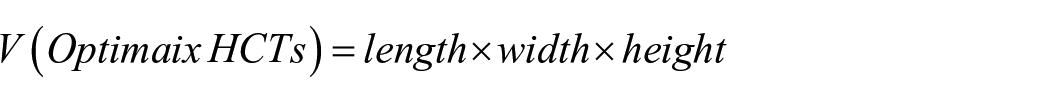Abstract
Tissue engineering approaches hold great promise in the field of regenerative medicine, especially in the context of pediatric applications, where ideal grafts need to restore the function of the targeted tissue and consider growth. In the present study, we aimed to develop a protocol to engineer autologous phalangeal grafts of relevant size for children suffering from symbrachydactyly. This condition results in hands with short fingers and missing bones. A previously-described, developmentally-inspired strategy based on endochondral ossification (ECO)—the main pathway leading to bone and bone marrow development—and adipose derived-stromal cells (ASCs) as the source of chondroprogenitor was used. First, we demonstrated that pediatric ASCs associated with collagen sponges can generate hypertrophic cartilage tissues (HCTs) in vitro that remodel into bone tissue in vivo via ECO. Second, we developed and optimized an in vitro protocol to generate HCTs in the shape of small phalangeal bones (108–390 mm3) using freshly isolated adult cells from the stromal vascular fraction (SVF) of adipose tissue, associated with two commercially available large collagen scaffolds (Zimmer Plug® and Optimaix 3D®). We showed that after 12 weeks of in vivo implantation in an immunocompromised mouse model such upscaled grafts remodeled into bone organs (including bone marrow tissues) retaining the defined shape and size. Finally, we replicated similar outcome (albeit with a slight reduction in cartilage and bone formation) by using minimally expanded pediatric ASCs (3 × 106 cells per grafts) in the same in vitro and in vivo settings, thereby validating the compatibility of our pediatric phalanx engineering strategy with a clinically relevant scenario. Taken together, these results represent a proof of concept of an autologous approach to generate osteogenic phalangeal grafts of pertinent clinical size, using ASCs in children born with symbrachydactyly, despite a limited amount of tissue available from pediatric patients.
Keywords
Introduction
Symbrachydactyly is a rare, congenital, and typically unilateral limb abnormality, presenting brachydactyly, cutaneous syndactyly and global hypoplasia of the hand which occurs in 1 in 20–30,000 live births. 1 Patients suffer from a variety of phenotypes, ranging from having only short and sometimes webbed fingers (syndactyly) to no hand at all. In some cases, patients have an underdeveloped skeleton, but sufficiently long skin pockets to accommodate stable bone (for review).1–4 Traditionally, the hands of these children are improved with surgery to lengthen fingers or create a grip. However, each patient is treated according to its functional and aesthetic needs and resources of the healthcare system to create a grip or to improve hand function. 5 Vascularized free toe transfers and free, non-vascularized phalangeal transfers as well as bone lengthening or skin deepening (web release) procedures have been used for this purpose. Each procedure has benefits and downsides. In particular, the autologous free non-vascularized phalangeal transfer is discussed intensively.6–8 On one hand, it helps to fill empty skin pockets and therefore lengthen the digit whereby it reasonably improves function in pinch and gross grip. On the other hand, it leads to an inacceptable donor site morbidity, especially if multiple toes are transferred, that worsens on the long term follow-up with the growth of the child. 6
Developmental engineering uses a cell’s potential to recapitulate certain steps of a naturally occurring process involved in development and regeneration. 9 Hence, we are proposing a developmentally inspired tissue engineering (TE) strategy that recapitulates endochondral ossification (ECO), the main pathway leading to bone and bone marrow (BM) development,10,11 to generate osteogenic grafts suitable for the treatment of 1- to 5-year-old children suffering from symbrachydactyly (i.e., 75–500 mm3—cylindrical grafts 4–8 mm diameter and 6–10 mm length). During ECO, mesenchymal stromal cells (MSCs) first condense, differentiate into chondrocytes and then progressively acquire a hypertrophic phenotype which is associated with the production of catabolic cytokines and angiogenic factors. As a result, the cartilage structure is invaded by vascular, osteogenic, and hematopoietic progenitors and is eventually remodeled into a bone organ with the presence of bone marrow elements.12,13
Several groups have successfully recapitulated ECO processes with various cell types including, (i) embryonic stem cells (ESCs), 14 (ii) ESCs-derived MSCs, 15 (iii) BM-MSCs,16,17 (iv) periosteum-derived cells (PDCs),18,19 (v) human adipose derived stromal cells (ASCs),20–24 (vi) chondrocytes25,26 or cell-free hypertrophic cartilage extracellular matrix (ECM).27,28 However, so far, the size of the resulting bone organs is often too small (i.e., a few mm3) for clinical translation. In 2015, Sheehy et al. 29 used a BM-MSC-laden alginate hydrogel to generate hypertrophic cartilage tissues (HCTs) anatomically shaped in distal phalanges. In this study, the shape and volume of the graft were clinically relevant (around 350 mm3), but the resulting ECO was not optimal, with only the outer part of the implanted grafts able to remodel into bone tissue after subcutaneous implantation in immunodeficient mice.
Despite the great promise that TE holds for pediatric patients (e.g., the generation of grafts that would not only restore the function of the targeted tissue but also grow in concert with the child’s development 30 ) reports in the literature using pediatric progenitor cells are scarce compared to the plethora of studies on adult stem/progenitor cells. One explanation for this lack of studies is the limited amount of tissue that can be used as cell source. 31 Some reports in the literature suggest that pediatric BM-MSCs could be good candidates for TE applications because they possess a faster proliferating rate, significant morphology differences, 32 lower senescence 33 and retain a better osteogenic potential than adult ones. 34 However, the BM-MSCs invasive harvesting procedure and limited abundancy 35 are serious limitations for their use in pediatric TE approaches.
In a possible clinical scenario, syndactyly of the patients is released first and a full thickness skin graft from the groin region is then used to cover skin defects. At that time, the underlying adipose tissue (2–5 mL depending on the age of the patient) is resected from the skin graft without additional morbidity. Therefore, it represents an attractive source of mesenchymal stromal progenitors contained within the stromal vascular fraction (SVF) of the adipose tissue.36,37 The potential use of SVF cells and their expanded progeny (ASCs) for bone regeneration following an ECO pathway has been demonstrated by our group both in ectopic20,21,23,24 and orthotopic 38 settings but so far only with material obtained from adult individuals.
In the present study, we thus wanted to generate the proof-of-concept of an autologous-based approach to generate osteogenic phalangeal grafts for the treatment of children suffering from symbrachydactyly following an ECO pathway. First, we investigated the ECO capabilities of pediatric ASC in a well-established model.20,24 Second, we developed a protocol using adult freshly isolated SVF cells to generate HCT grafts of clinical relevance (i.e., resembling the size and shape of a small phalangeal bone). Third, we validated our strategy using minimally expanded ASCs obtained from a 20-month-old donor.
