Abstract
The plasticity of blood mononuclear cells (MCs) and their role in vascular remodeling have been the focus of many studies; however, their in vitro differentiation efficiency remains poorly understood. Herein, we demonstrate that the inflammatory response accelerates the efficiency of MCs differentiation into endothelial-like cells through chemical cues in vitro. RT-PCR and RNA sequencing revealed that the differentiated cells exhibited upregulated pathways associated with vascular remodeling and regeneration. In contrast, MCs collected from normal blood showed a differentiation bias toward macrophages. Notably, under inflammatory conditions, primarily monocytes transitioned into the CD14++/CD16+/CD163+ subset, which contributed significantly to vascular remodeling. This transition was triggered by inflammation, as confirmed by in vitro cytokine treatment.

Introduction
Vascular tissue regeneration in transplanted decellularized tissue-engineered vascular grafts (TEVGs) is accomplished through the action of host cells. Endothelial progenitor cells (EPCs) are an appropriate cell source for the reconstruction of vascular tissues in cell-free TEVGs.1,2 The therapeutic efficacy of EPCs in limb ischemia has been investigated, and favorable clinical outcomes have been reported. 3 The blood circulation of EPCs may still be limited (0.01%–0.1%) for lumen endothelium reconstruction.4,5 Moreover, cell migration from transanastomotic sites may be counterintuitive in clinical scenarios with grafts longer than 1 cm. 6
Smith Jr. et al. recently reported that endothelialization of a 50 cm long graft was accomplished without cellular ingrowth from the anastomotic site in a large animal model and that blood circulating monocytes contributed to endothelial regeneration.7–9 Previously, our group reported that REDV peptide-modified acellular grafts with an inner diameter of 2 mm and lengths of 20–30 cm exhibited long-term patency in a mini-pig femoral-femoral bypass model. The luminal surface of the graft was covered within 7 days after transplantation, with cells expressing CD14, CD16, and CD163 representing the mononuclear cell subpopulation. The captured cells infiltrated into the tunica media layer and constructed CD31 and α-smooth muscle actin (α-SMA) positive layers after 3 months. 10 The evidence indicated that circulating MCs contribute significantly to vascular regeneration in vivo.
It is well documented that primary MCs are non-dividing cells when cultured in vitro under normal conditions. 11 Cells captured on the lumen surface of TEVGs within three days post-transplantation showed endothelial cell-like behavior in vitro. 10 Moreover, blood MCs have been reported to accelerate angiogenesis within inflammatory and hypoxic microenvironments such as tumors.12–14 Based on these reports, we hypothesized that MCs plasticity and its role in vascular remodeling are influenced by the inflammatory microenvironment.
In this study, we evaluated the effects of acute inflammation on MCs differentiation for vascular remodeling using chemical cues in vitro. Mini-pig tail arterial blood was processed using red blood cell (RBC) lysis to isolate peripheral blood mononuclear cells (PBMC), which were sorted using autologous platelet-rich plasma (PRP) on collagen I-coated Petri dishes to obtain MCs. We demonstrated that MCs can effectively differentiate into functional endothelial cell (EC)-like cells that form tubular networks. Additionally, these MCs can potentially differentiate into smooth muscle cell (SMC)-like cells. Notably, MCs collected after acute inflammation exhibited higher attachment behavior and a greater response to external chemical cues than pre-inflammatory MCs. In addition, in vitro treatment of MCs with inflammatory cytokines (such as interleukin (IL)-6) results in accelerated differentiation toward EC-like cells.
Results and discussion
In vitro differentiation of MCs cells toward EC- and SMC-like cells
Whole blood was collected from the minipig tail artery, followed by RBC lysis using an RBC lysis buffer (PluriSelect Life Science, Germany) to obtain a PBMC suspension. The PBMC suspension was incubated in a collagen-I coated petri dish with autologous PRP (0.5 mL) for 12 hours at 37°C as shown in

In-vitro differentiation of blood mononuclear cells toward EC-like cells: (a) Schematic showing whole blood processing for PBMC collection, and immunostaining images of attached mononuclear cells (MCs) indicating CD14 positive cells, (b) FSC/SSC analysis reveals that 70%, 26%, 4%, and 3% of the attached MCs expressed CD14/16, CD14/34, CD14/45, and CD14/31, respectively, (c) Immunostaining images of cells described in panel B, (d) MCs were treated with ROCK/CHIR, as outlined in the protocol timeline. The corresponding phase contrast and immunostaining images show differentiated cell morphology and cluster formation, (e) The differentiated cells were positive for CD14/31, and (f) The fraction ratio of vWF, CD31, and CD144-positive cells steadily increases over time, indicating effective differentiation toward EC-like cells. Data were expressed as means ± standard deviations of one representative experiment out of three experiments carried out in triplicate (one-way ANOVA, Fraction ratio of vWF: F3,8 = 8.5, p = 0.027; CD31: F3,8 = 6.9, p = 0.017; CD144: F3,8 = 4.2, p = 0.024, Tukey’s HSD post hoc test, *p < 0.05, **p < 0.01, ***p < 0.001).
