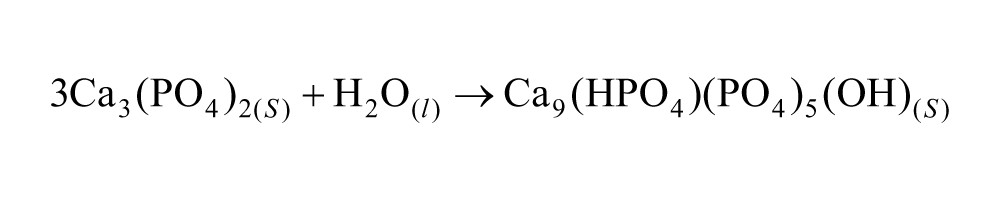Abstract
Developing appropriate cell culturing techniques to populate scaffolds has become a great challenge in tissue engineering. This work describes the use of spinner flask dynamic cell cultures to populate hydroxyapatite microcarriers for bone tissue engineering. The microcarriers were obtained through the emulsion of a self-setting aqueous α-tricalcium phosphate slurry in oil. After setting, hydroxyapatite microcarriers were obtained. The incorporation of gelatin in the liquid phase of the α-tricalcium phosphate slurry allowed obtaining hybrid gelatin/hydroxyapatite-microcarriers. Initial cell attachment on the microcarriers was strongly influenced by the speed of the dynamic culture, achieving higher attachment at low speed (40 r/min) as compared to high speed (80 r/min). Under moderate culture speeds (40 r/min), the number of cells present in the culture as well as the number of microcarrier-containing cells considerably increased after 3 days, particularly in the gelatin-containing microcarriers. At longer culture times in dynamic culture, hydroxyapatite-containing microcarriers formed aggregates containing viable and extracellular matrix proteins, with a significantly higher number of cells compared to static cultures.
Keywords
Introduction
Microcarriers (MCs) are widely used in tissue engineering, as a strategy to make a high surface area available to the cells, allowing high-yield culture of anchorage-dependent cells. 1 In addition, from a steric point of view, their sphericity results in a better filling of irregular-shaped defects with higher degree of packing. The ability of the cells to populate the MCs largely depends on the physico-chemical properties of the MCs, which need to provide the adequate microenvironment for new tissue formation in terms of roughness, surface chemistry, or even the exposure of cell adhesion motifs. 2 Moreover, once the MCs are loaded with cells under the appropriate conditions, they may secret specific extracellular matrix (ECM) molecules, which allow creating a cell–MC construct, with cells homogenously distributed. These constructs can eventually be injected into the defect site, obtaining a fully interconnected porous construct with high numbers of viable cells. 3
Cells can be cultured on MCs under dynamic or static conditions. In general, static conditions have low seeding efficiencies, and a small number of cells can be maintained in culture due to the limited culture medium. 4 Furthermore, static cultures have several other problems such as the presence of concentration gradients and inefficient gas–liquid oxygen transfer. 4 To overcome these limitations, dynamic culture conditions have been widely used for the seeding and proliferation of cells on MCs.5–8 The most common MC culture is based on the spinner flask system, where MCs and cells are gently agitated in high culture medium volumes. This technique has been shown to be very promising for polymeric materials, where the density of the MC is similar to that of the culture medium, 9 allowing for an homogenous suspension of the MC within the culture medium. These conditions enable an improved gas–liquid oxygen transfer, consequently increasing the rates of cell survival and reaching high cell numbers in culture. 10 Nevertheless, polymeric materials have very limited capacity as bone-forming material, lacking osteoconductive as well as osteoinductive properties.
