Abstract
Fetal bovine serum (FBS) plays a crucial role in the composition of animal cell culture medium. However, conventional serum-based medium face numerous challenges. The use of animal-derived free hydrolysate (ADFH) has garnered significant attention in research and applications as a viable alternative to FBS-containing medium in animal cell culture. This article provides a comprehensive overview of the effects, mechanisms of action, and applications of ADFH in animal cell culture. ADFH serves as an effective substitute for FBS-containing medium, enhancing various cellular processes, including cell proliferation, viability, protein synthesis, production, survival, and stability. Several mechanisms of action for ADFH have been elucidated through scientific investigations, such as nutrient provision, activation of signaling pathways, regulation of protein synthesis and folding, protection against oxidative damage and apoptosis, as well as cell cycle regulation. Researches and applications of ADFH represent a promising approach to overcoming the limitations of FBS-containing medium and advancing the field of animal cell culture. This review provides a theoretical foundation for promoting the development of sustainable and alternative hydrolysates, as well as the continued progress of animal cell culture.
Keywords
Introduction
Harrison’s groundbreaking work in 1907 involved the utilization of lymph as a medium for cultivating frog nerve fibers over an extended period of many weeks, thus marking the advent of animal cell culture. 1 The increasing focus on industrialized and large-scale cell culture can be traced from the initial vaccine revolution sparked by the rabies and Bacillus Calmette-Guerin vaccines, to the rapid subsequent advancements in diverse vaccine technologies during the global pandemic of the novel coronavirus in 2020. 2 Animal cell culture has become a prominent methodology employed in both research and practical applications within the life sciences. The production of numerous modern biotechnology products, such as recombinant proteins and vaccines, relies on the extensive cultivation of animal cell lines at a large scale. 3 This platform offers researchers significant opportunities to study cell biology, disease mechanisms, and drug development, thereby playing a crucial role in the biomedical industry. 4
Animal cells require a diverse range of nutrients to facilitate their growth and proliferation in laboratory cultures. Historically, Fetal Bovine Serum (FBS) has traditionally held significant prominence due to its richness in hormones, growth factors, and essential nutrients that support cellular growth, metabolism, and overall physiological functions. 5 However, FBS presents several challenges, such as batch-to-batch variability, undefined composition, and ethical concerns related to animal welfare.6,7 These issues, combined with the increasing demand for serum-free alternatives, have driven the development of a variety of serum-free medium (SFM).
Hydrolysates, one of the serum replacements, are crucial to the industrial animal cell culture process. The effects of hydrolysates on active ingredients, antioxidants, and cell development have been extensively studied by scientists (Figure 1). The initial serum substitutes used in mammalian cell cultures were hydrolysates derived from animal sources. Animal-source hydrolysate is a powdered substance derived from animal proteins using techniques such as enzymatic or acid hydrolysis, separation, filtration, concentration, drying, and packaging. Currently, commonly employed animal-derived hydrolysates in SFM include human platelet lysate (HPL), meat, whey protein, casein, salmon, and others.8–11 Among the FBS replacements, HPL and chemically defined medium (CDM) have gained significant attention. HPL has been shown to support the growth of various cell types, including mesenchymal stem cells and fibroblasts, with more consistent performance than FBS. 12 CDM, on the other hand, offers a fully synthetic composition free from animal-derived components, allowing for greater reproducibility and eliminating the risk of contamination. However, both HPL and CDM face limitations: HPL is constrained by high costs and limited supply, while CDM may not always fully replicate the complex nutrient environment that animal cells require for optimal growth.

Recent research trends related to hydrolysates in animal cell culture: (a) Web of Science’s core collection was searched for hydrolysate and animal cell culture, and 93 references from the last ten years were chosen and their keyword co-occurrence was examined. Includes Network visualization and Overlay visualization. (b) Co-occurrence analysis of keywords in the previous 20 years’ worth of literature on animal cell culture and ADFH. The results were conducted by VOSviewer version 1.6.20 of Leiden University, The Netherlands.
In contrast, animal-derived free hydrolysates (ADFH) represent a promising alternative that addresses many of the limitations associated with both FBS and its other replacements. ADFH is not only free from animal disease risks and ethical concerns but also offers significant advantages in terms of cost-effectiveness, stability, and scalability, particularly for industrial applications. 13 Furthermore, ADFH provides a more natural and balanced nutrient environment that mimics the complex interactions found in traditional serum, without the variability and undefined components of FBS. Research has shown that ADFH can enhance cell growth and productivity in biopharmaceutical production, matching, or even surpassing the performance of FBS and other alternatives. 14 However, due to the lack of characterization of most ADFH components, further research is required to determine their composition and understand their impact on cellular proliferation.
Thus, ADFH offers a unique combination of sustainability, efficiency, and scalability, making it a highly viable candidate for large-scale industrial use in animal cell culture. This review aims to systematically assess the potential and benefits of ADFH as an alternative in biopharmaceutical production medium. Additionally, a detailed investigation into the mechanisms by which ADFH promotes cell growth, alongside an analysis of its composition and key constituents, will provide valuable insights for advancing the development of more sustainable and cost-effective cell culture methods. This, in turn, will contribute to further progress in the field of animal cell culture.
ADFH: Composition, preparation, isolation, and purification
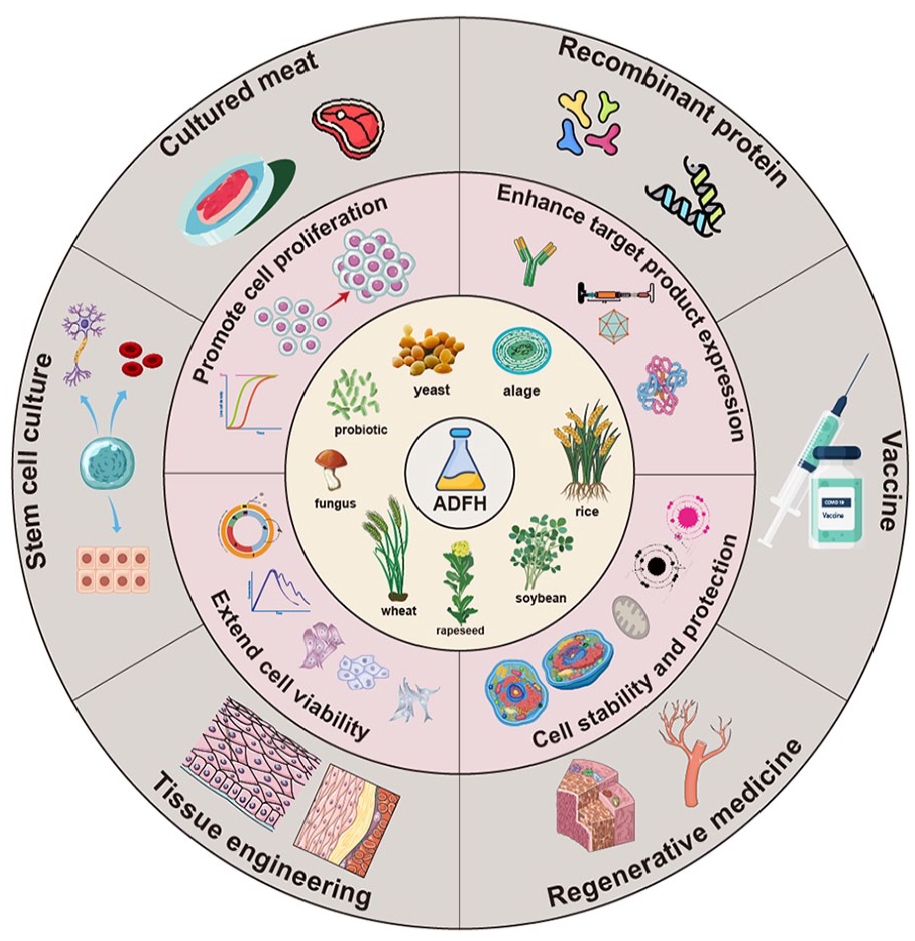
ADFH refers to a protein hydrolysate derived from plant and microbial origins, possessing comparable nutritional composition and biological functionality to hydrolysates sourced from animals. Yeast emerged as the predominant source of ADFH, constituting 54% of the overall research papers. Subsequently, soybean and wheat were also notable sources, accounting for 50% and 46% of the investigations, respectively. 15 To illustrate the advantages and limitations of various hydrolysate sources, a comparative analysis is presented in Table 1. The efficacy of ADFH as a cost-effective and low-risk option has been demonstrated in several cell lines, including Chinese hamster ovary (CHO) cells, Hela cells, and human lung cancer cell lines. These findings indicate its potential for wide-ranging applications in industries such as biomedicine and cell culture meat production. 16 Hence, a comprehensive examination of the various source types and the preparatory procedures employed for ADFH is advantageous to elucidate the core components of ADFH.
