Abstract
Spinal cord injury (SCI) causes tremendous harm to a patient’s physical, mental, and financial health. Moreover, recovery of SCI is affected by many factors, inflammation is one of the most important as it engulfs necrotic tissue and cells during the early stages of injury. However, excessive inflammation is not conducive to damage repair. Macrophages are classified into either blood-derived macrophages or resident microglia based on their origin, their effects on SCI being two-sided. Microglia first activate and recruit blood-derived macrophages at the site of injury—blood-borne macrophages being divided into pro-inflammatory M1 phenotypes and anti-inflammatory M2 phenotypes. Among them, M1 macrophages secrete inflammatory factors such as interleukin-β (IL-β), tumor necrosis factor-α (TNF-α), IL-6, and interferon-γ (IFN-γ) at the injury site, which aggravates SCIs. M2 macrophages secrete IL-4, IL-10, IL-13, and neurotrophic factors to inhibit the inflammatory response and inhibit neuronal apoptosis. Consequently, modulating phenotypic differentiation of macrophages appears to be a meaningful therapeutic target for the treatment of SCI. Biomaterials are widely used in regenerative medicine and tissue engineering due to their targeting and bio-histocompatibility. In this review, we describe the effects of biomaterials applied to modulate macrophage phenotypes on SCI recovery and provide an outlook.
Introduction
Spinal cord injury (SCI) is a serious injury, usually resulting in a transient or permanent loss of sensory, motor, and autonomic functions below the level of the injury. 1 In China, there are 127 cases of SCI per million people, the incidence rate amongst the elderly (64–75 years old) being higher than that of young people. 2 SCI results in quadriplegia and an impaired respiratory system, restricting activities and placing enormous physical, psychological, and financial stress on patients.1,3 The reported annual cost of SCI treatment is between $30,770 and $62,563. 4 SCI usually results from direct injury or compression of the spinal cord following trauma. 5 With age, cervical spondylosis, spinal stenosis, etc. compress the spinal cord and damage it, an injury defined as non-traumatic SCI, degenerative cervical myelopathy being considered the most common non-traumatic form of SCI. 6 However, traumatic SCI usually causes greater harm to patients due to its rapid onset and severity.
SCI treatment methods include surgical anastomosis, surgical decompression, drug therapy, local freezing, physical rehabilitation, and enzyme preparations amongst others, but there is still no truly effective treatment. 7 However, the extent of sensory, motor, and autonomic function recovery after an SCI correlates with the post-injury operative time. 8 Currently, there is no effective treatment for traumatic SCI other than improving the general condition, stabilizing the spine, and reducing the compression of the spinal cord. 9 Surgical decompression within 24 h of a SCI does, however, facilitate sensorimotor recovery.10,11 Studies have shown that surgical decompression within 6 h of injury results in ⩾1 American Spinal Injury Association grade improvement in approximately 70% of patients. 12 Moreover, surgical decompression prevents the spinal cord from being compressed, improves its surrounding environment, and helps nerve recovery in the damaged area, but it cannot regenerate necrotic nerve cells.1,13 SCI refractory occurs due to non-regenerating necrotic neurons, 1 thus, the re-establishment of closed pathways as a replacement for damaged neural tissue by cell transplantation is a promising treatment modality. 14 Additionally, stem cells—which secrete neurotrophic factors, improving nerve regeneration—are good candidates for SCI treatment. 15 Consequently, the use of human induced pluripotent stem cells, 14 bone mesenchymal stem cells, 16 Schwann cells, 17 neural stem cells (NSCs), 18 ensheathing cells, 19 and pluripotent stem cells (PSCs) 20 in SCI repair has been studied. Different kinds of stem cells or their derivatives can differentiate various cell types-that is, neurons, astrocytes, or oligodendrocytes-to support neuronal survival or regeneration. 21 Cell transplantation technology is also a promising method for the treatment of SCI. Although several such methods have entered clinical trials, transplanted stem cells have not progressed as expected. 22 Most NSCs differentiate into astrocytes after an SCI and ultimately lead to increased glial scarring, preventing axonal regeneration. 13 Conversely, an important factor affecting SCI repair is the microenvironment, which changes over time. 23
In addition, inflammation is often an important factor hindering cell transplantation. 21 Inflammation is a protective response of the body, which can effectively remove necrotic cells and replenish its own immunity. When SCI occurs, neutrophils are the first to be activated, migrating to the damaged area. 24 Subsequently, inflammatory cells—such as neutrophils, lymphocytes, and macrophages—are activated and release inflammatory factors—such as interferon-2 and tumor necrosis factor-α. With the increase of inflammatory markers, the microenvironment leads to the formation of syringomyelia and glial scar, inhibiting the recovery of neurological functions. 1 Different macrophage roles in the acute and chronic phases of SCI making macrophages a new target for SCI therapy. 25 In the acute SCI phase, M1-type macrophages with stronger phagocytic and antigen-presenting abilities are the first to polarize. 26 In the chronic phase, M2 macrophages—which promote tissue repair—play a major role. Consequently, regulating the macrophage phenotype in the SCI microenvironment is beneficial in promoting SCI repair.27,28
In recent years, biomaterial-assisted regenerative medicine has been developing rapidly. 29 Strategies for the repair of SCI based on biomaterials have been widely used, 7 biomaterials modulating the macrophage phenotype in the spinal cord microenvironment to repair SCI being a promising therapeutic target. In this review, we describe the SCI process and the role of macrophages in it (Schematic illustration and Table 1). Biomaterial-regulated macrophage phenotyping for delivery strategies are then introduced. Finally, we provide an outlook on biomaterials modulating macrophage phenotype to repair SCI.
