Abstract
Current materials used for adipose tissue reconstruction have critical shortcomings such as suboptimal volume retention, donor-site morbidity, and poor biocompatibility. The aim of this study was to examine a controlled delivery system of dexamethasone to generate stable adipose tissue when mixed with disaggregated human fat in an athymic mouse model for 6 months. The hypothesis that the continued release of dexamethasone from polymeric microspheres would enhance both adipogenesis and angiogenesis more significantly when compared to the single-walled microsphere model, resulting in long-term adipose volume retention, was tested. Dexamethasone was encapsulated within single-walled poly(lactic-co-glycolic acid) microspheres (Dex SW MS) and compared to dexamethasone encapsulated in a poly(lactic-co-glycolic acid) core surrounded by a shell of poly-
Introduction
Soft tissue defects, whether due to trauma, tumor resection, or congenital malformations, require extensive tissue repair. Standard care includes free tissue transfer or prosthetic components such as silicone or saline implants. Resection of tumors in the head and neck area, as well as trauma or congenital abnormalities, often result in contour defects from loss of soft tissue, which is largely composed of subcutaneous adipose tissue. 1 With breast cancer being one of the most common malignant conditions in the new era—one in eight women will develop breast cancer in the United States according to the American Society of Plastic Surgeons (ASPS)—there are over 100,000 breast reconstructive surgeries performed per year following a mastectomy. 2
Currently, the most common strategy used to repair soft tissue defects in these cases is by replacing lost volume with synthetic materials. A second major challenge is the deep tissue destruction and discontinuity that is often a result of trauma experienced during war, considerably facial traumas. The Joint Theater Trauma Registry alone showed 26% of all service members injured during battle and evacuated over a 6-year period in Iraq and Afghanistan suffered wounds to the cranio-maxillofacial region. 3 While the reconstruction of bone tissue has been achieved to some degree of precision, soft tissue reconstruction, which is responsible for the contours of the human form, falls short. Prosthetic restorations used as filler materials prove to not only be ineffective for soft tissue repair but also dangerous to the patient because of negative host reactions associated with local edema, lymphadenopathy, and scarring. Non-autologous materials are most often recognized as foreign bodies and are degraded by enzymes and inflammatory cell complexes. Repeated injections are required to maintain volume in even the smallest of defects. Although allergic reactions occur rarely—only in 3%–5% of restorative surgeries—hypersensitivity reactions are frequently observed.4,5 Allografts, also known as homologous tissue grafts, are not ideal due to the potential for viral transmission or immunogenic and allergic reactions to occur.
Autologous fat grafting is another option utilized in reconstructive and augmentative surgery.6,7 Current biomaterials used in restorative tissue surgery possess a number of limitations, including unpredictable outcomes, fibrous capsular contraction, allergic reaction, suboptimal mechanical properties, distortion, migration, and long-term resorption.
8
One promising strategy involves the controlled delivery of adipogenic factors, such as dexamethasone (Dex), within the fat graft.9,10 Our laboratory has been developing novel biomaterials—based on both native matrices and synthetic polymers—for regenerative medicine applications, specifically by encapsulating Dex.9–13 Not only has our laboratory previously shown the potential for Dex to improve adipose tissue retention in vivo, but Dex is also approved by the Food and Drug Administration (FDA) and a widely used corticosteroid clinically.9,10 Poly(lactic-co-glycolic acid) (PLGA) is the most commonly used polymer in tissue engineering because of the biodegradable properties and low toxicity. Similar to PLGA, poly-
Methods and materials
Dexamethasone encapsulation within single-walled microspheres
The encapsulation of dexamethasone within single-walled microspheres (Dex SW MS) was established in our laboratory. 9 PLGA (75:25) (400 mg) was dissolved in dichloromethane (DCM) (4.5 mL). Dexamethasone (20 mg) was dissolved in 0.5 mL of methanol. Following which, the Dex mixture was added to the polymer solution and vortexed until the solution was homogeneous. The solution was added to 600 mg of 2% poly(vinyl alcohol) (PVA) solution and stirred for 2 h at 1200 r/min. The MS were then collected by centrifugation, frozen at −20°C, and freeze dried for 12 h (LabConco Freezone 4.5). Empty single-walled microspheres were fabricated with the same protocol, without adding dexamethasone.