Unattached cells were removed by gentle washing with phosphate-buffered saline (PBS) and attached MCs were collected using trypsinization. Most MCs were positive for CD14 as shown in the immunostaining images. Flow cytometry revealed that the PBMC population contained 57%, 20%, 2%, and 1.5% fractions of CD14/16, CD14/34, CD14/45, and CD14/31-positive cells, respectively. In contrast, attached MCs showed 70%, 26%, 4% and 3% fractions of CD14/16, CD14/34, CD14/45, and CD14/31-positive cells, respectively, as shown in Figure 1(b) and (c). Moreover, only 3%–4% of the attached MCs were CD14/45/31-positive, indicating the presence of endothelial progenitor cells (EPCs). These data suggest that MCs were primarily composed of monocytes and a subset of granulocytes, and were predominantly positive for CD14 and CD16.
The MCs were cultured on collagen-I coated Petri dishes in porcine EC growth medium (5 mL) with a ROCK inhibitor (Y-27632, 10 µM) for 3 days, and the resulting cells were named ROCK_treat. The ROCK inhibitor Y-27632 has been used in previous studies to promote the spread and differentiation of human pluripotent stem cells toward ECs. 16 On day three, Y-27632 was removed, then a WNT pathway activator (CHIR-99021, 5 µM) was added for the next 3 days, and the resulting cells were named ROCK/CHIR. CHIR is necessary to coax MCs toward functional EC-like cells by inducing VE-cadherin junction and tube formation. 17 Subsequently, the medium was replaced with EC growth factor medium without ROCK or CHIR for another 3 days, and the resulting cells were named EC-like cells (Figure 1(d)).
Phase-contrast images revealed that initially cultured MCs displayed a spindle-like morphology that gradually transitioned to a cobblestone morphology as the cells proliferated and formed tight colonies, characteristic of ECs. Immunostaining (CD14/CD31) images showed that CD31 expression at cell junctions increased with the protocol timeline (Figure 1(e)). The fraction of vWF, CD31, and CD144-positive cells gradually increased to 11%, 14%, and 19%, respectively in EC-like cells, as shown in Figures 1f and S1.
Moreover, MCs differentiated toward SMC-like cells according to the timeline shown in Figure S2. The MCs were cultured on collagen-I coated petri dishes in porcine SMC growth medium (5 mL) with transforming growth factor (TGF)-β inhibitor (SB431542, 10 µM) for 3 days, and 5 µM for a further 3 days, and the resulting cells were named TGF_treat. The TGF-β inhibitor (SB431542) was used in previous studies to promote differentiation of induced pluripotent stem cells toward vascular SMCs. 18 Subsequently, the medium was replaced with SMC growth factor medium without TGF for another 7 days, and the resulting cells were named SMC-like cells. Phase-contrast images revealed that the initially cultured MCs displayed a round morphology that gradually transitioned to a spindle-like morphology. 11% of differentiated cell fraction was positive for α-SMA, indicating effective differentiation toward SMC-like cells (Figure S2). SMC-specific genes such as MYH11, ACTA2, ACTG2, and TAGLS were upregulated; however, functionally specific transcription, such as CNN1, was not upregulated. The primary data suggest that MCs have the potential to differentiate toward SMCs, although there is a need to improve this protocol to obtain functional SMCs from MCs differentiation.
To assess the function of EC-like cells, we performed in vitro assays for acetylated LDL (AcLDL) uptake and tube (neovessel) formation. EC-like cells were evaluated for AcLDL (PE-conjugated) uptake, which was quantified using relative intensity measurements. The relative intensities of MCs, ROCK_treat, and EC-like cells increased progressively, with values of 0.2, 0.5, and 0.7, respectively, as shown in Figure 2(a). Fibroblasts used as negative controls did not exhibit AcLDL uptake.