Materials and methods
In vitro cell isolation and expansion
Adipose tissue (2–7.5mL) from four pediatric donors were obtained (15–29-month-old). Pediatric SVF cells were recovered following a protocol previously described. 39 After isolation, pediatric SVF cells were immediately seeded at a density of 10,000 cells/cm2 in complete medium (CM), consisting of α-MEM (Gibco, 22571-038) supplemented with 10% fetal bovine serum (FBS, Gibco, 10270-106), 1% HEPES (Gibco, 15630-056), 1% sodium pyruvate (Gibco, 11360-039), 1% penicillin–streptomycin–glutamine (Gibco, 10378-016), as well as 5 ng/ml FGF2 (Bio-Techne, 233-FB-025) and further cultured at 37°C, 5% CO2 and 95% humidity with media change twice a week. Upon reaching confluency (90%), pediatric cells were detached with 0.05% trypsin/0.01% EDTA (Gibco, 25300-054) and seeded again at 3000 cells/cm2 for one additional cell passage. The resulting population was termed pediatric ASC P1. Adult SVF cells—obtained from 2 donors (37- and 77-years old)—used in the present study were isolated following the same protocol but not expanded in vitro.
In vitro generation of HCTs
Small HCTs were generated from pediatric ASC P1 (0.5 × 106 cells) associated with cylindrical (3 mm and a diameter of 4 mm, total volume 37.7 mm3) collagen sponge (Avitene™ Ultrafoam™ Collagen Sponge from Becton Dickinson, 1050050) as previously described 24 and serially exposed to chondrogenic factors for 3 weeks followed by hypertrophic factors for 1 week. For Zimmer phalangeal HCTs fabrication, 3 × 106 (2000 cells/mm3) or 9 × 106 (6000 cells/mm3) of either SVF adult cells or pediatric ASC P1 were seeded onto Zimmer plug® on a non-crosslinked collagen type I of porcine origin (Zimmer, 0102Z collagen plug) shaped in a cylindrical plug of 2 cm height and 1 cm diameter (total volume 1570 mm3). For Optimaix phalangeal HCTs fabrication, 0.3 × 106 (2000 cells/mm3) or 0.9 × 106 (6000 cells/mm3) of SVF adult cells were seeded onto a crosslinked collagen type I matrix of porcine origin with highly oriented fibers (Optimaix 3D®, Matricel GmbH), cut in rectangular bars 10 × 5 × 3 mm (volume 150 mm3). Pediatric ASC P1 were not used with the Optimaix 3D scaffolding material. Next, cell-laden sponges were serially exposed to chondrogenic factors for 3-,4- or 6-weeks followed by hypertrophic factors for 1 week. Briefly, the chondrogenic induction medium was composed of serum-free culture medium composed of Gibco Dulbecco’s Modified Eagle Medium (DMEM, Gibco, 10938-025), 1% HEPES (Gibco, 15630-056), 1% sodium pyruvate (Gibco, 11360-039), 1% penicillin–streptomycin–glutamine (Gibco, 10378-016), ITS+1 (Sigma, I2521), and 1.25 mg/ml human serum albumin (CSL Behring, 43075). The serum-free culture medium was further supplemented with 10−7 M dexamethasone (Sigma, D-2915), 0.1 mM ascorbic acid (Sigma, A-8960), 10 ng/ml transforming growth factor-β3 (TGF-β3, PeproTech, 100-36E), and 10 ng/ml bone morphogenetic protein 6 (BMP-6, PeproTech, 120-06). The hypertrophic induction medium was composed of serum-free culture medium supplemented with 0.1 mM ascorbic acid (Sigma, A-8960), 10 mM β-glycerophosphate (Sigma, G9422), 10−8 M dexamethasone, 50 mM L-thyroxin (Sigma, T-1775), and 50 pg/ml interleukin1-β (Sigma, SRP3083).
In vitro volumetric quantification
Volumetric quantification of the in vitro generated phalangeal HCTs was performed using the open-source software FIJI version 2.1.0. on macroscopic cell culture images. Briefly, for the Optimaix-based HCTs, the length, width, and height (mm) were measured and the volume (mm3) was calculated using the formula:
For the Zimmer-based HCTs, the length and diameter were measured (mm) and the volume (mm3) was calculated using the formula:
The Zimmer-based HCTs that were bend out of cylindrical shape were removed from the volumetric analyses.
In vivo ectopic implantation in nude mice
At the end of the cartilage maturation in vitro, HCTs grafts (maximum 4 per mouse) were implanted subcutaneously in athymic CD1 nu/nu female nude mice (Charles River Laboratories, Wilmington, MA). Mice were operated under the permission of the Federal Veterinary Office (permit BS 1797) as previously described. 20 In total, 12 mice were implanted with 4 pediatric donors and 2 adult donors as described in Table 1.
Summary of implanted HCTs.
Microcomputed tomography
After explantation, samples were retrieved and fixed overnight in 4% formalin (Formafix). Then, microcomputed tomography (microCT) data were acquired by using a high-resolution scanner (SkyScan1172, Skyscan, Belgium) and 0.5-mm aluminum filtered X- rays (applied voltage 50 kV; current, 200 uA). Transmission images were acquired during a 360° scan rotation with an incremental rotation step size of 0.25°. Reconstruction was performed using a modified Feldkamp algorithm at an isotropic voxel size of 4 μm for the ultrafoam®-based HCTs and 10 μm for the Zimmer- and Optimaix-based HCTs. Three-dimensional rendering, thresholding, segmentation, and 3D measurements were performed using VG Studio MAX 2.2 software (Volume Graphics, Heidelberg, Germany).
Histological assessments
For histological analyses, fixed samples were decalcified by 10% EDTA solution (MoL-Decalcifier, Milestone srl) if necessary, and embedded in paraffin. The scaffolds were cut into 5 µm thick sections using a Microtome HM 355S (Thermo Scientific) and the sections were placed onto Poly-lysine slides (Thermo Scientific, Waltham, US). The tissue sections were deparaffinized and rehydrated then stained with Haematoxylin-Eosin (both from Sigma Aldrich) and Safranin-O (Sigma-Aldrich, 84120) using the Epredia™ Gemini™ AS Automated Slide Stainer. Collagen type II (Reactivity: human, MP Biomedicals, 63171) and collagen type X (Reactivity: human; Cat. no. 14-9771-80, Invitrogen), and Bone sialoprotein (BSP, Reactivity: human; Cat. no. 52128, Abcam) stainings were performed with Ventana Discovery Ultra (RocheDiagnostics (Switzerland) SA) automated slide stainer. In brief, tissue sections were deparaffinized and rehydrated. Antigens were retrieved by a protease (Protease 3, ref. 760-2020, Ventana) digestion for 20–44 min at 37 °C. Primary antibody was manually applied and incubated for 1 h at 37°C. After washing, the secondary antibody was incubated for 1 h at 37°C. Detection step was performed with the Ventana DISCOVERY ChromoMap DAB (ref. 760-159 Ventana) detection kit. Afterward, the slides were counterstained with hematoxylin II, followed by the bluing reagent (respectively, Cat. no. 790-2208 and 760-2037, Ventana). Sections were then dehydrated, cleared, and mounted with permanent mounting and coverslips. Images of the histological sections were acquired with a Nikon Ti2 widefield microscope, a Nikon DS-Ri2 camera and a CFI Plan Apo Lambda NA 0.75, 20× objective. The software used was the NIS-Elements AR 5.21.03.