Dexamethasone encapsulation within double-walled microspheres
In order to create double-walled microspheres, 400 mg of PLGA (75:25) was dissolved in 4.5 mL of DCM. In a separate vial, a 10% solution of PLLA of equal polymer mass was prepared in DCM. After both polymers were dissolved, 20 mg of dexamethasone-sodium phosphate was added to the PLGA solution and vortexed until the solution was homogeneous. The PLGA was then added to the PLLA solution and vortexed for an additional 60 s. This solution was then added via pipette to 400 mL of aqueous 1% PVA solution and stirred at 500 r/min for 3 h. The microspheres were collected by means of centrifugation, followed by freeze-drying for a 24-h period (LabConco Freezone 4.5). Empty double-walled microspheres (Empty DW MS) were created following the same protocol as dexamethasone double-walled microspheres (Dex DW MS), omitting the addition of dexamethasone.
Dexamethasone single- and double-walled microsphere characterization
The morphology of both single- and double-walled microspheres was determined using scanning electron microscopy (SEM). Initially, the microspheres were gold coated using a Cressington 108 Auto (Cressington), followed by the usage of a JSM-6335F SEM (JEOL) operated at an acceleration of 3.0 kV for morphology characterization. The size distribution of microspheres was determined by measuring the diameters of 30–50 microspheres using SEM images. Microsphere loading capacity (LC) was determined using the equation LC = DL/P + DL, where DL is the amount of drug loaded and P + DL is the sum of polymer and amount of drug loaded. Encapsulation efficiency (EE) of the microspheres was determined using the formula EE = Ad/Td, where Ad is the actual dose of drug and Td is the theoretical dose of drug captured within the polymer.
Dexamethasone release from microspheres
The release kinetics of dexamethasone from both single- and double-walled microspheres were tested by incubating 10 mg of microspheres in 1 mL of phosphate-buffered saline (PBS) at 37°C. Supernatant was collected weekly for 45 weeks. Each week, the solution was replaced with fresh PBS after being vortexed for 15 s. The amount of dexamethasone released was analyzed using spectrometry. The peak wavelength of Dex was previously recorded as 242 nm (https://www.ncbi.nlm.nih.gov/pubmed/11915294). The data collected was calculated using a standard curve.
In vivo studies
Dexamethasone-loaded microspheres in vivo study design
Animals used in the study were approved by the University of Pittsburgh Institutional Animal Care and Use Committee. A total number of 80 female athymic nude mice (5–10 weeks old; Harlan Laboratories) were used for the study. Athymic nude mice were used due to their immunodeficiency to prevent the rejection of xenogeneic adipose tissue post-injection. Animals were separated into 16 groups, each containing five mice per group. The right side injection was the experimental (sample) group, and the left side consisted of the control group. Single- and double-walled dexamethasone microspheres were tested at two different time points, 6 weeks and 6 months. Two different combinations of dexamethasone microspheres were examined. The dosage of the drug is determined by the mass of the microspheres injected. The highest dose contained 50 mg of dexamethasone microspheres and the lowest dose consisted of 27 mg of dexamethasone microspheres. In addition to experimental groups, two control groups were tested. Control group “A” contained empty microspheres, whereas control group “B” consisted of only lipoaspirate. The doses were chosen based on our results in the preliminary data. 10 The doses correspond to doses currently used in humans that are within the safe and therapeutic range.14,15 Human lipoaspirate was processed as approved by the University of Pittsburgh Institutional Review Board, obtained by patients undergoing elective surgery using the Coleman method.7,10 Adipose tissue was aspirated using a two-holed blunt harvesting cannula in combination with a 10-mL Luer-Lok syringe. The syringe was then centrifuged at 1200g for 3 min. Following the removal of the upper oil and lower blood layers, the lower third of the center lipoaspirate layer was transferred via Luer-Lok connection to 1 mL syringes. Microspheres were then manually integrated into the tissue. Immediately following preparation, processed lipoaspirate was inserted subcutaneously bilaterally into the dorsal flanks of the mouse model.
