Abstract
We examined the recently discovered phenomenon of Adaptation-Induced Blindness (AIB), in which highly visible gratings with gradual onset profiles become invisible after exposure to a rapidly flickering grating, even at very high contrasts. Using very similar stimuli to those in the original AIB experiment, we replicated the original effect across multiple contrast levels, with observers at chance in detecting the gradual onset stimuli at all contrasts. Then, using full-contrast target stimuli with either abrupt or gradual onsets, we tested both the orientation tuning and interocular transfer of AIB. If, as the original authors suggested, AIB were a high-level (perhaps parietally mediated) effect resulting from the ‘gating’ of awareness, we would not expect the effects of AIB to be tuned to the adapting orientation, and the effect should transfer interocularly. Instead, we find that AIB (which was present only for the gradual onset target stimuli) is both tightly orientation-tuned and shows absolutely no interocular transfer, consistent with a very early cortical locus.
Neurons at the earliest stages of visual cortical processing respond preferentially to retinal image movement (De Valois, Yund, & Hepler, 1982; Hubel & Wiesel, 1962; Movshon & Newsome, 1996). Not only does retinal motion inform us about the relative speeds and trajectories of objects in our visual environment (including our own bodies), but it can also capture our attention (Cass, Van der Burg, & Alais, 2011), break object camouflage, and can even inform us about the surface properties of objects (Doerschner et al., 2011).
Prolonged exposure to ‘fast’ (∼10 Hz) movement or flicker, however, can temporarily alter visual processing and cause a range of perceptual effects. Movement at a recently adapted retinal location appears slowed (Thompson, 1981; see Hietanen, Crowder, Price, & Ibbotson, 2007 for an electrophysiological analogue of this effect). Under certain conditions, adaptation can even cause illusory reversals in perceived motion direction (Arnold, Pearce, & Marinovic, 2014). Notably, flicker adaptation produces subsequent elevation in detection thresholds, most significantly when adaptor and target stimuli are similarly oriented (Campbell & Kulikowski, 1966; Cass, Johnson, Bex, & Alais, 2012).
In 2010, Motoyoshi and Hayakawa introduced a compelling new illusion, Adaptation-Induced Blindness (AIB). Following prolonged exposure to ∼10 Hz motion, a target grating is then ramped on from zero to full (or near full) contrast, with either a gradual or an abrupt temporal profile. The slope of this onset ramp has a profound effect on subsequent perception: whereas high-contrast patterns with abrupt onsets are clearly visible (although still somewhat affected), gradually presented patterns become temporarily ‘invisible’. Motoyoshi and Hayakawa (2010) attribute this effect to relatively high-level processes, possibly involving parietal brain regions, suggesting that the visual transients associated with abrupt-onset stimuli are necessary to prompt visual awareness of the stimuli. They further suggest that even though the ‘invisible’ stimuli are not available to awareness, they can still cause low-level effects such as the tilt illusion, suggesting that there is some processing of the suppressed stimuli at lower levels of visual processing, such as primary visual cortex (V1). This study is designed to evaluate the relative contribution of low- and high-level visual processes to the AIB phenomenon.
An image presented to the retina generates a cascade of neural activity throughout the cortex, principally via lateral geniculate nucleus (LGN) and V1. LGN neurons are monocular, receiving exclusive input from the ipsilateral eye. They are selective for parameters such as spatial frequency, temporal frequency, as well as retinal location. They are not orientation-selective, however. That is to say, their receptive fields are roughly isotropic in the orientation domain (Alonso, Usrey, & Reid, 2001; Cheong, Tailby, Solomon, & Martin, 2013; Hubel & Wiesel, 1961; 1968; 1962). Consequently, adapting LGN neurons to a Gabor stimulus drifting or flickering in the alpha (8–12 Hz) range—similar to that used by Motoyoshi and Hayakawa (2010)—causes their subsequent responses to be strongly attenuated to an approximately equivalent extent across the driving orientation spectrum (Solomon, Peirce, Dhruv, & Lennie, 2004).
The transmission of signals from LGN to V1 is functionally significant for several reasons. For example, orientation selectivity first emerges in V1 (Ferster & Miller, 2000; Hubel & Wiesel, 1968). While neurons in both areas respond to luminance-defined edges, only V1 contains an abundance of highly orientation-selective cells (Ringach, Shapley, & Hawken, 2002); most LGN cells respond equivalently to all orientations. As observed in LGN, profound adaptation effects are observed in V1 following prolonged exposure to flickering gratings (Boynton & Finney, 2003). Consistent with their orientation tuning, and unlike in LGN, V1 adaptation effects are orientation specific (Anderson & Burr, 1985; Blakemore & Nachmias, 1971; Campbell & Kulikowski, 1966; Snowden, 1991). Therefore, if AIB is mediated at a cortical stage of processing, reports of invisibility following adaptation to rapid flicker should peak at the adapting orientation.