Assessment of EC-like cells function: (a) EC-like cells successfully uptake acetylated low-density lipoprotein (AcLDL), whereas fibroblast cells cannot, (b) EC-like cells form tubular networks and are positive for CD14 and CD31, as shown by immunostaining, and (c) Quantitative PCR analysis of endothelial transcription factors further confirms the effective differentiation of MCs into EC-like cells. Pig endothelial cells (PECs) were used as a positive reference for comparison. Data were expressed as means ± standard deviations of one representative experiment (one-way ANOVA, AcLDL uptake: F3,8 = 7.3, p = 0.010; CD31: F3,12 = 344, p = 0.013; Ang: F3,12 = 110, p = 0.024; vWF: F3,12 = 128, p = 0.024; Nos: F3,12 = 181, p = 0.024; CD14: F3,12 = 196, p = 0.024; CD163: F3,12 = 226, p = 0.024; Tgf-β: F3,12 = 119, p = 0.024; Notch1: F3,12 = 312, p = 0.024, Tukey’s HSD post hoc test, *p < 0.05, **p < 0.01, ***p < 0.001).
Although both macrophages and ECs are capable of AcLDL uptake, only ECs form neovessels. 19 Interestingly, EC-like cells formed a connected tube-like network within 24 h, as shown in Figure 2(b). Additionally, cells in the tube network expressed CD31, which was localized at cell-cell junctions. Surprisingly, these cells expressed CD14, as shown in Figure 2.
Furthermore, we analyzed multiple genes involved in EC differentiation and function as well as genes associated with macrophages. Endothelial-specific genes including CD31, vWF, Ang, and Nos were progressively upregulated during differentiation, as shown in Figure 2(c). Macrophage-related markers such as CD14 and CD163 were slightly downregulated during differentiation. Interestingly, Tgf-β, an M2-associated gene, was upregulated, indicating M2 activation of macrophages during differentiation. HEY1 and SOX17 exhibited the maximum upregulation, which closely resembled the levels observed in typical pig endothelial cells (PECs). Other endothelial marker genes such as TIE1, ERG, HEY2, and IL-6 were upregulated during differentiation (Figure S3.) Furthermore, heatmap analysis of the top 50 upregulated genes confirmed that EC-like cells highly expressed PEC-specific genes that were not upregulated in the PBMC, MCs, or ROCK_ treated groups (Figure 3(a)).

Analysis of genes involved in EC differentiation and function: (a) Heatmap analysis of the top 50 expressed genes across all groups, (b) K-map representation of gene expression patterns in all groups, based on K-means clustering, identifies four distinct clusters. (c), (d), (e), and (f) are annotation analyses of selected genes from clusters A, B, C, and D, respectively, summarized in the table. One sample is selected from each group for analysis.
To evaluate the global transcriptional changes during the differentiation toward EC-like cells, RNA sequencing was performed. The top 1000 ranked genes were divided into four clusters using k-means analysis (Figure 3(b)). Clusters C and D showed upregulation of immune system and cell activation pathway-related genes in PBMC and MCs. These genes were slightly upregulated in ROCK_treated and EC-like cells but were not upregulated in PECs. Following ROCK and CHIR treatments, upregulation of genes associated with chemical stimuli and organic substance transport pathways was observed in ROCK_ treated and EC-like cells, as shown in Cluster B. Interestingly, Cluster A was enriched with genes related to angiogenesis and vascular development pathways, which were upregulated in both EC-like cells and PECs (Figure 3).
Addition of CHIR activates the Wnt/β-catenin pathway, promoting β-catenin nuclear translocation and its interaction with TCF7, LEF1, and TCF7L2 to drive transcription of endothelial genes like VE-cadherin, VEGFR2, and NOS. Simultaneous upregulation of JUN and FOSL1 (AP-1 complex) supports cytoskeletal remodeling and angiogenic gene expression (Figure S4). In parallel, ROCK inhibition enhances cell survival, reduces actomyosin contractility, and promotes cytoskeletal. Overall, ROCK/CHIR treatment provides a synergistic environment for promoting endothelial differentiation from MCs.
These findings indicate that EC-like cells exhibit significant upregulation of genes responsible for their differentiation into ECs. Furthermore, the data suggest that efficient in vitro differentiation of MCs into EC-like cells on a collagen-I-coated surface can occur via biochemical cues.