Animal surgeries
Animals were anesthetized with 3% isoflurane in oxygen at the time of the surgery. The lipoaspirate/MS injections were performed using a 16-gauge surgical cannula, on both sides of the dorsal area of the animal. The right side was injected with the experimental group and the left side contained lipoaspirate exclusively. Photographs were taken immediately following the surgery and weekly throughout the study.
Histological analysis
Following extraction, the samples were fixed in 4% paraformaldehyde, incubated overnight in 30% sucrose (Sigma-Aldrich), and then embedded in paraffin, then cryosectioned at a thickness of 10 μm. Hematoxylin (Santa Cruz Biotechnology, Inc.) and eosin (Sigma-Aldrich) (H&E) staining was conducted using the same protocol as prescribed in previous study. 10 Vascularization of the extracted samples was determined by assessing a pan peroxidase-based CD31 antibody staining to confirm the presence of blood vessels using the same protocol as in the previous study. 10
Image analysis
The Olympus Provis microscope (Olympus) was used to image extracted adipose tissue slides. Adipose tissue architecture was assessed by analyzing the tissue morphology of H&E-stained slides. Blood vessels were counted using ImageJ (NIH). Blood vessel lumens were counted using CD31-stained slides, containing five slides per group. CD31-stained slides were used to evaluate 20 random cell surface areas. Beginning from the center of the slide and counting five cells in four directions, ImageJ was used to set the difference in cell areas between the control and treatment groups. In total, 10 random tissue sections were quantified for each sample and means were determined for the aforementioned vessels and vessel lumens.
Dexamethasone-loaded microspheres systemic effects
Dexamethasone is able to effectively suppress levels of corticosterone as negative feedback from adrenocorticotropic hormones.16–18 The levels of dexamethasone in treatment groups were tested by drawing blood from animals of the 6-week treatment groups. Before sacrifice, blood was collected and preserved in EDTA-coated tubes from four animals in each group under light anesthesia. The plasma was then separated and stored at −80°C until used. Corticosterone levels in the plasma were quantified using an ELISA kit following the manufacturer’s instructions (Enzo Life Sciences).18–21
Statistical analysis
Data were analyzed using SPSS Statistical Software. Paired t-tests, two-sample t-tests, and mixed way analysis of variance (ANOVA) tests were performed to determine if the differences between the experimental and control groups were statistically significant at the p ≤ 0.05 significance level. All results are presented as mean ± standard deviation.
Results
Dexamethasone microspheres characterization
Single- and double-walled microspheres were examined using SEM imaging. Single-walled microspheres—both dexamethasone loaded and empty—are an average of 100 ± 10.5 μm in diameter (Figure 1(a)). Double-walled microspheres are an average of 250 ± 8.5 μm in diameter (Figure 1(b)). Single-walled dexamethasone-loaded microspheres released the drug in 80 days, with an average of 30% ± 5.5% being released in the first week (Figure 2). After 85 days, around 95% ± 3.5% was released in total. Double-walled dexamethasone-loaded microspheres released the drug over an average period of 300 days, with around 10% ± 5.5% released in the first 40 days, resulting in a minimal burst release effect. Around day 300, 90% ± 2.7% of the drug was released. Double-walled dexamethasone loading capacity was found to be LC = 42 ± 3.6 mg Dex/g MS and the encapsulation efficiency was calculated as EE = 43% ± 2.3%.

Microsphere morphology imaged using scanning electron microscopy. (a) Single-walled microspheres—both dexamethasone loaded and empty—are an average of 100 ± 10.5 µm in diameter. (b) Double-walled microspheres are an average of 250 ± 8.5 µm in diameter.