Another functional change between LGN and V1 is the emergence of binocularly driven responses (Hubel & Wiesel, 1962). Whereas LGN neurons are driven monocularly, V1 is the first stage in visual processing to contain neurons which respond (in varying proportions) to both eyes. Because many V1 cells can be driven equally by either eye, many adaptation effects transfer interocularly (Bjorklund & Magnussen, 1981; Blake, Overton, & Lema-Stern, 1981; Gilinsky & Doherty, 1969), although some do not. For example, both Cass et al. (2012) and Baker and Meese (2012) reported that the loss of sensitivity following adaptation to a flickering Gabor of low spatial frequency (∼ < 1.5 cpd) failed to transfer when adapting and testing in different eyes (i.e., interocularly). Intriguingly, this monocular threshold elevation effect was insensitive to orientation differences between adaptor and target patterns (i.e., it was purely isotropic). However, since the earliest levels of V1 are both orientation-selective and monocular, it is possible that some forms of adaptation may show both eye- and orientation-selective effects.
Unlike early visual cortex, higher level parietal areas respond (amongst many other things) to the orientation of visual objects involved in behavioural interaction such as grasping (Murata, Gallese, Luppino, Kaseda, & Sakata, 2000) and to the slant of 3D stereoscopic surfaces (Shikata, Tanaka, Nakamura, Taira, & Sakata, 1996) We know of no physiological evidence, however, that parietal cortex is selective simply to the 2D orientation of the retinal stimulus.
In this article, we will investigate both the orientation tuning and interocular transfer of AIB. Given that AIB and classical threshold elevation result from similar adaptation paradigms (Baker & Meese, 2012; Cass et al., 2012), we might expect to observe a similar pattern of results: (a) An orientation-invariant component which is specific to the adapted eye (i.e., failing to transfer interocularly), and therefore possibly mediated at precortical locus (Solomon et al., 2004); and (b) an orientation-specific component which transfers between the eyes, and therefore likely to be cortical in origin.
By contrast, if AIB were a predominantly ‘high-level’ phenomenon, possibly involving parietal structures and mediated by attentional gating (Motoyoshi & Hayakawa, 2010), in the absence of any physiological evidence suggesting that parietal cortex might be selective to either 2D retinal orientation or eye of origin, we reason that if AIB were mediated within parietal cortex (Motoyoshi & Hayakawa, 2010), we would expect it to be weakly tuned (or completely untuned) for orientation and to exhibit complete—or at least partial—interocular transfer.
Experiment 1: AIB Across Contrast Levels
Experiment 1 aimed to replicate Motoyoshi and Hayakawa’s original (2010) findings. Key to these findings was that the gradually presented target stimuli were not visible even at high contrasts, unlike the abruptly presented stimuli, which are more similar to those used in measuring contrast detection thresholds (where detection typically improves as function of contrast). Thus, it was necessary to measure detection of target Gabors at several levels of target contrast. Using the method of constant stimuli, participants adapted to fast (8 Hz) flicker and in a 4AFC paradigm (a simplified version of Motoyoshi and Hayakawa’s original 8AFC paradigm) indicated the perceived location of a gradually on-ramped target stimulus. Target stimuli were presented at contrasts of 0.06, 0.09, 0.13, 0.18, 0.25, 0.35, 0.5, 0.71 and 1 (full contrast).
Methods
Participants
Six adults with normal or corrected-to-normal vision participated in the experiment. All participants, including three of the authors, were experienced psychophysical observers; three were naïve to the purpose of the experiment.
Stimuli and apparatus
Stimuli were programmed in MATLAB version 2015b using the Psychophysics Toolbox version 3.0.12 (Brainard, 1997; Pelli, 1997). Visual stimuli were presented on a ViewPixx custom LCD monitor with a screen resolution of 1920 × 1080 pixels and a vertical refresh rate of 120 Hz. The monitor was gamma-corrected to ensure linear luminance output and was controlled by a quad core Mac Pro computer. The ViewPixx monitor incorporated a digital-to-analogue converter that provided 12-bit resolution for measurement of low-contrast thresholds. Maximum and minimum luminances were 96.2 and 0.1 cd/m2, and mean luminance was 48.05 cd/m2. Participants sat in a darkened room with their head supported by a chin rest at a distance of 41 cm from the monitor and made responses on a standard keyboard.
The adaptor was a counterphasing grating composed of two superimposed sine-wave gratings drifting leftwards and rightwards at a speed which produced 8 Hz flicker, equal to the speed of the drifting gratings used by Motoyoshi and Hayakawa (2010), and was presented at full contrast. Counterphasing gratings were used as adaptors instead of drifting gratings because drifting gratings produce potentially confounding motion aftereffects (Pantle & Sekuler, 1969), and might reasonably be expected to induce saccades in the direction of the drift, thereby disrupting fixation and adaptation. The spatial frequency and orientation of the adaptor were kept constant at 1.5 c.p.d and 0°, respectively. Each adaptor was enclosed in a circular aperture whose diameter subtended 3.92° of visual angle and was edge-blurred by a cosine ramp that transitioned from minimum to maximum over 0.78° of visual angle.
The target stimulus was a static Gabor (phase was randomized) with equal spatial frequency to the adaptors but variable contrast. The Gaussian spatial envelope of the target had a standard deviation of 0.65° of visual angle and was equal in diameter to the adaptors. The target grating ramped on and off in a temporal Gaussian window of 1,000 ms with a standard deviation of 200 ms, starting 1,000 ms after adaptor offset. This was jittered by a period of 1 to 240 ms to increase temporal uncertainty.