Human nuclei immunofluorescence staining
Decalcified samples were cryoprotected with sequential incubations in 15% and 30% sucrose/PBS solutions at 4℃ overnight. Samples were embedded in OCT compound, cryosectioned into 10 µm sections, and stored at −80℃ until use. Sections were rehydrated with PBS for 10 min at room temperature, permeabilized with 0.3% Triton X-100/PBS, and then subjected to 1% BSA and 5% goat serum to block unspecific binding. Then, sections were incubated with a 1:100 dilution of anti-human nuclei antibody (Millipore, MAB1281) overnight at 4°C and further incubated with a 1:500 dilution of Alexa Fluor 546 goat anti-mouse IgG (H + L) secondary antibody (Invitrogen A11030) and 1:1000 DAPI (4′,6-diamidino-2-phenylindole, Invitrogen D1306) to stain nuclei prior to imaging.
Automated in vitro cartilage maturation score
To grade the maturity of in vitro engineered cartilage, the Automated Modified Bern Score was used to score histological images. 40 A python script to grade whole slide images was used along with the trained model for visualization of the scores within a construct (https://github.com/Lopo358/AMBS_whole_slide_image).
QuPath analyses
Tissue classification into four classes, namely: bone, bone marrow, cartilage and fibrotic tissue, was performed as previously described 24 using the open-source software QuPath v0.3.2. 41 In short, a training image was composed of multiple regions of interest from representative scans to account for tissue variability, and annotations for each class were manually drawn. First, tissue was classified into two regions: Cartilage/Bone area and Bone marrow/Fibrotic tissue area. Each of these regions were further classified into single region annotations, using parameters: Pixel_classifier_type: “OpenCVPixelClassifier,” Resolution: moderate: (2.93 μm/px), Channels selected (Red, Green, Blue); Scale: 1.0. Features used: “gaussian,” “weighted_std_dev,” “gradient magnitude,” “Laplacian,” “structure_tensor_eigenvalue_max. Results were inspected by an expert and manually corrected if needed. Corresponding training images and classifiers can be found on Zenodo (https://zenodo.org/records/10680128).
Results
Pediatric ASCs are suited for bone tissue engineering via the ECO pathway
We first addressed whether ASCs obtained from pediatric adipose samples (12–29-month-old) could recapitulate ECO in a similar fashion to what has been demonstrated for adult ASCs.20,24 To that aim, Pediatric ASCs were associated with collagen sponges (UltraFoam® with a cylindrical shape; h = 3 mm d = 4 mm V = 37.7 mm3) and cultured for 4W (serially exposed to chondrogenic factors for 3 weeks followed by exposure to hypertrophic factors for 1 week). The bone forming capacity of these in vitro generated HCTs was assessed in vivo in an ectopic immunodeficient mouse model for 12 weeks. Figure 1(A) offers a summary of the experimental set-up. Cells from each tested donor (N = 4) were capable of forming cartilage tissues in vitro as evidenced by round chondrocytes within lacunae embedded in ECM positive for glycosaminoglycans (GAG, in red), collagen II and collagen X, a marker of hypertrophy (Figure 1(B)). The periphery of each HCTs displayed mineralization (in blue) while their core was mostly comprised of non-chondrogenic cells and collagen sponge remanence (Figure 1(Ba–d)).
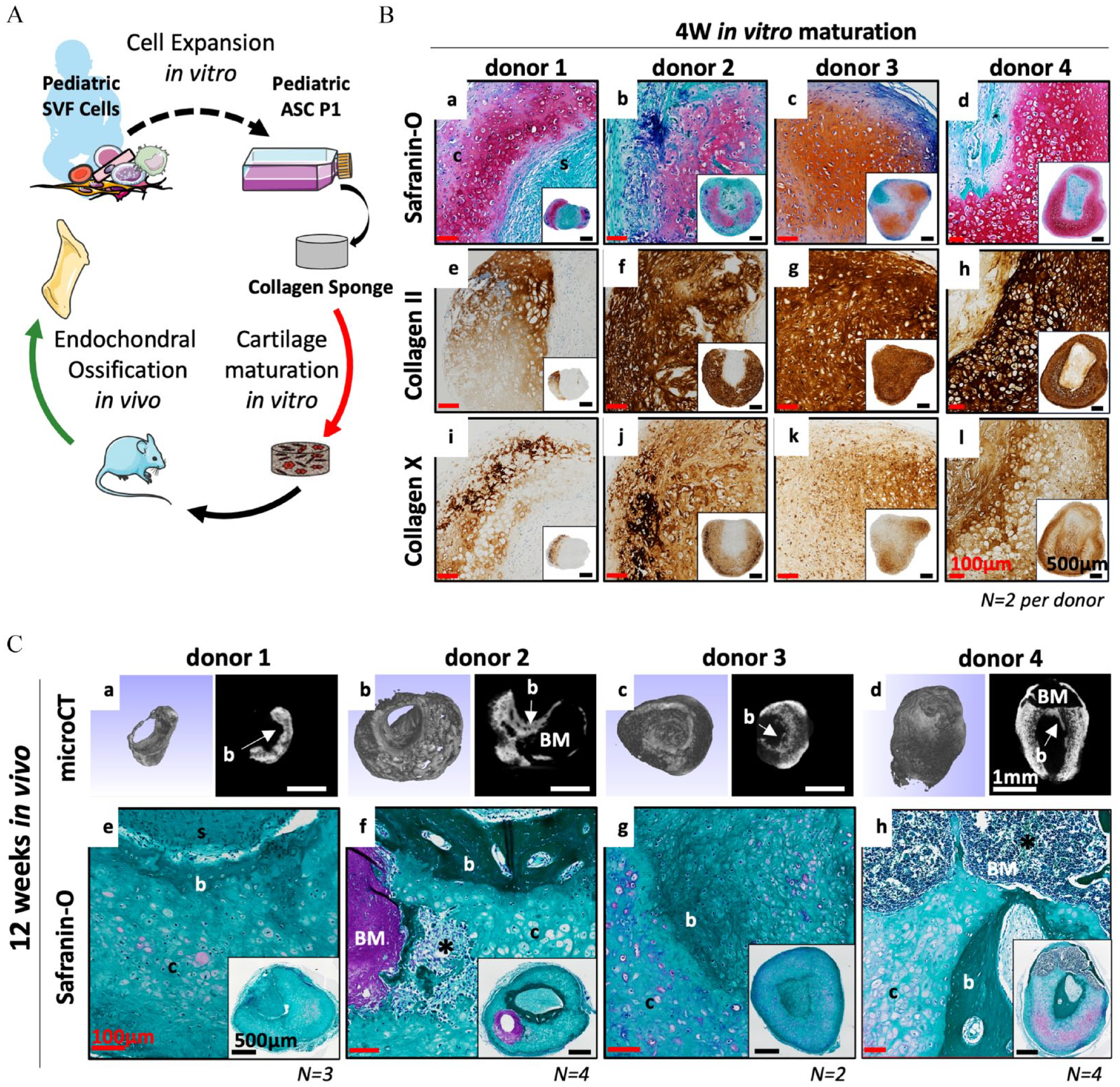
Pediatric ASCs recapitulate endochondral ossification in vivo when adequately primed in vitro. (A) Schematic overview of the experimental design. (B) 0.5 × 106 Pediatric ASC P1 cells are associated with a collagen scaffold (cylindrical shape, 4 mm diameter and 3 mm height; V = 37.68 mm3) and cultured in vitro for 4W (3 weeks chondrogenic + 1 week hypertrophy). Representative images of (a–d) Safranin-O staining, cartilage tissue in red, mineralized cartilage in blue and non-cartilage tissue in green, (e–h) collagen type II and (i–l) collagen type X of the in vitro generated cartilage tissues. (C) In vivo bone remodeling via endochondral ossification after 12 weeks of ectopic implantation in a nude mice model. Representative images of (a–d) Microcomputed tomography (µCT), Bone (dense signal with smooth surface) mineralized HCT (signal with a rough surface). (e–h) Safranin-O staining, bone (dark green), fibrotic tissue (light green), cartilage (red), bone marrow (purple). (N = 13, 2–4 biological replicates per donor, 4 donors tested). Black scale bar = 500 µm, white scale bar = 1 mm, red scale bar 100 µm. Symbols: b: Bone, BM: Bone Marrow, c: cartilage, s: scaffold, *: blood vessel.