Acute inflammation induced accelerated differentiation
To evaluate the effect of acute inflammation on MCs differentiation toward EC-like cells, blood samples were collected before and after vascular surgery-induced acute inflammation in a minipig model. The timeline of blood collection and further processing is shown in Figure 4(a). FSC-SSC analysis of PBMC and MCs indicated an increase in monocyte and granulocyte counts and a significant reduction in the lymphocyte population after acute inflammation (Figures 4(b), (c), and S5). Interestingly, MCs revealed that higher total cell count, with mostly granulocytes (56%) and monocytes (7%) were attached to collagen-coated Petri dishes after acute inflammation; however, a balanced attachment of granulocytes (20%) and lymphocytes (17%) was observed in pre-inflammatory MCs, as shown in Figure 4(d) and (e). PBMC analysis with different surface markers reveals a higher fraction of CD14/16+, CD14/163+, and CD14/VEGFR2+ compared to pre-inflammation. However, the EPCs marker-positive cell fraction (CD34+, CD45+, CD31+) decreased after acute inflammation, as shown in Figure S6. The EPCs count reduced in the blood sample collected 24–48 h following acute vascular injury.20,21 Interestingly, MCs analysis revealed a higher fraction of CD14/16+ cells and a lower fraction of EPCs attaching to the dish surface after acute inflammation, as shown in Figure S7. The attached MCs were processed for differentiation toward EC-like cells using a previously described ROCK/CHIR treatment protocol. The corresponding phase-contrast images are shown in Figure 4f. Notably, a higher fraction of EC-like cells and dense colonies were observed in a blood sample collected from an acutely inflamed minipig.

Analysis of accelerated differentiation induced by acute inflammation: (a) Timeline of blood collection from an acutely inflamed minipig model before and after surgery, and processed for MCs differentiation, (b) FSC/SSC analysis of PBMC and MCs revealed a, (c) significant increase in monocyte count and a decrease in lymphocyte count, (d) a Higher MCs attachment observed with (e) significantly higher monocyte and granulocyte count in acutely inflamed samples, and (f) Phase contrast images of all stages of differentiation from MCs to EC-like cells with ROCK/CHIR protocol showed significantly higher differentiation into EC-like cells observed within a blood sample collected from an acutely inflamed minipig. Unpaired t-tests (n = 3) were used to evaluate differences among the groups. Values of p < 0.05 are considered statistically significant, and are presented as follows: *p < 0.05, **p < 0.01, and ***p < 0.001.
RNA sequencing analysis was performed to evaluate global transcriptional changes in attached MCs before and 2 days after induction of acute inflammation. The top 1000 ranked genes were divided into three clusters using k-means analysis, as shown in Figure 5. MCs collected after acute inflammation showed upregulated genes related to pathways such as chemotaxis, response to external stimuli, and signal transduction. Moreover, the activity of integrins such as α4β1, α5β1, and α4β7 was more pronounced in MCs collected following acute inflammation (Figure S8). These integrins enhance adhesion to extracellular matrix components, such as collagen, fibronectin, and laminin.

Analysis of acute inflammation induced pathway enrichment: (a) Timeline of blood collection and processing, (b) Heat map representation of MCs and corresponding differentiated EC-like cells, genes based on the K-means analysis with three clusters. Annotation analysis of selected genes in respective clusters was summarized in the tables.
Following ROCK/CHIR treatment, MCs collected after acute inflammation exhibited an upregulated expression of endothelial-specific genes associated with pathways as tube development and angiogenesis, indicating more efficient differentiation toward EC-like cells. However, MCs collected pre-inflammation upregulated immune system-related genes, suggesting a differentiation bias toward macrophages.
Further, we have evaluated the long-term effect of acute inflammation on MCs differentiation toward EC-like cells, the timeline for blood collection and further processing is shown in Figure 6(a). The timeline is chosen because inflammatory cytokines such as TNF-α, IL-6, and IL-10 typically reach peak levels within 48–72 h following inflammation and then gradually decline, returning to baseline around day 14. 22 Phase-contrast images of EC-like cell colonies are shown in Figure 6(b), and the number of colonies and fraction ratio in a 10 mm petri dish is depicted in Figures 6(c), (d), and S9. Intriguingly, the number of EC-like cell colonies and the cell density within the colonies continually decreased with increasing durations of acute inflammation.