Comparison of FBS, HPL, and ADFH in animal cell culture.

Compositions and preparation of ADFH
Research findings indicate that the primary origins of ADFH predominantly encompass microorganisms and plants. Microbially-derived hydrolysates, such as yeast hydrolysates and algal hydrolysates, are frequently employed in animal cell culture. However, microbial sources pose potential contamination risks. To address these hazards, advanced purification techniques, including ultrafiltration, chromatography, and sterilization, are used throughout the ADFH production process.17,18 These methods help eliminate microbial contaminants while preserving key bioactive compounds, ensuring product safety and quality in biotechnological applications. The primary methods for preparing these hydrolysates are illustrated in Figure 2.

Simple processes for the preparation of ADFH: (a) The standard preparation processes of microbial-derived hydrolysates, consider yeast hydrolysate as an example. (b) The standard preparation processes of plant-derived hydrolysates, consider soybean hydrolysates as an example.The figure was created with BioRender.
The utilization of yeast hydrolysate in cell culture is a significant milestone as it marks the introduction of the first hydrolysate obtained from microbial sources. Traditional yeast hydrolysates consist of two primary components: yeast extract (YE) and yeast peptone (YP). 14 YE is mostly derived from yeast autolysis, whereas YP is primarily obtained through enzymatic hydrolysis using external enzymes. 19 These preparatory methods are designed to minimize contamination while ensuring the active compounds are preserved, maintaining the quality of ADFH for industrial applications. At present, YE serves as a prominent constituent employed in the cultivation of CHO cells for the purpose of producing recombinant proteins. 20 The augmentation of cellular volume and enhancement of lipid synthesis capacity in CHO cells can significantly facilitate the promotion of target protein synthesis by YE. YP is abundant in a diverse array of complimentary amino acids and oligopeptides. The combined supplementation of YE and YP might enhance the availability of substrates for the culture, hence leading to an increased production of desired recombinant proteins. 21 The manufacture of yeast hydrolysate has incorporated the utilization of high pressure and ultrasonic crushing techniques, together with the application of numerous enzymes, to enhance the release of active compounds present in yeast. 22
ADFH can also be found in significant amounts in marine-derived algae, which are high in lipids, proteins, vitamins, and other minerals. Research has substantiated the wide-ranging potential of algal hydrolysates in facilitating the proliferation of various cell types, particularly stem cells, as well as encouraging their differentiation and exhibiting antioxidant properties.23–25 In contrast to yeast, the production process of ADFH derived from algae shares similarities with plant-derived hydrolysate. Typically, it involves the utilization of methods such as strong acid treatment, strong alkali treatment, or high-temperature treatment after pulverizing the algae into a powdered form. 26 These techniques are employed to facilitate the liberation of nutrients.
In recent years, probiotics have emerged as a notable source of microbial-derived hydrolysate. Various preparation methods are frequently employed, such as ultrasonic fragmentation conducted in an ice water bath, centrifugation to get Cell-free supernatant (CFS), and high temperature and high pressure lysis, among others. Probiotic lysates encompass a diverse array of constituents that exert an influence on cellular proliferation and function. Frequently employed probiotic species encompass Lactobacillus, alongside several metabiotics that exhibit distinct physiological actions. The study observed that the consumption of L. Roy DS0384, supplemented with N-Carbamylglutamate (NCG), had a positive impact on the growth of intestinal stem cells and had a protective effect on intestinal organoids against inflammation induced by interferon-γ (IFN-γ) and Tumor Necrosis Factor α (TNF α). 27 Likewise, the fermentation of hawthorn juice by Lactobacillus paracasei subsp. tolerans, followed by high-temperature lysis, resulted in CFS that was able to efficiently stimulate the growth of human colorectal cancer Caco-2 cells. 28 Furthermore, the utilization of hydrolysates derived from certain culinary and medicinal fungi exhibits significant potential for application in animal cell culture. The hydrolysate obtained from Phellinus linteus has the potential to enhance the proliferation of bone marrow cells mediated by Peyer’s patches (PPs) and stimulate the synthesis of cytokines, including interleukin-2 (IL-2) and IL-6.29,30
Plant-derived hydrolysates are currently mostly made from soybean, rapeseed, wheat, and other source materials in cell culture.31–33 The most extensively researched hydrolysate obtained from plants in the field of cell culture is now soybean hydrolysate. Its production typically involves acid-base hydrolysis, enzymatic hydrolysis, or a combination of both methods (Figure 1). The majority of the functional active ingredients in soybean hydrolysates can be retained using enzymatic hydrolysis, one of the numerous available preparation techniques. Commonly employed enzymes include cellulase, papain, bromelain, and others. In addition to enzymatic hydrolysis, several studies have proposed that the utilization of ultrasound or bio-ionic liquid as aids in the manufacturing of hydrolysate can enhance the extent of hydrolysis of soy protein. 34 More free amino acids and tiny molecular peptides, which are more readily absorbed by cells, are present in ADFH made using these specific techniques.35,36 Moreover, it is noteworthy that plant seeds, including rapeseed, flaxseed, and cottonseed, are significant sources of ADFH. The hydrolysates derived from the seeds of these plants not only encompass vital nutrients necessary for cellular growth, but also exhibit a diverse array of bioactive constituents.37,38 In contrast to soybean, the preparation of plant seed hydrolysates often involves acid hydrolysis or enzymatic hydrolysis, both of which have been demonstrated to produce tiny molecular compounds possessing diverse biological properties. 39 Xiao used high pressure liquid chromatography analysis to determine that ADFH made from rapeseed contains a range of amino acids, vitamin E, sterols, peptides, and other active substances. Additionally, ADFH served as a means of selenium accumulation, which is a necessary trace element for cell growth. 40 Hence, it can be inferred that ADFH, which is generated from rapeseed, exhibits significant potential for utilization in cell culture.
