Abstract
Tendon disease and injuries carry significant morbidity worldwide in both athletic and non-athletic populations. It is estimated that tendon injuries account for 30%−50% of all musculoskeletal injuries globally. Current treatments have been inadequate in providing an accelerated process of repair resulting in high relapse rates. Modern concepts in tissue engineering and regenerative medicine have led to increasing interest in the application of cell therapy for the treatment of tendon disease. This review will explore the use of cell therapy, by bringing together up-to-date evidence from in vivo human and animal studies, and discuss the issues surrounding the safety and efficacy of its use in the treatment of tendon disease.
Introduction
Tendon disease and injuries have a huge impact on morbidity and disability of patients, mainly as a result of the prolonged recuperation period. Due to the relative avascular nature of tendons, the regenerative potential is limited. This has significant impacts on the quality and speed of its healing. Hence, the latest research into the treatment of tendon disease and injury puts more emphasis on the application of cell therapy to help overcome these treatment challenges.
Regenerative cell therapy is a subset of tissue engineering. Research data demonstrating the potential therapeutic use of exogenous cells within the discipline of tissue engineering to treat tendon pathology prove to be quite promising. 1 This review will focus on the use of cell therapy and its effectiveness as a form of treatment for tendon disease as well as injury. This would include the use of mesenchymal stem cells (MSCs) and differentiated cells such as tenocyte and fibroblast. 1
Background
Tendons are complex structures made up of tenocytes embedded within a dense extracellular matrix (ECM). Tenocytes synthesizes collagen (mostly type I) and other components of the ECM. 2 The organization of the matrix is key to the function of tendons. Therefore, the tendon’s structural stability via its matrix organization will have significant effect on the functional outcome as a whole.
Tendons connect muscle to bone and enable forces generated by the muscle to transmit to bone causing movement of the skeleton. The collagen fibers in tendon determine its material properties, giving it tensile strength and reducing stress exerted during muscle contraction. 3 Tendons are viscoelastic and can function as a shock absorber and as an elastic energy store. However, because of this function, tendons are constantly exposed to mechanical stresses. Additionally, very high loads as much as 12.5 times the body weight can be placed on tendons during high impact activities such as jumping and weight-lifting.4–7 This places enormous amounts of strain on tendons, especially in regions of repetitive loading, giving rise to an increased risk of injury to these tendon sites.
Tendon disease—defining terminology
Tendon disease (or tendinopathy) is hard to define particularly due to poor understanding of the underlying pathophysiology. The term “tendinosis” is used in histology to describe the pathological features seen in the ECM of the tendon, 8 where there is failure of the matrix to adapt to stresses due to imbalance in synthesis and degeneration of the matrix.9–12 Many clinicians have adopted the term “tendinitis” or “tendonitis” to describe tendon disorders, indicating it is inflammatory in origin. However, with no histological evidence of this, it would be inaccurate to say so. 13 Recent histopathological studies have shown that the presence of degeneration occurs before spontaneous rupture, and the inflammatory phase is not typically seen until rupture occurs.14,15 This indicates that inflammation is not causative but as a result of the injury. Tendon disease or tendinopathy describes a process of degeneration rather than inflammation. Sharma and Maffulli 16 recommended the use of the term “tendinopathy” to accurately describe tendon disease or disorders instead (see Table 1).
Defining tendon disease terminologies 8 .

Source: adapted from Wang et al. 18
Tendon injury
Tendon injuries range from a spectrum of acute tendon rupture to chronic degenerative tendinopathy. Tendons have a lower oxygen consumption compared to other tissues allowing tendons to function for longer periods of time without fatiguing. 19 Studies using laser Doppler have shown compromised blood flow at junctional zones, sites of torsion, friction, and compression. 20 Unsurprisingly, injuries often occur at these sites of strain, and the limited perfusion to these areas would undoubtedly compromise the healing process. As a result, recovery times of tendon damage can take up to 6 months with the first line treatment. 9 The prevalence of tendon injuries is approximately 14.2% in elite athletes 21 and also accounts for about 30%−;50% of musculoskeletal injuries affecting an estimate of 100 million people globally each year. 22 With potentially extended recuperation period, this will undoubtedly have a significant impact on the clinical management and financial implications of tendon injuries.
It is particularly important to understand the etiology of a disease so that patients can be treated correctly. However, the exact causes of such tendon rupture are still relatively unclear. 23 It is convenient to assume that such injuries are based entirely on mechanical damage, but this is not always true. There has been more research into the underlying pathology of tendon injury. Many studies have shown that spontaneous tendon rupture does not just occur acutely due to mechanical insult but over time with signs of an ongoing degeneration process.14,15 Preexisting degeneration has been implicated as a risk factor for acute tendon rupture due to reduced tensile strength caused by repetitive overstraining. 16 Common injury sites are the Achilles and patellar tendons, rotator cuff complex, and finger flexors, demonstrating a level of anatomical influence over injury as well.