In this sense, bone mimicking biomaterials such as hydroxyapatite (HA) have great potential for bone tissue engineering purposes.11,12 We previously reported the fabrication of microporous HA-MC at low temperature through a water-in-oil emulsion, taking advantage of the setting reaction of a calcium phosphate cement (CPC).13,14 Thus, an aqueous suspension of reactive ceramic particles was emulsified in an hydrophobic liquid, obtaining the microspheres. Moreover, the addition of gelatin in the liquid phase of the ceramic slurry allowed obtaining hybrid gelatin/HA-MC in a single step. 13 It is precisely the ability of the CPC to harden at room temperature that allows the incorporation of biological active molecules, for example, gelatin. This is a key issue that distinguishes these MCs from other types of calcium phosphate MCs/granules previously obtained. Previous works were based on a two-step process, forming first the microsphere, followed by a high-temperature sintering step, making non-viable the incorporation of biological molecules within the structure.15,16
We have previously shown that on the HA-MC and the gelatin/HA-MC under static conditions, cells were able to attach, proliferate, and differentiate properly, although the number of cells was shown to be significantly lower than the control. 13 Therefore, as represented in Figure 1, this work investigates the use of dynamic cell culturing techniques with the microporous HA and gelatin/HA-MC as a strategy to increase the yield of the cell culture, comparing their behavior in dynamic cultures with that of polymer MCs that are used as control. Although the density of the inorganic and hybrid MC is higher than that of the polymer MC, and therefore different behaviors are to be expected in dynamic cultures, our hypothesis is that the continuous agitation of the cell culture medium can increase cell attachment and consequently cell proliferation.

The 2D static culture versus 3D dynamic culture in a spinner flask.
Materials and methods
α-Tricalcium phosphate
The powder phase of the ceramic slurry was composed of α-tricalcium phosphate (α-Ca3(PO4)2, α-TCP), which was obtained from the stoichiometric mixture of calcium hydrogen phosphate (CaHPO4; Sigma–Aldrich C7263) and calcium carbonate (CaCO3; Sigma–Aldrich C4830), sintered at 1400 °C for 15 h (Hobersal CNR-58) followed by quenching. The α-TCP was milled in a Pulverisette 6 planetary ball mill (Fritsch GmbB) following standardized protocols to obtain a specific particle size distribution (median size: 5.8 ± 0.6 µm, Brunauer–Emmett–Teller (BET)-specific surface area: 1.23 ± 0.01 m2/g). A 2 wt% precipitated hydroxyapatite (PHA; Alco ref. 1.02143) was added as a seed.
MC preparation
The HA-MCs were prepared as previously described. 13 Briefly, the powder was mixed with a 10× phosphate buffer solution (PBS) at a liquid-to-powder ratio of 0.8 mL/g. Three milliliters of the paste was added into a beaker containing 300 mL of olive oil and stirred at 900 r/min (Heidolph BDC 2002). Once the setting reaction had concluded, a Ringer’s solution (0.9% sodium chloride (Panreac 121659.1214) aqueous solution) with 0.01% surfactant solution (Triton X-100; Sigma–Aldrich 23,472-9) was added into the beaker. The MCs were then allowed to sediment at the bottom of the flask and the supernatant was discarded. The MCs were cleaned with ethanol and immersed in a Ringer’s solution for 7 days in order to allow the α-TCP hydrolysis to a calcium-deficient HA to be completed, according to the following equation
Composite gelatin/hydroxyapatite MCs (GEL/HA-MC) were obtained by the addition of gelatin (Rousselot ref. LB-B, Bloom 250) in the liquid phase at 5 w/vol%. 13 The gelatin solution was mixed with the ceramic powder at a liquid-to-powder ratio of 1.2 mL/g. The olive oil was heated at 60°C, and the ceramic slurry was added and stirred at 900 r/min. Once the microspheres were formed in the oil, an ice-cold washing solution with the same composition stated before (0.01% surfactant in 0.9% NaCl aqueous solution) was added to the oil in order to extract the MCs and simultaneously decrease the temperature below the gelling point of gelatin. The MCs were cross-linked by immersing them in an aqueous solution of 1-ethyl-3,3(3-dimethylaminipropyl)carbodiimide (EDC; Fluka 39391) (1.15 w/v %) and N-hydroxysuccinimide (Aldrich 13,067-2) (0.03 m) at a molar ratio of 2:1 for 2 h and rinsed afterward three times with a 0.3 m Na2HPO4 solution. The MCs were then immersed in Ringer’s solution for 7 days to allow the complete transformation into HA.
Both MCs were sieved between 100 and 297 µm and sterilized by 30-min ethanol immersion previous to the cell culture experiments and subsequently washed three times with PBS to remove any remnants of ethanol. As a control, dextran microspheres (Cytodex 3, Percell Biolytica AB) were used, coded as CTRL-MC. CTRL-MCs were initially hydrated in PBS for 3 h and then sterilized with autoclave, according to manufacturer’s instructions (GE Healthcare; Instructions 18-1119-79AD).