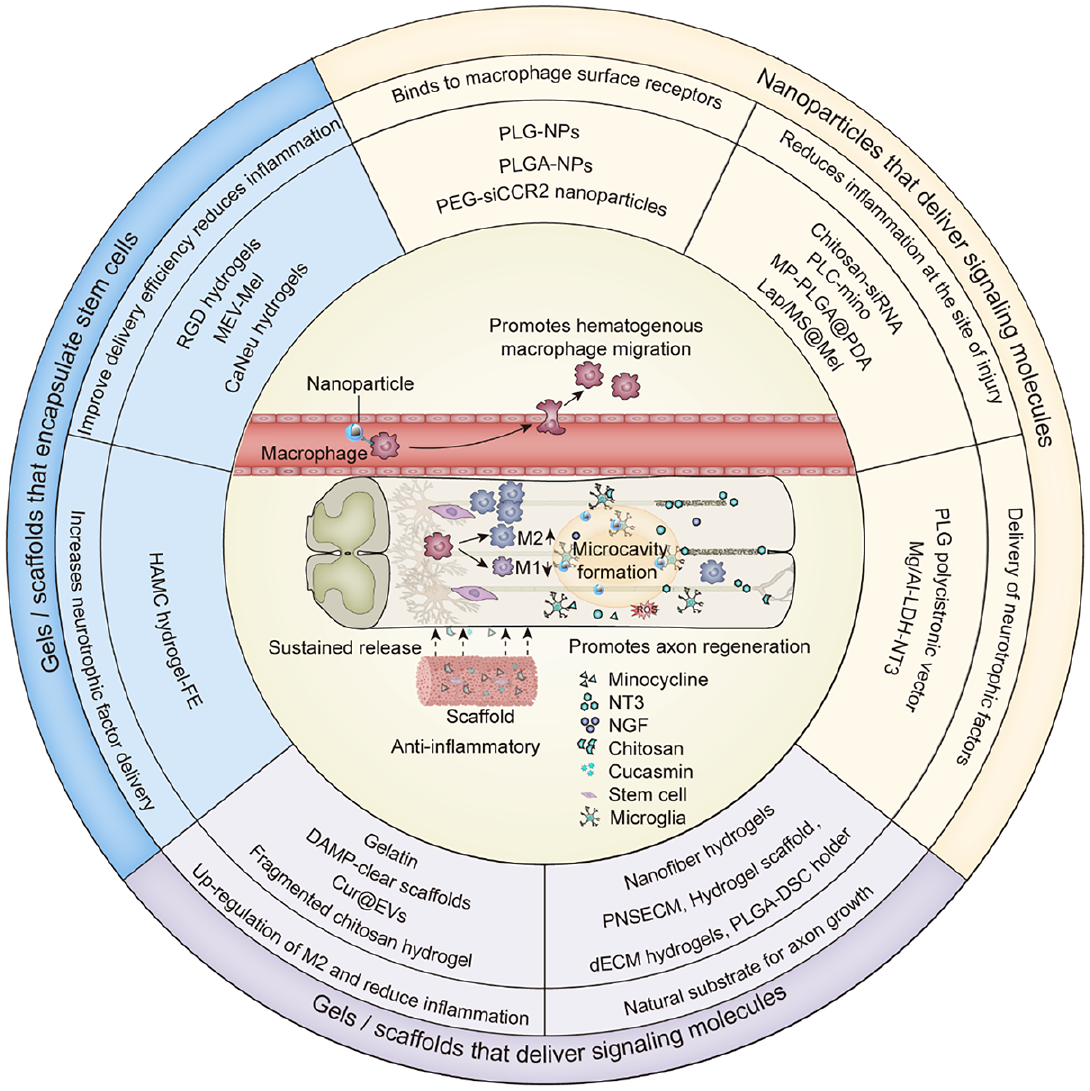
Biomaterials delivery strategies to repair spinal cord injury by modulating macrophage phenotypes.

Schematic illustration of biomaterials delivery strategies to repair spinal cord injury by modulating macrophage phenotypes. (1) The nanoparticles bind to the surface receptors of macrophages to promote the migration of bloodborne macrophages to the site of principle damage. (2) The gel scaffold continuously releases delivery substances to reduce the inflammatory response of the damage microenvironment, releases molecules to promote the polarization of macrophages to M2 type, and releases neurotrophic factors to promote axon regeneration.
Spinal cord injury (SCI)
Spinal cord injury (SCI) process
The spinal cord is enclosed in the spinal canal along with the capsules, blood vessels, and nerve roots. The spine plays a protective role through its bony structures, ligaments, and dura mater. Physiologically, the spinal cord is surrounded by a plasticity-regulated dynamic extracellular matrix, composed of hyaluronic acid, connexin, and chondroitin sulfate proteoglycan (CSPG) called the perineural network (PNN). 30 In addition, the blood-spinal cord barrier (BSCB) is a physical barrier between the blood and spinal cord parenchyma that prevents toxins, blood cells, and pathogens from entering the spinal cord. 31 The BSCB is composed of endothelial cells, cell-to-cell junctions, basement membranes, and astrocyte foot processes and helps to maintain ion homeostasis in neural signaling. 31 Under normal circumstances, the spinal cord maintains functional stability under the maintenance of the PNN and BSCB.
SCI usually involve two mechanisms—that is, primary (mechanical) injury and secondary injury. 32 Primary injury is irreversible, usually caused by external mechanical forces causing damage to the integrity of the spine resulting in direct neuronal damage—such as contusion, compression, or tears. 33 Secondary injury is based on the primary injury, a series of cells and cytokines causing inflammation and cell death in the injury microenvironment. 34 When the spinal cord is traumatized, the fracture fragments or trauma directly cause neuronal cell death and blood vessel destruction. 1 Following the breakdown of the BSCB, inflammatory cells—such as neutrophils and macrophages—are recruited to the injury site to exacerbate the SCI. 35
Based on the time of injury, an SCI can be divided into acute, subacute, and chronic phases. SCI in the acute phase occurs 2–48 h after injury. Acute inflammation is the primary manifestation of this stage, and neutrophils are considered to be the first inflammatory cells to migrate to the injury site after SCI. 1 Neutrophils have a short half-life and resolve rapidly after releasing IL-1β at the site of injury. The acute inflammatory response that occurs after SCI is often caused by microglia and macrophages triggering a dysregulation of the homeostasis of the lesion and the spinal cord parenchyma immediately adjacent to the lesion.36,37 Studies show that activation of microglia and astrocytes favors neural tissue protection during the first week after injury. 38 Microglia are innate immune cells that, once activated, produce pro-inflammatory factors such as IL-1β and TNF. 39 During the early stage of SCI, all kinds of inflammatory cells gather at the lesion, clear the damaged tissue by actively phagocytosing myelin sheath and dead cell debris, and limit further expansion of the injury. 40 However, the aggregation of these inflammatory and immune cells causes a strong immune response, and the secretion of a large number of inflammatory factors and toxins, further aggravating the SCI.