Isolation and purification of ADFH
In order to determine the precise active ingredients, ADFH typically needs to be separated and purified after preparation. To improve the stability of ADFH production between batches, the active substance content in the next batch’s production is then determined. Currently, the prevailing methods employed for the separation and purification of ADFH mostly encompass membrane separation technology and chromatographic purification technology. The membrane separation technology encompasses many techniques, including nanofiltration, ultrafiltration, and microfiltration, which are categorized based on their distinct pore sizes. The utilization of ultrafiltration membranes has been prevalent in the field of protein hydrolysate or peptide separation and purification. This is mostly attributed to the numerous benefits it offers, such as its straightforward operational procedures, minimal energy requirements, and positive impact on the environment. 41 Further optimizing these techniques to better isolate target peptides and bioactive compounds will be crucial for enhancing product consistency. To optimize the efficiency of cell culture, it is frequently required to utilize ultrafiltration or nanofiltration to intercept peptides with varying molecular weights in order to separate and collect the target active peptides after yeast hydrolysate has been generated and concentrated. 22 After fractionating YE by nanofiltration, Mosser et al. discovered that adding low-molecular peptides, polysaccharides, nucleic acids, and other YE components below 500 Da to the medium could greatly enhance CHO cell performance without compromising the target recombinant protein’s subsequent purification. 42
Typically, further purification steps are necessary to achieve products of high purity in the context of ultrafiltration. Chromatographic methods that can accomplish a more precise classification of complicated active compounds include size exclusion chromatography (SEC), hydrophobic interaction chromatography (HIC), and ion exchange chromatography (IEC); following the purification process, mass spectrometry and tandem mass spectrometry can be employed to further investigate the peptide sequence and other active constituents of ADFH.43–46 Furthermore, isolates with greater purity and activity can be obtained using omics techniques including Western blot analysis, computer virtual screening, and molecular conformational analysis; however, these approaches are currently costly and challenging to implement for large-scale industrialization. 41 ADFH can be further prepared by vacuum, freeze, or spray drying to guarantee the stability of the obtained material after separation and purification.47,48
To improve ADFH’s utility in various applications, future research should focus on tailoring hydrolysate compositions to specific cell lines or applications. This may involve adjusting the peptide and amino acid profiles to better support the growth requirements of particular cells, such as CHO cells for biopharmaceutical production or stem cells for regenerative medicine. 49 Customizing ADFH formulations could lead to more efficient culture processes and potentially reduce the need for supplementary growth factors. Exploring novel microbial and plant sources with unique bioactive properties may also expand the scope of ADFH’s applications. In particular, sourcing ADFH from species with high levels of specific nutrients or bioactive compounds could enhance its efficacy in specialized cell culture systems. 50 Last but not the least, scaling up these processes for industrial applications remains a priority, necessitating the development of cost-effective and scalable purification techniques.
Effects of ADFH on animal cell culture
The selection of nutrients in the culture medium is critical for animal cell culture, and standard animal cell lines can exhibit significantly improved performance when the medium’s composition is optimized. While FBS and HPL both promote cell proliferation due to their rich content of growth factors, their animal origins raise concerns about potential contamination from viruses or prions, as well as ethical issues. ADFH, being entirely non-animal-derived, offers a safer, more consistent alternative, free from these risks, making it a preferable choice for cultures requiring high levels of consistency and safety. ADFH is an affordable supplement to cell culture medium, derived from abundant raw materials. It aims to provide the necessary nutrients for cell growth, reduce reliance on animal-derived components, enhance cell division, improve recombinant protein synthesis in animal cells, and bolster culture stability.
Maintenance of cell proliferation and viability
ADFH is a protein-containing hydrolysate that provides a rich source of amino acids and peptides essential for the growth and multiplication of mammalian cells. These peptides and amino acids support essential processes such as protein synthesis, metabolism, and cell growth. They serve as the fundamental building blocks of cellular structure and function. The effects of ADFH on cell formation and proliferation are summarized in Table 2.
ADFH promotes cell proliferation, increases production, and improves cell performance.
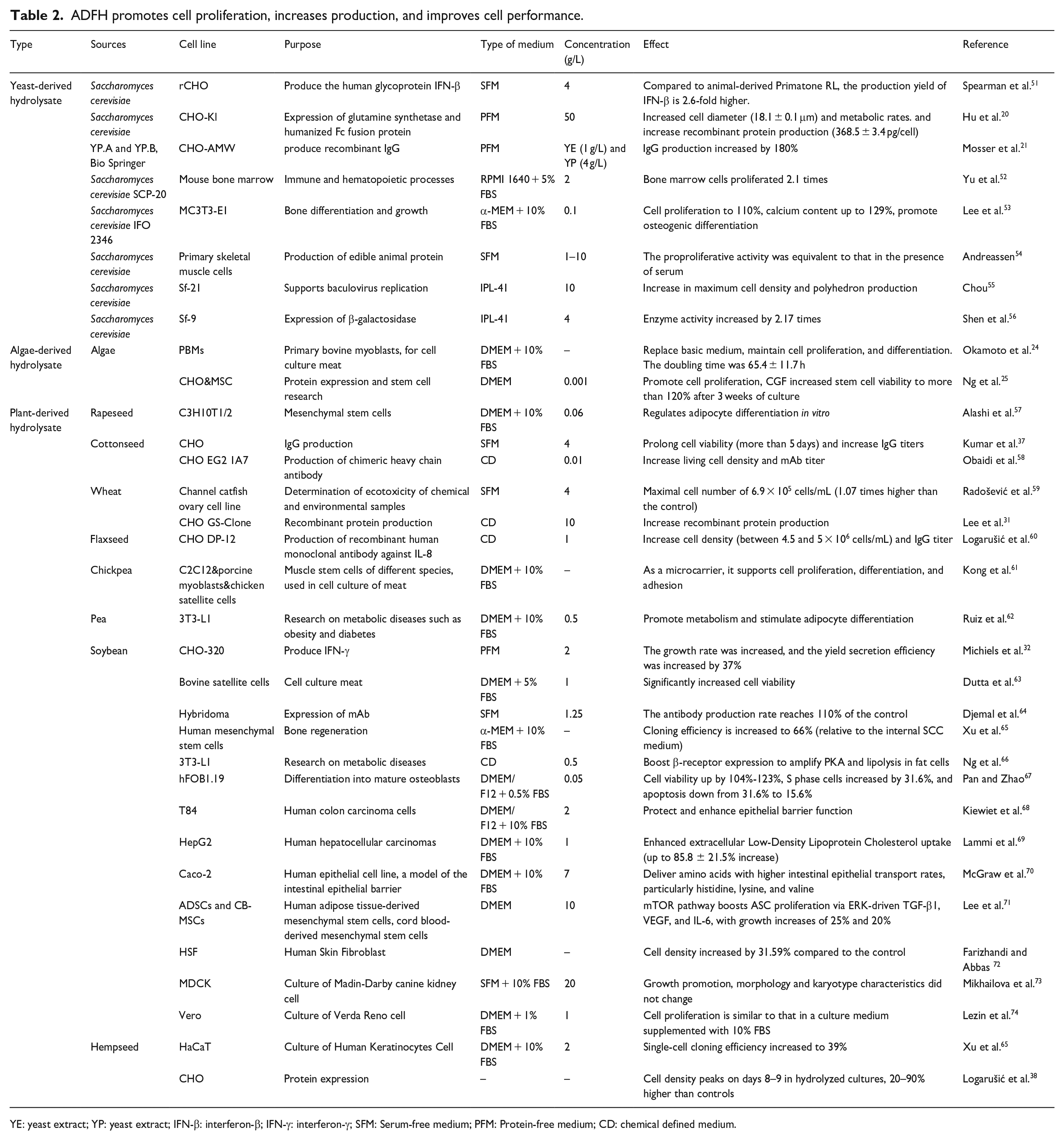
YE: yeast extract; YP: yeast extract; IFN-β: interferon-β; IFN-γ: interferon-γ; SFM: Serum-free medium; PFM: Protein-free medium; CD: chemical defined medium.
