Abstract
Three-dimensional (3D) bioprinting has emerged as a promising strategy for fabricating complex tissue analogs with intricate architectures, such as vascular networks. Achieving this necessitates bioink formulations that possess highly printable properties and provide a cell-friendly microenvironment mimicking the native extracellular matrix. Rapid advancements in printing techniques continue to expand the capabilities of researchers, enabling them to overcome existing biological barriers. This review offers a comprehensive examination of ultraviolet-based 3D bioprinting, renowned for its exceptional precision compared to other techniques, and explores its applications in inducing angiogenesis across diverse tissue models related to hypoxia. The high-precision and rapid photocuring capabilities of 3D bioprinting are essential for accurately replicating the intricate complexity of vascular networks and extending the diffusion limits for nutrients and gases. Addressing the lack of vascular structure is crucial in hypoxia-related diseases, as it can significantly improve oxygen delivery and overall tissue health. Consequently, high-resolution 3D bioprinting facilitates the creation of vascular structures within three-dimensional engineered tissues, offering a potential solution for addressing hypoxia-related diseases. Emphasis is placed on fundamental components essential for successful 3D bioprinting, including cell types, bioink compositions, and growth factors highlighted in recent studies. The insights provided in this review underscore the promising prospects of leveraging 3D printing technologies for addressing hypoxia-related diseases through the stimulation of angiogenesis, complementing the therapeutic efficacy of cell therapy.
Introduction
In tissue engineering, the introduction of 3D printing technologies has led to a significant development in traditional biofabrication methods. This progress is characterized by the ability to exert precise spatiotemporal control over the positioning of cells and biomaterials, facilitating the creation of intricate constructs. 1 Its application extends to reproduce the anatomy, biology, and physiology of tissues. From the first demonstration of cell cytoscribing in 1986, bioprinting has experienced significant advancement and has found extensive use in constructing living tissues for diverse applications.2,3 Recent studies have shown that traditional monolayer cell culturing systems experience difficulties in replicating the genetic complexities of the native tissue microenvironment. As a result, cells cultured in two-dimensional (2D) environments do not adequately represent in vivo functionality, phenotype, morphology, and differentiation potential compared to those cultured in 3D environments. 4 To this day, 3D bioprinting has advanced to the point where constructs approaching sizes and geometries relevant to clinical applications can be created by patterning multiple biomaterials and cell types. The technology’s potential is evident in 3D bioprinted tissues such as cartilage, bone, and skin. 5 Moreover, 3D bioprinting can effectively stimulate intricate models such as vascular network and neural networks yielding impressive outcomes.6 –10 By accurately allocating the position of biomaterials, cells, and additives (such as VEGF, a growth factor secreted by cells when they want to form new vasculature), 3D bioprinting facilitates tissue regeneration through the creation of vascular networks.11,12 A rising number of studies have successfully integrated neural stem cells directly into bio-printed scaffolds, showing no adverse impact on viability and cellular activity.13 –15
Among the various 3D printing methods, photocuring 3D printing is one of the first methods introduced in this domain. It relies on the photopolymerization technique, utilizing photosensitive liquid resin as its material. 16 Photopolymerization occurs when light-sensitive chemicals known as photoinitiators are irradiated with various light sources (such as UV, visible light, near-infrared light), causing them to form covalent bonds between them, resulting in a polymer mixture.17 –22 The most basic form of 3D bioprinting that employs UV light is inkjet printing, a method first used in early bioprinting experiments. 23 Despite its versatility and advantages, it comes with certain drawbacks when compared to alternative technologies.24 –26 One limitation is its restricted resolution, typically with a minimum feature size exceeding 100 µm, which may limit its applications. 27 Although recent studies have shown that the resolution of this technique can reach between 10 and 60 μm in droplet diameter, it can be considered as a typical range 3D bioprinting. 28 Digital Light Processing (DLP) is notable among 3D printing methods for its impressive precision. 16 The technology behind most current light-based 3D printers, employing DLP, traces its roots to the invention of the digital micromirror device by Larry J. Hornbeck at Texas Instruments in 1987. 29 Due to the light-based characteristics of these printing platforms, a crucial aspect of advancing angiogenic therapy involves integrating the right cell type with suitable bio-ink and subjecting it to the correct light exposure. Although the used of visible light to induce photopolymerization had create the foundation of DLP, recent researchers prefer the maneuver of UV light to control the crosslinking process in photopolymerization. DLP has advantages in terms of faster speed, convenient manipulation of bioink mechanical properties, and a superior practical resolution of up to 10 μm when contrasted with alternative techniques.24,30
DLP bioprinting is favored for microtissue applications due to its high fabrication speed (100–1000 cubic millimeters per second) surpassing conventional methods, as well as its efficient photopolymerization process at various wavelengths, enabling the creation of complex structures with micrometer-sized resolutions for microvascularized tissue models. 31 Figure 1 presents the schematic demonstration of the general working process of the latest UV-based 3D printing method.
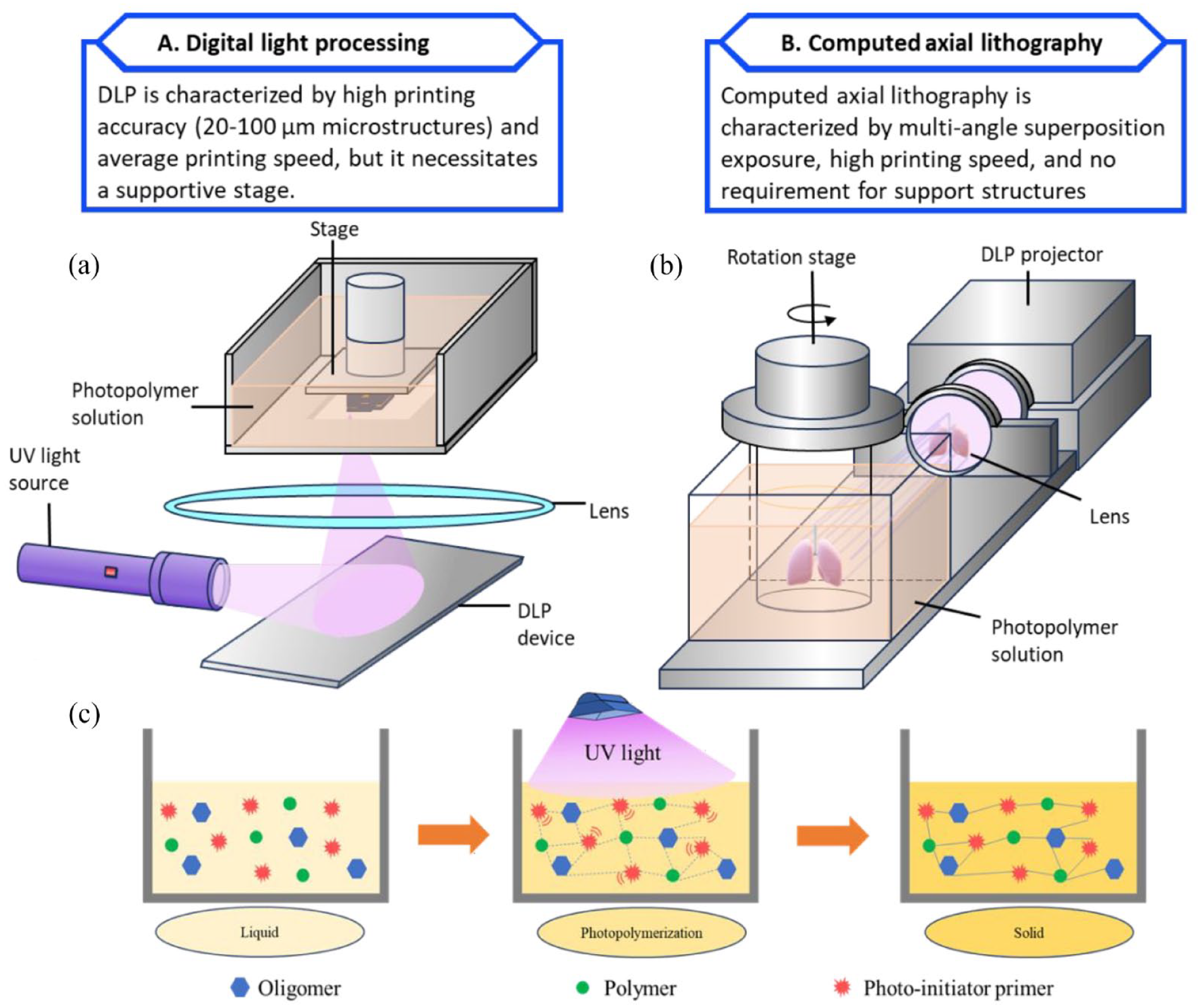
The DLP method is capable of incorporating a higher cell density compared to the volumetric-based method while maintaining the same level of printing resolution. 24 Because of this feature, DLP is preferred for mimicking vascular network structure in, as it enhances cell-cell interactions, which are crucial for the treatment of hypoxia-related disease. DLP bioprinting is favored for microtissue applications due to its high fabrication speed (100–1000 cubic millimeters per second) surpassing conventional methods, as well as its efficient photopolymerization process at various wavelengths, enabling the creation of complex structures with micrometer-sized resolutions for microvascularized tissue models. 31 By applying UV-based 3D printing in cell therapies, the growth and regression of blood capillaries can be modulated to enhance cell proliferation in tissues that are essential to the body (musculoskeletal system, cardiac system, central nervous system, etc.).1,8,32 Noteworthy examples include the development of a model featuring functional living organs based on skin fabrication, human tissues with decellularized extracellular matrix, as well as anatomically accurate trabecular bone models incorporating angiogenic sprouts and meniscal grafts.17,25,33 UV-based 3D printing enabled researchers to examine the geometry of capillaries seeded with endothelial cells and its impact on the spatial patterning of diffusive gradients. Consequently, this influenced the invasion of endothelial cells during angiogenic sprouting induced by a combination of angiogenic growth factors and chemokines. 34 DLP 3D printing has demonstrated a very important prospect to construct bone tissue engineering scaffolds that are required to fit the defect site, allow transport of nutrient and growth factor, and degrade over time. 35 UV-based 3D printing is mainly used for skin tissue engineering with specialized cells like fibroblasts and keratinocytes. However, these cells have limited reproduction capability and are hard to obtain in large quantities, which makes it challenging to use them for treating large skin wounds. 36 Recently, 3D bioprinting methods, multi-photon polymerization (MMP) and volumetric 3D bioprinting have overcome the low resolution and slow speed limitations of existing 3D bioprinting techniques.25,37 –40
The detailed methodology for DLP varies depending on the purpose and protocol of each laboratory. Consequently, the type of printer used, the resolution, the printing speed, and the specific bioinks and cell types employed can differ. We have selected a representative example of DLP, which is presented in this Table 1:
Representative devices and properties for an 3D bioprinting procedure.