For example, neutrophils phagocytose peripheral cells destroy nerve tissue and inhibit oligodendrocyte activity, producing proteases and reactive oxygen species to inhibit nerve repair. 41 Moreover, astrocytes are the main cause of glial scarring 7–9 days after injury. 42 Glial scars are produced primarily by activated astrocytes, the main component of which is chondroitin sulfate proteoglycan (CSPG). During axon regeneration, the glial scar causes the collapse of the axon growth cone, which is not conducive to neuronal regeneration. 43 Infiltrating leukocytes and T cells are cleared 14 days after the SCI, but macrophages remain in the injury environment for some time. 44 Toxins—such as TNF-α, inducible nitric oxide synthase (iNOS), reactive oxygen species (ROS), and pro-inflammatory factors released by macrophages during the subacute and chronic phases—are often key mechanisms of secondary injuries, 36 which can persist for days and weeks after SCI, leading to increased cavitation and glial scarring at the lesion site, exacerbating neurological dysfunction. Consequently, altering the inflammatory environment surrounding the injury is critical for SCI repair.
The role of macrophages in spinal cord injury (SCI)
Based on their origin, macrophages can be divided into resident microglia and blood-derived macrophages. 25 Microglia originate from the embryonic yolk sac, tissue-retaining cells involved in central nervous system homeostasis. 45 After an SCI, neuroinflammation is mediated primarily by microglia. 46 Compared to macrophages, microglia are activated first and are distributed in the center of injury. 25 Blood-derived macrophages are derived primarily from monocytes and can be divided into pro-inflammatory M1 and anti-inflammatory M2 type macrophages. M1-type macrophages are polarized first during the early stages of inflammation and exhibit stronger phagocytic and antigen-presenting abilities at the site of the SCI.47,48 M2 macrophages inhibit neuronal apoptosis and inflammatory responses, thereby promoting the repair of neural tissue. 26 Studies have shown that after an SCI, M1 macrophages are derived primarily from microglia, a small portion of them being derived from blood-derived macrophages. 49 In an SCI, the activated microglia secrete inflammatory factors—such as IL-β, TNF-α and IL-6—to promote the infiltration of blood-derived macrophages into the surrounding injury. 50 The macrophages are then polarized to M1 type macrophages under the stimulation of IL-β and TNF-α, and further secrete IL-β, TNF-α, IL-6, interferon-γ, NO and reactive oxygen species. 50 Unlike microglia, M1 macrophages are more phagocytic.48,51 These factors engulf necrotic tissue and cells during the initial phase of inflammation, but in the subacute and chronic phases, they act synergistically to cause endothelial, neuronal, and axonal damage.46,52
Compared with M1-type macrophages, M2-type macrophages release anti-inflammatory cytokines and neurotrophic factors—such as IL-4, IL-10, IL-13, transforming growth factor beta (TGF-β), insulin-like growth factor (IGF), epidermal growth factor, and nerve growth factor (NGF). 53 Anti-inflammatory cytokines and neurotrophic factors provide an environment for neuronal regeneration. Although studies have shown that M2 macrophages can induce astrocyte activation through TGF-β in vitro to promote glial scarring, this does not appear to be conducive to SCI repair. 43 However, during the early stages of SCI, glial scars can act as an anti-inflammatory barrier to hinder the damage of toxic substances to the SCI microenvironment. 54 Overall depletion of macrophages in the injured microenvironment without distinguishing between M1 and M2 phenotypes can be detrimental to SCI recovery. 25 As such, down-regulation of M1 macrophages and up-regulation of M2 macrophages have positive effects on SCI repair.52,55
Biomaterials modulate macrophage phenotypes
Nanoparticles that deliver signaling molecules
SCI induces elevated production of C-C chemokine ligand 2 (CCL2), CC-chemokine receptor 2 (CCR2) being expressed on potent pro-inflammatory mononuclear M2 macrophages. 56 Inhibition of the CCL2/CCR2 signaling pathway by inhibiting CCR2 expression impedes the migration of blood-derived monocytes to the injury site. 57 However, systemic administration of CCR2 inhibitors may lead to side effects. To better target hematogenous monocytes for the purpose of reducing their infiltration to the site of injury. Zhang et al. prepared a CCR2siRNA-encapsulated nanoparticle delivery system by means of a double-emulsion method using polyethylene glycol (PEG) and polylactide as matrices (Figure 1(a)). 58 CCL2 expression was up-regulated after SCI (Figure 1(b)), which induced macrophage infiltration to the injury site (Figure 1(c)). CCR2siRNA-encapsulated nanoparticle were specifically absorbed by bloodborne monocytes, preventing these cells from infiltrating to the site of injury. 12 h after injection of CCR2siRNA-encapsulated nanoparticles, 94.2% of blood-derived macrophages detected a decrease in the expression of CCR2 by flow cytometry. Down-regulation of CCR2 expression resulted in a reduction in the number of monocytes at the injury site and inhibition of pro-inflammatory cytokine production (Figure 1(d)). CCR2 siRNA nanoparticles play a role in reducing CCR2 expression in bloodborne macrophages. However, the effect on macrophages in the center of injury was not reported. Park et al. developed negatively charged PLG nanoparticles. 59 Nanoparticles (diameter 500 nm) bound to scavenger receptors on immune cells at the injury site, encouraging them to move to the spleen, thereby indirectly reducing inflammation at the injury site. Immunofluorescence showed that PLG nanoparticles increased M2 macrophages and IL-10 at the site of injury for up to 84 days after the SCI, (IL-10 is an anti-inflammatory factor secreted by M2 macrophages, which induces M2 macrophage polarization). Jeong et al. prepared poly(lactide-co-glycolide) (PLGA) immune-modifying nanoparticles (IMPs). 60 IMPs were highly negatively charged synthetic particles of diameter 500 nm that bound to macrophage scavenger receptors with collagenous structure. The bound macrophages did not migrate to the injury site but to the spleen for apoptosis. Although this approach reduces M1-type macrophage levels around the injury, it has not been shown whether reducing blood-borne macrophage levels results in systemic damage.