In the case of microbially produced ADFH, it was found that adding 10 g/L yeast hydrolysate could support the sustained proliferation of Sf-9 insect cells in serum-free environments. 55 Yeast hydrolysate in serum-free medium (SFM) maintained 50% of skeletal muscle cells’ proliferative capacity and metabolic functionality. 74 ADFH generated from algae provides key sources of fatty acids, free amino acids, trace elements, and minerals required for animal cell development. Research indicates that 50% spirulinum hydrolysate can replace serum to support the cultivation of human lung cancer H460 cells with similar viability, morphology, and proliferation rates. 26 Additionally, spleen cell proliferation and cytokine release can be stimulated by Curtobacterium proimmune lysate, isolated from ginseng beverage. 75
In studies on plant-derived ADFH, Dhara discovered that adding cottonseed hydrolysate to serum-free medium could raise the peak density of CHO cells to 3.7 × 106 cells/mL, significantly enhancing cell survival. 76 Similarly, the addition of 4 g/L wheat gluten hydrolysate to serum-free medium substantially increased the maximum viable cells of the channel catfish ovarian cell line. 59 Alkaline hydrolase-treated soybean and aloe vera hydrolysates can efficiently nourish human skin fibroblast cells, enabling them to achieve their maximum viable cell density. 72 The density of HaCaT cells increased by 120% with the addition of 1 g/L soy hydrolysate to a medium containing bovine pituitary extract. 77
Furthermore, ADFH derived from plants can promote animal cell proliferation and enhance in vitro cell density. Research has shown that soy protein degraded by pepsin can significantly boost the immune system by stimulating lymphocyte proliferation in mice. 78 The addition of 2 mg/L hemp seed hydrolysate significantly increased HaCaT cell proliferation in MTS experiments, resulting in a 61.8% increase in cell viability. 79 Additionally, chickpea hydrolysate, when used as a serum substitute in THP-1 cell line cultures, effectively maintained cell proliferation. 80 Finally, Vero cells, which are primarily used in vaccine production, have been shown to achieve and maintain maximum density under low-serum conditions, including 1% FBS and 0.1 g/L soybean and yeast hydrolysate. 81
In addition to preserving the ability of stem cells to differentiate in culture, ADFH serves as a safe biopromoting additive that supports the maintenance of cell proliferative activity. Research has shown that soybean hydrolysate effectively stimulates adult stem cell (ASC) multiplication while preserving their capacity for differentiation. 71 Ahmed employed an amidation reaction to graft soybean protein hydrolysate onto TEMPO-oxidized cellulose nanofibers (T-CNF), a highly biocompatible method to promote the proliferation of human bone marrow mesenchymal stem cells. 82 Similarly, to enhance the proliferation of porcine muscle stem cells while maintaining their normal differentiation capacity, soybean meal hydrolysate fermented with Aspergillus oryzae and Bacillus subtilis could substitute for up to 50% of FBS. 83 Furthermore, pea globulin hydrolysate can induce lipid accumulation in mouse preadipocyte 3T3-L1 cells during differentiation, thereby promoting adipocyte proliferation and differentiation. 62
Enhancement of protein production and quality
In the biopharmaceutical industry, the use of non-animal-derived materials is increasingly favored to avoid contamination risks and meet stringent regulatory requirements. 84 While HPL supports high recombinant protein yields, ADFH offers a similarly effective yet animal-free alternative. This not only ensures safety but also positions ADFH as a more sustainable and scalable solution for protein production processes. To increase recombinant protein production, yeast is commonly used to produce ADFH. Research has shown that the production of human thrombopoietin (hTPO) in CHO cell suspension cultures can be significantly enhanced when the yeast hydrolysate concentration in SFM exceeds 5 g/L, with production levels reaching up to 11.5 times higher than in SFM alone. 85 Hu achieved a two-fold increase in protein production by modifying the fed-batch culture process through the addition of yeast hydrolysate to the CHO cell medium for the manufacture of Fc fusion proteins. 20 While soybean hydrolysate derived from plants can sustain high cell growth density, yeast hydrolysate maintained high cell growth activity in the IFN-β producing CHO cell system and yielded superior recombinant protein compared to medium supplemented with animal hydrolysate. 51 Saccharomyces cerevisiae IFO 2346 hydrolysate can robustly stimulate CHO cells, bone marrow cell proliferation, macrophage activation, and increased cytokine secretion, including IL-6. 52 The NS0 cell line, a monoclonal antibody (mAb) producing cell, demonstrated a 20% increase in mAb titer with the addition of yeast hydrolysate, while the number of medium components in the mixture was reduced from 67 to 25, resulting in a 20% cost reduction compared to the control. 86
Plant-derived ADFH has also been widely used to enhance recombinant protein expression in animal cells, similarly to microbially produced ADFH. In hybridoma cells, viable cell density was significantly higher in culture medium supplemented with soybean hydrolysate, leading to 1.11 times more monoclonal antibody production. 64 Hydrolysis of flaxseed hydrolysate with alkaline phosphatase significantly increased IgG production in CHO DP-12 cells. 60 Additionally, rice protein hydrolysate effectively stimulated protein synthesis, resulting in a 30% increase in recombinant IFN-γ production in CHO-320 cells, while wheat hydrolysate raised secreted IFN-γ concentration by 60%. 87 Soy peptone addition to ExCell™ 293 medium led to a 3.5-fold increase in recombinant IgG titer in HEK293 cells. 88
Studies have demonstrated that recombinant protein production can be markedly enhanced by adding ADFH from various sources to the culture medium. Lee reported a 3.3-fold increase in the maximal antibody concentration against 4-1BBCHO cells when PFM was combined with soybean and faba bean hydrolysates. 89 An animal-free medium containing 11 g/L soybean hydrolysate and 5 g/L yeast extract (YE) outperformed serum-based medium in supporting the transient infection of Sf-9 cells to express green fluorescent protein and supported both static and suspension cultures of Sf-9 insect cells. 90 Similarly, CHO cell cultures supplemented with 0.1 g/L of soybean and wheat hydrolysates doubled the anti-IgE mAb concentration to 2.6 g/L compared to the control. 91 Murayyan’s screening and optimization of a formula containing four plant hydrolysates (two from soybean and two from wheat) confirmed its potential as a replacement for animal-derived components in CHO cell cultures. 92
Biosimilars and reengineered monoclonal antibodies are critical therapeutic products. Research indicates that post-translational modifications, particularly microscopic heterogeneity in protein alterations, significantly impact the safety, pharmacodynamics, pharmacokinetics, and immunogenicity of these products. 93 Cell biology studies have shown that this heterogeneity is regulated by intracellular and extracellular pathways influenced by the nutrients present during cell culture. 94 Zheng demonstrated that wheat and soybean hydrolysates dramatically altered the distribution of charge variants in monoclonal antibodies produced by CHO-K1 cells, which generate recombinant humanized IgE antibodies. At a subphysiological temperature of 34°C, this led to a significant reduction in acidic variants and an increase in major variants, improving product safety, reducing immunogenicity, and enhancing antibody pharmacodynamics and efficacy. 91 Moreover, recombinant proteins produced by CHO-S-RTX and CHO-EG2 cells exhibited improved galactosylation and sialylation when treated with cotton hydrolysate HP7504, leading to increased yield and quality stability. 58 Monoclonal antibody glycosylation is a critical quality attribute that affects the safety and efficacy of the product, and it is essential to evaluate how medium supplements impact the glycan profile. Comparative analyses of CHO cell-produced monoclonal antibodies in serum-free medium revealed that the addition of yeast or soybean hydrolysates altered the types and distribution of galactosylated glycans, with minimal impact on high-mannose, fucosylated, or sialylated glycans. 8 These findings underscore the ability of ADFH to influence recombinant protein glycan modifications, ultimately affecting antibody functionality.
Extension of cell survival and lifespan
Target protein yield and associated benefits can increase by extending the duration of animal cell culture with the use of ADFH. Yao Ng discovered that Chlorella hydrolysates could successfully maintain the stem cell phenotype for up to 21 days and increase the viability of cells cultured with ADFH derived from adipose tissue and umbilical cord blood by 120% under both 2D and 3D culture conditions. 25 These results demonstrate that microalgae-derived ADFH is highly effective in preserving cell survival, particularly stem cell survival. Chun found that CHO DG 44 cells maintained 50% cell viability for over 8 days while attaining maximum cell density when 4 g/L soybean hydrolysate was added to the culture medium. 95 Haddani significantly increased recombinant IFN-γ production and doubled the incubation duration of CHO cells by adding peptides from rapeseed hydrolysate to PFM. 96 Additionally, the use of green tea hydrolysate containing epigallocatechin gallate (EGCG) enhanced the overall yield of CHO-K1 cells by approximately 50% and extended the batch culture incubation period by 2–3 days by reducing the rate of cell growth. 97 Although HPL effectively prolongs cell viability in culture, its animal-derived nature limits its application in specific bioprocesses that aim to eliminate animal components. ADFH, by comparison, supports extended culture durations with greater assurance of purity and stability, making it an optimal choice for long-term cultures, particularly in animal-free systems. 98
Protection of cellular stability and integrity
Cellular functions and proper physiological processes depend on the stability of cell shape. The inclusion of ADFH has been shown in multiple studies to increase CHO cell diameter from 14–18 µm to 16–20 µm.20,99 Morphological and structural alterations in cells can influence intracellular protein expression, folding processes, membrane permeability, metabolic activity, and recombinant protein productivity. These phenotypic alterations may also have a significant impact on future recombinant protein yields. 100 HPL has been shown to protect cells from environmental stresses, its animal-derived composition introduces variability and potential risks in sensitive applications. 101 In contrast, ADFH offers a stable and reliable solution, ensuring cellular integrity without the variability associated with animal-based materials.