This review aims to present the fundamental knowledge and experimental results that support recent advancements in UV-based 3D bioprinting. Furthermore, this review delves into the cell behaviors of recent studies, encompassing the integration of various disciplines in the context of 3D bioprinting using photo-curing devices and its prospective applications in cell therapy. Not only we aimed to summarize important discoveries, but we also wanted to highlight the current challenges and outline the future directions.
Employed cells in 3D bioprinting
The majority of tissues and organs bioprinted in 3D are of small-scale, typically incorporating one or two cell types and manifesting relatively simple structures. 5 For example, when non-immunogenic cell sources are required, mesenchymal stem cells, induced pluripotent stem cells, and adipose-derived cell can be applied.41 –43 Natural tissues are intricate systems comprising two or more types of cells that communicate through vital movement. 44 The cell types utilized in 3D bioprinting to demonstrate hypoxia-related disease models are summarized in Figure 2. The cells selected for in vivo and in vitro experiment must be suitable for its conditions, as each cell type possesses unique characteristics corresponding to the study’s purpose. Stem cells are promising candidate to satisfy tissue model requirements due to their ability to differentiate into many other cell types and their self-renewal properties. 45

Representative cell types employed in 3D bioprinting.
Mesenchymal stem cells (MSCs)
MSCs have emerged as a preferred experimental subject for utilization in 3D printing technology in the past decade. 46 Demonstrating self-renewal capacity, MSCs have the capability to undergo differentiation into various cell types, including chondrocytes, osteocytes, myocytes, and adipocytes. Additionally, they exhibit immunomodulatory properties by releasing bioactive factors like growth factors, chemokines, and cytokines, thereby influencing the immune response, and promoting tissue regeneration. 47 Because of such properties, MSCs are integrated with biomaterials to investigate the biocompatibility of suggested 3D bioprinting procedure. There are numerous factors that can put an impact on the angiogenic process of MSCs in 3D printing model such as cell-cell distance, secretion of bioactive molecules, differentiation of seeded MSCs.48,49 In addition, MSCs can also be combined with other cell types to further enhance this effect. 50
Bone marrow derives stem cells (BMSCs) are the most abundant cells in bone marrow, existing in the body connective tissue and organ stroma. Reports on cell proliferation conditions indicated that BMSCs exhibit proliferation under a wavelength of 808 nm (near-infrared red) irradiation, while they do not proliferate under a wavelength of 470 nm (blue) irradiation.51,52 Given that BMSCs undergo apoptosis after more than 5 minutes of low-wavelength irradiation, it is reasonable to conduct the 3D printing process, which involve with photopolymerization by UV irradiation, within this time frame. 52 After culturing into the 3D printed materials, BMSCs have been observed to enhance motor function recovery by up-regulating vascular endothelial growth factor (VEGF) mRNA expression and promoting angiogenesis. 53
One of the latest studies reported the use of BMSCs in 3D bioprinting technology, specifically on a meticulously designed and biocompatible hydrogel made of methacrylated gelatin/polymethacrylic acid (GelMA/PMAA). 54 The printing technology employed was DLP. The researcher theorized the carboxyl functional group of PMAA would efficiently chelates iron ions, promoting the expression of hypoxia-inducible factor-1 alpha (HIF-1α). The microporous structure of the 3D-printed scaffold actively supports blood vessel growth and bone tissue regeneration. Although GelMA/6% PMAA had a lower optical density compared to GelMA/3% PMAA, GelMA/3% PMMA demonstrated higher cell proliferation through Live/Dead staining, indicating that GelMA/3% PMAA was the more suitable bioink. Co-culturing with GelMA/3% PMAA for 5 days significantly increased the expression of HIF-1α, COL-II, and ACAN, corroborating previous research on chondrocyte formation and specific extracellular matrix secretion. The characteristics of GelMA were not affected by the additive, but its biocompatibility with BMSCs was enhanced. In conclusion, it was shown that the GelMA/3% PMAA hydrogel scaffold could chelate iron ions and induce the endochondral ossification pathway by upregulating HIF-1α expression to facilitate bone tissue regeneration.
Integration of collagen-binding domain-bone morphogenetic protein 2 (CBD-BMP2) -collagen microfibers into BMSC-laden GelMA scaffolds via a customized 3D printer can be served as a differentiation-control module. 55 The authors used growth factor and protein to induce progenitor cells differentiated into osteocyte differentiation of progenitor cells. Despite of using UV light to induce photopolymerization, the procedure used in the study was not DLP, but inkjet printing (as shown in Figure 3). The results indicated that BMSCs showed high cell viability (>90%) during printing. The differentiation into osteocytes in the printed scaffolds was confirmed by RT-PCR method after seeding BMSC on the hydrogel for 14 days. RT-PCR results revealed a higher expression of osteogenic markers, including alkaline phosphatase (ALP), bone sialoprotein, osteocalcin, and collagen type I, in the protein-enhanced samples compared to the samples without CBD-BMP2-collagen microfibers in the growth medium. The elevated gene expression demonstrated the successful osteogenic differentiation of BMSCs in the 3D printed GelMA scaffolds with CBD-BMP2-collagen microfibers, resulting in a mature osteoblast phenotype more efficiently than the osteogenic medium.
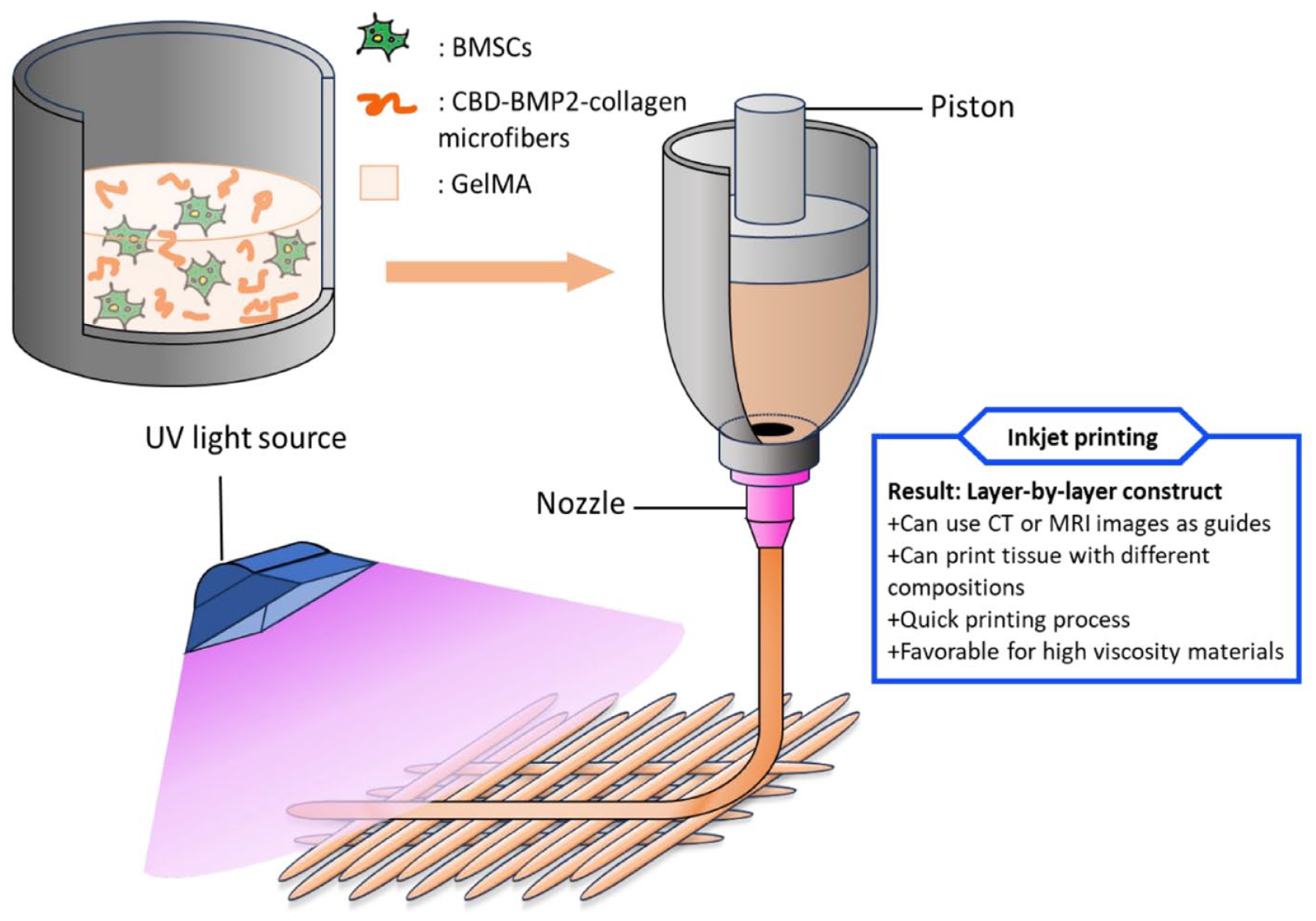
Schematic of the printing process of inkjet bioprinting. 55
In vitro assessments of BMSCs cellular behaviors on resulting biphasic calcium phosphate (BCP) ceramics, focusing on osteogenic activity, were performed in order to find out which is the optimal structure for repairing bone tissue defects. 56 The study observed strong adhesion and similar proliferation of BMSCs on both BCP-Foam (commercial foam BCP) and BCP-3D (3D-printed BCP) samples. BCP-3D and BCP-Foam have different pore structures, BCP-Foam has a non-uniformly distributed spherical macropore structure, whereas BCP-3D has a regular square macropore structure and good connectivity. And the macropore diameter and the interconnected pore size of BCP-3D was greater than BCP-Foam. Notably, BCP-3D exhibited significantly larger spreading areas and higher osteogenic activity than BCP-Foam, as supported by more intensive ALP staining area. This enhancement could have been achieved by two major reasons. BCP-3D has a flatter surface than BCP-Foam, which is conducive to cell spreading. BCP-Foam has a non-uniformly distributed spherical macropore structure, whereas BCP-3D has a regular square macropore structure and good connectivity. The implementation of the DLP technique has significantly improved the curing rate of CaP green bodies from 69.78% to 93.91%, resulting in a 46.34% increase in the mechanical strength of CaP ceramics. In vivo rat cranial defect implantation studies demonstrated that 3D-printed CaP ceramics exhibit superior osteogenic ability compared to commercial alternatives, achieving a level of effectiveness comparable to autografts. The results provided meaningful suggestions, encouraging the scientists to develop mechanically consistent material when culturing cells in 3D models. Overall, BCP-3D demonstrated superior biological performance, fostering cellular responses, and aiding in regenerative repair for large calvarial defects. This may be attributed to its flatter surface compared to BCP foam, which promotes cell spreading. Additionally, BCP-3D possesses a more uniform and wider pore structure than BCP Foam, further enhancing its regenerative properties.