Tendon healing
Tendon healing is a complex process influenced by several factors. It is mainly described in distinctive stages: inflammation, proliferation, and remodeling.16,24 The inflammatory stage is characterized by an acute inflammatory response with infiltration of extrinsic cells such as fibroblast, neutrophils, and macrophages. There is also synthesis of chemotactic agents to encourage the process of cellular migration as well as release of bioactive factors to promote angiogenesis. 25 The proliferative phase begins after 4–7 days of inflammatory phase. This is mainly a reparative process where a substantial increase in synthesis of types I and III collagen is observed. The granulation tissue forms a scar-like structure around the wound site and contains the components which make up the ECM. At this point, the immature cells of the ECM would synthesize mainly type III collagen in a haphazard randomized manner. 26 The final remodeling stage commences at 6–8 weeks after injury. It is characterized by a decrease in cellularity and reorganization of the collagen architecture where type III collagen will be replaced with type I collagen. As the scar tissue matures, the collagen units interact in such a way that they lead to greater tendon stiffness and tensile strength. 25
Despite the remodeling, healed tendons never fully restore the ECM structure and material properties to match those of normal intact tendons.16,27 This can be due to a number of reason such as poor vascular supply to tendons which leads to poor scar tissue formation which is then further exacerbated by the inevitable biomechanics burden to healing tendons. All of these factors compromise healing which in turn reduces efficiency of tendon performance. 16 In addition, studies on tendinopathic and ruptured Achilles tendons demonstrated abnormal healing response where the healing tenocytes showed a reduction in type I collagen and a significant increase in type III collagen. 28 This resulted in reduced tensile strength due to fewer number of cross links in type III collagen as compared to type I. This property is thought to be responsible for the biological inferiority of the repaired tissue’s strength and poor healing of tendons. 29
Current treatments for tendon disease
Managing tendon disease is clinically challenging, as the exact pathophysiology of tendon disease is still unclear and treatments for tendon disease are inconsistent and highly variable in many centers. Therefore, most treatment programs do not yield very high success rates in general.
There have been many proposed therapeutic strategies for treatment of tendon disease which can be broadly classified into surgical and nonsurgical. Surgical treatments are more likely to be advocated for acute tendon injury or rupture where damaged tendons are sutured together with or without the use of autografts, allografts, or prosthetic devices to close the rupture site. For chronic tendinopathies, surgery is often only used as the last resort after all other treatment options have been exhausted. Where conservative management is indicated in acute tendon injuries, the affected tendon will be placed in a cast and the repair process will be entirely dependent on the body’s natural healing ability.
Several nonsurgical therapies have also been advocated, such as the use of steroidal and nonsteroid anti-inflammatory medication in short-term treatment of tendinopathies. 30 Active physiotherapy via eccentric exercises has been shown to be effective in supporting rehabilitation.31,32 There are also several commercial treatment modalities in the market such as therapeutic ultrasound, inotophoresis, phonophoresis, and low-level laser. All but ultrasound treatment, which has been shown to have positive effect in treatment of lateral epicondylitis, has no firm evidence to support its widespread use clinically. More research into its therapeutic benefits is required. 30 Other available alternative therapies include topical glyceryl trinitrite, therapeutic hyperthermia, injectables for sclerosing treatment, and extracorporeal shock wave therapy. Some have shown promising effect on treatment of tendinopathy, but similar to the other proposed therapies above, there is currently very little evidence in literature to fully support its application. Going into detail of all these clinical therapies would go beyond the scope of this article. One thing is clear; there is a need for more well-designed randomized control trials to accurately prove its clinical effectiveness. Despite all these available treatment options, injured tendons still never fully restore to its original condition.16,25,27 As a result, current research has concentrated much of its efforts into regenerating tendons to as close to its original structure and function as possible via cell-based therapies.
Cell therapy in tendon disease
Rationale
During the natural healing process of tendons, fibroblastic cells have been observed at the site of repair and are known to play a role in tendon regeneration by synthesizing collagen. 16 It has been widely accepted that these cells arise from undifferentiated MSCs. However, the source of these MSCs, whether they originate from the surrounding tissues or via the blood supply from the bone marrow, still remains controversial. 1 The critical question is whether there are sufficient cells recruited into the repair site in the first instance to generate enough healing potential. This uncertainty has led to research around the application of exogenous cells to facilitate tendon healing. This is the fundamental principle of cell therapy.