Studies indicate that ADFH preserves intracellular stability by shielding cells from stress and mitigating the negative effects of environmental factors such as oxidative stress. For instance, Lactobacillus fermentans XJC60 can effectively synthesize nicotinamide and protect HaCaT cells from UV-induced oxidative damage. 102 Similarly, selenium-rich yeast hydrolysate with a molecular weight of less than 3 kDa has been shown to protect HaCaT cells. 103 Additionally, we showed that the antioxidant hydrolysate from Shoenu mushroom protein had neuroprotective effects in our previous study by simulating gastrointestinal digestion. 104 This hydrolysate showed significant pre-protective effects against oxidative damage in rat adrenal medulla pheochromocytoma cells (PC12). Further research revealed that the hydrolysate functions as a gene regulator, controlling the expression of endogenous antioxidant enzymes and scavenging intracellular reactive oxygen species. 104 CHO cells and their monoclonal antibodies are protected from free radical damage in the culture medium by natural antioxidants such as resveratrol and catechin, found in some plant hydrolysates, which increases antibody stability. 105
Furthermore, ADFH use can mitigate inflammation, providing cytoprotection, as inflammation often contributes to cell damage (Table 3). High-nucleotide yeast hydrolysate (60–150 μg/mL) significantly improved RAW264.7 cells’ phagocytic capacity and inhibited the release of inflammatory factors such as NO, TNF-α, IL-1β, and IL-6, while affecting the mRNA expression of related genes. 106 Our previous research also demonstrated that polysaccharide fraction F31, isolated from Ganoderma lucidum, reduced liver inflammation and maintained liver cell morphology. 107 Additionally, L. angustiphylla enzyme hydrolysate has been shown to inhibit thrombin and reduce inflammation in several cell lines, including THP-1, ARPE-19, BV2, monocytes, and monocyte-differentiated osteoclasts. 108
Effects of common ADFH on the protection and maintenance of cell stability.
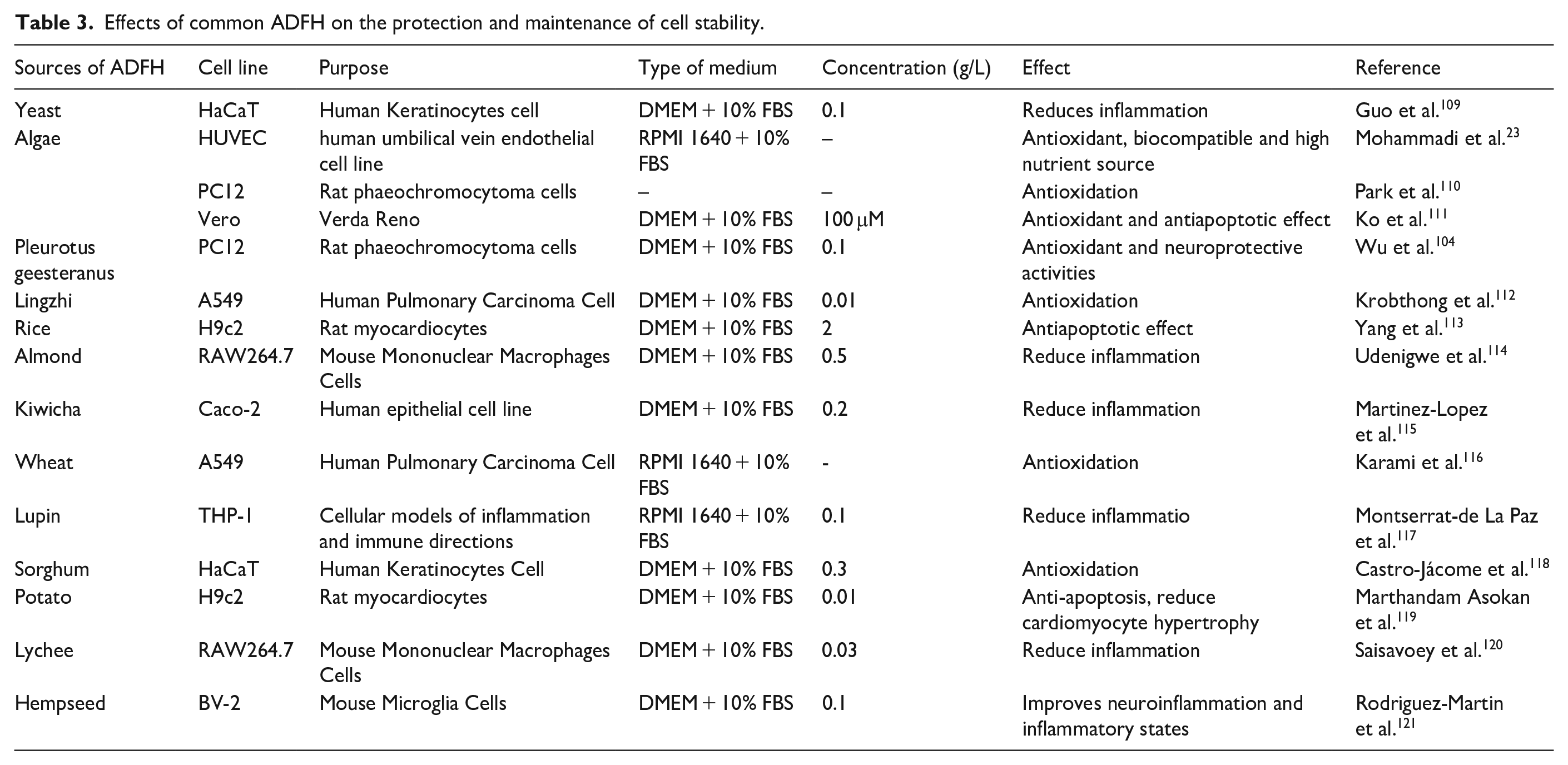
ADFH prevents cell death, maintains cell stability, and shields recombinant proteins from proteolytic enzymes. 51 For example, rice hydrolysate treatment of H9c2 cardiomyocytes reduced H2O2-induced cell death and proliferation suppression, whereas alkaline protease-hydrolyzed potato hydrolysate’s active dipeptide prevented mice’s high-fat diet-induced hepatocyte apoptosis.113,122
The impact mechanism of ADFH
Animal cells can benefit from ADFH through various mechanisms, such as the provision of vital nutrients, regulation of signaling pathways associated with cell proliferation, and promotion of protein synthesis. Additionally, ADFH can regulate animal cell metabolism, the cell cycle, and oxidative stress. By interacting with these systems, ADFH helps to optimize cell function (Figure 3).

Mechanisms of action of ADFH on animal cell culture: (1) Provision of amino acids, peptides, trace elements, vitamins, inorganic salts, nucleosides, fatty acids, and other essential nutrients; (2) Activation of metabolic pathways to stimulate cellular proliferation (e.g. mTOR and NF-κB signaling); (3) Enhancement of the expression of proteins involved in proliferation, metabolism, structure, and other related processes; (4) Antioxidant compounds capable of inhibiting intracellular oxidative stress and reducing free radical-induced damage; (5) Regulation of the cell cycle to promote increased product expression.