To examine the impact of biomaterial porosity on cell migration, Tao et al. explore the potential of a void-forming hydrogel, GelMA/dextran. 18 The authors conducted the experiment with the purpose to compare the osteogenic differentiation of BMSCs in two physically different environments from the same biomaterial. The study suggested that the porosity of biomaterials had the potential as the matrix for encapsulating BMSCs in bone tissue engineering. BMSCs within the void-forming hydrogels exhibited extended spreading morphology. In contrast, the BMSCs remained spherical in the standard hydrogels. This implies that the proliferation and migration of BMSCs may vary depending on the mechanical properties of the supporting material. The larger macropore diameter is proposed to facilitate the growth of bone tissue, primarily due to the critical role of angiogenesis in bone formation. 57 Biomaterials with good angiogenic potential can supply nutrients essential for tissue growth, thereby aiding in the repair of bone defects. Post-printing Live/Dead fluorescent staining showed that both 3D-printed hydrogels were non-toxic to the encapsulated cells over 5 days. Compared to standard hydrogels, the encapsulated BMSCs in void-forming hydrogels displayed approximately three times higher RUNX2 expression at day 7 and one and a half times higher at day 14. This angiogenic capability is influenced by factors such as macropore and interconnectivity size, permeability, and pore geometry.
Various strategies have been employed to study the behavior of BMSCs in 3D printed materials, with the most common involving the assessment of BMSCs osteogenic differentiation. Moreover, the angiogenic effect of BMSCs contributed to the overall proliferation and differentiation in the materials they imbedded in. 3D printing of hydrogels for BMSCs delivery is gaining attention in bone regeneration, highlighting the technology’s capability to pattern cells and biomaterials at high resolution for custom structures resembling bone tissues. Because of their off-the-shelf availability, non-immunogenicity, and stability after in vitro expansion, BMSCs are a promising cellular source for enhancing therapeutic outcome when combined with other biomaterials. Implanted BMSCs contribute to the fracture healing process through both cellular and paracrine effects, while biomaterials create an optimized 3D space to preserve BMSC functionality, offering enhanced therapeutic efficacy with physiological regulatory capacities. BMSCs exhibited a notable ability to migrate in response to various angiogenic factors and cytokines, including hepatocyte growth factor (HGF), platelet-derived growth factor (PDGF), EGF, VEGF, basic fibroblast growth factor (bFGF), insulin-like growth factor-1 (IGF-1), macrophage inflammatory protein-3 beta (MIP-3β), macrophage inflammatory protein-3 Alpha (MIP-1α), regulated upon activation normal T cell expressed and presumably secreted RANTES, stromal cell-derived factor 1 alpha (SDF-1α), interleukin 1 beta (IL-1β), interleukin 6 (IL-6), interleukin 8 (IL-8), and tumor necrosis factor alpha (TNF-α). 58 Therefore, an 3D bioprinting biomaterial engrafted with BMSCs should not only promote the migration and differentiation of MSCs to the injury site but also facilitate angiogenesis at the site of implantation.
Induced pluripotent stem cells (iPSCs)
Since the first report on the creation of iPSCs through the introduction of four critical transcription factors (
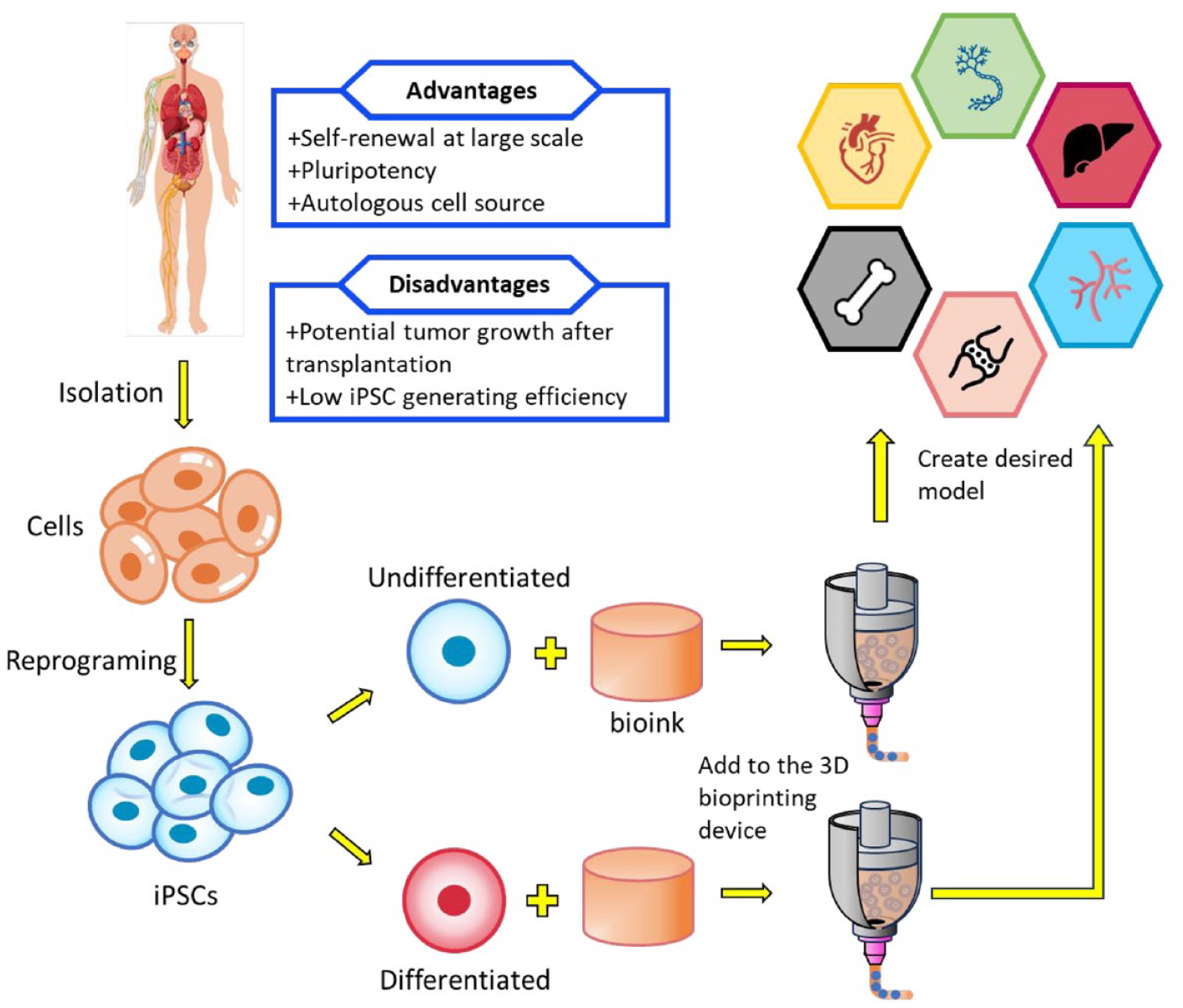
General process of employing iPSCs in 3D bioprinting. First, embryonic cells are isolated from the human body and reprogramed into iPSCs. These iPSCs can then be directly incorporated into the bioink or differentiated into fully matured cells for specific applications before being incorporated into the bioink. Finally, the bioink-cell mixture is loaded into 3D bioprinting devices to fabricate the desired models.