Cell therapy and tissue engineering techniques
To date, there are various techniques that fall under the umbrella of cell therapy. These are as follows: 33
Cell replacement therapy where existing cells with or without gene defects or mutations are replaced by healthy donor cells via local infiltration of stem cells to site of injury;
Tissue engineering technique using a three-dimensional scaffold incorporated with cells to replace tendon tissues in vivo;
Use of stem cells as cytokine and growth factor pumps to stimulate growth and repair of tendons.
Literature search
We performed a comprehensive literature for the above subtopics of cell therapy to obtain animal studies pertaining to each field. Our search strategy for available literature made use of MEDLINE via PubMed search engine using the various combinations following terminologies with relevance to the topic sought: “tendon,” “tendon injury,” “tendon repair,” “tendon healing,” “cell therapy,” “stem cell,” “mesenchymal cell,” “tenocyte,” “fibroblast,” “bone marrow–derived,” “adipose-derived cell,” “tendinopathy,” “tendinosis,” “tissue engineering,” “regeneration,” “scaffold” (and use of commercial names), “growth factors” (and specific growth factors), “cytokines,” “gene therapy,” “gene transfer,” and “biological augmentation.” Articles were screen first by title and then by their abstract to see whether they provided relevant information. Reference list from systematic review articles was also used to source out relevant articles not already included in the existing list of articles found via PubMed.
Cell replacement therapy
Cell replacement therapy involves implanting healthy cells into areas of tendon loss or severe degeneration to optimize healing of tendons. The ideal cell type should be easy to isolate and should have the ability to proliferate rapidly in vitro. 34 Once implanted, they should not induce immunological response from the host. Obtaining cells to promote such healing can come from various sources: embryonic stem cells, adult stem cells, and autologous differentiated cells.
Stem cells are known to regenerate and replace damaged and diseased tissues in vivo. There are two types of stem cells that can be used for cell therapy—embryonic and adult stem cells. Due to the difficulty in controlling differentiation, potential tumorigenesis, and ethical controversy surrounding the use of embryonic stem cells, adult stem cells are a preferred source for regenerative purposes. 35 Adult stem cells reside in adult tissues and are relatively easy to isolate. Despite the fact that they have lesser proliferative capacity to that of embryonic stem cells, research has shown that under the right conditions, they are relatively easy to culture and expand.36–41 In vitro and in vivo assays have identified adult MSCs which take multipotency lineages capable of differentiating into specialized tissue cells such as bone, cartilage, muscle, fat, tendon, and ligament.36,38,41,42 Hence, MSCs can be extracted from bone marrow and other sources such as adipose tissue, synovium, muscle, tendon, ligament, periosteum, subcutaneous scalp tissue, circulating peripheral blood, and umbilical cord blood.43,44 Given the abundant source of MSCs in the body combined with its potential regenerative effects, MSCs prove to be a powerful tool for cell therapy in tendon disease.38,39,45,46 This is evident from the large amount of published literature discussing the use of MSCs over other cell types.
Several studies have also demonstrated the successful use of autologous differentiated cells, such as tenocytes and fibroblasts, either directly injected into tendon defects or seeded in constructs for culture and implantation into tendon defects of animal models. Histological and biomechanical assessment demonstrated successfully bridging of gaps in the tendon with the new tendon construct closely resembling that of the native form in terms of structure and tensile strength.47–49 This has promising implications, but there are a number of concerns such as the availability of donor sites for cell harvest, the long in vitro culture expansion times, and donor site morbidity which restrict the practicality of such autologous cell-based techniques. 34
More recently, scientists have discovered another promising cell source in the form of induced pluripotent stem cells (iPSCs). This breakthrough method, first discovered by Takahashi and Yamanaka 50 in 2006, consists of “reprogramming” differentiated cells with genetic modifications in laboratory conditions to become more pluripotent in nature, resembling features similar to embryonic stem cells. This has led to new avenues of cell therapy research and may solve the problem of cell source and abundance in tissue engineering as well as the ethical issues surrounding the use of embryonic stem cells. However, due to problems with tumorigenesis and potential problems with the use of viruses to induce pluripotency, there is still further work required in this field to improve the efficacy and safety issues surrounding the use of iPSCs in the clinical setting. 51
For clinical application of cell therapy in tendon disease, it is important to acknowledge the fundamental differences in the treatment of tendinopathies and acute tendon ruptures. They are essentially two separate physiological and biomechanical events. In acute tendon rupture, there is either partial or complete loss of tissue interface. Due to the inability for tendon to regenerate, the application of use of cell therapy may be more relevant. In tendon ruptures, the use of scaffolds combined with cell therapy is possible but not in tendinopathies unless the degenerated section is surgically excised and replaced.