Temporal effects of acute inflammation on MCs differentiation: (a) A timeline illustrating blood collection, processing, IL-6 treatment, and differentiation into EC-like cells from an acutely inflamed minipig model, (b) phase-contrast images of EC-like colonies, (c) a graphical representation of the total colony counts, and (d) EC-like cell fraction at different time points of blood sample processing. EC-like cells fraction, and colony number significantly increase after acute inflammation (day 2) and are reduced with longer time to blood collection (day 16) from acute inflammation. Interestingly, in vitro treatment with IL-6 significantly enhances the EC-like cell fraction. Data were expressed as means ± standard deviations of one representative experiment out of three experiments carried out in triplicate (one-way ANOVA, Number of EC-like cell colonies: F3,8 = 7.6, p = 0.0021; EC-like cells fraction ratio: F3,8 = 6.8, p = 0.0037, Tukey’s HSD post hoc test, *p < 0.05, **p < 0.01, ***p < 0.001).
MCs analysis using CD14 and CD16 markers revealed a shift in surface marker levels in a specific cell population at different periods of acute inflammation, as shown in Figure 7(a). Cell-specific gating, as previously reported23–25 showed that majorly monocytes have a shift in intensity toward a CD14++/16+ state following acute inflammation. Interestingly, marker intensities of these monocytes return to the initial state after prolonged periods of acute inflammation (Figure 7(b)). After inflammation, surface markers for monocytes are in the CD14++/CD16+ state, representing a pro-active monocyte state that is characterized by the upregulation of chemokine receptors (e.g., CCR2, CXCR1) and cytokine receptors,26,27 which enable them to respond more effectively to external chemical stimuli. Therefore, in the presence of EPCs, activated monocytes effectively differentiated toward EC-like cells by external chemical stimulation in vitro. 9 Consequently, there are more EC-like colonies in blood collected after acute inflammation, even though the EPC count was lower than before the inflammatory conditions. Additionally, the fraction of CD14++/16+ monocytes exhibited a profile similar to that of in vitro differentiated EC-like cells at various stages of acute inflammation (Figure 7(c)). This suggests that these monocytes significantly contribute to the differentiation of EC-like cells in vitro. However, similar profile was not observed in granulocytes or lymphocytes, as shown in Figure 7(c).