(a) Schematic diagram of siRNA-NP synthesis and siRNA-NP particle size distribution. (b) The expression level of CCL2 on the first, third, and fifth day after the SCI microenvironment. (c) The number of macrophages and microglia recruited by the injured microenvironment after SCI. (d) Percentage of macrophages and microglia at 72 h after SCI. (e) Spinal cord immunostaining 3 days after spinal cord hemisection model. (f) Determination of inflammatory cell content in the injured area (miR-CTRL: non-functional miRNA fragment). Reproduced by permission of Zhang et al 58 and Louw et al. 64
Microglia are resident cells in SCIs. Activated microglia are the main source of chronically damaging substances such as TNF-α, ROS. 61 Therefore, maintaining the quiescent state of microglia is an important way to reduce damage. Studies have shown that miRNA-124 maintains the quiescent state of microglia via CCAAT/enhancer binding proteins (a transcription factor). 62 Maintenance of microglia quiescence reduces microglia recruitment to blood-derived macrophages and reduces the inflammatory response surrounding SCI. However, the delivery of miRNA-124 into endogenous microglia is difficult. Chitosan is a naturally charged polysaccharide with a positive charge. 63 Chitosan binds to negatively charged RNA strands to increase the efficiency of RNA delivery. Louw et al. developed chitosan/siRNA nanoparticles by adjusting the ratio of positively charged chitosans to negatively charged miRNA-124 components (n/p ratio) to control the surface charge and particle size. 64 The diameter of the composite particles was 158.1 ± 19.4 nm. In in vitro experiments, the composite particles were endocytosed by microglia 2 h after their addition, and no cytotoxicity of the composite particles was found within 48 h. In vivo experiments in mice found that composite particles reduced TNF-α and ROS production in macrophages. Immunostaining showed that siRNA-NP reduced the macrophage content around the spinal cord in the rat spinal cord hemisection model (Figure 1(e) and (f)). Although studies have shown that composite particles reduced acute inflammation in SCIs, long-term effects of chitosan composite particles on SCI recovery are unknown.
Minocycline being a spectrum antibiotic that reduces pro-inflammatory responses associated with microglia/macrophage activation. 65 Using potassium sulfate as an initiator, Papa et al. prepared nanoparticles using polyethylene glycol (PEG) and polycaprolactone, encapsulated minocycline into nanoparticles to make PCL-mino.66,67 PCL-mino increase pharmacological activity and reduce potential drug-related side effects by enhancing drug targeting and concentration at the site of injury. PCL-mino releases minocycline after being engulfed by microglia/macrophages to reduce the inflammatory response. Confocal microscopy revealed that PCL mino was able to target microglia and be endocytosed by microglia. PCL mino inhibited microglia activation and released pro-inflammatory factors, thereby reducing the recruitment of M1-type macrophages. PCL-mino substantially reduced microglial hyperplasia and macrophage infiltration detected on the seventh day after the SCI. Through quantitative analysis, M1-type macrophages peaked on day three and declined on day seven, and M2-type macrophages peaked on day seven, substantially reducing the content of M1 macrophages by the seventh day compared to the treatment group. Moreover, the study also found that the mRNA and protein levels of CCL2 were considerably reduced after PCL-mino treatment by qRT-PCR. PCL-mino reduced the recruitment of M1-type macrophages around the SCI. PCL-mino treatment induced considerable durable recovery for up to 63 days after the SCI.
Methylprednisolone (MP) is a synthetic glucocorticoid with potent long-acting anti-inflammatory, anti-allergic, and immunosuppressant properties. 68 MP can promote nerve recovery in acute SCI. However, high doses of MP increase the risk of adverse events. 69 In order to reduce the adverse events caused by high concentration of MP and increase the targeting of MP to the site of injury. Wei et al. developed MP-PLGA@PDA—composed of a MP-incorporated poly(lactic-co-glycolic acid) (PLGA) inner core and a biocompatible polydopamine (PDA) outer shell (Figure 2(a)). 70 The PDA shell captures the released cytokines, and MP inhibits new cytokine synthesis, which can effectively inhibit the inflammatory cascade. The MP-PLGA@PDA had an average diameter of 237 nm and a sustained slow release of MP, releasing only approximately 26.3% of MP within 4 h; approximately 90% of MP was released within 3 days (Figure 2(b)). The advantage of MP-PLGA@PDA was that it captured the released cytokines, but also released MP to inhibit the synthesis of cytokines. In vitro experiments showed that MP-PLGA@PDA substantially reduced TNF-α and IL-6 and decreased the recruitment of macrophages within 6 h. In vivo, it was showed that MP-PLGA@PDA reduced the damage to neurons and axons by 21.5% by day 28 (Figure 2(c)), indicating that MP-PLGA@PDA had a protective effect on nerve regeneration after the SCI.

(a) MP-PLGA@PDA simultaneously captures released cytokines and inhibits the secretion of new cytokines, thereby inhibiting neuroinflammation to promote functional recovery of the injured spinal cord. (b) MP release profiles of MP-PLGA and MP-PLGA@PDA. (c) Representative immunohistochemical staining images of GFAP (green) and NF200 (red) in longitudinal sections of injured spinal cord at day 28 after trauma (neurofilament (NF200) is considered to be a potential surrogate marker for neuronal and axonal damage). The MP-PLGA@PDA group has a considerably smaller reduction in NF200 intensity than that of the MP-PLGA and PLGA@PDA groups. Reproduced by permission of Wei et al. 70
The transcription factor interferon regulatory factor 5 (IRF5) has received much attention for its role in inflammatory and immune responses, playing an important role in the M1 phenotype of macrophages. 71 IRF5 expression in macrophages is reversibly induced by inflammatory stimuli and contributes to the plasticity of macrophage polarization. 72 siRNA-mediated inhibition of IRF5 expression reduces pro-inflammatory M1 macrophage markers and down-regulates inflammation. 73 Li et al. developed lipidoid nanoparticles (LNPs) using distearoylphosphatidylcholine and cholesterol for siRNA delivery to SCI sites. 74 siRNA-mediated IRF5 knockdown leads to decreased expression of M1 macrophage markers. Nanoparticles ranging in size from 50 to 100 nm substantially increased the percentage of M2 macrophages within 3 days of injury in SCI mice. IRF5 silencing resulted in a sharp reduction in TNF-α levels (starting from day 1) after SCI and induced a considerable increase in IL-10 levels (starting from day 1). Nonetheless, this study only demonstrated this effect in mouse macrophage phenotypes, and it was unclear whether IRF5 would have the same effect in human macrophage phenotypes. Targeting the TGF receptor also modulates the macrophage phenotype to promote SCI repair.