In animal studies, the use of MSCs has been applied to horse tendon disease and defects with encouraging results.52–58 Tendon injuries are very common in horses, and because of biological similarity of their superficial digital flexor tendon to that of the Achilles tendon in humans, success in use of MSCs repair of tendons in horse can have significant implications on the clinical application in humans.59–61 Table 2 summarizes the results from animal model cell therapy studies which have been published. We performed a literature search to look for animal studies using cell therapy interventions only. Any study that included any other intervention such as scaffold, specific growth factors, or gene therapy was not included here but will be discussed later in this article.
Animal models using cell replacement therapy.

BMMSC: bone marrow mesenchymal stem cells; fdESC: fetal-derived embryonic-like stem cells; BMSC: bone marrow stromal cell; BMMNC: bone marrow mononucleated cell; BMMSC: bone marrow mesenchymal cell; AdMSC: adipose tissue–derived MSC; SDFT: superficial digital flexor tendon; ECM: extracellular matrix; ESCs: embryonic stem cells; hESC: human embryonic stem cells; cBMSC: cultured bone marrow mesenchymal cell; PRP: platelet-rich plasma; TdSC: tendon-derived stem cell; MSC: mesenchymal stem cell.
Scaffolds
A study by Wakitani et al. 72 in 1994 showed that when cells are cultured in a free-floating collagen gel, they grew in a disorganized manner, eventually deforming the gel structure. But when two fixed posts were placed in the collagen gel, cells and collagen fibers aligned along the axis of contraction between the two posts, demonstrating the need for additional mechanical support such as suturing in scaffold constructs.72,73 Therefore, cells need a suitable scaffold to provide mechanical support for three-dimensional growth as well as a vector for implantation. One early in vivo study by Young et al. in 1998 delivered cultured MSCs from rabbit bone marrow in collagen type I gel to large tendon defects in rabbit Achilles tendons. This study demonstrated significant improvement to the biomechanics and structure post injury as compared to controls. 74 Juncosa-Melvin et al.75,76 later showed that the introduction of mechanical stimulation to the collagen scaffolds increased the gene expression of type I and type III collagen which improved the biomechanical properties of the construct in vivo. This demonstrates that tissue engineered and fabricated scaffold combined with the use of stem cell can successfully restore mechanical integrity and biological healing of the torn and ruptured tendon. Hence, using tissue engineered scaffolds to treat tendon injuries can offer significant therapeutic potential.
The fabrication of tendon scaffolds requires the following characteristics in order to yield success in vivo: (1) biocompatible and biodegradable to be able to provide a short-term stability of the cell transplant and allow for integration into host tissue with higher porosity for cell attachment. (2) They should match the mechanical properties of the host tissue and respond to its kinetics. (3) Fibril alignment influencing the architecture of the scaffold is critically important to enable functional transmission of force across the tendon.77–79 Problems with scaffold design that researchers often face with are usually due to any of the above failings. This results in decreased biocompatibility and mismatched degradation kinetics which leads to the inability of scaffolds to fully integrate into host tissue. Where there are differences in biomechanical properties to native tendon, this can cause incongruous forces to be transmitted across the tendon and scaffold, increasing the failure rate of repairs. In addition, due to lack of necessary bioactive properties within the scaffold, this may result in decreased capacity to induce cell adhesion and proliferation. 80
Scaffolds that are off the shelf have inherent advantages over traditional tendon repair/grafts. These are often synthetically made. However, synthetic materials can also carry an increased risk of infection in the long term. 81
Tendon scaffolds are divided into two major categories: natural and synthetic polymers. Synthetic polymers have equivalent biomechanics and physical properties as tendons, but the main drawbacks are graft rejection, inflammation reaction, and cell toxicity.