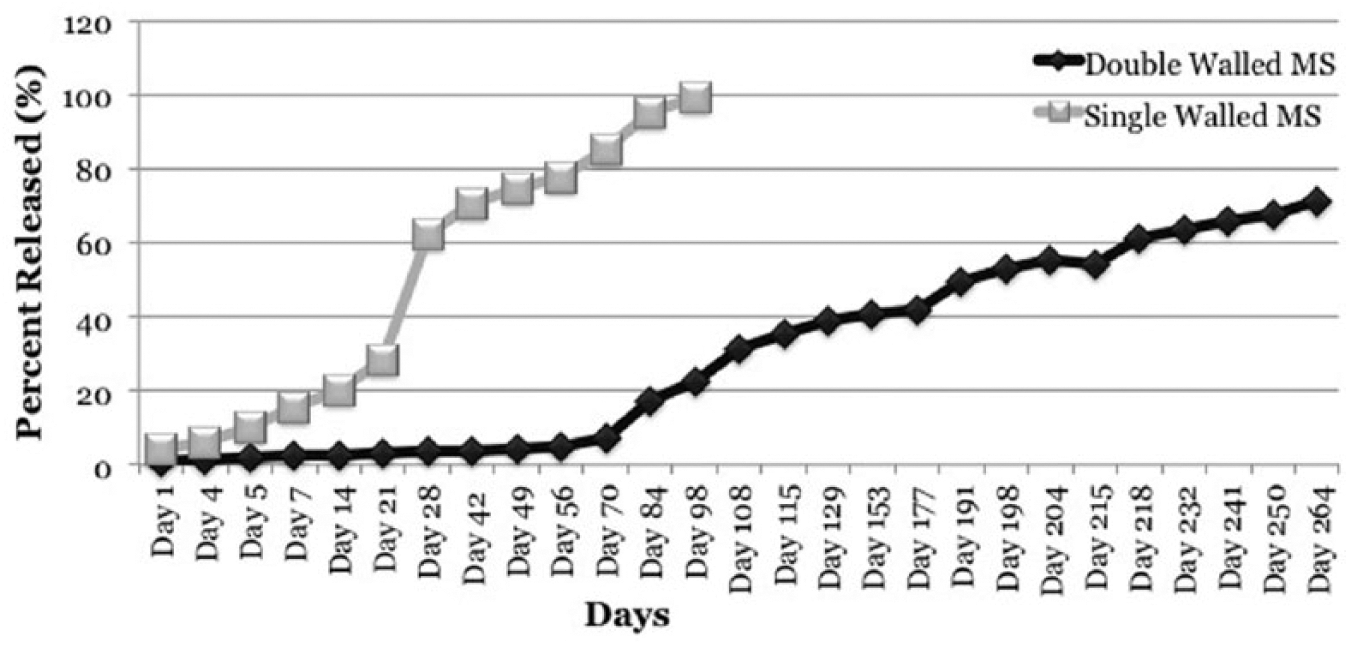
SW and DW MS release kinetics. A burst release of dexamethasone was observed from the SW MS on Day 21. Dexamethasone was continuously released until Day 98, when the drug was no longer detectable by the spectrometer. The release of dexamethasone from the DW MS was significantly slower than that of the SW MS, showing a similar burst of drug on Day 70.
In vivo studies
Animals were sacrificed at two different time points: 6 weeks and 6 months. Dexamethasone-loaded microspheres were mixed with 0.3 mL of lipoaspirate, whereas the control consisted of 0.3 mL of only human lipoaspirate (C = Control without microspheres and S = sample with Dex MS). All animals were also compared with a control group that consisted of empty microspheres injected on one side and lipoaspirate on the other side, as well as with a group that had lipoaspirate injected on both sides of the animal. A significant difference can be seen between the control and the single-walled microsphere treatment groups in the photographs taken at the 6-week time point (Figure 3(a)). On the contrary, no noticeable difference can be seen between the double-walled microsphere treatment group and the control side at the earlier 6 weeks time point (Figure 3(b)). Photographs taken at the 6-month time point, however, show significant differences between the treatment group and the control group in both single-walled (Figure 4(a)) and double-walled dexamethasone microspheres (Figure 4(b)).

Macroscopic images of explanted adipose tissue at 6 weeks. (a) A significant difference was seen between the control and the single-walled microsphere treatment groups in the photographs at the 6-week time point. (b) No noticeable difference was observed between the double-walled microsphere treatment group and the control at the earlier 6-week time point. C: no microsphere control; S: Dex MS group.