The dynamic development of 3D bioprinting had encouraged many researchers to recreate a liver model to study how hepatocyte assembly can affect major liver functions. In the standard study of this viscera, the authors printed human iPSC-hepatic progenitor cells and supportive cells embedded in glycidyl methacrylate-hyaluronic acid and GelMA, using a DLP bioprinting system. 64 The hepatic lobule structure was recapitulated, and the obtained data demonstrated its advanced morphological organization, increased liver-specific gene expression levels, and the metabolic product secretion in bioprinted human iPSC (hiPSC)- derived hepatic progenitor cells (hiPSC-HPCs), compared to a 2D culture and a 3D HPC-only model (with no supporting cells). 3D hydrogel encapsulation at hepatic progenitor stage exhibits a sustained higher level of albumin production compared with encapsulation at maturation stage. Upon initiation of differentiation, cells at each of the four major stages exhibited stage-specific markers, as confirmed by immunofluorescent staining. Notably, the expression levels of HNF4a, the fetal hepatic marker AFP, and TTR after 10 days of differentiation were significantly elevated compared to those at the endodermal stage, indicating that the differentiated cells had progressed into hepatic lineages after day 10. These findings imply that after several days of in vitro maturation, HPCs may emerge as a more suitable candidate for an in vitro liver model than hepatocyte-like cells. The 3D triculture model presented in the study demonstrated the practical application of DLP-based bioprinting technology in liver tissue engineering. This marks a notable advancement in the field, allowing for the study of liver microarchitecture and cell composition in a physiologically relevant model. The liver organ model has attracted significant interest among researchers, and similarly, the heart organ model has captured the attention of those seeking to replicate it using 3D bioprinting with iPSCs. However, the process is complex, as it allows for printing only one part of the heart at a time. In efforts to replicate native heart valve tissue, a traditional yet efficient approach involves synthesizing and remodeling valve interstitial cells (VICs) within the extracellular matrix (ECM).
To create a fabricated tissues which suitable for the individual patient conditions, Yu et al. developed a novel direct method to produce highly tunable tissue-specific dECM (decellularized extracellular matrices)-based constructs possessing biomimetic microarchitectures with iPSCs. 17 This approach was designed to create biomimetically patterned cell-laden 3D dECM heart and liver tissue constructs to guide cellular organization and provide a complex biochemical microenvironment for promoting the maturation of hiPSC-derived cardiomyocytes (hiPSC-CMs) and hiPSC-hepatocytes (hiPSC-Heps), respectively. For the heart tissues, the hiPSC-CMs in the dECM construct within these parallel regions also revealed a denser cellular network. Cellular localization along the direction of the patterned lines was also more visible in the heart dECM constructs with greater co-staining of actin filaments and sarcomeric α-actinin markers. For the liver tissues, larger hiPSC-Hep aggregates and multicellular spheroids were present throughout the liver dECM constructs. Live/Dead fluorescent images indicated high cell viability with minimal cell death in both types of constructs at all time points, consistent with the observed metabolic activity. In summary, these findings offer a promising foundation for future research aimed at exploring the functionality of hiPSC-derived cells within 3D-printed dECM constructs, aiming to establish a resilient and mature cell population suitable for drug testing and disease modeling purposes.
While numerous novel bioink biomaterials have been developed, there are still natural limitation which these materials are unable to overcome to satisfy the interdependent conditions that will lead to ideal cell proliferation. Though, many researchers were able to confront these problems by tailoring post-print modifications through the conjugation of biologically active molecules. Considering the orthogonality and photoreactive nature of thiol-ene click chemistry, Yu et al. developed the first integrated 3D bioprinting and orthogonal bioconjugation platform that utilizes a rapid DLP-based approach to fabricate biomimetic tissues. 65 The theory regarding how orthogonal bioconjugation can improve material properties was examined within a system comprising a pure gelatin thiol-ene prepolymer. This prepolymer consisted of two precursors: norbornene-functionalized gelatin (GelNB) and thiol-functionalized gelatin (GelSH), utilized as a bioink for DLP-based bioprinting. The emergence of novel materials for integration into 3D printing techniques can leverage iPSCs as in vitro test subjects. Confluent regions of the printed construct also showed the cells adopting a cobblestone-like endothelial cell morphology near the surface of the hydrogel. iPSC-derived cardiomyocytes (iPSC-CMs) were encapsulated in a soft hydrogel formulation chosen to fall within the stiffness range of the developing heart (i.e. ~5 kPa) to evaluate the biocompatibility. GelNB-GelSH hydrogels were able to support greater than 86% viability across all time points for the encapsulated iPSC-CMs. Utilizing a DLP-based orthogonal photoconjugation technique, proteins and peptide growth factors can be immobilized to incorporate additional biochemical properties into 3D bioprinted cell matrices with precise spatiotemporal control, in a scalable manner. The mentioned procedure allows the materials to surpass their original characteristics, thereby increasing the proliferation and migration of cultured cells.
iPSCs emerge as the most potential option for the future progression of bioprinted tissues and organs. Additionally, these cells can be expanded in sufficient quantities suitable for bioprinting, although determining the optimal stage of differentiation prior to bioprinting remains uncertain. The primary challenges associated with employing such cells and their respective differentiation protocols include the significant costs linked to the cell culture medium and the required growth factors. Furthermore, these iPSCs necessitate intricate differentiation protocols, requiring hydrogels that do not hinder the diffusion of essential growth factors guiding these cells toward specific lineages. Extracellular vesicles derived from iPSCs is reported to have angiogenic effects and were used to treat diabetic wounds in mice. 66 Exosomes derived from iPSCs had proven to be protective against ischemic injury in an experimental mouse hind-limb ischemic model through the stimulation of angiogenesis in ischemic muscle. 67 While advancements in cell culture techniques have been made to maintain and expand specialized cells for bioprinting, the inherent biological complexity still imposes limitations on their application. Mature cells derived from iPSCs could potentially overcome these challenges if properly maintained in culture and scaled up for expansion. Nonetheless, the potential of iPSCs in 3D bioprinting has not been fully explored.
Muscle-derived stem cells, adipose-derived stem cells, and endothelial cells
Because Muscle-derived stem cells (MDSCs) exhibit mild angiogenic characters, the utilization of MDSCs in 3D bioprinting is usually neglected in recent research. Nonetheless, several attempts have been made using this cell type in exotic application. An et al. designed HIF-1α overexpressing MDSCs through lentiviral transfection, aiming to regenerate injured corpus cavernosa using 3D-printed hydrogel scaffolds. 68 Compared to control groups receiving normal MDSCs, scaffolds modified with heparin and loaded with HIF-1α-overexpressing MDSCs significantly enhanced the expression of angiogenesis-related genes and proteins, such as VEGF, PDGF, and SDF-1, and promoted neovascularization in vivo under both hypoxic and normoxic conditions. Additionally, 3D Live/Dead imaging demonstrated that the cells maintained high viability and were uniformly and densely distributed across the heparin-coated scaffolds. These bioengineered scaffolds have good biocompatibility with more than 94% survival rate of cells on them, support the adhesion, migration, proliferation, and differentiation of cells. Moreover, the incorporation of heparin onto the surface of 3D-printed scaffolds facilitates the selective binding of angiogenic factors, thereby prolonging their efficacy. This mechanism expedites angiogenesis regeneration and increases vascular density within tissues. Notably, this study represents a rare exploration of MDSCs’ angiogenic potential in the context of hypoxia-related disease treatment.
Adipose-derived stem cells (ADSCs) have the capability to differentiate into multiple cell lineages in 3D culturing environment. 69 It can secrete numerous growth factors and cytokines that are critical for tissue generation. One of the models that has been highly favored by scientists attempting to replicate using 3D bioprinting is human skin. Eke et al. proposed the effective crosslinking of gelatin and hyaluronic acid to form a GelMA/hyaluronic acid biocomponent network (BCN) hydrogel structure, suitable for incorporating ADSCs. 70 The hydrogel solution was grafted for delivering ADSCs to wound beds to stimulate new blood vessel formation and later use as the dermal layer of a bilayer skin substitute. Live-Dead Cell staining revealed that live cell coverage was 99%, 97%, 94%, and 96% on days 3, 7, 14, and 21, respectively, indicating that over 90% of the cells remained viable, capable of elongating, and able to move freely within the hydrogels over a period of 3 weeks. The study revealed that ADSC-laden hydrogels promoted angiogenesis in vivo three time better compared to cell-free hydrogels. The inclusion of hyaluronic acid in the hydrogel mimics the in vivo cellular environment, playing a crucial role in promoting early inflammation, which is essential in overall process of skin wound healing.