Synthetic polymers such as poly(lactide-co-glycolide) (PLGA), polyglycolide (PGA), and polylactide (PLA) are Food and Drug Administration (FDA) approved and are widely used in tendon tissue engineering. Advantages of these polymers are the easily engineered and predictable biomechanics they inherently possess as well as the fact that electrospun alignment of these fibers can easily mimic native tendon. Combination of these polymers with bone marrow stromal cells (BMSCs), tenocytes, and growth factors has been shown to significantly improve repaired tendon function in vivo and in vitro.82–85 Moshiri et al. were able to repair large tendon gap of 2 cm in the Achilles tendon in a rabbit model using synthetically derived electrospun collagen. Results show that it restored 50% of its biomechanics and cellular properties within the neotendon by the end of 3 months. 86
Natural polymers have the advantage of being biocompatible and can possess native biological ECM such as collagen. Collagen is a highly conserved protein in the tendon with almost 60% of tendon being made up of it; of which, 95% is type 1 collagen.77,87,88 Collagen itself does not exhibit strong mechanical properties, but in combination with other polymers and/or with MSCs, this can increase its biomechanics as well as provide a suitable environment for stem cell differentiation to the tenocytes within the scaffold matrix.89,90 Sawadkar et al. 91 demonstrated that by sharing suture tension away from tissue engineered collagen graft, biomechanics of the repair to the clinical standard in a tendon defect of 2 cm increased. Chitosan–hyaluronan, alginate–chitosan hybrid polymer, and chitosan scaffolds have also shown significant collagen type I synthesis with cellular affinity and ECM production similar to that of living tendons.92–95 Funakoshi et al. 96 successfully repaired 10-mm rotator cuff defect by chitin nonwoven fabric acellular matrix after 12 weeks infiltration of cells with collagen fibril alignment. Bioengineered tendon graft fabricated from porcine small intestine submucosa (SIS) also demonstrated the ability to repair and strengthen flexor tendon defect by collagen deposition and increase in the transforming growth factor-β (TGF-β) levels in the rabbit model. 97 Another approach to maintain mechanical integrity of the natural polymer is through the use of decellularized tendon scaffolds. This is an attractive technique as it retains the tendon’s normal tissue architecture. Several research groups are now focusing on using decellurized tendon to treat tendon defects.98,99 A recent study used decellularized porcine tendon which was recellularized with human hamstring tendon, and the results showed successful tenocyte and glycosaminoglycan (GAG) recovery. 100 This is a very promising finding which would certainly pave the way for further use of xenogeneic material in humans which in itself would have a distinctive advantage over allogeneic means due to the more abundant supply of tissue available for transplant. However, effective decellularization of tissue itself is a challenging process, and further research into the methodology is still required to refine it for clinical use.
Engineered scaffold can have therapeutic complications, and unfortunately, there is no gold standard for the use in tendon repair. Some researchers propose the use of scaffold-free arrangements or cell-produced constructs as potential alternatives. A study has shown that scaffold-free aligned tendon structure can be produced by using rat Achilles tendon cells to produce neotendon with satisfactory biomechanics properties. 101 In another study, tendon constructs were produced by tendon-derived stem cells which demonstrated the ability to repair rat patellar tendon in window defects at 12 weeks. 66
However, with so many different scaffolds advocated in literature, it is difficult to see which scaffold truly champions over the rest. To assist with this comparison, it would be necessary for future research to conduct larger animal studies in order to produce more statistically significant results for the ideal scaffold to be appreciated. Table 3 summarizes the list of published animal model studies where scaffolds were used in combination with cell therapy.
Animal models using cell therapy and scaffolds.

AdSC: adipose-derived stem cells; PHBHHx: poly(3-hydroxybutyrate-co-3-hydroxyhexanoate); TdSC: tendon-derived stem cell; PGA: polyglycolic acid; MSC: mesenchymal stem cell; BMSC: bone marrow stromal cell; BMMSC: bone marrow–derived mesenchymal stem cell; PLGA: poly(lactide-co-glycolide).
Tissue engineering with cell therapy and growth factors
Cytokines and growth factors are signaling molecules involved in cell migration, proliferation, differentiation, and matrix synthesis. They play a key role in stimulation and regulation of stem cell differentiation, hence influencing the process of wound healing as a whole. 117 During tendon repair, these factors are expressed by cells, such as fibroblast and inflammatory cells, to activate specific cell signaling pathways leading to transcription of regulatory genes. This stimulates the appropriate cells to migrate and proliferate or to synthesize collagen, matrix components, and angiogenesis as part of the reparative phase in tendon repair. 25 The identification of these growth factors was shown in rat and human models which demonstrated the release of growth factors with different phases in tendon healing and showed how these factors interacted with one another.118–120 The key growth factors involved and their role in tendon healing are shown in Figure 1.