After implantation, both the micro-computed tomography (µCT) and the histological analyses demonstrated successful and reproducible bone formation in vivo. For every donor tested, newly formed bone tissue was located at the core of the implanted constructs and characterized by a smooth surface on the µCT images (Figure 1(Ca-d)) and by a darker green color on the safranin-O images. The persistence of cartilage tissues was observed and characterized by chondrocytes embedded in a faintly GAG positive ECM (in red) (Figure 1(Ce–h)). In contrast, the ECO was most advanced for donor-2 and -4, where a larger amount of bone as well as the presence of bone marrow compartment within the bone ossicles formed were observed (Figure 1(Cb,f and 1Cd,h)). These results are in line with our previous report using a similar experimental set-up with adult cells 24 and demonstrate that ASCs derived from pediatric adipose tissues can similarly generate hypertrophic cartilage in vitro that remodel into bone tissue via ECO in vivo.
Adult SVF cells in larger collagen sponges generate phalangeal HCT grafts suited for the treatment of children suffering from symbrachydactyly
To generate phalangeal HCT grafts, we tested the influence of (i) the initial size and composition of the scaffolding material, (ii) the cell seeding density and (iii) the in vitro culture duration. Specifically, we tested the commercially available Zimmer Plug® (a non-crosslinked collagen type I of porcine origin similar to the UltraFoam® shaped in a plug—h = 20 mm d = 10 mm V = 1500 mm3) and Optimaix 3D® (a crosslinked collagen type 1 matrix of porcine origin with highly oriented fibers shaped in form of a bar—l = 10 mm w = 5 mm h = 3 mm V = 150 mm3) associated with either 2000 or 6000 SVF cells/mm3 and cultured for 4W, 5W, or 7W in vitro (chondrogenic factors for 3–6 weeks followed by hypertrophic factors for 1 week). For the two adult donors tested, both scaffolding materials were capable of supporting a gradual cartilage formation in vitro (Figure 2). Interestingly, with the higher cell density, the strongest cartilage formation (i.e., more area positive for GAG, collagen II, and collagen X) occurred in the Optimaix-based grafts, whereas cartilage formation was impaired in Zimmer-based grafts (Figure S1). As expected from his crosslinked nature, the Optimaix-based grafts were able to retain their shape and size throughout the in vitro maturation (Figure 2(A)). In addition, histological analyses revealed that the cartilage formation was located exclusively at the periphery while the core of these grafts was comprised of non-chondrogenic cells attached to the scaffolding material (Figure 2(B)).

Adult SVF cells associated large collagen sponge scaffolds generate phalangeal HCT grafts in vitro. (A) Schematic overview of the experimental design and macroscopic views of the phalangeal HCT grafts (a) (i) before and (ii) after cell seeding and (b) throughout the in vitro cartilage maturation. Histological assessments of (B) 0.9 × 106 SVF cells (6000 cell/mm3) associated with Optimaix® collagen scaffold (bar shape, 10 mm length 5 mm width and 3 mm height; V = 150 mm3) or (C) 3 × 106 SVF cells (2000 cell/mm3) associated with Zimmer Plug® collagen scaffold (cylindrical shape, 10 mm diameter and 20 mm length; V = 1500 mm3) and cultured in vitro for (a–c) 4W (3 weeks chondrogenic + 1 week hypertrophy), (d–f) 5 W (4 weeks chondrogenic + 1 week hypertrophy) or (g–i) 7W (6 weeks chondrogenic + 1 week hypertrophy). Representative images of (a,d,g) Safranin-O staining, (b,e,h) collagen type II and (c,f,h) collagen type X obtained from the in vitro generated HCT grafts. (N ⩾ 1, 1–2 biological replicates per donor, 1–2 donors tested). Red scale bar = 10 mm, black scale bar = 1 mm, yellow scale bar = 150 µm.
In contrast, the Zimmer-based grafts had a behavior more similar to the ones based on smaller UltraFoam-based constructs used in previous studies,20,24 contracting rapidly during the first 4 weeks of in vitro maturation, with initial formation of cartilage at the periphery of the grafts (Figure 2(Ca–c)). At later time-points, the cartilage pockets were larger and also present in the inner parts of the constructs. However, even after 7 weeks of in vitro maturation, non-cartilaginous areas remained (Figure 2(Cg–i)).
When quantifying the contraction of each graft throughout the in vitro phase, a 70% shrinkage of Zimmer-based grafts was observed during the first weeks of culture followed by a plateau concomitant with the appearance of cartilage tissues which occurred around week-4 or -5, depending on the donor (Figure 3A). The Optimaix-based grafts, due to their highly oriented fibers, showed a much more limited contraction (10%) during the in vitro phase. After 7 weeks of maturation, the final volume ranged from 59 to 136 mm3 for Optimaix-based grafts, and 108 to 390 mm3 for the Zimmer-based ones (Figure 3A). To quantify the cartilage maturation in the phalangeal grafts, we used a modified version of an automated deep-learning method previously developed in our laboratory. 40 In a previous report, this maturation index was shown to be predictive of the bone remodeling via ECO, with an optimal result for scores ranging from 1 to 3 (out of a maximal score of 6). 24 While the overall maturation index was similar for both materials, with a gradual score increase reaching 1.7 after 7 weeks, their cartilaginous maturation profiles were different. The Optimaix-based grafts displayed a more heterogenous profile with a marked difference between non cartilaginous areas (62 ± 19.8% 0-scored tiles) and strongly differentiated regions (19.5 ± 4.9% 6-scored tiles). Whereas, the Zimmer-based grafts were more homogeneous and contained fewer non cartilaginous areas (35.5 ± 14.8% of 0-scored tiles) (Figure 3B).