Cell source
Human osteoblast-like Saos-2 cells were used as cell model. The cells were maintained in McCoy medium (Sigma; M8403) supplemented with 0.75%
Cell culture on the MCs
Dynamic cultures were performed in 125-mL capacity spinner flasks (Corning), which were previously coated with Sigmacote (SL2; Sigma) to prevent cell adhesion on the walls of the flask. As recommended by Cytodex manufacturer, 300 mg of CTRL-MCs was introduced in the spinner flasks. Identical amounts of HA-MCs and GEL/HA-MCs were also used for the dynamic cell culture. Three replicates were performed for each material. All MCs were left in contact with 50 mL of medium for 24 h prior to cell culture. The following day, 107 cells were introduced in the spinner flask containing the MCs, followed by repeating agitation cycles according to previous studies.8,17 For the initial 24 h after cell seeding, the agitation remained on for 2 min and off for 30 min. Two different speeds, 40 and 80 r/min, were tested for the initial 24 h in order to verify the optimum speed for cell attachment on the MCs. Cell viability for the two different speeds was assessed by means of fluorescent microscopy after fluorescein diacetate (FDA) staining.
After the initial 24 h, the medium volume was adjusted to 100 mL, and the repeating cycles consisted of 120 min of no agitation, followed by 2 min of agitation at 40 r/min. The medium was changed every second day, replacing 50 mL of each flask with fresh medium.
In order to assess the efficiency of the dynamic culture method at short and long culture periods, two sets of experiments were performed. In the short-term, initial cell attachment was studied at 6, 24, and 72 h. Cell number was quantified by the lactate dehydrogenase (LDH) assay (described in a subsequent sub-section), and cell morphology was assessed by scanning electron microscopy (SEM) and fluorescence microscopy using FDA staining. The performance of HA-MC, GEL/HA-MC, and CTRL-MC was compared. For the long-term study, the ability of the dynamic culture to sustain cell proliferation and allow for the formation of tissue engineered clusters was assessed at 1, 7, and 14 days in the HA-MC and GEL/HA-MC.
In both short- and long-term studies, static cultures were performed as previously described, 12 and the yield was compared to the dynamic cultures. In order to maintain constant the surface area in the static cultures, 0.039 g of the HA-MC and GEL/HA-MC and 0.024 g of CTRL-MC were placed in 24-well ultra-low attachment plates (Costar 3473) to prevent cell attachment at the bottom of the well. Each well was seeded with 200,000 cells. The volume used in the static cultures was 1 mL, which was replaced for fresh medium every other day. For comparative purposes, the specific values for the dynamic and static cultures are displayed in Table 1.
Comparison between the different parameters of cell culture in static and dynamic cultures.

HA: hydroxyapatite; MC: microcarrier.
The values for the HA-containing MCs and the CTRL-MCs are identical unless otherwise specified.
Cell quantification
The relative cell numbers were evaluated by the LDH assay. At each time point, 1 mL of culture medium containing MCs in suspension was extracted in triplicates from the spinner flask and introduced in a 24-well plate. The MCs were then washed twice with PBS and left with 1 mL of PBS, freezing the plate afterward. The cells were lysed by freeze–thawing the PBS with the cells, following at least two repeating cycles. The lysates were centrifuged at 1500 r/min for 10 min. The LDH activity was determined spectrophotometrically with a commercially available LDH kit (Roche) in a plate reader (Power WaveX, Bio-Tek Instruments; 490 nm). A calibration curve with decreasing concentrations of cells was created to express results in cell number. For comparison purposes, the cell numbers were normalized by the surface of the MCs available.
Cell morphology
In order to visualize the overall morphology of adhering living cells, fluorescence images were obtained after FDA staining (Invitrogen F1303) at different times. One milliliter aliquots were extracted with a pipette to determine cell morphology. The cells were labeled with FDA by adding 10 µL of a 1-mg/mL stock solution of FDA in acetone. Representative pictures of the adhered cells were then taken with a fluorescent microscope (Nikon; Eclipse E600) using the green channel.