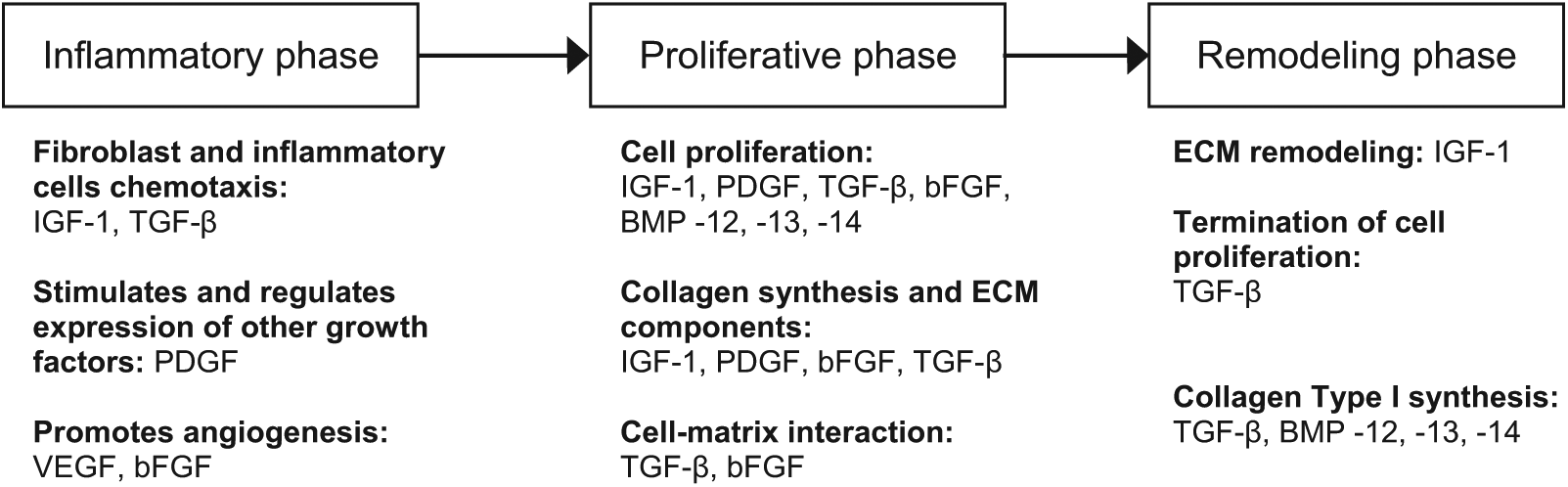
Several studies have taken this concept to the laboratory by determining the biological significance of growth factors in regenerating tendons in vitro and in vivo. Insulin-like growth factor 1 (IGF-1), which is expressed in large amounts during the inflammatory phase of healing, has been shown to stimulate migration and proliferation of many different cells including fibroblast, tenocytes, and inflammatory cells such as neutrophils and macrophages. 122 It can also stimulate collagen production and matrix synthesis. 123 In in vivo studies, IGF-1 has been shown to improve the rate of healing in rat Achilles tendon model. 124 TGF-β has been shown to be active in practically all stages of tendon healing. 125 It exerts a stronger effect on matrix synthesis and collagen production than on cell chemotaxis and proliferation. Klein et al. 126 showed that TGF-β isoforms significantly increased collagen types I and III in cultured rabbit sheath in vitro. Platelet-derived growth factor (PDGF) assists in the synthesis with other growth factors such as IGF-1 in the initial stages of tendon repair. 127 It is also involved in cell proliferation and overall collagen synthesis; an in vitro study demonstrated 300% increase in collagen synthesis with PDGF stimulus. 128 Basic fibroblast growth factor (bFGF) is a potent factor in angiogenesis and cell migration and proliferation. 129 In vitro and in vivo study models show significant effects on wound healing and acceleration of the rate of tendon healing, but this is only evident in the initial stages of healing.130,131 Vascular endothelial growth factor (VEGF) is another crucial angiogenic factor responsible for producing vascularity at the site of repair of tendons. Therefore, it plays an important role in tendon healing. Zhang et al. 132 showed a significant increase in tensile strength of postoperative tendons treated with VEGF in rat Achilles tendons compared to controls. Bone morphogenetic protein (BMP) is known to promote bone formation and can also produce tendon-like connective tissue when delivered to an ectopic area. 25 There are various forms of BMP: BMP-12, -13, and -14 are most commonly found in human tendons. 133 It is also known as growth differentiation factor (GDP)-7, GDP-6, and GDP-5 or cartilage-derived morphogenetic protein (CDMP)-3, CDMP-2, and CDMP-1 in other literatures. 25 In vivo studies have shown that the delivery of BMP can improve tensile strength of transected tendons.134–137
An increasing number of studies have described the use of MSCs for growth factor application via gene transfer techniques. 35 MSCs have been shown to preferentially survive and proliferate in regenerative tissues when injected into the circulation. 40 This property can be exploited by using MSCs as vectors for gene therapy (see Table 4). Transduced MSCs are genetically modified by viral vectors such as adenovirus and retrovirus to house genes that specifically express growth factors known to promote tendon formation. They are injected into the host and are targeted to site of tendon healing. They then release a steady production of growth factors which are known to aid tendon regeneration by promoting cell proliferation and matrix synthesis.126,137,146,147 This has been shown to improve the structural repair of tendinopathy animal models when transgene MSCs expressing IGF-1 were compared to control groups treated with just MSCs alone. 141 In vitro studies have also shown increase in growth factor expression which in some cases have demonstrated increase in abundance of type I collagen synthesis. But this was not always consistent among all the studies as there are variability in the use of different types of growth factors and viral carriers.142–145 The use of tendon fibroblast transfected with IGF-1 via retrovirus in one study showed improved tendon repair in in vivo rat model. 128 In separate study, using adenovirus BMP-13 gene transduced bone marrow mesenchymal stem cells (BMMSC) did not show any improvement in rotator cuff repair rat model. 139 However, it appears that combining growth factors such as PDGF-β and IGF-1 into genetically modified fibroblast held in a synthetic scaffold showed improvement in cell proliferation and collagen production in vitro and accelerate rate of repair in vivo. 128 Therefore, much more research is still required in order to determine the appropriate growth factor combination and timing of growth factor release from transduced cells which will produce the most effective tendon repair model in vivo.