Endothelial cells have traditionally served as an in vitro model for investigating endothelial cell function and their responses to various stimuli. When subjected to appropriate conditions, human umbilical venous endothelial cells (HUVECs) can be differentiated into 3D spheroid cultures or 3D co-cultures, serving as advanced models to enhance our understanding of endothelial cell behavior.71,72 A innovative method called Organ-on-a-chip (OoCs) utilizes systems containing engineered or natural miniature tissues cultured within microfluidic chips. 73 These chips are designed to mimic human physiology more precisely by controlling cell microenvironments and maintaining tissue-specific functions. DLP-based bioprinting was employed for the creation of GelMA-PEGDA (methacrylated gelatin-polyethylene glycol diacrylate) microfluidic chips with functional potential as OoCs. 74 The bioink’s purpose is to serve as an ECM for cell growth. Subsequently, HUVECs were cultivated on the chip to establish vascular channels. To assess the bioink’s toxicity, cell viability was analyzed both post-fabrication and after 24 h, revealing that in the absence of PEGDA, 3% and 5% GelMA had approximately 74% and 83% viable cells at 24 h, respectively, while in the presence of a light absorber, the viability was around 96% and 94% post-bioprinting. Convenient transportation of nutrients, solutes, and chemotactic responses to cells was achieved through the bioprinted vasculature. The sustained dynamic flow induced physiological shear stresses, leading to the formation of lumen structures and the generation of angiogenesis biomarkers such as CD31 within 10 days. This biofabrication approach enables the creation of platforms for drug discovery and therapeutic screening. OoCs boast small sample sizes, low reagent consumption, affordability, and ease of handling. By employing microfluidic chips, they integrate multiple biological materials, physiological stiffness, and diverse biological processes via the mass transportation of biological cells. The HUVEC-employing model, representing the human vascular endothelium physiologically, enables the study of physiological and pathological effects of various stimuli, both in isolation and in co-culture with other cell types.33,74
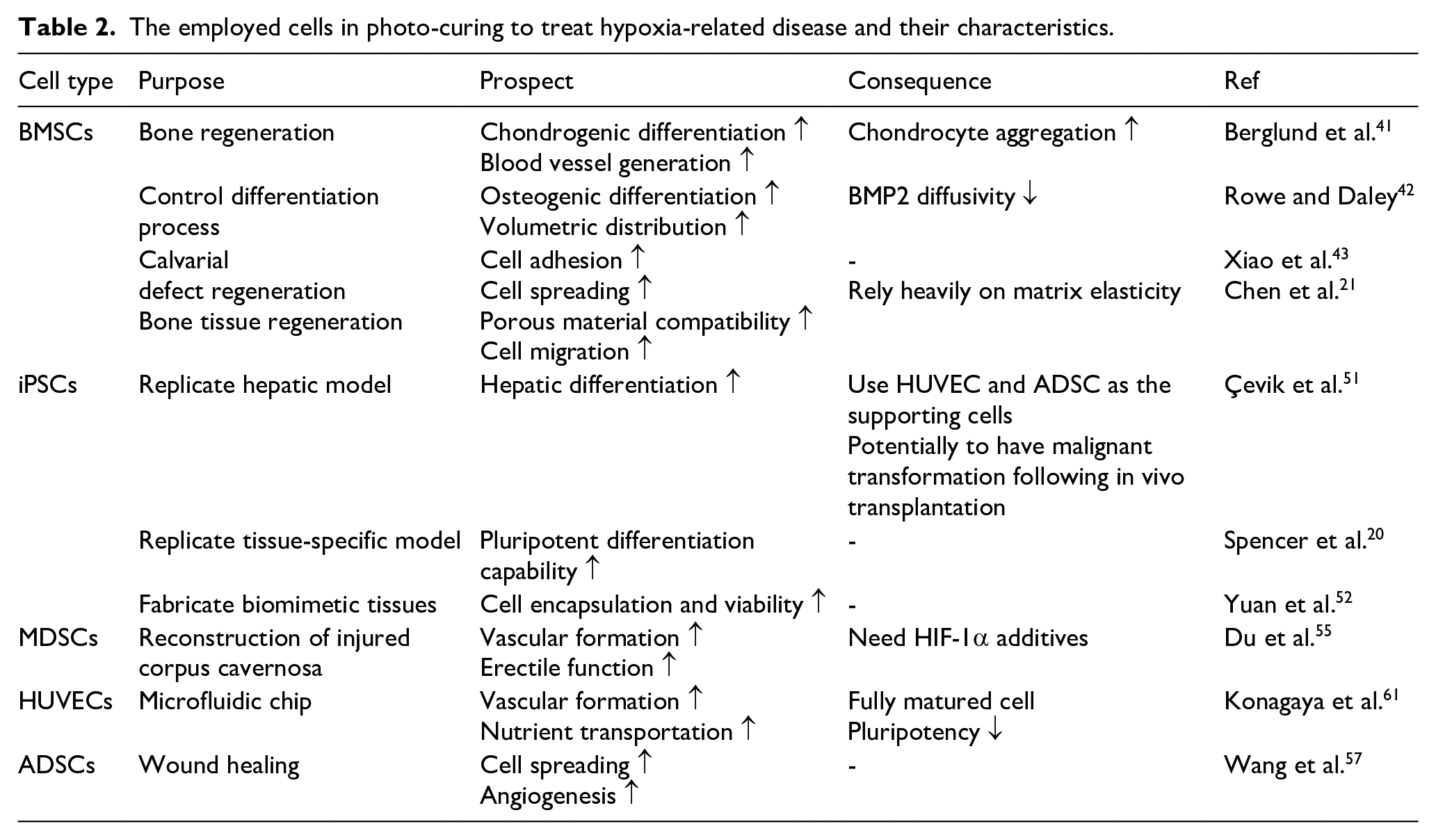
The investigation of various stem cell types in 3D bioprinting has produced significant insights and advancements in tissue engineering. While bone BMSCs and iPSCs dominate research on hypoxia-related diseases due to their established ability to activate angiogenic responses in murine models of hindlimb ischemia, other cell types are increasingly being explored in recent studies on angiogenesis.75,76 ADSCs have emerged as strong contenders in angiogenesis-related research.77,78 Although both ADSCs and BMSCs are mesenchymal stem cells, ADSCs have demonstrated superior blood flow recovery in ischemic models, which may prompt further attention and investigation into their potential in future studies. 52 Human umbilical vein endothelial cells (HUVECs) play a crucial role in angiogenesis due to their capacity to form capillary-like structures known as tubes. 79 However, as fully differentiated cells, HUVECs lack multipotency and are typically combined with other stem cells, such as BMSCs or ADSCs, to enhance their angiogenic properties (Table 2).80,81
The employed cells in photo-curing to treat hypoxia-related disease and their characteristics.
Scaffold free 3D bioprinting for angiogenesis
Recently, scaffold-free 3D bioprinting methods containing living cell-only aggregations without biomaterial carriers have emerged as a promising alternative way to replace the downsides of scaffold-based 3D bioprinting, such as low cell engraftment rate, cytotoxicity, and toxic degradation byproducts. 82 Bioink is an essential element in the 3D bioprinting process, acts as a carrier for living cells, provides structural support, has biocompatibility, biodegradability, supports cell viability, function, mechanical stability, and complies with the bioprinting process with shape fidelity to avoid deformation during printing.83 –87 Before the emergence of scaffold free 3D bioprinting method, scaffold-based 3D bioprinting was represented as a groundbreaking approach in the field of tissue engineering, combining advanced printing technologies with biomaterial science. Scaffold based 3D bioprinting method uses a scaffold made from synthetic or natural biomaterials. By freeze-drying, computer-based method, or 3D printing, scaffold can get porous structure, mimicking the extracellular matrix (ECM) of natural tissues.88 –91 On the scaffold, cells can be seeded, and the type of cell is chosen differently depending on the therapeutic purpose.90,92 While scaffold-based 3D bioprinting holds great promise for various applications, there are several challenges and disadvantages associated with the technology such problems with biocompatibility, cytotoxicity, toxic degradation byproducts, and low cell engraftment rate.82,93,94 Poly-L-lactic acid (PLLA) is commonly used material to make scaffold for bone regeneration, but PLLA has acidic degradation byproduct that can cause inflammatory response after implantation. 95 Also, after scaffold implantation, host phagocytes attached to the scaffold surface and scaffold surface is covered with host tissue proteins such as fibrinogen. Absorbed fibrinogen interact with host leukocytes, triggering inflammatory response. 93 While polymers such as collagen, hyaluronic acid, and alginate based bioinks are biocompatible, these bioink-based 3D bioprinted structures do not make rejection after implantation.96,97,107 Ensuring that the scaffold is biocompatible and does not cause inflammatory response is crucial because some scaffold materials may trigger immune rejection in the host body, impacting the success of the tissue engineering process. Also, natural polymers such as gelatin, hyaluronic acid, and alginate based bioinks with non-toxic properties for encapsulated cells are essential for scaffold-free 3D bioprinting for regenerative medicine.97,98 Non-homogeneously and non-precisely loaded cells on the scaffolds result in substantial cell loss after scaffold engraftment, which is the major cause of low efficacy of cell therapy.82,99 By encapsulating cells within bioinks, the limitation of the cell loading method of scaffold-based methods can be overcome without damage during the printing process. 100 Furthermore, cell encapsulation within polymer based bioinks such as PEG and gelatin can act as a barrier to external stimuli, resulting in improved cell viability and cell retention rate. 82 To overcome these limitations of scaffold-based 3D bioprinting, several components of scaffold free 3D bioprinting bioink and 3D bioprinting technologies have been studied.