Cell morphology was further characterized by means of SEM. For that purpose, the samples were washed in 0.1 m phosphate buffer, pH 7.4, and cells were fixed with a 2.5% glutaraldehyde (Sigma–Aldrich G400-4) solution in PBS, washed, and maintained in 0.1 m phosphate buffer. Osmium tetraoxide (Sigma–Aldrich 201013) was added after fixation and graded ethanol solutions were used to dehydrate the samples (50%, 70%, 90%, 96%, and 100% ethanol). Finally, hexamethyldisilazane (HDMS; Fluka 52620) was used to complete dehydration of the samples, being air-dried afterward.
Statistical analysis
All the experiments were performed in duplicate with three samples per group. Statistical analysis was carried out with significance of 5%. One-way analysis of variance (ANOVA) with Fisher’s post hoc test was conducted. The data were expressed as mean ± standard deviation.
Results
Effect of rotating speed
The morphology and microstructure of the HA-MC and GEL/HA-MC are shown in Figure 2. The sphericity was higher in the gelatin-containing MCs. The surface was porous in both the materials, although the HA-MC showed a rougher surface, with HA crystals clearly visible on the surface.

SEM images of (a, b) HA-MC and (c, d) GEL/HA-MC.
During the dynamic culture at 40 r/min, HA-containing MCs had higher tendency to sediment at the bottom of the flask than CTRL-MC due to their higher skeletal density (~2.85 g/mL for HA-containing MCs as opposed to 1.02 g/mL for CTRL-MCs). In order to avoid sedimentation, the culturing speed was increased to 80 r/min. Nevertheless, cell attachment was reduced when the rotation speed increased, as shown in Figure 3. Moreover, dynamic culture at 80 r/min had a detrimental effect on MC integrity, resulting in multiple breaking of HA-containing MC. Therefore, according to these results, further experiments were performed at 40 r/min.

FDA staining for the GEL/HA-MC in dynamic culture for 24 h at two different speeds: (a, b) 40 r/min and (c, d) 80 r/min. Bar = 200 µm. (a, b) The 40 r/min speed showed higher amount of viable cells on the surface of the MC compared to (c, d) the 80 r/min speed as well as lower MC disintegration, probably arising from collisions.
Short-term cell cultures
Figure 4 shows the fluorescence microscopy images obtained for the cell culture in the different MCs after 6 h, comparing the static and dynamic cultures. Despite the qualitative nature of the images, it appears that both the number of cell-loaded MCs and the number of cells in each MC were higher in the dynamic cultures, in the three different materials. The differences seemed to be more pronounced in the CTRL-MCs, suggesting an increase in cell number and cell-loaded MCs in dynamic culture. After 72 h, cell number tended to increase in the HA-MC and GEL/HA-MC for the dynamic cultures, whereas no differences were observed for the static cultures.

Comparison of FDA staining images for HA-MC, GEL/HA-MC, and CTRL-MC after 6 and 72 h of cell attachment in static and dynamic cultures. The number of MCs with cells present on the surface tends to increase in the dynamic cultures. Bar = 200 µm.
The morphology of the HA-MC and GEL/HA-MC after different culture periods is shown in Figure 5. As time advanced from 6 h (Figure 5(a)) to 24 h (Figure 5(b)) and 72 h (Figure 5(c)), fragments detached from the HA-MC and several MC were broken. GEL/HA-MC (Figure 5(d)–(f)) maintained better integrity.

SEM images for HA-MC at different times: (a) 6 h, (b) 24 h, and (c) 72 h and for GEL/HA-MC (d) 6 h, (e) 24 h, and (f) 72 h. Bar = 100 µm. At low magnifications, it can be observed that as time advances, the HA-MCs tended to disintegrate while the GEL/HA-MCs maintained better integrity.
Figure 6 shows cell proliferation at 6, 24, and 72 h measured by LDH. In the static culture, no statistically significant differences (p > 0.05) were found between HA-MCs and GEL/HA-MCs. Although cells attached, no proliferation was registered in the first 72 h. Cell proliferation was significantly higher on the CTRL-MCs, with statistically significant differences compared to the HA-containing MCs at 24 and 72 h (p < 0.05). A different trend was observed in the dynamic cultures. The HA-MC showed significant increase in cell number, presenting a saturation value at 24 h without any increase at 72 h. This could be due to cell confluence on the surface of the few HA-MCs that were initially loaded with cells. Cell proliferation was higher in GEL/HA-MC, with increasing cell numbers at 24 and 72 h, presenting statistically significant differences with respect to CTRL-MC at 24 h (p > 0.05).