In vitro evolution of the size and cartilage maturation of phalangeal HCTs grafts generated from adult SVF cells. (A) Time course of the estimated volumes of the phalangeal HCT grafts for both the Zimmer Plug® and Optimaix® groups. Data are expressed as mean ± SD (mm3). (N = 6, 6 biological replicates per donor, 2 donors tested per scaffolding material). (B) Automated histological assessment of the in vitro cartilage maturation for the 4W-, 5W- and 7W-groups. Safranin-O-stained pictures are cut into square tiles (336 pixels by 336 pixels) and scored 0-non cartilaginous tissue; (2) poor cartilage maturation; (4) moderate cartilage maturation; (6) high cartilage maturation. Distribution of the number of tiles for each cartilage maturation category expressed as a percentage of the total number of tiles and plotted as a histogram. (N ⩾ 1, 1–2 biological replicates per donor, 1–2 donors tested).
Next, we tested the bone forming capacity of these phalangeal HCT grafts in an ectopic, immune-compromised mice model mimicking the clinical scenario of an empty phalangeal pocket. Briefly, Zimmer- and Optimaix-based grafts matured for either 5W or 7W were implanted subcutaneously for 12 weeks in nude mice. Samples were then collected and the bone formation was analyzed by µCT and histological analyses. All constructs tested were successful in achieving bone formation via ECO. The in vivo results obtained for 7W-Optimaix- and -Zimmer-based grafts are presented in Figure 4, whereas the ones of 5W-Optimaix- and -Zimmer-based grafts are presented in Supplementary Figure 2. At time of explantation, we could macroscopically observe vascularization of the implanted cartilage constructs both for the 7W-Optimaix and -Zimmer groups (Figure 4(A)). In addition, µCT analyses showed strong mineralization and the presence of bone marrow pockets for both groups (Figure 4(Aa and Ba)). Endochondral ossification of the implanted cartilage was highlighted by the presence of interlaced bone and cartilage tissue (Figure 4(Ab–c and Bb–c)). For the 7W-Optimaix group, the bone formation was located solely at the periphery of the construct (where cartilage was initially present at the time of implantation) and contained bone marrow pockets (in 2 out of 8 constructs) while the vast majority (67%) of the construct was still comprised of non-remodeled Optimaix sponge (Figure 4(Ab–d)). In contrast, the 7W-Zimmer group showed better remodeling in vivo, with most of the construct comprised of either bone, bone marrow (in 3 out of 8 constructs) or cartilage tissues (Figure 4(Bb–d)). In both cases, the cartilage remanence was characterized by a faintly positive safranin-O staining still present after the 12 weeks of implantation (Figure 4(Ab and Bb)). Similar results (i.e., presence of bone and cartilage tissues after 12 weeks of implantation) were obtained for the 5W-groups, however we could not detect bone marrow tissues in any of them (Figure S2).

Phalangeal HCT grafts remodel into bone organs via endochondral ossification in vivo. Bone remodeling phenotypes observed for (A) 7W-Optimaix- and (B) 7W-Zimmer-based grafts after 12 weeks of implantation in vivo. (a) Macroscopic and Micro-µCT views. Representative images of (b) Safranin-O and (c) H&E sections. (d) Automated tissue segmentation performed H&E sections using a semi-automated script on QuPath. (C) Evolution of mineralized tissue (mm3) obtained by µCT after 12 weeks in vivo. Data are expressed as mean ± SD (N ⩾ 4, 4 biological replicates per donor, 1-2 donors tested). Evolution of (D) Bone and Bone Marrow, (E) Cartilage and (F) Other tissues within the H&E sections obtained after 12 weeks in vivo. Data are expressed as a percentage of the total tissue mean ± SD. (N ⩾ 4 biological replicates per donor, 1–2 donors tested). b: Bone, BM: Bone Marrow, c: cartilage, s: scaffold, *: blood vessel. Red scale bar = 10 mm, black scale bar = mm, yellow scale bar = 150 µm. *p < 0.05 Mann Whitney test (Zimmer group vs. Optimaix group); ##p < 0.01 Mann Whitney test (7 weeks vs. 5 weeks).
Volumetric quantification of the mineralized tissues by µCT demonstrated a superior osteogenic potential of the Zimmer-based grafts as compared to the Optimaix ones, specifically, with 26.4 ± 2.4 vs 4.0 ± 1.6 mm3 and 27.2 ± 14.3 vs 13.3 ± 5.6 mm3 of mineralized tissues obtained for the 5W- and 7W-in vitro cartilage maturation, respectively (p < 0.05; Figure 4(C)). On the H&E sections analyzed for tissue segmentation, we found no difference in the amount of bone and bone marrow present in the 5W-grafts, however, there was significantly more bone and bone marrow tissues in the 7W-Zimmer group (4.9 ± 3.2 mm3) compared to the 7W-Optimaix group (2.1 ± 1.2 mm3; Figure 4(D)). Conversely, for these two groups we could observe a significant increase in other tissues (i.e., non-cartilaginous and non-bony) for the 7W-Optimaix group (13.7 ± 3.6 mm3) compared to the 7W-Zimmer group (7.8 ± 5.0 mm3; Figure 4(F)). Finally, while we observed an increase in cartilage content in the 7W-constructs when compared to the 5W-constructs, no significant differences were observed between the Optimaix and Zimmer groups (Figure 4(E)). We thus were able to generate grafts shaped in the form of a phalanx from adult SVF cells with both scaffolding materials. However, after 12 weeks of implantation, an optimal bone formation via ECO characterized by (i) the maintenance of shape and size of the implanted cartilage, (ii) the presence of bone tissue and bone marrow, and (iii) the remodeling of the collagen scaffold, was obtained for the 7W-Zimmer group.
Pediatric ASCs in Zimmer plug sponges generate phalangeal HCT grafts in vitro that remodel into bone tissue via ECO in vivo
Next, we wanted to demonstrate the feasibility of an autologous approach for the construction of phalanx in children suffering from Symbrachydactyly. We tested this clinical approach with a pediatric donor, 2.5 mL of adipose tissue were recovered from a 15-month-old donor and 1.95 × 106 SVF cells were isolated by enzymatic digestion and cultured as monolayer for 2 weeks (3250 cells/cm2). After 2 weeks of expansion, more than 200 × 106 cells (referred to as ASCs P1) were generated and the Zimmer plug collagen sponges were seeded with 3 × 106 ASCs P1 (2000 cells/mm3). Following the same protocol as the one used for adult SVF cells, Zimmer-based cartilage grafts were generated in vitro and their bone forming capacity was evaluated in vivo in an ectopic immunodeficient mouse model. Figure 5(A) offers a summary of the in vitro manufacturing protocol. After 7 weeks of in vitro cartilage maturation, a contraction of the Zimmer collagen sponge was observed (Figure 5(B)) as well as the presence of cartilage tissue, which formed a shell (pink and red areas on the safranin-O staining) located at the periphery of the Zimmer constructs, while the inner part of the constructs was mostly composed of compacted collagen sponge and few cells (Figure 5(Ca–c)). Likewise, the presence of collagen II and collagen X was limited to the periphery of the constructs (Figure 5(D)). The contraction of the construct throughout the in vitro cartilage differentiation was similar to the one observed for the adult-SVF cells (Figure 5(E)). Finally, using the automated in vitro maturation score, we could show a reduced chondrogenic capacity for the pediatric ASCs P1 (maturation score of 1 ± 0.2) when compared to the adult SVF (maturation score of 1.7 ± 0.4; Figure 5(F)).