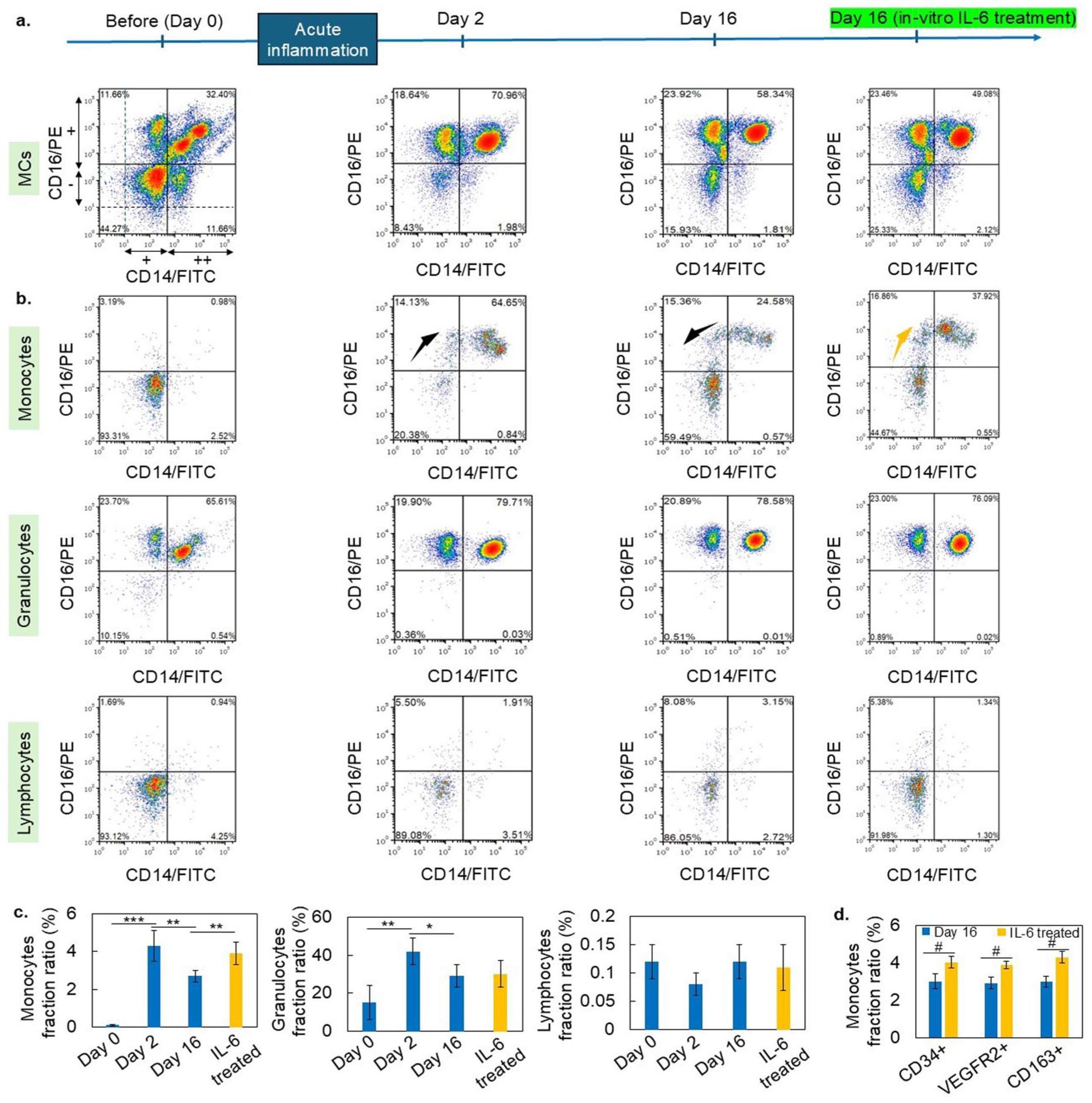
Flow cytometry analysis of MCs collected before and after acute inflammation: (a) MCs’ analysis using CD14 and CD16 markers revealed the shift in surface marker intensity of a specific cell population at different stages of acute inflammation. (b) Cell-specific gating showed that primarily monocytes transited toward a CD14++/16+ state following acute inflammation. Interestingly, these cells marker intensity come to initial state after prolonged periods of acute inflammation. (c) The fraction of CD14++/16+ monocytes shows a profile similar to that of in vitro differentiated EC-like cells at different periods of acute inflammation. This implies that monocytes in CD14++/16+ state significantly contribute to the differentiation of EC-like cells in vitro. However, no significant intensity shift was observed in granulocytes or lymphocytes. In vitro treatment of PBMC with IL-6 also promotes monocyte transition toward a CD14++/16+ state and increases the EC-like cell fraction. (d) In vitro IL-6 treatment significantly enhances the population of CD34+, VEGFR2+, and CD163+ monocytes. Data were expressed as means ± standard deviations of one representative experiment out of three experiments carried out in triplicate (one-way ANOVA, Fraction ratio of monocytes: F3,8 = 9.1, p = 0.0011; granulocytes: F3,8 = 6.3, p = 0.0030; lymphocytes: F3,8 = 1.2, p = 0.247, Tukey’s HSD post hoc test, *p < 0.05, **p < 0.01, ***p < 0.001). Paired t-tests (n = 3) were used to evaluate differences among the groups in (d). Values of p < 0.05 are considered statistically significant, and are presented as follows: # p < 0.05.
Furthermore, in vitro treatment of PBMC with IL-6 also shifts the surface marker intensity of monocytes toward a CD14++/16+ state and increases the fraction of EC-like cells. IL-6 treatment significantly shifts the surface marker intensity of CD34+, VEGFR2+, and CD163+ monocytes, as shown in Figures 7(d) and S10. These data suggested that IL-6 may be one of the inflammatory cytokines affecting MCs differentiation toward EC-like cells in vitro.
Additionally, an acellular graft covered with EC-like cells effectively prevented autologous platelet attachment during blood circulation (Figure S11). However, there are limitations to this experiment, such as shorter circulation time, periodic heparin injection, and removal of EC-like cells during circulation. Moreover, MCs attachment behavior in vitro potentially leads to early detection of vascular abnormalities (Figure S12).