Essential nutrients supplementary
The integration of amino acids and peptides within ADFH can enhance cellular nutrient absorption and metabolism, thereby creating favorable conditions for animal cell proliferation and facilitating cellular reproduction and growth. The identification of the precise nutritional components provided by hydrolysate can be achieved through the examination of cellular amino acid and glucose uptake following the addition of hydrolysate, using appropriate metabolic tests such as high-performance liquid chromatography (HPLC). 123 The significance of wheat hydrolysates in CHO cell cultures lies in their ability to provide a plentiful source of amino acids, which is crucial for sustaining a phenotype characterized by rapid cell growth and targeted antibody production. Among these factors, the heightened metabolic activity of serine and glycine may contribute to cell proliferation in cancer contexts. Additionally, the increased flux of the malate dehydrogenase reaction and elevated tricarboxylic acid cycle activity may be associated with optimizing cellular redox balance and energy metabolism. 31
In hybridoma and CHO cells, the use of protein hydrolysate with higher molecular weight peptides can enhance cellular metabolism and optimize amino acid utilization. Research findings indicate that wheat protein hydrolysate has a notable positive impact on monoclonal antibody synthesis in hybridoma cells. 124
Trace elements play critical roles in various aspects of cellular function and have wide-ranging effects on metabolism, morphology, and product quality in mammals. These elements are crucial for modulating cellular development and proliferation. Prior research has shown that ADFH derived from soybean can enhance cellular proliferation by sustaining iron solubility and converting iron ions into ferrous ions. 125 The use of soybean hydrolysate in SP2/0 cell line cultures proved significant due to its abundance of amino acids, peptides, carbohydrates, and trace elements. 64 Subsequent investigations revealed that soybean hydrolysate contains peptides, phytate, and compounds capable of chelating cations, such as Mg²⁺, Ca²⁺, and Zn²⁺. 126 This chelation process facilitates cell proliferation and enhances protein expression. In contrast, Chabanon found that rapeseed hydrolysate shows promise as a viable alternative to trace elements in cultivating the CHO C5 cell line.127,128
The hydrophilic portion of yeast cell lysate, known as YE, contains nucleotides and base components, which can modulate intracellular nucleotide metabolism, supporting recombinant protein synthesis. Additionally, YE can enhance energy provision and facilitate protein glycosylation. 14
Signaling pathway activation and proliferative stimulation
ADFH potentially contains physiologically active constituents, including peptides and growth factors, that can initiate intracellular signalling cascades upon binding to cell surface receptors. Consequently, these interactions can induce cellular proliferation and division. The mechanistic/mammalian target of rapamycin (mTOR) is a serine/threonine protein kinase with a molecular weight of 289 kDa. It is a member of the phosphatidylinositol-3-kinase (PI3K) family, a group of related protein kinases known as PIKKs. mTOR plays a crucial role in coordinating signals that regulate cell development, growth, and proliferation. 129 Another signalling pathway linked to cellular proliferation is p44/42 MAPK (ERK), part of the mitogen-activated protein kinase (MAPK) family, which regulates cellular growth, differentiation, and viability. Research indicates that soy hydrolysate stimulates the mTOR-related signalling pathway through a cascade of molecular and enzymatic processes, leading to the activation of p44/42 MAPK (ERK) and the generation of transforming growth factor-β1 (TGF-β1). TGF-β1 subsequently induces the proliferation of ASCs and promotes the synthesis of cytokines, including vascular endothelial growth factor (VEGF) and IL-6, even in the absence of serum. 71 The addition of yeast hydrolysate induces the up-regulation of mTOR and PI3K proteins in CHO-K1 cells, enhancing cell growth, stabilizing cell characteristics, boosting recombinant protein production, and modulating changes in cell size, composition, and metabolite breakdown via the regulation of cell signal transduction pathways.20,130,131 Peptides generated from Selenium-rich yeast hydrolysate (SeP) have been observed to inhibit the activation of MAPK and NF-κB signalling pathways in HaCaT cells and macrophage cell lines. 109
Cell growth can be stimulated by the interaction between microbial and plant-derived peptides and cell surface receptors. Additionally, specific peptides can serve as carriers or antidotes for lipids, metal ions, or other toxic substances, facilitating their transport. These peptides can also interact with larger peptides to stabilize fragile cell membranes. Plant-derived peptides, particularly high molecular weight oligopeptides, exhibit effects comparable to growth factors in CHO cell cultures. 124 Many oligopeptides positively impact cell viability. For example, certain oligopeptides in hybridoma and CHO cell cultures enhance peak cell density and product yield (Gly-Phe-Gly, Ala-Ala-Ala-Ala, Ser-Ser-Ser, and Thr-Thr-Thr), while others inhibit growth but increase product yield (Gly-Lys-Gly, Lys-Lys-Lys, and Gly-His-Lys), or increase peak cell density (Gly-Gly-Gly-Gly and Gly-Phe-Gly).132,133 YE contains alkaline and hydrophilic peptides consisting of arginine and lysine, which promote cellular growth and metabolism, thereby facilitating cell proliferation. Additionally, cationic peptides can enhance nutrient transfer within cells, thereby promoting CHO cell proliferation. 46
Protein synthesis and folding regulation
The regulation of protein synthesis and folding is critical in cellular activities, as it directly affects cellular function and the overall well-being of the organism. During this process, certain proteolytic products can regulate the expression of cell phenotypic markers, thereby inducing cell proliferation and differentiation. Alkaline phosphatase (ALP) serves as a key indicator of osteoblast development. Research shows that ALP activity significantly increases in osteoblasts when Saccharomyces cerevisiae hydrolysate enzymolysis by bromelain is applied at concentrations of 25–100 μg/mL. Osteoblast differentiation is triggered by upregulation of bone matrix protein expression at the mRNA level. 53
The growth rate of CHO DG44 cells is highest when supplemented with a mixture of soybean and wheat hydrolysates. This supplementation leads to the up-regulation of metabolically related proteins (PGK, ENO, PK, and PSAT), cytoskeletal-related proteins (EBP50, PG, and AnxA2), and proliferation-related proteins (SNEV, CNBP, EBP1). Based on this, yeast hydrolysate can further up-regulate CRP55 and PDI expression, thereby facilitating protein folding and recombinant protein synthesis. 134
Hydrolyzed wheat gluten (HWG) counteracted the suppressive effects of deoxynivalenol (DON) on the expression of Ki 67/PCNA and KRT 20, both markers of cellular proliferation and differentiation. This promotes the proliferation and specialization of intestinal stem cells. Additionally, HWG contains high levels of glutamate and glutamine, which promote the proliferation of small intestine mucosal epithelial cells and regulate tight junction protein-1, safeguarding the integrity of the intestinal barrier. 135 Similarly, transcriptomic analysis revealed that cottonseed hydrolysate significantly up-regulated key genes involved in cell growth (such as Atf family and homeobox proteins), anabolic pathways (such as gluconeogenesis), translation extension, and protein folding, enhancing CHO cell culture performance. 37
ADFH can facilitate cellular proliferation and promote product expression through its regulatory influence on the cell cycle. The use of spirulina animal cell culture solution (SACCS) derived from Spirulina maxima has been effective in promoting cell growth, proliferation, and stimulating immunological activity. Proteomic analysis revealed an increase in ubiquitin expression when FBS concentrations were reduced to 3% or below. Increased ubiquitin production may indicate cell cycle irregularities and heightened autophagy in response to reduced FBS levels. The addition of SACCS enabled normal cell proliferation in the G0/G1 phase, preventing cells from being arrested in the early phase of DNA synthesis (G1 phase), even under low FBS conditions. 26 Similarly, by preventing CHO cells from entering G1, yeast extract can stimulate the transcription of the Fc fusion protein gene and increase target protein expression. 136
Resistance to oxidative damange and apotosis