The components of 3D bioprinting bioink can vary depending on the intended therapeutic applications. However, some common components of bioinks include cell, hydrogel, and supportive additive. For cartilage regeneration, primary chondrocytes laden silk-gelatin bioink can be used. Silk-gelatin bioinks not only enhanced the printability but also increased the production of ECM components such as sGAG, collagen, the most important substances for cartilage regeneration. The poly-alanine sequence in silk can improve the mechanical properties of bioinks result in appropriate characteristics for 3D bioprinting process such as enhanced printability and shear thinning behavior. Also, the existence of the cell binding RGD peptides in silk fibroin and gelatin within printed structures result in stimulation of cellular signaling and enhanced ECM formation. 101 Furthermore, pro-angiogenic cell such as HUVECs laden bioinks printed structure showed enhancement of vascularization abilities in in vivo test. Bioinks added with Li, Mg, and Si particle with HUVECs showed enhancement of cell survival and proliferation, while also demonstrating lasting capabilities in promoting blood vessel formation. 102 Enhancing angiogenesis via the improved angiogenic effects of cells embedded in bioinks, along with increased cell survival and cell engraftment rate through porous structural patterning using 3D bioprinting, could significantly improve the therapeutic outcomes for hypoxia-related diseases. 103 Also, porous structure of the printed constructs can mimic microenvironments of natural tissue, facilitating the adequate supply of oxygen and nutrients. 104 Hydrogel of bioink provides the structural support for the bioink and mimics the ECM in natural tissues. Hydrogel of bioinks can be made from biocompatible polymers that printable, harmless, and provide a suitable environment for cell growth.83,84,105,106 Various polymers for 3D printing bioinks, including natural polymers (e.g. alginate, collagen, hyaluronic acid, and gelatin) and synthetic polymers (e.g. polylactic acid (PLA), polyethylene glycol (PEG), and polycaprolactone (PCL)), have been explored as bioink components.83,105 –107 Furthermore, bioink mixed with other natural polymers can be used to overcome limitations of a single natural polymer based bioink such as the low stability. Gelatin, sodium alginate, and oxidized sodium alginate based bioink showed enhancement of stability of printed structures. In in vivo test, porous structure, and fast degradation time of gelatin, sodium alginate and oxidized sodium alginate based bioink printed structures showed enhancement of tissue regeneration and angiogenesis. 108 The use of synthetic polymers in bioink has several advantages such as mechanical stability and flexibility, but it has several disadvantages related to relatively low cell adhesion properties. Compared to natural polymer, synthetic polymer has less cell-binding motifs result in low cell adhesion and proliferation within printed structure. To overcome limitations, loading additives such as arginine-glycine-aspartic acid (RGD) and tyrosine-isoleucine-glycine-serine-arginine (YIGSR) peptides can be used. Supportive additives such as growth factors and crosslinking agents play a crucial role in bioink formulation, particularly in the field of 3D bioprinting for tissue engineering and regenerative medicine. Growth factors, signaling molecules are proteins that regulate various cellular processes, including cell proliferation, differentiation, and migration. Growth factors such as vascular endothelial growth factor (VEGF) and transforming growth factor-β (TGF-β) can be added in bioinks to regulate cell proliferation and differentiation.105,109
Photocrosslinking is a crucial technique in scaffold free 3D bioprinting, because photocrosslinking takes part in creating stable connections between polymer chains in the bioink to enhance the structural integrity of the printed structures. Under light irradiation, polymers in bioink can absorb light energy result in formation of covalent bonds. 110 Therefore, crosslinking process is essential for maintaining the shape of the printed structure, preventing deformation, and supporting the viability and functionality of encapsulated cells within printed structures. Among the various photocrosslinking methods, the most common and effective way is the UV based photopolymerization method. UV photopolymerization method involves the use of UV light to initiate the polymerization and cross-linking of the bioink, allowing precise control over the printing process. UV photopolymerization not only provides mechanical stability, but also has the advantage about not negatively affecting the viability of encapsulated cells. After UV photopolymerization, cell viability remained high with enhanced cell proliferation. 111 Also, UV photopolymerized printed structures can be connected with vasculogenic peptides (e.g. QK peptide) resulting in enhancement of angiogenesis. 112 Furthermore, UV photopolymerization based 3D bioprinting bioinks with photoinitiator (PI) and UV absorber (UA) was successfully applied to print cells loaded structures with improved resolution without cytotoxicity. 113 In addition, two types of polymers can be added in bioinks. For example, gelatin methacryloyl (GelMA) and collagen type I mixed bioink showed enhanced stability of printed structure and cell migration. GelMA and collagen added bioink showed higher elastic shear modulus because of gelation by collagen result in mechanical stability after printing. Also, GelMA and collagen added bioink have a fibrillar collagen network, which can offer a guide for cell migration result in enhanced cell spreading. Besides, GelMA and collagen added bioink based printed structures showed enhancement of vascular structure formation. 114 Various combinations of bioink components showed better tissue regeneration effects and printability. In this paragraph, we will discuss about several components of bioink and their applications.
UV based 3D printing system with various bioink for angiogenesis
In scaffold-free 3D bioprinting, bioink modified by UV photopolymerization play a significant role in creating stable and well-defined structures. UV photopolymerization is the process that uses UV light to initiate chemical reaction within the bioink, leading to the crosslinking of polymers in the hydrogel of bioinks. This crosslinking provides mechanical stability to the printed structures, allowing them to maintain their shape and integrity after printing. GelMA, PEGDA, Collagen-Methacryloyl (ColMA), HAMA (methacrylated hyaluronic acid), and Alg-MA (alginate methacrylate) are widely used polymers in bioinks for angiogenesis, all known for their biocompatibility and tunable mechanical properties.
GelMA
GelMA is a widely used bioink hydrogel in scaffold-free 3D bioprinting that undergoes UV photopolymerization. GelMA is derived from gelatin, a natural protein, and modified with methacrylate groups, enabling it to undergo photocrosslinking when exposed to UV light in the presence of photoinitiators such as lithium phenyl-2,4,6-trimethylbenzoylphosphinate (LAP) and Irgacure 2959.19,115,116 The photopolymerized GelMA based bioink can provide a way to place encapsulated cells accurately and enhance mechanical stability with high resolution of printed structure. When the UV exposure time increases, the storage modulus value of bioink is increased, resulting in the stabilization of printed structures. 117 Also, after UV photopolymerization, the GelMA based bioinks preserved its bioactive properties, providing a favorable microenvironment for cell behaviors and survival.19,118 This is crucial for supporting angiogenesis, as the hydrogels interact with cells involved in blood vessel formation. 119 Also, photopolymerized GelMA based bioink application showed enhancement of cellular activities such as cell migration and proliferation result in enhanced skin tissue regeneration.19,120 3D bioprinted structures of GelMA based bioink can mimic the ECM of natural tissues, enhancing interaction between encapsulated cells and printed structures or interation between each type of encapsulated cell, resulting in enhanced angiogenesis.103,121 The degree of crosslinking can be controlled by concentration of GelMA and photoinitiator, degree of methacryloylation, intensity of light, and exposure time.120,122 –125 Also, GelMA is known for its biocompatibility and the presence of cell-adhesive motifs such as RGD peptides derived from gelatin.105,126 The combination of GelMA based bioink and UV photopolymerization in scaffold-free 3D bioprinting demonstrates promising results in achieving both structural stability and promoting cellular activities.
PEGDA
PEGDA is a common polymer used in bioink formulations for 3D bioprinting. PEGDA is synthesized by dissolving PEG in a methylene chloride solvent with a triethylamine catalyst and adding acryl chloride. PEGDA hydrogel is attractive for use as bioink component due to its tunable mechanical properties, biocompatibility and ability to form stable crosslinked structures when exposed to UV light in the presence of a photoinitiator (e.g. Irgacure2959, diphenyl(2,4,6-trimethylbenzoyl)phosphine oxide (TPO), LAP).113,125,127 The mechanical properties of PEGDA based bioink can be easily tuned by adjusting factors such as the concentration of PEGDA, the degree of crosslinking, and the molecular weight of PEG.125,128,129 The UV photopolymerization process in PEGDA bioink is crucial for creating stable 3D structures and enhancing printability with increased shear thinning properties, stiffness, and fidelity. 128 The degree of photopolymerization can be controlled by adjusting parameters such as UV exposure time and light intensity. 113 Tunable properties of PEGDA based bioink enables researchers to tailor the mechanical properties of the printed structures to match the requirements of specific applications. Also, PEGDA can be easily modified and combined with other materials to enhance its properties or introduce specific functionalities. For example, PEGDA can be blended with other materials (e.g. methacrylated tilapia collagen (MATC), PCL, and chitosan) to enhance cell adhesion, proliferation, migration, printability and mechanical strength of printed structures.130 –132 Furthermore, in bioprinting applications, PEGDA based bioinks can be used to encapsulate living cells within a supportive hydrogel, allowing for the creation of structures that mimic the natural ECM by including additives such as peptides and growth factors. 133 Sustained releasing of growth factors from 3D printed structures of PEGDA based bioinks can stimulate cellular activities of encapsulated cells, such as cell migration, which is essential for effective angiogenesis. 134
After UV photopolymerization, implantation of printed structures showed high cell viability, resulting in enhancement of pro-angiogenic effects by encapsulated cells such as HUVECs and endothelial cells.135,136 With formulation of various types of materials, UV photopolymerized PEGDA based bioinks have shown promise for applications in tissue engineering and regenerative medicine.
