous vessels within muscle tissue, providing a potential explanation for the complete vascularization observed as early as 7 days. Although blood vessels are known to support Schwann cell migration, 26 our findings suggest that muscle-derived vessels do not significantly accelerate long-term Schwann cell migration. Similarly, regrowing axons do not exhibit a clear preference for growth along these vessels. Previous studies have shown that muscular fibers may provide an alternative pathway for Schwann cell migration, 22 which may explain the limited impact of the vascular architecture on the migration process. Instead, these vessels seem to be more critical for resolving the hypoxic state within the graft, thus supporting cellular activities necessary for nerve repair, as further supported by gene ontology analysis and discussed later.
A second critical observation emerges from the comparison between MIV and MIT grafts. At 7 days post-surgery, MIT grafts showed absent or only partially formed vascular structures. Notably, regions lacking vascularization also exhibited few or no macrophages. Although our data do not provide functional evidence of causality, the association between reduced macrophage presence and limited angiogenesis is consistent with previous reports describing a pro-angiogenic role of macrophages during tissue regeneration. 26 These findings suggest that macrophages may contribute to the early vascularization process, while also highlighting the importance of the venous conduit in supporting this regenerative microenvironment. While previous studies have demonstrated that the vein can prevent fibroblast infiltration and subsequent scar formation, 48 our findings show that it also facilitates macrophage migration to the lesion site, thus supporting the maintenance and sprouting of muscle local vessels. This raises important questions about how the cellular permeability of biomaterials affects regeneration, particularly in nerve repair, where reconnecting a long nerve gap is a slow process, initially supported by the immune system.26,49 Recent advances in tissue engineering, including modification to chitosan, aimed at creating pores of sufficient size to house cells, thereby enhancing tissue regeneration. 50 Future research could focus on characterizing degenerated vein properties to identify morphological features to functionalize biomaterials, shifting the concept of conduit as a cellular isolated environment to a selective interface that enables communication with the surrounding tissue.
As a third consideration, the sequencing results from this study offer a detailed and supportive overview of the molecular events that occur during the early stages of nerve injury and repair. Notably, the development of vasculature emerges as a key factor in these early stages, as highlighted by the gene ontology enrichment analysis. By employing RNA-seq, a set of differentially expressed genes (DEGs) was identified in the comparison between the 3-day (MIV_3d) and 7-day (MIV_7d) post-injury samples. These findings contribute valuable insights into nerve regeneration, particularly regarding vascularization and endothelial cell behavior. Our data showed that in the MIV_7d group, the vascularization was complete and the vessels were undergoing early stages of maturation. In fact, the downregulation of critical pro-angiogenic factors 38 such as VegfA, Pgf, Eno1 and Igfbp-3, combined with the upregulation of Igfbp-4, -5, and -6, and Angptl1, known as inhibitors of angiogenesis, 39 suggests that the system might be transitioning from an early, active pro-angiogenic phase toward a more controlled and stabilized vascular state. Notably, although these pro-angiogenic genes were down-regulated in MIV_7d compared with MIV_3d, their expression levels remained within the physiological range observed in healthy nerve tissue, consistent with the maintenance of a mature vascular network rather than the induction of new vessel formation. This interpretation is supported by the immunofluorescence data showing completed vascularization in the MIV_7d group. Additionally, the up-regulation of genes associated with arterial phenotypes, such as GJa4, Sema3g, and Sox17, 40 reflects a transition from initial endothelial cell proliferation and sprouting to later stages of vascular differentiation and stabilization. Although the upregulation of pro-angiogenic factors as Fgf1, and Igf1, may seem to contradict the aforementioned hypothesis, FGF1 is known to be an essential factor expressed in DRG neurons, where it plays a role in the maintenance and survival of the nerve, 42 as well as IGF-1 is secreted from various sources such as Schwann cells, monocytes and skeletal muscle and is known to promote regenerative processes. 