This study demonstrated that the differentiation phenotype of MCs varies depending on the collection conditions, highlighting the significant impact of acute inflammation on MCs differentiation behavior in vitro. Moreover, it provides clear evidence of MCs differentiation toward EC-like and SMC-like cells in vitro by chemical cues and enhances our understanding of neovascularization, vascular diseases, and remodeling under acute inflammation.
Conclusion
We demonstrated that acute inflammation could accelerate MCs-induced vascular remodeling via the activation of integrins and chemotaxis. MCs plasticity can be controlled in vitro by chemical cues and cytokine treatment. Monocytes that transition to CD14++/CD16+/CD163+ states are primarily responsible for accelerated vascular remodeling.
Methods
Whole blood processing to obtain PBMC
Whole blood was collected from the tail artery of the mini-pigs, and RBC lysis was performed using RBC lysis buffer (catalog number 60-00050-13, PluriSelect Life Science, Germany) according to the manufacturer’s protocol to obtain peripheral blood mononuclear cells (PBMC). Briefly, 5 mL whole blood was mixed with 45 mL lysis buffer, incubated at room temperature for 15 min, and centrifuged at 300 × g for 10 min to obtain a suspended cell pellet. The supernatant was removed using suction. The collected cell pellet was washed with PBS and resuspended in a cell medium (catalog number P211K-500, Porcine EC Growth Medium Kit, Cell Application Inc., USA).
Isolation of MCs
The PBMC suspension (4 × 106 cells) was incubated in a 10 cm collagen-I coated petri dish with porcine endothelial basal media (5 mL) and autologous platelet-rich plasma (0.5 mL) for 12 hours at 37 °C. Unattached cells were removed by gentle washing with PBS and attached MCs were collected using trypsinization.
Acute inflammation mini-pig model
All animal experiments were conducted in accordance with the Guidelines for Animal Experiments established by the Ministry of Health, Labor, and Welfare of Japan and the National Cerebral and Cardiovascular Center Research Institute in Japan. The study protocol was approved by the Committee on Ethics of Animal Experiments of the National Cerebral and Cardiovascular Center Research Institute (Permit Number: 009017). Göttingen mini-pigs (male), purchased from Ellegaard Göttingen Minipigs A/S (Dalmose, Denmark), were anesthetized with intravenous injection of 1% propofol (100 mg/h; Diprivan; AstraZeneca, Wilmington, DE). After a 1 h evaluation, a decellularized vascular graft was transplanted in the femoral-femoral bypass position. Whole blood collected from the tail artery before transplantation was defined as pre-inflammatory blood. After graft transplantation, blood was collected from the same pigtail artery and was defined as acute inflammation blood. Total three different mini-pigs are used for transplant and blood collection. Blood was collected according to the experimental timeline, as previously described.
Immunocytochemistry
MCs and EC-like cells were stained with FITC-conjugated CD14 (MCA1218F), PE-conjugated anti-CD31 (MCA1746PET), and PE-conjugated CD16 (MCA1971PE) supplied by Bio-Rad Laboratories (Montreal, Quebec), and CD34 (bs-0646R-PE, Bioss Antibody Inc., Boston, MA, USA). In brief, cells incubated in a petridish were washed with PBS, then cells on the surface were fixed with 5% formaldehyde solution at 25°C for 20 min. Then, 0.1% Triton X-100 was added at room temperature for 5 min. The dish was washed with PBS, and the staining antibody was added and incubated at 4°C overnight. The cells were then washed with PBS and the nuclei were stained with DAPI (4′-6-diamidino-2′-phenylindole, 3 μM in PBS; Molecular Probes) for 5 min. The samples were examined using a FV4000 CLSM system (Olympus, Tokyo, Japan). FITC fluorescence was excited at 488 nm, and the emitted fluorescence was measured at 491–586 nm. PE fluorescence was excited at 488 nm and measured at 575–675 nm. DAPI fluorescence was excited at 405 nm and measured at 420–480 nm.
Flow cytometry
Surface marker expression in MCs and EC-like cells was evaluated using flow cytometry. The cells were collected using trypsinization (trypsin/EDTA solution, Lonza, Basel, Switzerland) according to the experimental timeline. MCs were suspended in PBS containing 10% normal pig serum to block Fc receptors. Fluorescence minus one (FMO) controls for FITC and PE channels have been included in the Supporting Information. Additionally, unstained MCs were used to assess baseline autofluorescence. The Cell labeling was performed for surface markers CD14, CD16, CD45, CD31, CD34, VEGFR2, and CD163 according to the manufacturer’s instructions. Data were acquired using a FACSverse cell analyzer (BD Biosciences, San Jose, CA, USA) and analyzed using FlowJo software. The fraction ratio was calculated by quadrat statistics analysis using FCS Express flow cytometer software (De Novo software, US).