Animal models using cell therapy and growth factors.

PDGF-β: platelet-derived growth factor β; VEGF: vascular endothelial growth factor; TGF-β: transforming growth factor β; IGF-1: insulin-like growth factor-1; bFGF: basic fibroblast growth factor; AAV: adeno-associated viral; BMMSC: bone marrow mesenchymal stem cells; BMP-12: bone morphogenetic protein 12; MT1-MMP: membrane type 1 matrix metalloproteinase.
The same concept can also be applied for replacing cells with genetic defects, but its relevance to tendon disease has not yet been established. Due to its labor intensive process, this technique can only be recommended for treatment of degenerative processes instead of acute tendon injuries.
Current translational research: going into clinical trials
As animal models establish more evidence in the benefits of cell therapy in tendon disease, the number of clinical trials using MSCs is growing. Performing a literature search via PubMed and clinicaltrials.gov using combinations of terms “tendon,” “epicondylitis,” “tennis elbow,” “rotator cuff,” “tendinopathy,” “Achilles tendon,” “patellar tendon” with “stem cell,” “autologous cell,” “cell therapy,” “cell transplantation,” “tenocyte,” “fibroblast” and “humans,” “adult,” “clinical,” “treatment outcome,” and “double-blind method” yielded five published articles demonstrating the use of cell therapy in human trials (see Table 5).
Clinical studies.

PRTEE: patient-rated tennis elbow evaluation; UCLA: University of California-Los Angeles; VISA: Victorian Institute of Sport Assessment; VAS: visual analog scale; QuickDASH: quick disabilities of the arm, shoulder, and hand; BMMC: bone marrow mononuclear cells; MRI: magnetic resonance imaging; USS: ultrasound scan.
Four of the five trials had tendinopathy as the primary pathology of clinical concern. Wang et al. and Connell et al. carried out similar studies with lateral epicondylitis patients where patellar tendon-derived autologous tenocyte and skin-derived fibroblast, respectively, were infiltrated into the affected area. Both studies demonstrated therapeutic benefit from this treatment in the form of a patient scoring system at follow-up. In addition, both studies also showed objective improvement to patient’s imaging appearances on magnetic resonance imaging (MRI) at 12 months for Wang et al. 148 and ultrasound scan (USS) at 6 months for Connell et al. 149 However, with no comparative group, it is hard to measure these improvements statistically.
In the trial by Clarke et al., 46 patients were randomly allocated into two groups where one group received an injection of autologous dermal fibroblast suspended in autologous plasma and the other received autologous plasma only. At 6-month follow-up, the group treated with cell therapy had significantly improved Victorian Institute of Sport Assessment (VISA) scores compared to the plasma group. Only one patient from the treated group developed late tendon rupture requiring surgical intervention. Interestingly, the rare opportunity to perform a histological analysis of a cell-treated tendon post rupture revealed normally appearing tendon cells where degenerative features were expected. 150 This indicates promising finding that the injected fibroblasts were able to differentiate successfully into tenocytes in vivo, producing a better histological repair of an injured tendon at microscopic level compared to the fibrosis and scarring that would have ensued if left to nature’s devices. Obaid et al. carried out a double-blind randomized controlled trial using dermal fibroblast compared to a control group using local anesthetist infiltration and physiotherapy in 32 patients with Achilles tendinopathy. Bilateral and unilateral involvement patient groups were analyzed separately for VISA and visual analog scale scoring at 6-month follow-up. The patients who had unilateral Achilles tendinopathy showed significant improvements in the scores compared to the control group. However, in the patient group with bilateral involvement, there was no significant difference detected. The author postulates that this may be a result of the small sample size in this group. 151
In the trial performed by Ellera Gomes et al., 152 the infiltration of autologous bone marrow mononuclear cells prior surgery for management of operative rotator cuff tears was investigated. This study primarily demonstrated safe use of autologous cells in vivo in humans which may aid healing of tendon postoperatively after rupture. However, due to the lack of a control group, no significant improvement in functional outcome can be concluded from the study.
These five clinical trials were carried out in a nonrandomized or double-blinded fashion due to the relative small number of patients recruited. Overall, results showed good response to treatment from the patient’s perspective and low rupture rates. There were no reported safety issues with the use of autologous cell therapy. However, more research is still required to accurately determine the safety and efficacy of the use of cell therapy in tendon disease especially if stem cells are to be considered.