43 Moreover, endothelial cell differentiation toward arterial and venous phenotypes is closely linked to cell cycle regulation, which was the pathway most significantly enriched in DEG analysis. A qualitative assessment of gene signatures associated with different endothelial cell subtypes also revealed that the signature of proliferating endothelial cells was up-regulated in the MIV_3d group compared to healthy muscle tissue, but was down-regulated when compared to the MIV_7d group. Furthermore, in the MIV_7d group, an increase trend in differentiated endothelial cell genes was observed, particularly those associated with arteries and veins, suggesting that most of the endothelial cell proliferation takes place within 3 days post-injury and repair, while the following differentiation, contributing to restore the blood flow and to solve the hypoxia condition, occurs between 3 and 7 days post-injury and repair. Regrettably, the evaluation of the signatures of the various endothelial subtypes yields only qualitative information, likely due to the limited number of samples in each group. In fact, a major limitation of this preclinical study is the high surgical complexity. The rat median nerve is much smaller than a human nerve, requiring specialized microsurgical instruments and a skilled microsurgeon, which necessitates minimizing the number of surgeries. Moreover, the rat model presents only a few veins suitable for the MIV technique without compromising the well-being of the animals. These challenges are substantial, which accounts for the limited number of preclinical studies investigating this approach. 24 For this reason, we suggest further investigation in larger animal models, such as sheep, which has been recently employed in peripheral nerve regeneration studies. 51
Conclusions and further directions
These findings advance our understanding of the MIV technique and its potential in nerve repair. The muscle, supported by the vein, appears crucial in maintaining vascularization, resolving hypoxia and enabling communication with the external environment. Notably, an effective method for treating painful peripheral nerve neuromas consists of relocating the resected nerve stump into an adjacent vein with an established blood flow.52–55 This flow is thought to dilute factors that contribute to neuroma formation. Our findings suggest that the MIV technique, through early revascularization, may similarly help prevent neuroma formation and reduce neuropathic pain.
By elucidating the biological mechanisms involved in the early stages of nerve regeneration using the MIV technique, this research offers valuable insights for improving and innovating tissue engineering approaches, ultimately leading to more effective treatments for peripheral nerve injuries. Ideal conduits should facilitate the influx of soluble growth factors, nutrients and supportive cells from the surrounding environment while allowing waste removal.56,57
A more in-depth evaluation of degenerating vein wall properties could further refine tissue engineering strategies. Additionally, the data suggest that the use of fresh skeletal muscle represents a promising strategy as a pre-vascularized conduit if provided with adequate permeable support, as could be a vein.
Supplemental Material
sj-docx-1-tej-10.1177_20417314261437253 – Supplemental material for Early vascularization as a key feature of muscle-in-vein grafts for peripheral nerve repair
Supplemental material, sj-docx-1-tej-10.1177_20417314261437253 for Early vascularization as a key feature of muscle-in-vein grafts for peripheral nerve repair by Federica Zen, Giulio Ferrero, Alessandro Crosio, Debora Molinaro, Giulia Ronchi, Stefano Geuna, Giovanna Gambarotta and Stefania Raimondo in Journal of Tissue Engineering
Footnotes
Acknowledgements
Chitosan conduits were supplied by Kerimedical (Mainz, Germany). All authors have read the journal’s policy on disclosure of potential conflicts of interest, and all authors have disclosed any financial or personal relationship with organizations that could potentially be perceived as influencing the described research. All authors have read the journal’s authorship statement.
Glossary/Abbreviations
ORCID iDs
Author contributions
Conceptualization: F.Z., G.F., G.R., S.G., G.G., S.R.; Project administration: F.Z., G.G., S.R.; Investigation: F.Z., D.M., G.R., A.C.; Data curation and Methodology: F.Z., D.M., G.F.; Formal analysis and Visualization: F.Z., G.F.; Supervision: S.G., G.G., S.R; Writing-Original Draft, F.Z.; Writing-Review and Editing, F.Z., G.R., G.F., S.G., G.G., S.R., A.C., D.M.; Funding acquisition: G.R., G.G., S.R.
Funding
The author disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The publication was funded by the University of Turin.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The raw data supporting the findings of this study will be made available by the authors upon reasonable request. Raw and processed RNA-Seq data were deposited in Gene Expression Omnibus with the identifier GSE298178.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