Envisioned manufacturing strategy for autologous phalangeal HCT grafts production to treat children suffering from Symbrachydactyly. (A) Schematic overview of the manufacturing protocol. Pediatric SVF cells are expanded for 2 weeks (ASC P1). 3 × 106 pediatric ASCs P1 are associated with Zimmer Plug® collagen scaffolds and cartilage maturation is induced for 7 weeks. (B) Macroscopic view of the cartilage constructs after (a) 1 week and (b) 7 weeks of cartilage maturation. (C) Representative images of Safranin-O staining, (D) (a) collagen type II and (b) collagen type X obtained from the in vitro generated cartilage. (E) Time course of the estimated volumes of the HCTs. Data are expressed as mean ± SD (mm3). (N ⩾ 4, 4–6 biological replicates per donor, 1-2 donors tested per cell group). (F) Automated histological assessment of the in vitro maturation of the cartilage tissues after 7 weeks of induction. (N ⩾ 3, 1–3 biological replicates per donor, 1–2 donors tested). Red scale bar = 10 mm, black scale bar = 1 mm, yellow scale bar = 150 µm.
After 12 weeks of implantation, similarly to what was observed for the adult SVF cells-based Zimmer cartilage constructs, bone formation via ECO was obtained for the pediatric Zimmer-grafts (Figure 6). In some cases, we could macroscopically observe a reduction in the size of the implanted constructs (Figure 6(Aa)). For all constructs tested (N = 8), the presence of newly formed bone (characterized by a smooth surface with hollow sinuses) adjacent to the mineralized cartilage tissue was visible on the µCT images (Figure 6(Abc)). On H&E- and Safranin-O-stained sections of the explanted pediatric cartilage constructs, we observed not only the presence of vascularized bone tissue but also the presence of cartilage tissue containing numerous chondrocytes, some likely transitioning towards an osteoblastic lineage (Figure 6(B–D)). In addition, even after 12 weeks of implantation, the cartilage tissues exhibited a relatively high GAG content in the ECM and the presence of hypertrophic chondrocytes evidenced by the Safranin-O and Bone sialoprotein (BSP) staining, respectively (Figure 6(C,D) and Supplementary Figure 3). To investigate the contributions of the implanted cells (human) and the host cells (mouse) on the in vivo bone- and cartilage-formation, a human-nuclei staining on was performed on cryosections of pediatric Zimmer-based constructs (Figure 6(E)). The multi-nucleated osteoclastic cells present in small areas containing blood vessels inside the cartilage matrix were of mouse origin (Figure 6(Eb)), thus evidencing the host-mediated bone remodeling that is taking place (Figure 6(Bb and Be)). In the inner part of the constructs, mostly filled with human cells, a distinctive interphase between newly formed bone and the calcified cartilage matrices was observed with chondrocytes gradually replaced by osteoblasts (Figure 6(Bc and 6Ec)). In addition, a progressive loss of BSP expression was observed in the human implanted chondrocytes from the calcified cartilage towards the newly formed bone (Figure 6(Cb) and Supplementary Figure 3(c and d)). In contrast, for the newly formed cortical bone tissue obtain at the periphery of the constructs, the cells embedded in the bone matrix were of mouse origin, demonstrating that they were the one responsible for the endochondral ossification located on the outer part of the implanted constructs (Figure 6(Bd and Ed)).
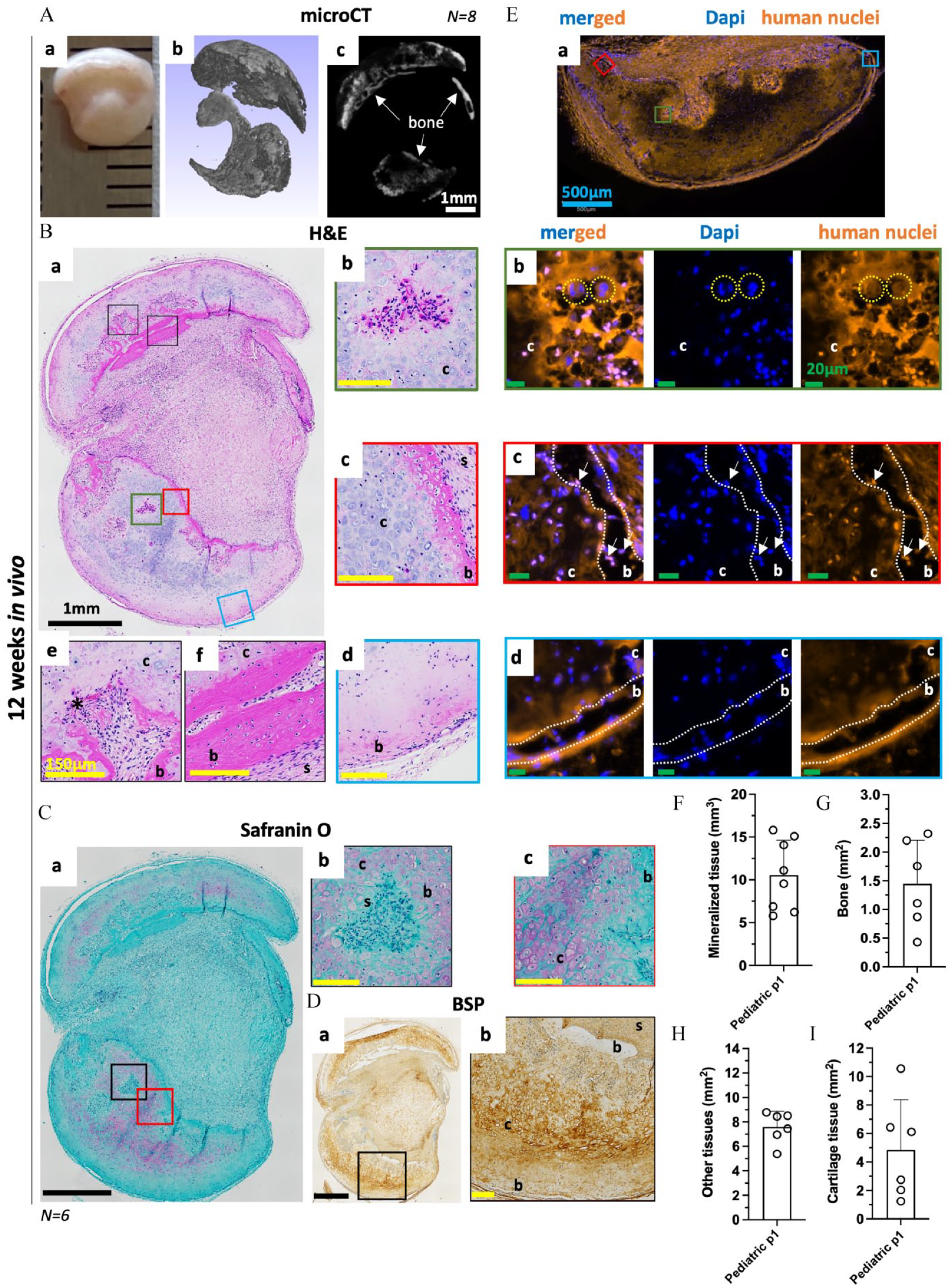
Pediatric phalangeal HCT grafts remodel into bone tissue via endochondral ossification. 7W Pediatric ASCs P1 based Zimmer constructs were implanted in an ectopic nude mice model for 12 weeks. (A) (a) Macroscopic and (b,c) Micro- µCT views post-implantation. Representative images of (B) H&E and (C) Safranin-O staining showing evidence of ECO in the implanted constructs (a) whole section, (b) early cartilage remodeling with the presence of multinucleated cells, (c) Chondrocyte to osteoblast transition, (d) outer cortical bone, (e) intermediate cartilage remodeling with the presence of bone and blood vessels and (f) mature bone located at the core of the constructs. (D) Bone Sialoprotein (BSP) staining (a) whole section, (b) early cartilage remodeling. (E) Mouse and human cells contribution to the ECO. Representative images of human nuclei and Dapi staining of (a) large section, (b) early cartilage remodeling with the presence of murine multinucleated cells, (c) chondrocyte to osteoblast transition with the presence of human chondrocytes and osteoblasts and (d) outer cortical bone with the presence of murine osteoblasts. Mouse nuclei in blue, and human nuclei in pink on the merged pictures. (F) Evolution of mineralized tissue in mm3 for pediatric phalangeal HCT grafts obtained by µCT after 12 weeks in vivo. Data are expressed as mean ± SD (N = 8, 1 donor tested). Evolution of (G) bone and bone marrow, (H) other and (I) cartilage obtained by automated tissue segmentation performed H&E sections using a semi-automated script on QuPath. Data are expressed as a percentage of the total tissue mean ± SD. (N = 6, 1 donor tested). Symbols: b: bone, c: cartilage, s: scaffold, *: blood vessel, yellow circles murine multinucleated cells, white arrows human osteoblasts. White scale bar = 1 mm, black scale bar = 1mm, blue scale bar = 500 µm, yellow scale bar = 150 µm, green scale bar = 20 µm.
In conclusion, we were able to demonstrate the feasibility of a clinical strategy using pediatric adipose derived cells to generate tissue engineered phalanx grafts for children suffering from symbrachydactyly.
Discussion