Macroscopic images of explanted adipose tissue at 6 months. Significant differences were observed between the treatment groups and the control group in both (a) single-walled and (b) double-walled dexamethasone microspheres. C: no microsphere control; S: Dex MS group.
Macroscopic differences observed within the injections that contained dexamethasone microsphere treatment compared to tissue without Dex were confirmed by measuring mass and volume displacement of the extracted lipoaspirate in all groups. Tissue extracted from the earlier time point (6 weeks) show increase in mass and volume in single-walled dexamethasone microspheres treatment (Figure 5(a) and (b)) (p-value ≤ 0.05), likely due to faster release of the drug from the single-walled microspheres. Adipose tissue from the second time point, 6 months respectfully, show a greater increase in mass measurements and volume displacement in double-walled dexamethasone microspheres treatment groups compared to control groups of empty microspheres and lipoaspirate only as shown in Figure 6(a) and (b) (p-value ≤ 0.05).

(a) Mass and (b) volume measurements of extracted adipose tissue at 6 weeks. Tissue treated with single-walled dexamethasone microspheres extracted at 6 weeks shows an increase in both mass and volume (p-value ≤ 0.05). No significant difference was seen between the 27 and 50 mg groups. Adipose tissue from the 6-month time point showed a larger increase in mass and volume in the double-walled dexamethasone microsphere treatment groups compared to the empty microsphere and lipoaspirate-only control groups (p-value ≤ 0.05).

(a) Mass and (b) volume measurements of extracted adipose tissue at 6 months. The extracted adipose tissue showed a larger increase in mass and volume in the double-walled dexamethasone microsphere treatment groups compared to the empty microsphere and lipoaspirate-only control groups (p-value ≤ 0.05).
Plasma collected from four animals (n = 4) per group of the 6-week time point was tested for the presence of dexamethasone in the systemic blood stream using the Corticosterone ELISA kit and by following the manufacturer’s instructions (Enzo Life Sciences). Corticosterone levels were similar in all groups (≈1600 pg/mL), including the treatment and control groups, showing no significant difference in corticosterone levels on a systemic scale in any of the groups (p-value ≥ 0.05).
Histological analysis
Three tissue sections from each lipoaspirate extraction were stained with H&E in order to analyze gross architecture of the tissue. Blood vessels were analyzed using ImageJ to view CD31-stained slides. The differences in vascularization between treatment and control samples are demonstrated in Figure 7. The greatest number of blood vessel lumens were observed in the 6-month 27 mg DW MS group as 120 ± 12 lumens, whereas the same treatment in the 6-week time point resulted in 60 ± 10 lumens. A similar trend, however much less pronounced, was seen between the 6-month and 6-week 50 mg SW MS groups, which were 70 ± 8 and 60 ± 5 lumens, respectively. All treatment groups had greater blood vessel lumen counts than the 6-month empty MS and lipo only samples, which were 40 ± 10 and 35 ± 10 lumens correspondingly. The difference in mass measurements between all groups was analyzed statistically with SPSS software, showing a statistically significant difference between the treatment group from the 6-month time point, which had results similar to the volume displacement measurements. All treatment groups had significant volume retention, with the 27 mg Dex DW MS group retaining up to 80% of the initially injected lipoaspirate (Figure 6(b)). Measurements from the 6-week time trial showed mass and volume retention of up to 60% for tissue treated with single-walled microspheres compared to the empty microsphere and lipoaspirate groups (Figure (6)).

CD31 images of single-walled microsphere (top row) and double-walled microsphere treatment (bottom row) groups to demonstrate vascularization in the tissue. Images are sections from the center of the tissue. The error bars are representative of 100 µm.