The cellular antioxidant system primarily involves scavenging hydroxyl radicals and reducing reactive oxygen species (ROS). Hydroxyl radicals are highly reactive free radicals that engage in non-selective oxidation reactions with biomolecules, causing cell membrane degradation and impaired cellular function. ROS include molecules such as superoxide anion (O₂·⁻), hydrogen peroxide (H₂O₂), and hydroxyl radicals, which lead to oxidative stress, resulting in pathological processes like cellular damage and inflammation. Antioxidant compounds can reduce ROS levels by either blocking their generation or enhancing the activity of ROS-scavenging enzymes. 137 As a result, these compounds play a crucial role in protecting cells from oxidative damage caused by ROS. Research shows that various short peptides derived from plants possess antioxidant capabilities by scavenging free radicals or suppressing the generation of oxidants and pro-inflammatory cytokines. 138 Screening of compounds in defatted walnut meal hydrolysate revealed that bioactive peptides P16 and P22, rich in glutamic acid, arginine, aspartate, and glycine, demonstrated effective scavenging of hydroxyl radicals and reduced ROS levels. 139 The hepatoprotective effects of Ganoderma lucidum polysaccharide hydrolysate F31 have been shown to mitigate diabetes-induced oxidative damage through upregulation of glutathione peroxidase and superoxide dismutase (SOD) activity in liver cells. 140
The cellular anti-oxidation system primarily encompasses the process of scavenging hydroxyl radicals and decreasing reactive oxygen species. The hydroxyl radical is classified as a highly reactive free radical that engages in non-selective oxidation reactions with biomolecules, leading to the detrimental effects of cell membrane degradation and impaired cellular function. Reactive oxygen species (ROS) encompass several molecules, including superoxide anion (O2·-), hydrogen peroxide (H2O2), and hydroxyl radical. These species elicit an oxidative stress response within cells, subsequently resulting in pathological processes such as cellular damage and inflammation. Antioxidant compounds have the ability to decrease the levels of reactive oxygen species (ROS) within cells by either blocking ROS generation or improving the activity of enzymes responsible for scavenging ROS. As a result, these compounds play a crucial role in safeguarding cells against oxidative damage caused by ROS. Numerous studies have indicated that a variety of short peptides derived from plant sources possess antioxidant capabilities through their ability to scavenge free radicals or suppress the generation of oxidants and pro-inflammatory cytokines. It was shown that the antioxidant mechanism of various peptides may include varying degrees of hydroxyl radical scavenging and ROS reduction while screening the compounds present in the hydrolysate of defatted walnut meal that had effective protective effects on nerve cells SH-SY5Y. The bioactive peptides P16 and P22, characterised by a high content of glutamic acid, arginine, aspartate, and glycine, exhibited effective hydroxyl radical scavenging activity and a reduction in reactive oxygen species. The hepatoprotective effects of Ganoderma lucidum polysaccharide hydrolysate F31 have been shown in the context of diabetes-induced oxidative damage. This protective mechanism involves the upregulation of glutathione peroxidase and superoxide dismutase (SOD) activity in liver cells.
The hydrolysate of Kluyveromyces marxianus contains the antioxidant peptide VLSTSFPPK (VL-9) and other peptide analogues, which showed significantly increased DPPH and ABTS scavenging activity. The activation of the Keap1-Nrf2 signalling pathway can induce the overexpression of genes encoding antioxidant enzymes. It can also interact with enzymes such as catalase (CAT) and SOD, leading to conformational changes that enhance their enzymatic activity. Consequently, this molecular mechanism effectively mitigates apoptosis and promotes cellular viability. 141 The incorporation of yeast extract (YE) can enhance mRNA expression of recombinant proteins by increasing gene transcription efficiency in CHO-K1 cells. The presence of intracellular recombinant protein can lead to endoplasmic reticulum (ER) stress and an accumulation of unfolded proteins. However, calcium ions (Ca²⁺) and arginine-rich oligopeptides in yeast extract can mitigate these detrimental effects on cellular organelles. 136 Additionally, mannanase-hydrolyzed yeast extract down-regulates immune signalling genes, including IRAK1, IRF7, and JUN, in human colon cancer HT-29 cells, reducing bacterial adhesion and the inflammatory response induced by E. coli infection. 142
Application of ADFH in animal cell culture
Since the development of serum-free hybridoma cell culture in the 1980s, researchers have discovered several alternative serum substitutes that effectively sustain the growth and proliferation of animal cells. These options include recombinant proteins such as growth factors, interferons, insulin, and transferrin, as well as hormones like dexamethasone, hydrocortisone, and growth hormone. 143 Moreover, trace elements and lipid mixes are considered potential alternatives. ADFH, known for its abundance of nutrients and affordability, was first used in microbial culture and industrial strain fermentation. Recently, due to the substantial demand for biological products like monoclonal antibodies, ADFH has been incorporated into serum-free mammalian cell culture. 144 Research shows that ADFH contains growth factors and nutrients similar to FBS, thus supporting the proliferation of mammalian cells and promoting the expression of target products. 13 As a result, ADFH can be used in place of animal-derived ingredients, reducing the use of animal serum, improving the sustainability of cell culture, and better meeting the demands of clinical and industrial applications. This is crucial for mitigating the risks and unpredictability associated with animal sources in biopharmaceuticals and cell culture. The creation of a culture environment suitable for various cell types and applications can be achieved by modifying the quantity and composition of hydrolysate supplied. ADFH has significant potential applications in tissue engineering, recombinant protein and vaccine production, cultured meat, stem cell culture, and regenerative medicine.
Cultured meat and stem cell culture
Cultured meat involves the in vitro proliferation and differentiation of stem cells to generate tissue suitable for human consumption. It represents a promising protein alternative for future agricultural practices. Compared to conventional meat, cultured meat production addresses sustainability concerns, including greenhouse gas emissions and animal welfare issues, positioning it as a more environmentally friendly alternative. 145 Adult stem cells, muscle satellite cells, induced pluripotent stem cells, and other cell sources are commonly used to generate cultured meat. 146 The process relies on cultivating substantial quantities of muscle, fat, or other tissue from a limited number of stem cells or cell lines. The culture medium plays a crucial role in this production process. It must contain essential nutrients, hormones, and growth factors to support cell proliferation and differentiation into fully developed tissues. 147 FBS is used as a supportive culture component in nearly all cultured meat research to date. 148 However, the high cost of FBS has posed a significant obstacle to the commercialization of cultured meat, accounting for approximately 80% of the culture medium costs. 149 Additionally, FBS contains components unsuitable for consumption, further complicating cultured meat development (Figure 4).

Production process of FBS and its role and limitations in cell culture.
Serum components are extracted from fetal bovine blood and undergo a series of filtration and sterilization processes to become commercial fetal bovine serum, which has important applications in mammalian cell culture and biopharmaceutical engineering, yet it has several limitations. The figure was created with BioRender.
Studies show that microbial or plant-derived ADFH could serve as a viable medium supplement for cultured meat production. Hydrolyzed products derived from Chlorella contain glucose, free amino acids, and B vitamins essential for animal cell culture. These components enhance the survival rate of primary bovine myoblasts and have shown comparable efficacy to standard medium in promoting the differentiation of muscle cells. 24 Rapeseed protein hydrolysate-prepared bovine satellite cell medium, Beefy-R, can fully replace recombinant albumin in the original Beefy-9 medium while maintaining normal cell phenotype and myogenicity. 150 Thus, the use of ADFH not only reduces costs but also provides a foundation for the advancement of PFM.
Microcarriers are microbeads ideal for adherent cell development, offering a substantial surface area for cell attachment and proliferation, thereby increasing cell density. 151 One of the main challenges in cultured meat production is the availability of suitable microcarriers to support cell adhesion, proliferation, and differentiation. Kong engineered a practical microcarrier coated with hydrolysate derived from chickpea protein. Experimental results showed favorable proliferative activity and strong affinity toward various cell lines, including C2C12, porcine myoblasts, and 3T3-L1 cells. 61 This substrate material is both cost-effective and scalable. Similarly, microspheres coated with hydrolysate from pumpkin seeds demonstrated similar potential for these cell types. 152 Moreover, the use of a 3D gel cell culture meat substrate, combined with hydrolysates derived from soybeans and peas, can provide a robust, nutrient-rich environment that facilitates rapid myoblast proliferation under low serum conditions. 63 Based on these results, it is evident that combining ADFH with microcarriers can improve production efficiency and quality in cultured meat. Additionally, there is significant market potential for cultured meat in the future.