In this study, we showed for the first time the ECO potential of pediatric ASC associated with small collagen sponge scaffolds and of phalangeal HCT grafts. These were generated from either adult SVF cells or pediatric ASCs associated with large collagen sponges in an ectopic, preclinical immunocompromised mouse model aiming at modeling both an autologous approach—with limited immune response—and the empty phalangeal pocket of children suffering from symbrachydactyly. We observed a small reduction in chondrogenicity for expanded pediatric ASCs as compared to adult SVF cells. However, they both remained capable of generating HCT grafts that successfully remodeled into bone organs, demonstrating the feasibility of an autologous approach to generate phalangeal substitutes.
Remarkably, after implantation, HCTs derived from pediatric donors not only displayed signs of ECO but also maintenance of cartilage tissues characterized by the presence of proliferating chondrocytes and a high content in GAG within the ECM (Figures 1 and 6). The latter features were absent from all the adult SVF-cells implanted HCTs tested. Similarly to what was previously observed for HCT based on BMSCs, 42 the bone located at the core the implant and in direct contact with the cartilage tissue was of human origin (Figure 6), suggesting that some implanted hypertrophic chondrocytes likely could give rise to osteoblasts. Further studies are needed to confirm and understand the underlying mechanisms of this in vivo cartilage homeostasis and hypertrophic chondrocytes to osteoblasts transition observed in pediatric ASCs-derived HCTs.
The manufacturing protocol developed here was adopted to ensure a timely and feasible clinical translation. In particular, it involves (i) limited numbers of cells (3 × 106 per graft), (ii) a simple, straightforward in vitro culture system, and (iii) a manufacturing time of less than 10 weeks from cell isolation to implantation of the graft. We chose ASCs as the source of autologous chondroprogenitor because adipose tissue would be available at the time of the syndactyly release without additional morbidity. While SVF cells and their expanded counterpart (ASCs) are not skeletal cells (SSCs), several studies have demonstrated that these cells can acquire SSCs traits when adequately primed in vitro 43 and form bone organs via ECO when implanted in vivo.20,21,23,24,44 Moreover, a rapid isolation and expansion process makes ASCs a well-suited cell source for pediatric TE application where donor sites are very limited. In this study, for the three pediatric donors, we obtained on average 2.1 ± 1.1 × 106 SVF cells per gram of adipose tissue (adipose tissues ranging from 2 to 6g), consistent with a recent report on pediatric patients with osteogenesis imperfecta. 45 After in vitro expansion, we generated more than 150 × 106 ASCs within 2 weeks which—using the manufacturing protocol developed here—could potentially generate up to 50 phalangeal grafts.
One limitation of this work is the reduced number of pediatric donors tested in the herein study (n = 3 for the ECO potential of pediatric ASC and n = 1 for the phalangeal HCTs grafts). The lack of additional donors is linked to the fact that pediatric biopsies are rare and that we could not obtain additional donors over the course of the study. However, the high reproducibility of ECO using ASCs and SVF-cells associated with collagen sponges demonstrated in a recent report from our group by Chaaban et al. 24 and the number of in vivo replicates used in the present study mitigate the impact of the single pediatric donor used for validation of the manufacturing protocol.
For bone regeneration, TE strategies following an intramembranous ossification (IMO) pathway—usually comprised of a biomaterial mimicking the bone ECM associated with progenitor cells (e.g., BM-MSCs, SVF cells, ASCs)—are the only ones currently evaluated in clinical trials. While promising, this approach has not yielded the expected results. A major limitation for their success remains the lack of vascularization of such large grafts leading to the development of a necrotic core after implantation.46,47 Therefore, targeting bone regeneration via the ECO pathway appears to be a more promising approach to generate up-scaled grafts. In fact, hypertrophic chondrocytes are not only more suited for the post-implantation avascular environments but also actively promoting angiogenesis. In addition, transplanted hypertrophic chondrocytes can be replaced by host osteoblasts when they undergo apoptosis or necrosis, mimicking the developmental process.10,46,47 Nevertheless, despite reports highlighting the reproducibility and efficacy of ECO-based strategies in preclinical animal models (for review47,48), they have not yet reached the stage of clinical trials. This is mostly because the upscaling required to produce grafts of clinically-relevant size remains a major hurdle for the translation of such ECO strategies, where chondroprogenitors first condense and later deposit ECM which gets enriched in GAG over time. 49 Especially during the condensation phase, maintaining a stable, desired shape and size for a TE graft is challenging. Several approaches are being explored to tackle this issue. A modular strategy consisting in aggregating cartilage microtissues to achieve either larger or more complex tissue architecture, requiring a large number of cells. For example, Hall et al. 50 report the fabrication of osteochondral grafts by combining iPSC-derived cartilage microtissues in combination with pre-hypertrophic cartilage organoids derived from PDCs. Whereas, Burdis et al. 51 describe the biofabrication of osteochondral constructs obtained by spatially localizing phenotypically distinct cartilage microtissues within an instructive 3D printed polymer framework. In this case the authors estimated that a human clinical situation would require approximately 330 × 106 BMSCs, 112 × 106 Fat-pad stromal cells and 37 × 106 chondrocytes.