ColMA
Collagen is one of the main components constituting an ECM in natural tissues and plays a role in regulating structural support of tissues and cell signals. Collagen has a cell adhesion characteristic by having GFOGER binding sites, which relies on collagen’s helical structure. 137 By collagen’s cell adhesive motif, collagen based biomaterials can interact with cells, resulting in enhancement of cell viability and growth rate. 138 Furthermore, the connection between collagen and integrins of the cell surface significantly influences the process of blood vessel formation. 139 Also, this natural component can offer biocompatibility and biodegradability without rejection, making it suitable for enhancing cell growth and tissue regeneration. 140 Although collagen based bioinks have great potential, it has limitations about their insufficient mechanical strength and printability needed for a successful 3D bioprinting process and after printing.141,142 To overcome these limitations, UV photopolymerization method can be used. The collagen functionalized by the addition of methacrylic anhydride is called ColMA. 143 Added methacryloyl groups crosslink when it is under UV light in the presence of photoinitiators (e.g. LAP, Irgacure 2959) result in stable hydrogel structure.144,145 This photopolymerization process is crucial for enhancing the mechanical stability of printed structures and printability with enhanced storage modulus and viscosity.144,146,147 By controlling factors such as concentration of ColMA, amount of crosslinking agent, light intensity, and exposure time, mechanical properties of printed structures can be adjusted. 145 In general, more crosslinked printed structures tend to show stronger mechanical properties and long degradation time. 146 This fine-tunable and natural ECM mimicking properties make ColMA based bioinks suitable for regenerative medicine and tissue engineering applications.
HAMA
Hyaluronic acid (HA) is a natural polymer found in the ECM of connective tissues, renowned for its biocompatibility, biodegradability, and bioactivity.96,148 Also, hyaluronic acid can interact with stem cell’s CD44 surface receptors, resulting in enhancement of regeneration efficacy of HA scaffold-based stem cell therapy. 149 Interaction between HA and CD44 surface receptors can induce an increase in cell migration, adhesion, differentiation, and angiogenesis, which are essential for effective tissue regeneration.150,151 However, pure HA is not suitable for using as a bioink hydrogel for 3D bioprinting. HA in aqueous solutions forms viscous shear-thinning mixtures with a high viscous modulus, which means there is no ability to retain shape after printing and there is possibility to induce shear forces on loaded cells in HA based bioinks.149,152 Also, hydrophilic properties and rapid degradation of HA in the human body result in limited usage in the clinical field. 149 Consequently, HA alone cannot withstand the stress of the printing process. Therefore, HA is typically combined with other materials to improve its printability and structural integrity.150,152 HAMA is an innovative bioink material for 3D bioprinting applications. The methacrylation of HA introduces methacrylate groups, enabling HAMA to undergo UV photopolymerization and form stable crosslinked networks when exposed to UV light in the presence of photoinitiators such as LAP or Irgacure 2959.152,153 This crosslinking process enhances the mechanical stability and printability of HA-based bioinks while preserving the biological performance of HA.148,154 After methacrylation, HA can get shear thinning properties, fine fidelity, and prolonged degradation time after the 3D printing process, making HA based bioink more suitable as bioink for 3D bioprinting. 148 Moreover, the incorporation of cell-adhesive motifs such as RGD peptides makes HAMA based bioink a versatile material for tissue engineering and regenerative medicine applications. 150
Alg-MA
Alginate is a natural bipolymer extracted from brown seaweed, widely utilized in bioink formulations for 3D bioprinting due to its biocompatibility, non-toxicity, and gelation properties. Alginate undergoes ionic crosslinking in the presence of divalent cations (e.g. Ca2+, Sr2+), forming hydrogels that provide a supportive environment for encapsulated cells and tissue regeneration.97,155 However, alginate has limitations in mechanical strength and cell adhesion, requiring modifications to enhance its functionality. 156 Alg-MA is a chemically modified form of alginate, and methacrylate groups are introduced to enable UV photopolymerization. This modification enhances the mechanical properties and printability of alginate-based bioinks by forming stable, crosslinked networks when exposed to UV light in the presence of photoinitiators such as LAP or Irgacure 2959. The degree of crosslinking in Alg-MA can be finely tuned by adjusting the concentration of photoinitiator, and the parameters of UV exposure, such as intensity and UV exposure time.157,158 Also, reaction time to make Alg-MA can affect the mechanical properties of printed structures. High functionalized Alg-MA bioink printed structures showed smaller pores, lower swelling capacity, and faster degradation time. 158 The photopolymerized Alg-MA bioinks retain the biocompatible and non-toxic characteristics of native alginate while providing improved mechanical stability. These properties make Alg-MA based bioinks more suitable for creating 3D structures that support cell encapsulation. 159 Moreover, Alg-MA can be blended with other biomaterials or functionalized with bioactive molecules to further enhance the loaded cell functions and tissue regeneration capabilities.159,160 The photo-curing bio-inks and their associated characteristics are comprehensively summarized in Table 3. In summary, the combination of alginate’s natural biocompatibility with the enhanced mechanical properties provided by methacrylation and UV photopolymerization makes Alg-MA a promising material for advanced applications in tissue engineering and regenerative medicine.
Mechanical properties and printability of bioink depending on the type of hydrogel.

UV based 3D printing with additives
In scaffold-free 3D bioprinting, hydrogels such as GelMA and PEGDA provides mechanical support in printed structures. Also, this mechanical support can be enhanced by photopolymerization based on crosslinking by UV irradiation. Furthermore, these materials provide characteristics of mimicking the microenvironment of natural tissue result in enhancement of cellular activities such as cell adhesion, proliferation, and differentiation of encapsulated cells. 162 Recently, additives such as nanoparticles and cell-responsive materials (e.g. growth factors, peptides) are loaded in bioinks to improve the function of bioinks and printed structures. The choice of additives depends on the specific requirements of the intended application and the desired properties of the printed structure.87,163 Incorporation of bioinks with additive materials provides improved mechanical stability, printability, and biomimetic environments formation. Nanoparticles loaded bioink’s printed structures had porous structure, and this porous structure can enhance spreading of encapsulated cells with high cell viability, which is an essential factor for angiogenesis.118,164,165 Also, controlled release of encapsulated growth factors from bioinks can contribute to the enhancement of cell adhesion and proliferation without cytotoxicity. 166 Sustained release of VEGF from printed structure result in enhancement of angiogenic effects in in vivo test. 167 These additive materials contribute to the overall success of the bioprinting process by creating structures that closely resemble natural tissues and support the viability and functionality of encapsulated cells. In this part, we will discuss various kinds of additives for bioink formulation and their advantages and limitations. The beneficial and detrimental characteristics of these additives when utilized in 3D bioprinting are outlined in Figure 5.
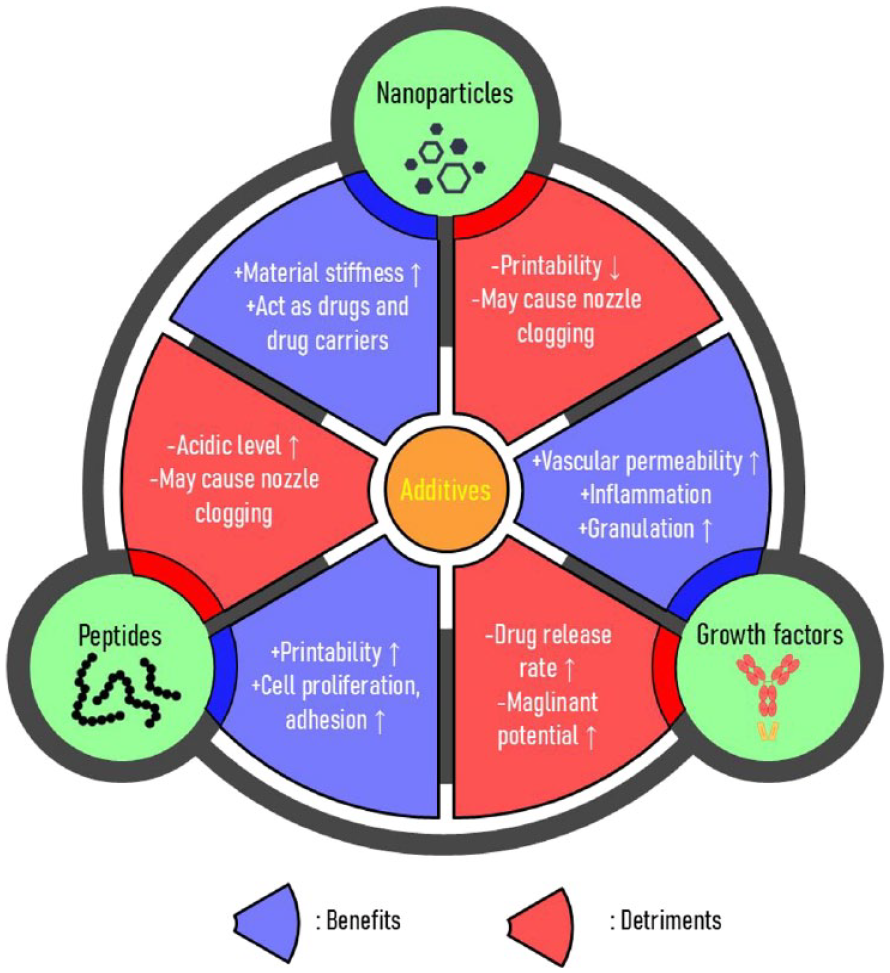
Benefits and detriments of additives in bioinks for 3D bioprinting.