Recombinant proteins and vaccines
Recombinant therapeutic proteins and monoclonal antibodies are in high demand as novel treatments for immunological disorders and cancer, while vaccines are essential public health tools for controlling the spread of infectious diseases. 153 In addition to their widespread use in the production of viral vaccines, mammalian cell lines undergo complex modifications for the synthesis of immunological and biotherapeutic compounds. 154
Studies have shown that supplementing ADFH with basic nutritional medium can effectively enhance cell proliferation and biological production. This approach offers a potential solution to meet the growing market demand for recombinant proteins. Research shows that supplementing CHO III PFM with ADFH effectively enhances recombinant protein production in CHO-K1 cells, while maintaining the usual transfection capacity of cationic lipids. 155 Furthermore, PFM containing ADFH consists exclusively of hydrolyzed or low molecular weight proteins, such as insulin, which supports the downstream processes of protein separation and purification. 49
ADFH is a production-enhancing ingredient that provides robust support for dependable and efficient vaccine manufacturing. The HEK 293 cell line, derived from human embryonic kidney cells, is widely used for viral vector production, making it a preferred choice among mammalian cell lines. Research shows that the amino acid metabolism of HEK 293 cells can be modified with rice or yeast hydrolysate. The addition of rice hydrolysate to HEK 293 serum-free medium NSFM-13 mitigates the cell density effect and increases viral production threefold.156,157 Similarly, OptiPRO-SFM enhances reovirus generation in Vero cells with the addition of ADFH. Baculovirus-insect cell expression systems are also frequently used for the rapid production of bioactive proteins. 155 Hypep 1510™, a plant-derived hydrolysate, was developed for application in industrial production and is widely used for insect cell cultures. 158
Tissue engineering and regenerative medicine
The advancement of novel biodegradable biomaterials is a key focus in tissue engineering and regenerative medicine. 159 This field aims to facilitate tissue regeneration or replace malfunctioning organs by integrating scaffolds, cells, and bioactive molecules. Recent studies have shown that producing soy-based biomaterials is straightforward, economical, and biocompatible, making these materials promising as novel degradable biomaterials. Nanogel encapsulation of soybean and lupine protein hydrolysates can effectively delay protease breakdown for cell therapy and tissue engineering. 160 Additionally, nanogel can regulate hydrolysate release, thus supporting cell proliferation and tissue functionality restoration. Due to its ability to promote tissue regeneration, soy-derived ADFH shows significant promise for use in dentistry, maxillofacial surgery, and plastic surgery. These biomaterials can integrate with blood clots and promote collagen deposition, providing a cost-effective alternative to using growth factors in wound dressings that stimulate cellular activity and tissue generation. 161 Hydrolyzed polysaccharides from microbes and plants also present significant potential in tissue engineering, wound healing, and drug delivery due to their biocompatibility, biodegradability, and targeted therapeutic activity.162,163
Human embryonic stem cells (hESCs) are an important cell type in tissue engineering and stem cell research, with great potential for current research and future clinical applications. Comprehensive collections of hESCs for scientific and therapeutic uses require affordable, standardized, and consistent cryopreservation protocols. Soybean hydrolysates (Ultrapep™ Soy) and wheat hydrolysates (Hypep™ 4601) can provide cells with sufficient growth-promoting peptides, nutrients, and osmotic protection, making them suitable for the standard cryopreservation of hESCs. 164
Summary and prospects
ADFH has demonstrated potential utility in animal cell culture and offers a wide range of application opportunities. However, several limitations still need to be addressed. Although it has been shown to support cell viability and proliferation, enhance protein synthesis and yield, extend cell survival, and maintain cell stability, there may be variations in ADFH composition and quality. Variability between batches, particularly in nutrient content, peptide profiles, or bioactive components, can affect cell growth, protein production, and gene expression, impacting reproducibility and scalability. 165 Therefore, it is critical to further optimize and standardize the ADFH production process. ADFH’s complex preparation process and wide range of sources could lead to the generation of impurities or byproducts that negatively affect cell performance. 166 Controlling batch-to-batch consistency through standardized sourcing, refined hydrolysis, and targeted quality control measures (e.g. amino acid content, peptide distribution) is crucial. Establishing clear guidelines and requirements is essential to ensuring product quality and safety.
Contamination hazards are more prevalent in microbial-derived substances compared to plant-based alternatives. To mitigate these risks, industry practices rely on microbial strains certified as “Generally Recognized as Safe” (GRAS) by the FDA, ensuring safety through established risk control measures.167,168 As understanding of ADFH bioactive molecules improves, it will be possible to monitor production more precisely, expanding the range of microbial species used while enhancing risk management strategies.
From a regulatory perspective, the adoption of ADFH for commercial-scale production faces significant challenges related to compliance with safety and quality standards. 169 Regulatory frameworks in biopharmaceutical manufacturing, such as those set by the U.S. Food and Drug Administration (FDA), the European Medicines Agency (EMA), and other authorities, require strict control over raw material consistency, traceability, and contamination risks. 101 Variability in ADFH composition and the potential presence of impurities or unidentified bioactive components complicate meeting these regulatory demands. Robust analytical methods must be developed to detect and quantify critical quality attributes (CQAs), facilitating batch equivalence and meeting regulatory requirements for reproducibility. 170 Ensuring batch-to-batch consistency will be essential to comply with Good Manufacturing Practices (GMP) and to maintain positive cell culture outcomes. 171 Additionally, the absence of standardized protocols for characterizing ADFH poses challenges in meeting regulatory requirements for raw materials in biologics production. Further refinement of the manufacturing process and the development of standardized analytical techniques for ADFH characterization will be necessary to facilitate its regulatory acceptance and commercial adoption.
The regulatory landscape for non-animal alternatives is shifting toward chemically defined medium, with authorities promoting animal component-free materials due to safety concerns such as prion transmission.172–174 Although ADFH provides a non-animal-derived alternative, establishing its safety profile and regulatory acceptance will require validation to ensure safety from unknown bioactive substances. Integrating ADFH into bioproduction will thus require collaboration with regulators for risk assessments and validation strategies. Chemically defined medium will be the main focus of future SFM research. ADFH samples should be analyzed using various identification techniques to determine the active substances present. Additionally, animal cells should be used as biological models to evaluate the effects of these active substances on cell proliferation, metabolic activity, gene expression, as well as separation and purification processes (Figure 5). Finally, a scientific foundation should be established to develop an effective chemically defined medium.

Future research directions for ADFH.
Our review addresses ADFH’s applicability across multiple disciplines, outlines its types and components, and examines its effects on animal cell culture and its underlying mechanisms. Hydrolysates generated from non-animal sources are valuable with diverse applications in animal cell culture. To fully capitalize on ADFH’s wide range of applications, researchers must have a comprehensive understanding of its functional elements and mechanisms of action. This knowledge will help advance animal cell culture, biomedicine, and related industries through enhanced innovation and development.
Footnotes
Acknowledgements
Guanghan Fan and Ying Li contributed equally to the work.
Author contributions
All persons who meet authorship criteria are listed as authors. Guanghan Fan: Writing—original draft preparation; Writing—review and editing; Conceptualization; Ying Li: Writing—review and editing; Conceptualization; Qinghua Ye: Project Administration; Qinya Niu: Conceptualization; Xinyu Zhao: Project Administration; Ling Chen: Funding Acquisition; Qihui Gu: Funding Acquisition; Youxiong Zhang: Funding Acquisition; Xianhu Wei: Supervision; Shi Wu: Supervision; Qingping Wu: Conceptualization; Supervision; Funding Acquisition; Yuwei Wu: Conceptualization; Supervision. All the authors read and approved the final manuscript.
Availability of data and materials
Data availability is not applicable to this article as no new datawere created or analyzed in this study.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Key Research and Development Program of China (2023YFF1104100), Science and Technology Program of Guangzhou, China (2024A04J6592), and GDAS’ Special Project of Science and Technology Development (2022GDASZH-2022020402-01).