Tube formation assay
Differentiated MCs on day 14 were trypsinized, replated on Matrigel with Porcine EC Growth Medium, and cultured for 24–48 h. Then, the cells were fixed in 4% formaldehyde and permeabilized with 1% Triton-X100 prior to immunocytochemistry.
LDL uptake
DiI-Ac-LDL Kit from Cell Application, USA, is used for this test. Differentiated MCs on day 14 were incubated with acetylated LDL according to the manufacturer’s protocol. Cells were exposed to DiI-labeled LDL for 4 h, washed three times with PBS, and then fixed in 4% paraformaldehyde before imaging and quantification.
RNA extraction, reverse transcription, and RT-qPCR
The total RNA from MCs, ROCK_treat cells, EC-like cells, and PECs were extracted using the High Pure RNA Isolation Kit (Qiagen, Hilden, Germany), and quantified using a NanoDrop system, reverse-transcribing 2 μg of RNA to cDNA using the High-Capacity cDNA Reverse Transcriptase kit (Applied Biosystems, Thermo Fisher, Waltham, MA, USA) and random primers. This cDNA was used to perform RT-qPCR with Power SYBR Green PCR mix (Applied Biosystems), and the primers (Thermo Fisher) are listed in Table 1. The RT-qPCR expression data were quantified according to the 2−ΔΔCt formula.
List of primers used for quantitative PCR.

RNA sequencing
Total RNA was extracted from PBMCs, MCs, ROCK_treat cells, EC-like cells, and PECs using an miRNeasy-micro/mini kit (Qiagen) and treated with RNase-free DNase I (Qiagen). To assess the quality of the isolated total RNA, RNA integrity was measured using an Agilent 4200 TapeStation (Agilent, Santa Clara, CA, USA). The mRNA sequencing libraries were prepared from high-quality total RNA using the TruSeq Stranded mRNA Library Prep Kit (Illumina, San Diego, CA, USA). After evaluating the library quality, sequencing was performed with 75 bp paired-end reads using the NextSeq 500 High Output Kit (Illumina). After demultiplexing with Bcl2fastq (Illumina), the quality of the raw reads was checked, trimmed, and aligned to the mini-pig (Sus scrofa) reference genome (Sscrofa 11.1) using the CLC Genomics Workbench ver. 20.0 (Qiagen). K-means, functional annotation, and pathway enrichment analyses were performed using iDEP 0.96.
Statistics %
Data were expressed as means ± standard deviations of one representative experiment out of three experiments carried out in triplicate. Statistical analyses were performed using GraphPad Prism. One-way and two-way analyses of variance (ANOVA), and t-tests were used to evaluate differences among groups. Values of p < 0.05 is considered statistically significant, presented as: *p < 0.05, **p < 0.01, and ***p < 0.001.
Supplemental Material
sj-docx-1-tej-10.1177_20417314251381716 – Supplemental material for Blood mononuclear cells induce accelerated vascular remodeling under acute inflammation in vitro
Supplemental material, sj-docx-1-tej-10.1177_20417314251381716 for Blood mononuclear cells induce accelerated vascular remodeling under acute inflammation in vitro by Raghav Soni, Manabu Shirai, Tetsuji Yamaoka and Atsushi Mahara in Journal of Tissue Engineering
Footnotes
Ethics approval
All animal experiments were conducted in accordance with the Guidelines for Animal Experiments established by the Ministry of Health, Labor, and Welfare of Japan and the National Cerebral and Cardiovascular Center Research Institute in Japan. The study protocol was approved by the Committee on Ethics of Animal Experiments of the National Cerebral and Cardiovascular Center Research Institute (Permit Number: 009017).
Consent for publication
All authors read, approved, and consented to submit the final manuscript.
Author contributions
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Japan Agency for Medical Research and Development (AMED) under Grant Number JP23he2822006j0001, the Japan Society for the Promotion of Science (JSPS) KAKENHI Grant-in-Aid for Early Career Scientists (No. 24K21101), and the Japan Science and Technology Agency (JST) Grant [Grant number JPMJPF2018].
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