Neurotrophic factors in the spinal cord need neurotrophic factors to survive and develop to re-establish connections to the original target. 75 However, endogenous neurotrophic factors at the site of injury are insufficient to maintain nerve repair. 76 The strategy of delivering exogenous neurotrophic factors is more conducive to nerve repair. Smith et al. dissolved 75:25 lactide: glycolide in dichloromethane, added sucrose, and then froze it to make sugar fibers to form PLG bridges and combine IL-10 and neurotrophic factor 3 (NT3) cistron piggybacking on the PLG bridge 77 —IL-10 promoting the M2 macrophage phenotype, and NT3 providing nutrients for nerve regeneration. IL-10—provided by the PLG—increased the number of M2 macrophages threefold by week 12 (Figure 3(a)). Immunohistochemical staining showed that IL-10+NT-3 delivery also increased oligodendrocyte and Schwann cell axon formation (Figure 3(b) and (c)). To enhance the anti-inflammatory and vegetative-neurological properties of the injury site, Zhu et al. developed magnesium-aluminum layered double hydroxide nanoparticles (Mg/Al-LDH) for loading NT3. 78 LDH is a type of clay with anionic and anion exchange properties with good biocompatibility and anti-inflammatory properties. The average hydrated particle size of LDH-NT3 (112.6 ± 8.2 nm) targeted the TGF receptor to up-regulate the TGF-β2 signaling pathway and promoted the expression of TGF-β and IL-10. Following the loading of NT3, LDH-NT3 further accelerates axonal growth and neuron-to-neuron synaptic transmission.

(a) Ctrl, NT-3, IL-10, and IL-10+NT-3 macrophage density from bridge implants. White arrows indicate F480+ macrophages. Yellow arrows indicate F480+/Arg+ macrophages. Scale: 100 μm. (b) Ctrl, NT-3, IL-10, and IL-10+NT-3 sources of myelination from bridge implants. White arrows indicate oligodendrocyte myelinated axons. Yellow arrows indicate Schwann cell myelinated axons. Scale: 100 microns. (c) Density of myelinated axons in oligodendrocytes (NF200+/MBP+/P0-) and myelinated axons in Schwann cells (NF200+/MBP+/P0+). (d) Staining of M1 macrophages and M2 macrophages in PNSECM and empty ducts. M1 macrophages (CD68+CCR7+) M2 macrophages (CD68+CD206+). The presence of PNSECM also increases the M2:M1 ratio within the duct. Reproduced by permission of Smith et al. 77 and Prest et al. 87
Zhang et al. developed a novel drug delivery method for SCI repair. 79 First, 10 mg of melatonin (Mel) and 100 mg of PLGA were phacoemulsified under ice bath conditions to obtain sustained-release microsphere (MS) nanoparticles MS@Mel. Mel exerted neuroprotective effects by attenuating inflammatory and oxidative stress responses. 80 MS has several advantages—including improved drug solubility, permeability, and bioavailability—and Mel was a free radical scavenger and broad-spectrum antioxidant. Mel also promotes the M2-like polarization of macrophages. 81 The MS@Mel was encapsulated in the platelet membrane to form PM@MS@Mel. Platelet membranes naturally targeted bleeding and inflammatory sites without the need for ligand-dependent passive and active targeting and external stimuli. The new sustained-release system facilitated the transformation of PM@MS@Mel from a pro-inflammatory M1 phenotype to an anti-inflammatory M2 phenotype of macrophage subsets. The MS (of diameter approximately 280.7 nm) and PM/MS@Mel sustained Mel released for at least 7 days in vitro, promoting the polarization of macrophages M1 to M2 in the SCI rat model.
Gels/scaffolds that deliver signaling molecules
Gelatin, possessing biodegradability and biocompatibility, is used as a substitute for tissue engineering. Studies have shown that gelatin can reduce local inflammation after implantation to the SCI and brain injury. 82 Ma et al. prepared a gelatin hydrogel by photocrosslinking gelatin methacrylate and lithium phenyl-2,4,6-trimethylbenzoylphosphinate (LAP) under the induction of photoinitiators. 83 Hydrogels have a porous structure, with pores ranging in diameter from 20 to 200 μm. Transplantation of hydrogel to a lesion site has been shown to reduce inflammation around the SCI injury. However, the application of gelatin alone only reduced inflammation in the center of the injury, cannot inhibit microglia and macrophages that have been activated. Therefore, gelatin scaffolds alone are not sufficient to address the inflammatory response caused by overactivation of macrophages and microglia. The colony stimulating factor 1 receptor (CSF1R)-the macrophage colony stimulating factor (M-CSF) receptor-is a transmembrane tyrosine kinase present on the surface of macrophages, microglia, and monocyte receptors. 84 The CSF1/CSF1R axis is associated with the proliferation, differentiation, and function of macrophages. PLX3397, a CSF1R inhibitor, reduces the number of macrophage and microglia. Transplantation of hydrogel to a injury site combined with feeding the CSF1R inhibitor PLX3397 to SCI mice inhibited the activation of microglia and macrophages, especially the macrophage content in the hydrogel scaffold. 83 Moreover, the mRNA levels of cytokines IL1β, IL6, TNFα, and IFNγ detected by quantitative real-time PCR (qRT-PCR) decreased within 60 days. Relying on the advantages of gelatin’s adhesion to cells, gelatin scaffolds induced endogenous NSCs to migrate to the injury site. 85 Gelatin hydrogels provided bridges to connect damaged tissues, and provided endogenous NSCs with a substrate to induce migration, while reducing inflammation caused by macrophages and microglia. However, CSF1R is also expressed on the surface of peripheral myeloid cells, CSF1R inhibitors causing peripheral blood mononucleosis and bone marrow suppression. 86