Discussion
According to the ASPS, there were a total of 5,814,998 reconstructive surgeries conducted in 2015 alone. 22 With such a large number of reconstructive surgeries occurring each year, it is critical that an optimal substance be used in tissue repair. Autologous fat, or mature adipose tissue, has been investigated as a key source of material for soft tissue reconstruction. Fat tissue is easily harvested and many patients possess excessive amounts of mature adipose tissue that can be collected without producing significant contour defects. Despite the incredible potential advantages of using adipose tissue in restorative tissue surgery, autologous fat transplantation has demonstrated poor results, with a 40%–60% reduction in graft volume due to resorption.23,24 Such high rates of adipose tissue resorption are theorized to be related to insufficient revascularization and mechanical damage. 25 Recent work has demonstrated that microvascular endothelial cells play a critical role in protecting adipose cells from hypoxia. 26 Moreover, adipose tissue mass is governed by the formation of healthy vasculature. 27 Dexamethasone, a synthetic glucocorticoid, promotes adipogenesis by increasing the gene expression of CCAAT-enhancer-binding protein (C/EBP) and peroxisome proliferator-activated receptor-gamma (PPAR-gamma).9,10,25,26 The sustained delivery of adipogenic drugs, such as dexamethasone, to the healing tissue not only promotes cell differentiation and vascularization but also acts to support long-term retention of the injected tissue. In this study, we demonstrated the ability to encapsulate and deliver dexamethasone within biodegradable double-walled microspheres to the injected adipose tissue for a period of over 6 months. The long-term delivery of an adipogenic drug to the surgery site is critical for tissue repair and retention. According to the release studies, Dex was released steadily from the Dex DW MS for the entirety of the 6-month trial. In both the 6-week and 6-month trials, the injected adipose tissue treated with Dex DW MS resulted in increased overall mass and volume retention when compared to the lipoaspirate and empty MS control groups. In addition, a greater number of blood vessels were observed in the treatment group samples following CD31 and H&E staining, suggesting improved vascularization of the treated tissue.
Scaffolds comprising synthetic materials are another means of supporting tissue regeneration by offering mechanical support. Yuksel et al. 28 successfully described the delivery of adipogenic agents such as insulin and insulin-like growth factor-1 via PLGA/polyethylene glycol microspheres for the purpose of creating de novo fat tissue, which supported adipogenesis for a period of 4 weeks. Adipocyte differentiation is multistep process requiring the sequential activation of several groups of transcription factors, including C/EBP gene family and PPAR-γ.29–31 Dexamethasone as a highly potent synthetic glucocorticoid works through activation of the glucocorticoid receptor, which is a nuclear hormone receptor in the same superfamily as PPAR-γ. Dexamethasone induces C/EBP-δ adipogenic activity and reduces the expression of pref-1, a negative regulator of adipogenesis.31,32
Although these findings are promising, to date, research has shown limited data on creating a proficient system for delivering adipogenic drugs over a prolonged period of time. By encapsulating an adipogenic drug, such as dexamethasone in double-walled PLGA/PLLA microspheres, we have shown that one can achieve slow and sustained drug delivery in a local environment, resulting in a long-term effect of dexamethasone in the implanted fat tissue for over 6 months.
Conclusion
By encapsulating an adipogenic drug, such as dexamethasone within double-walled PLGA/PLLA microspheres, we have demonstrated a slow and sustained drug delivery in a local environment, resulting in a long-term effect. While the sustained and controlled release of dexamethasone from double-walled microspheres significantly increased adipose tissue retention, it also contributed to an increase in blood supply in the injected tissue. The immunohistochemistry staining demonstrated more positive staining in the double-walled microsphere treatment groups compared to the single-walled microspheres, empty microspheres, and lipoaspirate only. Blood drawn from the animals showed an absence of dexamethasone in the blood stream, suggesting that the systemic effects of the dexamethasone double-walled microspheres are minimum.
Encapsulation of dexamethasone in double-walled PLGA/PLLA microspheres with the purpose of releasing the drug at a sustained rate presents an improved drug delivery system for prolonged adipose tissue retention. In this study, it was demonstrated that the sustained and local release of dexamethasone is successfully achieved in grafted adipose tissue.
Footnotes
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Opinions, interpretations, conclusions and recommendations are those of the author and are not necessarily endorsed by the Department of Defense.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Army, Navy, NIH, Air Force, VA and Health Affairs to support the AFIRM II effort, under Award No. W81XWH-14-2-0004. The U.S. Army Medical Research Acquisition Activity, 820 Chandler Street, Fort Detrick MD 21702-5014 is the awarding and administering acquisition office.