Proliferation after 6, 24, and 72 h with LDH under dynamic and static conditions for HA-MC, GEL/HA-MC, and CTRL.
Long-term cell cultures
One of the objectives of extending the cell cultures was to study the feasibility of using this type of systems as platforms for the development of tissue engineering constructs with homogenously distributed cells and ECM proteins. For this reason, CTRL-MCs were not taken into account and only the HA-containing MCs were compared as possible bone tissue engineering microscaffolds. Figure 7 shows fluorescence images after FDA staining of the HA-MCs and GEL/HA-MCs after 1, 7, and 14 days of culture. At short culture times, the MCs were able to move easily around the spinner flask and were re-suspended when the spinner flask was stirred. However, after 7 days, as evidenced by the FDA images, the MCs tended to agglomerate (Figure 7). This was visually observed by sedimentation of big clusters of MC at the bottom of the flask. Cells proliferated in the interface between the different MCs and tended to aggregate them, making it more difficult the re-suspension upon stirring. This aggregation was also visualized by SEM after 14 days (Figure 7).

Fluorescence images after FDA staining images obtained at 1, 7, and 14 days for the HA-MCs and GEL/HA-MCs. It is shown that cells were able to contact cells on other MCs and aggregate the different MCs. Representative SEM image after 14 days of dynamic culture for the HA-MC. Bar = 200 µm.
Figure 8 shows the cell proliferation measured by LDH in static and dynamic conditions. Low numbers of cells were found for the HA-MC at 1 and 7 days in static culture, increasing considerably after 14 days. A higher cell number at 1 and 7 days was found in the GEL/HA-MCs, but no significant differences between HA-MCs and GEL/HA-MCs were observed after 14 days in static conditions.

Proliferation after 1, 7, and 14 days with LDH under dynamic and static conditions for HA-MCs and GEL/HA-MCs.
In the dynamic cultures, the trend was different. At 1 day, the number of attached cells was significantly higher on the GEL/HA-MC (p < 0.05). After 7 days, the amount of cells was higher in the HA-MCs, whereas after 14 days, the amount of cells was higher for the GEL/HA-MC, the differences being statistically significant (p < 0.05) in both cases. The fact that the cell number did not increase on the HA-MC after 7 days was probably related with the lower number of cell-loaded MCs, together with the higher MC fragility when subjected to the stresses generated during the dynamic culture.
Discussion
The use of MCs under dynamic cell culture conditions is not only used in the field of tissue engineering. It has also been used extensively as an improved methodology to expand cells compared to the conventional two-dimensional (2D) culture flask.18,19 The MCs used as cell expansion substrates are generally made of polymeric materials, which have similar densities to the culture medium, which allows them to be suspended upon stirring. In this work, the dynamic cell culture conditions were applied to MCs containing an inorganic phase, HA, therefore having a higher density, this representing a challenge for their use in spinner flask cultures. However, the benefits associated with the bioactivity and osteoconductivity of HA justify the effort of trying to enhance cell colonization, as they are good candidates as bioactive microscaffolds for bone tissue engineering.
A key issue was to determine the optimum conditions that ensured high cell attachment and proliferation. Previous works have revealed the importance of selecting the proper settings, according to the MC and the cell type used, in order to ensure the highest yield of survival. In this sense, parameters such as the amount of culture medium, 20 the impeller shape and size, 21 the rotating speed,21,22 as well as the ratio between cell–MC 21 have been shown to influence cell viability. In addition, having intermittent rotating culture regimes as opposed to constant rotating regimes has also been shown to enhance cell adhesion in dynamic cultures.1,23,24
Among the different parameters that may have an effect on cell attachment and proliferation, we studied the rotating speed, which was shown to be of great importance for proper initial cell attachment. High speeds (80 r/min) gave rise to a higher re-suspension of the MCs in the culture medium but showed a decrease in both MC integrity and cell viability, and for this reason, the lower speed (40 r/min) was selected. It has been previously documented that speed influences cell adhesion and proliferation on MC.21,22 It was reported that speeds around 40 and 60 r/min gave the highest values of proliferation for Vero cells on Cytodex MC, whereas lower or higher speeds decreased the number of proliferating cells.25,26 This can also be related to the fact that cells can be damaged by strong collisions during agitation at high rotating speeds. 27 We suggest that this effect was even more pronounced in our culture system due to the fragmentation of the MCs at high speeds, which increased the number of particles in culture medium, subsequently increasing the chances of collision with the cells.