Acellular tissue engineering can repair many tissues—including cardiac muscle, bone tissue, and peripheral nerves—making it an engineering field on the rise. 87 Decellularized extracellular matrix (dECM) provides a living environment similar to that of the original tissue structure. However, prior to application, the ECM needs to be freed of cells and harmful extracellular matrices. Prest et al. removed the cellular structure of a dog’s spinal cord ECM and stirred in gastric acid for 48 h, after which the product was frozen to form a peripheral nerve-specific extracellular matrix-based hydrogel (PNSECM). 87 PNSECM has the advantage of providing a natural substrate for axon growth over other implanted stents. After filling PNSECM at the site of injury, PNSECM were also found to cause an increase in the number of M2 macrophages to protect axon regeneration. Immunofluorescence staining showed that PNSECM induced the migration of M1-type macrophages and M2-type macrophages, the migration of M2-type macrophages being more than that of M1-type macrophages (Figure 3(d)). As pigs are genetically similar to humans, multiple applications of porcine dECM hydrogels have been reported. 88 Hong et al. decellularized porcine brain-derived ECMs and cleared glycosaminoglycans (GAGs) to form porous dECM gels. 89 A brain-derived dECM is softer and more suitable for cell growth than other collagens. To match the mechanical strength of the gel, collagen fibers were added to the dECM, with in vitro studies confirming that the dECM promoted hippocampal neuronal proliferation and increased the number of M2 macrophages by day 2. After 8 weeks in a rat SCI model, M2 macrophages were elevated by immunohistochemical staining, and there was improved exercise capacity in rats. Although porcine dECMs have a certain repair effect after SCIs, the high extraction cost and difficult purification methods hinder their development. Electrospun polycaprolactone (PCL) fibers and hyaluronic acid (HA) covalently bond to form hydrogels. 90 To a certain extent, the hydrogels makes up for the high extraction cost of dECM and the difficulty of purification methods. The advantage of hydrogel was that it resembled the native spinal cord ECM to provide mechanical support in the contused spinal cord segment while promoting cell penetration. Immunostaining showed that the hydrogel promoted macrophage infiltration and macrophage polarization to M2 phenotypes in the rat SCI model by day 28. The hydrogel promoted a substantial 2.6-fold increase in the number of immature neurons in the SCI. However, the induction of macrophage polarization toward M2 by PCL alone was weak. Ma et al. fabricated PLGA-DSC scaffolds using an electrospun PLGA shell and decellularized spinal cord (DSC). 91 Implantation of the DSC into the injured spinal cord resulted in improved spinal cord structure or function after the SCI. 92 The PLGA electrospun nanofibers had an average diameter of 300 nm, and the DSC had a porous structure. Consequently, more than 90% of NSCs survived 7 days after orthotopic transplantation of the SCI in vivo. By week 8, M2-type macrophages had infiltrated the PLGA-DSC scaffolds, ameliorated chronic inflammation, and formed a pro-regenerative microenvironment conducive to tissue repair.
Damage-associated molecular patterns (DAMPs) are diverse set of host molecules responsible for initiating innate immune activation after SCI. 93 DAMPs are produced at the injury site and release pro-inflammatory factors such as TNF-α and IL-1β that worsened SCI. 94 Shen et al. developed a poly amidoamine-gelatin photocrosslinked hydrogel scaffold that could clear DAMPs around lesions and release IL-10. 95 The porous microstructures of hydrogel scaffolds of similar pore sizes (60–180 μm) provide conditions for cell growth and differentiation. The scaffold has a dendritic network of cationic structures that adsorb negatively charged DAMPs molecules around the damage. Consequently, the loaded IL-10 remained in the scaffold for 21 days, continuously releasing IL-10 around the SCI. At the same time, the scaffold removed DAMPs and reduced the inflammatory environment around the SCI, providing a basis for neuronal regeneration. A reduction in the syringomyelia was observed by the eighth week in a mouse model of SCI, as was improved exercise capacity in the mice.
Chitosan is a natural cationic polysaccharide that modulates local inflammation. 96 Transplantation of chitosan hydrogels with SCI sites promotes neural tissue and revascularization, axonal regeneration, and reduce glial scarring. 97 von Boxberg et al. improved inflammation by applying fragmented chitosan gel to the site of SCI. 98 The particles were predominantly 20–100 μm in size, and in vitro experiments confirmed that the chitosan hydrogel could substantially up-regulate the M2 macrophage phenotype at 48 h. However, the study found that up-regulation of M2 macrophages and promotion of SCI repair only occurred when the acetylation of chitosan was less than 6%. Curcumin is a natural polyphenol that promotes the polarization of M1 macrophages to M2 macrophages. 99 To increase the delivery of curcumin to the site of injury, Zhang et al. transported curcumin and nerve growth factor (Cur@EVs) using M2 macrophage extracellular vesicles. 55 NGF was coupled to EVs surface extracellular vesicles (EVs-cl-NGF) via a matrix metalloproteinase 9 (MMP9) cleavable linker (cl), which has the advantages of high biocompatibility, low cytotoxicity, immune inertness, and long circulation time, becoming a new drug delivery carrier. 100 NGF bound to macrophage vesicles via MMP9 and is precisely released at the site of injury. In vitro experiments confirmed that EVs-cl-NGF had no toxic effect on neural cells (Figure 4(a)). EVs-cl-NGF substantially improved SCI repair by day 28 in the SCI mouse model (Figure 4(b) and (c)). Cur@EVs of diameter 120 nm could accurately target the inflammatory site in mice within 2 h, peaking at 12 h. In vitro experiments confirmed that Cur@EVs induced a considerable reduction in the expression of pro-inflammatory factors—that was, TNF-α, IL-1β, and IL-6—and an increase in the expression of anti-inflammatory factors (TGF-β), the proportion of M1 macrophage subsets decreasing to 26.90%, and the proportion of the M2 macrophage subset increasing to 31.77% (Figure 4(d)).