Nanoparticles
In the field of 3D bioprinting bioinks, the incorporation of nanoparticles, such as nanoclay and nanofiber emerges as a novel strategy to fortify mechanical properties. Nanoparticles play a crucial role in enhancing the capabilities of bioinks for 3D bioprinting, such as mechanical strength, printability, and functionality of the printed structures. For example, laponite nanoclays reinforced the stiffness, printability, and shear thinning properties of bioinks, ensuring the stability of 3D-printed structures.168,169 Besides nanoclays did not affect the viability of encapsulated cells within the bioinks and biocompatibility of printed structure.168 –171 Also, surface-modified bioactive nanoparticles preserved good biocompatibility, cell viability of bioink, and stemness of encapsulated cells. 172 Beyond mechanical support, nanoparticles can be used to facilitate drug delivery systems (DDS), acting as carriers for cell-responsive materials such as growth factors, thereby controlling release of growth factors within the printed structures.168,170,171,173 By encapsulating growth factors within nanoparticle before printing, it’s possible to shield growth factors from the mechanical stress encountered during the 3D printing process. Moreover, embedding the growth factors loaded nanoparticles within the bioink provided precise placement of nanoparticles and growth factors after printing. 174 Furthermore, loaded silver nanoparticle can impart bioink with antibacterial properties without cytotoxicity and UV light tolerance.175,176 The addition of nanofibers can significantly improve the mechanical strength and integrity of the printed structures without affecting the viability of encapsulated cells.86,164 Furthermore, encapsulating nanofibers in bioinks can enhance cell growth and proliferation, mimicking the collagen fibrils of natural tissues. Poly(lactic-co-glycolic acid) nanofibers in bioink can provide attachable area for cell and this results in improved cell viability and proliferation. 177 Also, silk fibroin (SF) and 2,2,6,6-tetramethylpiperidine-1-oxyl (TEMPO)-oxidized bacterial cellulose (OBC) nanofibrils loaded bioink showed increased printability and creation of biomimetic environment result in enhanced cell proliferation. 178 Artificial ECM by formulating nanofibers can promote replicating the fibrous structures of the ECM in tissues. Fibrous structures simulated by adding nanofibers to bioinks can provide more biomimetic environments for cells, influencing their behaviors such as cellular attachment and proliferation, thereby promoting tissue regeneration.177,178 These advantages are important for application of cell laden bioink based tissue engineering. Furthermore, the addition of nanofibers within bioink can induce porosity changes of printed structures result in controlled release of encapsulated bioactive molecules.179,180 Besides as the amount of nanofibers in bioink increased, the swelling capacity and disintegration rate of printed structures decreased because of the reduction in porosity. 179 By adjustable properties of nanofibers loaded bioinks, it will be possible to control drug release kinetics to customize drug delivery systems in patient specific way. The addition of nanoparticles to bioink has various advantages, but high concentration of nanoparticles in 3D bioprinting bioink can cause a decrease in printability and cell viability and increase occurrence of nozzle clogging during 3D printing process. So, before using bioinks supplemented with nanoparticles, an optimization process is needed to find the optimal nanoparticle concentration. 172
Cell-responsive materials
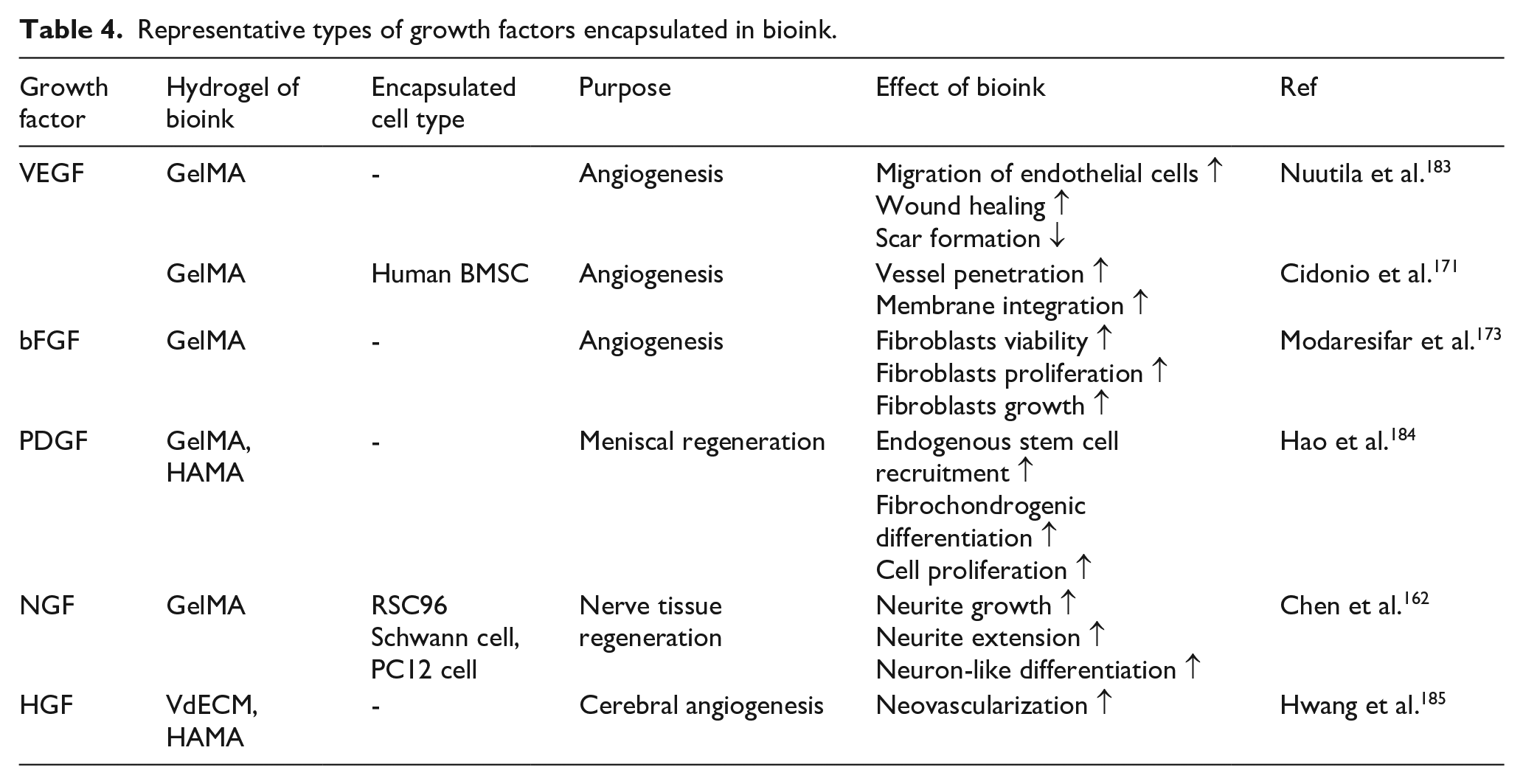
The cell-responsive materials such as peptides and growth factors interact with cells and influence cellular behaviors within printed structures. The inclusion of cell-responsive materials in bioink facilitates improved cell signaling and interaction, contributing to enhanced tissue formation and regeneration.109,171,181,182 Moreover, growth factors can be encapsulated in nanoparticles to regulate release and increase retention capacity. 171 Encapsulated growth factors within nanoparticles in bioink is possible to preserve growth factors from the mechanical stress during the 3D printing process. Also, growth factors loaded nanoparticles can provide precise placement of nanoparticles and growth factors result in enhanced cell proliferation and differentiation. 174 Table 4 encapsulates the growth factors and their effectiveness when incorporated into the bioink.
Representative types of growth factors encapsulated in bioink.
Conclusion
In the field of tissue engineering, scaffold-free 3D bioprinting, supported by advanced bioinks incorporated with cells, has emerged as an alternative to traditional scaffold-based methods through fine mechanical stability and flexibility with improved cell viability and cell functionalities. A critical factor that should be considered in this innovative approach is the bioink, which serves as a versatile carrier for cells, providing essential structural support while ensuring biocompatibility, biodegradability, and mechanical stability during the printing process. Unlike scaffold-based methods, scaffold-free methods reduce concerns about biocompatibility and toxic byproducts, offering a promising avenue for tissue regeneration. Bioink formulations, comprising cells, hydrogels, and supportive additives, drive tissue regeneration by mimicking the ECM of natural tissues. Hydrogels, made from polymers like GelMA, PEGDA, ColMA, HAMA, and Alg-MA, provide a suitable microenvironment for cellular growth, providing biocompatibility and mechanical stability. The inclusion of supportive additives such as growth factors and crosslinking agents further enhances bioink functionality, regulating cell behavior and ensuring tissue-specific ECM production and mechanical stability of printed structures. However, clinical applications with 3D bioprinting still have several limitations, such as biocompatibility, toxic byproducts, and insufficient physical stability. As researchers explore innovative methods to enhance tissue regeneration efficiency and mechanical stability in scaffold-free 3D bioprinting, continual refinement of bioink compositions and printing techniques holds the promise of advancing tissue engineering and leading to a new era of regenerative medicine.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Research Foundation of Korea (NRF) grant funded by the Korean government (the Ministry of Science and ICT) (No. NRF-2022R1A2B5B02001237, 2021K1A3A1A74099704, and RS-2023-00213691 Republic of Korea), the Ministry of Trade, Industry & Energy (MOTIE, Korea) the Korea Evaluation Institute of Industrial Technology (KEIT 20023353, NTIS 1415187291), the Ministry of Trade, Industry & Energy, Republic of Korea, the KIST Institutional Program (Project No. 2E32351, 2E32350 “Development of cellular bloc platform technology,” Republic of Korea).