During the literature search for published clinical trials in clinicaltrials.gov database, we found three ongoing clinical trials which are currently still recruiting patients for application of cell therapy in tendon diseases. This includes a double-blind randomized control trial held in the Netherlands using autologous tenocyte implantation in patients affected by chronic Achilles tendinopathy 153 and a nonrandomized pilot study in South Korea investigating safety and efficacy of the use of allogeneic adipose-derived MSCs in lateral epicondylitis. 154 Another promising clinical trial in Spain involves the use of MSCs embedded in a construct (under the commercial name of OrthADAPT) for rotator cuff repairs which started in May 2010 and was scheduled to conclude in December 2013. 155 The scientific and medical circle will anticipate the future publications of these results as they will no doubt contribute significantly in the quest for successful clinical implementation of cell therapy in tendon injuries.
Discussion
Practical considerations: limitations and issues surrounding the use of cell therapy
There are several factors which affect the success of cell therapy procedures such as good cell–scaffold interaction, presence of cell adhesions on matrix surface, and adequate ECM production. 40 Intact tendons have adequate vascular and nervous supply to maintain function; hence, effective revascularization and innervation in engineered tissues is key to viability and function in vivo. 16
Several animal and clinical trials have highlighted the issue of effective dosage of implanted cells for optimal results in vivo. Timing of cell-based therapy between injury and treatment has been shown to have effect on the outcomes. Modulating the early stages of tendon healing is crucial in the treatment and outcomes of tendon injuries; 156 hence, the timing of implantation is important in the success of treatment. 59 Therefore, more comparative studies will need to be carried out to determine best cell source and treatment regimen.
Translatability of the characteristics of tendinopathy in animal subjects to human is also a potential problem that needs to be addressed. Animal models are not currently validated for defining tendinopathy in humans, which puts into question just how much we can gain from animal research alone. 61 However, as we see increasing number of clinical trials being carried out internationally, the transferability and accuracy of such research will certainly become more evident with time.
Practically, researchers are also faced with the question of the accountability of the injected cells in relation to the regenerative findings in the tendon healing process. There needs to be careful interpretation of results as researchers need to take into account the local and systemic effects of cell therapy on the body. It is important to differentiate this from the potential effects of exogenous stem cells producing signaling molecules which may initiate migration of endogenous cells to site of repair. Therefore, future studies in this field ought to address the issue of tracking and differentiating the effects of the exogenous cells.
The safety of stem cell–based treatment in the clinical setting is also of chief concern for all disciplines working in this field. Several studies have reported cases of ectopic bone formation in vivo;28,114,157 Harris et al. 157 found ectopic bone formation in 28% of MSC-treated rabbit tendons in their study. This demonstrates that control variables are still not perfected, and further research is still required before clinical application with the use of MSCs. The use of stem cell also carries a risk of tumorigenesis which can result in cases of sarcoma and teratoma formation in vivo. Tasso et al. 158 found sarcoma formation with the use of MSCs seeded in bioscaffolds in syngeneic and immunodeficient mice, raising the concern of immunogenic response that cell-based therapy can induce. In terms of safety issues surrounding the culturing of MSCs in vitro, some literature have raised concerns of chromosomal abnormalities159,160 and possible tumorigenicity161,162 that can result in in vitro expansion of MSCs. This triggered a recent review by the Cell Products Working Party and the Committee for Advanced Therapies in the European Medicines Agency in 2013. In response to the need to tackle safety concerns, they organized a meeting with leading experts to discuss the safe use of MSC and to address the risk of inducing chromosomal abnormalities in culturing MSC. They have since published their review in a comprehensive article detailing their current recommendations. 163
In conclusion, there is a need for more well-designed long-term studies with adequate controls and blinding measure and larger sample sizes to help accurately determine the overall effectiveness and safety of cell therapy in tendon disease.
The future
The prospect of widespread clinical use of cell therapy is becoming closer to reality with more studies demonstrating its effectiveness in restoring form and function. However, we are still faced with many challenges. Further research to help identify precise conditions to optimize tendon repair and improve success of cell therapy for tendon disease is crucial. In the future, more researchers will be seen combining cell therapy with other tissue engineering techniques such as the use of growth factors, biomaterials, and gene therapy to help the complete regeneration of tendon tissues in vivo. Combining these bioengineering methods to act synergistically seems to be the most thorough and novel approach to facilitate the goal of returning normal function to a damaged tendon. Using a multidisciplinary scientific approach is likely to be the way forward for the successful implementation of functional tissue engineered tendon repair. 164 Research into these methods is currently in its infancy, and we still have some way to go before such therapies can be safely introduced into clinical trials.
Footnotes
Declaration of conflicting interests
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