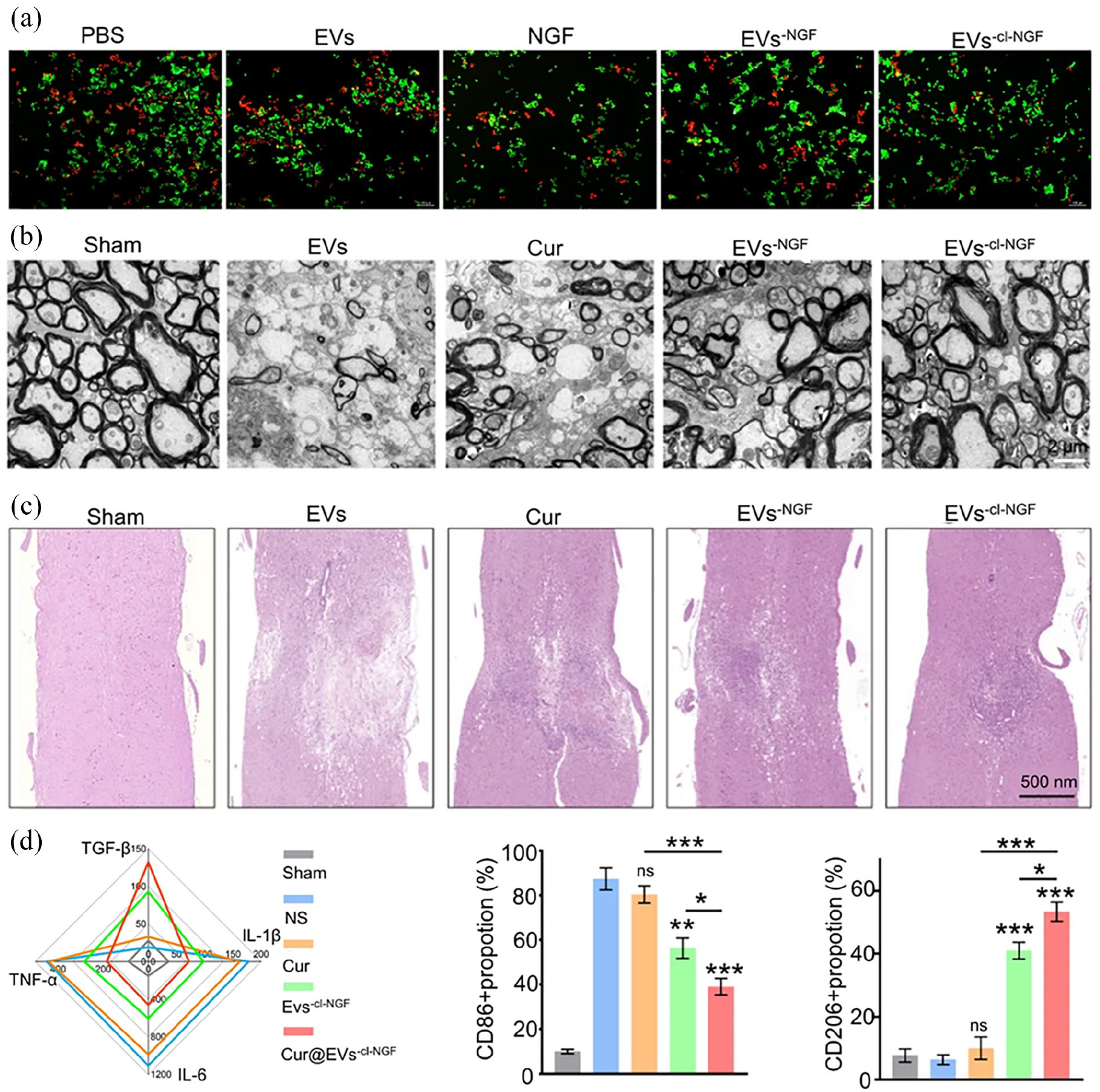
(a) Live-dead cell staining assessing the neuroprotective effect of EVs-cl-NGF on PC12 cells cultured on top of the culture system. (Green: live cells; Red: dead cells). (b) Ultrastructure of myelin sheaths from different groups as measured using TEM 28 days after SCI. (c) H&E staining image of spinal cord 28 days after SCI. (d) ELISA-based detection of pro-inflammatory factors (TNF-α, IL-1β, IL-6) and anti-inflammatory factors (TGF-β) in injured spinal cord tissue 7 days after SCI. M1 macrophages (CD80) and M2 macrophages Phage (CD206) expression content. Reproduced by permission of Zhang et al. 55
Gels/scaffolds that encapsulate stem cells which can secrete signaling molecules
Mesenchymal stem cells secrete anti-inflammatory factors to reduce the inflammatory response around injury. 101 Mesenchymal stem cells (MSCs) exhibit major autocrine and paracrine activities that stimulate the proliferation and differentiation of other cells and themselves. 93 Transplantation of mesenchymal stem cells into an injured area results in increased numbers of M2 macrophages and decreased M1 cells. 102 hMSCs can modulate immune system responses by releasing exogenous trophic factors. 103 However, direct transplantation of stem cells to the site of injury could results in extensive cell death, exacerbating the SCI by releasing harmful factors. 104 Caron et al. synthesized arginine-glycine-aspartic acid (RGD) and mixed it with azide groups to form multifunctional RGD hydrogels for loading hMSCs. 105 To increase the deposition of hMSCs, the hydrogel edge liquid was removed after culturing hMSCs on RGD for 14 days. Progressive deposition of ECM onto RGD from days 1 to 14 was observed, while the high viability of hMSCs was retained. In vivo experiments in animals confirmed that hMSCs on RGD+hMSCs+ECM scaffolds recruited more M2 macrophages at the injured site through the paracrine effect, while local injection of hMSCs did not cause an increase in M2 macrophages. This maybe because the locally injected stem cells did not stay in place, and the harsh injury environment further limited the function of the hMSCs due to tissue necrosis. 105
Although stem cells have advanced significantly in promoting regeneration, the inflammatory factors released by dead cells (IL-1, IL-6, and IL-8) released by dead cells can also exacerbate the surrounding inflammatory response when delivered to the site of injury. 106 With the development of tissue engineering and regenerative medicine, extracellular vesicles (EVs) have made great progress as active biological substances for intercellular communication due to their ability to promote tissue regeneration. 101 The paracrine-derived EVs of hMSCs have been reported in many preclinical models. 102 Liu et al. used Mel to pretreat hMSCs to generate EVs, before using Evs and Mel to form a delivery system (MEV) to deliver Mel to the SCI site for continuous release. 107 MEVs easily passed through the BSCB by virtue of their membrane fusion properties, a marked increase in M2 macrophage expression, and a marked reduction in M1 macrophage expression being observed 7 days after SCI in mice. The study found that MEVs combined with nuclear factor-like 2 (NRF2) to down-regulate the expression of NRF2 in macrophages and induced the polarization of macrophages from M1 to M2.