Once the speed was determined, dynamic conditions were compared with static ones in the initial cell seeding on the MCs. Significant differences were found between the two systems. In the HA-MCs and GEL/HA-MCs in static cultures, no significant increase in cell number was found in the first 72 h. In contrast, CTRL-MCs presented good proliferation values in static conditions, since these MCs have an optimized surface for anchorage-dependent cells to attach and proliferate. It is in fact known that cell proliferation on low-temperature calcium phosphates is low, due to the surface chemistry and specifically surface roughness.13,14,28–30 Proliferation is usually delayed and the amount of cells on the material is low at short times. For this purpose, polymeric additives such as gelatin are added into CPC not only to enhance their physico-chemical properties but also to enhance their biological properties. 31 Specifically, the presence of gelatin is clearly enhancing cell adhesion and proliferation as compared to the fully inorganic MC, which can be attributed to the availability of cell-attaching moieties contained in gelatin, as the Arginine-Glycine-Aspartic Acid (RGD) sequences. 13 Because of the low ability for cells to attach on these types of surfaces, its seeding efficiency is even more reduced under static conditions on microspheres since cells may fall in the gaps between the monolayer of MCs. 19 This is because once the cells are seeded, the cells that are able to attach on the MCs survive, whereas the ones that sediment on the bottom of the non-adhesive well die. The dynamic culture was able to counteract this limitation, achieving significantly higher values of cell proliferation (p < 0.05). Due to the repeating cycles of agitation and non-agitation, the chances of cells to attach on the MC increased, therefore showing higher cell numbers after 3 days of culture in dynamic cultures, particularly in the gelatin-containing MC.
In the long-term cell culture study, it was observed that even though the system was subjected to periods of gentle agitation, the MC aggregated among them. Clusters of MC were formed after 7 and 14 days with high amounts of cells. This aggregation took place once the cells achieved confluence on the surface of the MC. The degree of aggregation can be controlled by regulating different culture conditions such as mixing intensity,32,33 oxygen tension, 34 and duration of culture 33 among others. The aggregation is actually demonstrating that cells are being able to proliferate and create ECM proteins. This phenomenon has been previously described for chondrocytes seeded on MCs and cultured for less than 2 weeks in a spinner flask culture. Constructs containing MCs, cells, and ECM proteins were formed, which were suitable for injection; these chondrocyte–MC aggregates could be delivered, arthroscopically, into the cartilage defect or used for meniscus reconstruction.33,35
The efficiency of the MC suspension was strongly dependent on their composition. Thus, the CTRL-MCs were easily suspended during the stirring periods, allowing for close interactions between cells and MCs. Once the agitation stopped, both sedimented and remained in static for a period of time, allowing the remaining cells to attach to the MCs. Opposite to this, the HA-containing MCs were only partially suspended during the early stages of the culture. However, since the cells that were not successfully attached to the MCs were re-suspended at each stirring cycle, the chances for them to attach to the MCs increased, compared to the static cultures. For this reason, in spite that the denser HA-MCs and GEL/HA-MCs were only partially suspended during the spinner flask culture, it proved to be an efficient way to populate the HA-containing MCs, due to the increased chances of cell–MC interaction.
Conclusion
Cell attachment and proliferation on the HA-containing MC significantly increased in dynamic spinner flask cultures as compared to static culture conditions. The presence of gelatin in the HA-MC enhanced both cell attachment and proliferation. Three-dimensional (3D) tissue engineering constructs containing MC, cells, and ECM proteins were obtained after 7 days of dynamic culture. Further in vivo implantation studies are needed to prove their osteogenic potential.
Footnotes
Declaration of conflicting interests
The authors declare that there is no conflict of interest.
Funding
This study was supported by the Spanish Government through project MAT2012-38438-003-01. Support for the research of M.P.G. was received through the prize ICREA Academia for excellence in research, funded by the Generalitat de Catalunya.