In this study, we chose to modulate the size and architecture of collagen type I sponges in order to achieve HCT-grafts resembling small phalangeal bones (in size and shape). First, we selected the Zimmer plug® because of the similarity in composition with the Ultrafoam® collagen sponge used in previous studies20,24,42 and a 40-fold increase in volume. Second, we chose the Optimaix 3D®, which owing to its highly oriented fibers was capable to retain its shape and size during the in vitro phase. When associated with chondroprogenitors, both of these large collagen sponges supported chondrogenesis with an earlier cartilage onset in the Optimaix 3D® group (Figure 3). However, one major limitation of those Optimaix-based constructs was the lack of remodeling of the collagen sponge in vitro and in vivo that appeared much less permissive to host cells upon implantation and thus strongly reduced the amount of bone formation (Figure 4). This result validated the Zimmer plug® as the suitable scaffolding material for our proof-of-concept with pediatric ASCs. When used in combination with adult SVF cells, we generated clinically relevant phalangeal grafts (up to 390 mm3) containing large cartilage pockets, comparable in size to the one reported by Sheehy et al., 29 engineered by using distal phalanx-molded BM-MSC-laden alginate hydrogels, primed toward hypertrophic cartilage and supplemented with a layer of engineered hyaline cartilage. One obvious limitation of this work is the reduced number of pediatric donors—linked to extreme scarcity of pediatric biopsies—tested in the herein study (n = 4 for the ECO potential of pediatric ASC and n = 1 for the phalangeal HCTs grafts). However, the number of in vivo replicates (N = 8) used for the phalangeal HCT grafts aimed at mitigating the impact of the single pediatric donor used for validation. A relevant drawback of our upscaling method was the inferior cartilage maturation obtained as compared to the one obtained with Ultrafoam® scaffolds of smaller sizes. Nonetheless, when implanted for 12 weeks in an ectopic mouse model mimicking the clinical scenario of an empty phalangeal pocket, such large HCTs were able to de novo generate a full bone with preserved shape and size. An interesting aspect of this study was the extent of bone remodeling obtained in these large HCTs, that surpasses the one described in previous reports using ASCs20,21,23 or BM-MSCs. 29 Not only significant amounts of bone and BM tissues were observed, but also large pockets of vascularized cartilage. At that stage also, the collagen scaffolding material was mostly remodeled. Therefore, it is reasonable to speculate that the endochondral ossification was still ongoing rather than yet fully completed.
Conclusion
In the present study, we generated the proof of concept of an autologous TE strategy to generate osteogenic phalangeal grafts following a developmentally inspired approach for the treatment of children suffering from symbrachydactyly. Specifically, we could show the ECO potential of pediatric ASCs capable of generating de novo bone organs of size and shape suited for the targeted clinical scenario. In this context, the in vitro generated HCTs phalangeal grafts—owing to their biomechanical properties—would restore the hand pinching capability shortly after the surgery. However, the reduced chondrogenicity associated with the necessary expansion of ASCs should prompt us to better refine the in vitro manufacturing protocol in order to achieve better cartilage formation and bone formation. For example, the use of optimized scaffolding materials promoting cell distribution, cartilage formation and bone remodeling would enhance ECO processes. Finally, in the field of pediatric bone repair further research is needed in order to generate TE grafts that would not only (re)generate bone but also support the child development by growing in concert.
Supplemental Material
sj-docx-1-tej-10.1177_20417314241257352 – Supplemental material for Engineered phalangeal grafts for children with symbrachydactyly: A proof of concept
Supplemental material, sj-docx-1-tej-10.1177_20417314241257352 for Engineered phalangeal grafts for children with symbrachydactyly: A proof of concept by Romain Schaller, Adrien Moya, Gangyu Zhang, Mansoor Chaaban, Robert Paillaud, Ewelina M Bartoszek, Dirk J Schaefer, Ivan Martin, Alexandre Kaempfen and Arnaud Scherberich in Journal of Tissue Engineering
Supplemental Material
sj-png-2-tej-10.1177_20417314241257352 – Supplemental material for Engineered phalangeal grafts for children with symbrachydactyly: A proof of concept
Supplemental material, sj-png-2-tej-10.1177_20417314241257352 for Engineered phalangeal grafts for children with symbrachydactyly: A proof of concept by Romain Schaller, Adrien Moya, Gangyu Zhang, Mansoor Chaaban, Robert Paillaud, Ewelina M Bartoszek, Dirk J Schaefer, Ivan Martin, Alexandre Kaempfen and Arnaud Scherberich in Journal of Tissue Engineering
Supplemental Material
sj-png-3-tej-10.1177_20417314241257352 – Supplemental material for Engineered phalangeal grafts for children with symbrachydactyly: A proof of concept
Supplemental material, sj-png-3-tej-10.1177_20417314241257352 for Engineered phalangeal grafts for children with symbrachydactyly: A proof of concept by Romain Schaller, Adrien Moya, Gangyu Zhang, Mansoor Chaaban, Robert Paillaud, Ewelina M Bartoszek, Dirk J Schaefer, Ivan Martin, Alexandre Kaempfen and Arnaud Scherberich in Journal of Tissue Engineering
Supplemental Material
sj-png-4-tej-10.1177_20417314241257352 – Supplemental material for Engineered phalangeal grafts for children with symbrachydactyly: A proof of concept
Supplemental material, sj-png-4-tej-10.1177_20417314241257352 for Engineered phalangeal grafts for children with symbrachydactyly: A proof of concept by Romain Schaller, Adrien Moya, Gangyu Zhang, Mansoor Chaaban, Robert Paillaud, Ewelina M Bartoszek, Dirk J Schaefer, Ivan Martin, Alexandre Kaempfen and Arnaud Scherberich in Journal of Tissue Engineering
Footnotes
Acknowledgements
We acknowledge the contribution of Dr Laura Power for the development of the automated cartilage maturation score. We would like to thank both the Microscope and Histology Core Facilities of the Department of Biomedicine (University of Basel), especially Dr. Diego Calabrese and Mylène Toranelli for assistance and support
Data availability
Data will be made available on request.
Declaration of conflicting interests
The authors declared that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Swiss National Science Foundation (SNF Project Grant 310030_179444, to Arnaud Sherberich and Dirk Shaefer) and by the University of Basel (Spezialprogramm Nachwuchsförderung Klinische Forschung, to Adrien Moya).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.