Adipose-derived stem cells (ADSCs) secrete a large number of growth factors—including vascular endothelial growth factor, insulin-like growth factor (IGF)-1, and basic fibroblast growth factor (bFGF)—which exhibit good effects in SCI repair. 108 In order to enhance the ability of stem cell delivery, Yuan et al. developed a hydrogel. 106 Triethanolamine (TEA) solution was added to β-cyclodextrin and evaporated, after which acetone solution was added to obtain Ac-β-CD. Methacrylic acid (12 mL) was then added to a 10% gelatin solution, and the mixture was dialyzed and dried to obtain CaNeu hydrogels for the delivery of adipose stem cells (ADSCs). 106 The stiffness of the CaNeu hydrogel was controlled at ~900 Pa for compatibility with the spinal cord stiffness (500–1500 Pa). ADSCs maintained ADSC cell viability in the CaNeu hydrogels. Confocal microscopy revealed that CaNeu/ADSCs secreted more M2 macrophages 1 week after SCI (Figure 5(a)). The study also found that CaNeu/ADSC hydrogels promoted nerve and remyelination after 8 weeks (Figure 5(b) and (c)). The study found that extracts from fat (FE) contained a large quantity of brain-derived neurotrophic factors, glial cell-derived neurotrophic factors, and TGF-β. 109 FE maybe also promoted SCI repair when applied to it. Xu et al. developed a hydrogel using hyaluronic acid and methylcellulose (HAMC) to carry the FE. 110 The hydrogel had a porous structure and effectively loaded the FE. In vitro studies confirmed that the hydrogel could release FE rapidly within 16 h. In vitro immunofluorescence staining showed that HAMC-FE reduced inflammatory factors—such as IL-1β, IL-6, and TNF-α—and promoted macrophage polarization to M2 by activating the STAT6/Arg1 signaling pathway (69.6%).

(a) Confocal immunofluorescence microscopy images showing increased presence of M2 macrophages, (M2 macrophages: CD68 and CD163) in the CaNeu/ADSCs group. Scale bar: 20 μm (b) Representative confocal immunofluorescence images of spinal cord sections showing the distribution of TUBB3+ (TUBB3, green) and astrocytes (GFAP, red) in all groups 60 days after injury. Scale bars: 1 mm and 50 μm, respectively. (c) Electron microscopy image showing that ADSC-engrafted CaNeu induces remyelination after SCI. Yellow arrows indicate remyelination. Scale bar: 2 μm. Reproduced by permission of Yuan et al. 106
Conclusions
SCI is a serious condition, usually causing severe consequences leading to quadriplegia, an impaired respiratory system, and restricting the activities of daily living, placing enormous physical, psychological, and financial stress on patients. Although the medical community has developed a variety of methods to treat SCIs—including surgical anastomosis, surgical decompression, drug therapy, local freezing, physical rehabilitation, and enzyme preparations. These treatments only relieve pain but do not repair necrotic nerve cells. Inflammation appears to be beneficial in early SCI. Inflammatory cells phagocytose necrotic tissues and cells. However, the hyperinflammatory environment surrounding an SCI is not conducive to SCI repair. Macrophages are usually paradoxical in SCI. M1 macrophages secrete inflammatory factors—such as IL-β, TNF-α, IL-6, and interferon-γ—at the injury site, which aggravates the SCI. M2 macrophages secrete IL-4, IL-10, IL-13, and neurotrophic factors to inhibit inflammatory responses and neuronal apoptosis. Consequently, modulating the macrophage phenotypes to repair SCIs appears to be a promising therapeutic target. Some drugs or molecules that regulate macrophage polarization have limited effects on inducing macrophage polarization in SCI due to their low targeting properties.
Biomaterials have been widely used in tissue engineering and regenerative medicine due to their bio-histocompatibility and targeting.
At present, nanoparticles, gels / scaffolds—such as chitosan nanoparticles and gelatin—as vehicles to modulate the phenotype of macrophages to repair SCI have been demonstrated in vitro. Nanoparticles can precisely deliver miRNAs, anti-inflammatory drugs and molecules to the periphery of injury, induce macrophage phenotypes, and reduce the inflammatory response at the site of injury. Transplanted gels and scaffolds in situ deliver neurotrophic and growth factors, as well as natural polysaccharides (chitosan and curcumin) to the site of injury, modulating not only macrophage phenotypes but also promoting axon regeneration. The development of dECM scaffolds can provide a similar environment to the surrounding of injury, reduce and reduce immune rejection. The porous structure of the stent is also conducive to the regeneration of damage to the inside of the stent, providing a good foundation for SCI repair. In addition, transplanted gels and scaffolds in situ deliver stem cells to the injured site for anti-inflammatory and pro-repair effects. Stem cells also achieve anti-inflammatory and pro-regenerative properties at the site of injury through paracrine-derived EVs.
Nevertheless, current research into delivery biomaterials for SCI remediation is still in the experimental stage in animals. This may be due to the complex microenvironment changes after SCI, and damage repair cannot be achieved with a single condition change. There are currently no clinical reports of biomaterial-induced SCI repair in patients. Considering the complex microenvironmental changes after SCIs, it will take some time for biomaterials used for SCI repair to be applied clinically. Given the effects of polymer biomaterials, the application of polymer biomaterials in the treatment of SCIs will eventually have good clinical prospects.
Footnotes
Acknowledgements
We would like to express our appreciation to everyone who was involved in the drafting and preparation of the manuscript.
Author contributions
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (Grant Nos. 82071391), and the Achievement Transformation Fund of the First Hospital of Jilin University (Grant No. JDYYZH-2102052).