The visual display comprised four virtual display windows. The counterphasing adapting gratings appeared within these windows, such that the centre of each adapting grating was at 5.9° eccentricity, as in Motoyoshi and Hayakawa (2010), and appeared on a grey background held at mean luminance.
Procedure
Participants pressed a key to initiate trials and were presented with four counterphasing adaptors, one appearing in each aperture. Adaptors were displayed for 30 s for the initial two trials, with 10 s “top-ups” for subsequent trials. Following offset of the adaptors, the target appeared in one of the four locations (fully randomized), and a spatial four-alternative forced-choice task required observers to use a standard keyboard to indicate in which aperture it had appeared. Before the sessions, participants were reminded to keep their eyes fixated on the central cross and to avoid blinking as much as possible. Each target contrast was presented 10 times, randomly interleaved, for two blocks, meaning each participant completed 20 trials per target contrast level, a total of 180 trials. Targets were ramped on and off in a temporal Gaussian envelope with a standard deviation of 200 ms.
Participants were instructed to maintain fixation on the central fixation cross and to guess if they failed to observe the target. The fixation cross appeared white during presentation of the adaptors and grey during target onset and offset, before changing back to white to prompt the participant to respond. The next trial began immediately after response. Each of the adaptation blocks comprised 90 trials and took approximately 40 minutes to complete.
Results
Although some subjects showed slightly above-chance performance (see Figure 1(a)), on average, there was no difference in performance across contrast levels (in other words, unlike in conventional contrast adaptation experiments (Ross & Speed, 1991), there was no increase in detection levels at higher contrasts); F(8.40) = 1.18, p = .335; see Figure 1(b). In addition, there was no significant difference from chance performance (25% detection) across all the contrast levels (see Table 1). Thus, we closely replicated the results of Motoyoshi and Hayakawa (2010).
Results from Experiment 1, testing detection of gradually-onset target stimuli after adaptation to fast (8 Hz) flicker at the full range of target contrasts. (a) individual results; (b) mean results across the six participants (error bars show ± 1 standard error). As this was a 4AFC task, chance detection threshold was .25. There was no statistical difference from chance at any of the contrast levels, suggesting full adaptation-induced blindness. t-Tests Comparing Detection Thresholds to Chance (0.25) Across All the Contrast Levels. Note. Student's t-test. For all tests, the hypothesis was that the population mean is different from 0.25 (i.e., a two-tailed hypothesis). Tests are not corrected for multiple comparisons.

Experiment 2: Interocular Transfer of AIB
In Experiment 1, we replicated Motoyoshi and Hayakawa’s basic AIB effect, ensuring that we tested equal numbers of trials at all contrast levels. It is clear from the results that this effect does not resemble classical contrast adaptation, in that the effect is equal across the entire range of target contrasts. Thus, we went on to test whether it differed in other ways from classical contrast adaptation; would it transfer between the eyes, or to target stimuli at different orientations from the adapting stimulus? If, as Motoyoshi and Hayakawa suggest, the effect is a higher level, parietal effect involving attentional gating, it should be occurring at levels beyond binocular combination, and so we should expect to see full interocular transfer and, potentially, little or no orientation tuning.
Methods
Participants
Five adults with normal or corrected-to-normal vision participated in the experiment. Three participants, including two of the authors, were experienced psychophysical observers, whilst the remaining participants were naive to the purposes of the study.
Apparatus and stimuli
Stimuli were programmed in MATLAB version 7.9 using the Psychophysics Toolbox version 3 (Brainard, 1997; Pelli, 1997). Visual stimuli were presented on a Sony Triniton CPD-G500 22-in. CRT monitor with a screen resolution of 1024 × 768 pixels and a vertical refresh rate of 100 Hz. The monitor was gamma-corrected to ensure linear luminance output and was controlled by a quad core Mac Pro computer connected to a Cambridge Research Systems Bits++ digital-to-analogue converter that provided 14-bit resolution for measurement of low-contrast thresholds. Maximum and minimum luminances were 67.3 and 0.26 cd/m2, and mean luminance was 33.8 cd/m2. Participants sat in a darkened room with their head supported by a chin rest at a distance of 57 cm from the monitor and made responses on a standard keyboard. Adapting and target stimuli were as described earlier, except that there were now separate displays for each eye (see Figure 2). Stimuli for this experiment were viewed through a mirror stereoscope, with adaptors and targets presented either to the same or to different eyes.
A schematic illustration of the procedure for Experiments 2 and 3. Observers first adjusted the stereoscope using the fixation squares, which then disappeared. They adapted to four counterphasing high-contrast gratings in one eye and were then tested either in the same or in the other eye, with gradual-onset or abrupt-onset target gratings. Because images were viewed through a mirror stereoscope, images on the left were seen by the left eye and images on the right by the right eye. Target gratings were always at full contrast, and the number of incorrect judgments was recorded. Responses were always 4AFC, with participants choosing which of four locations contained the target grating.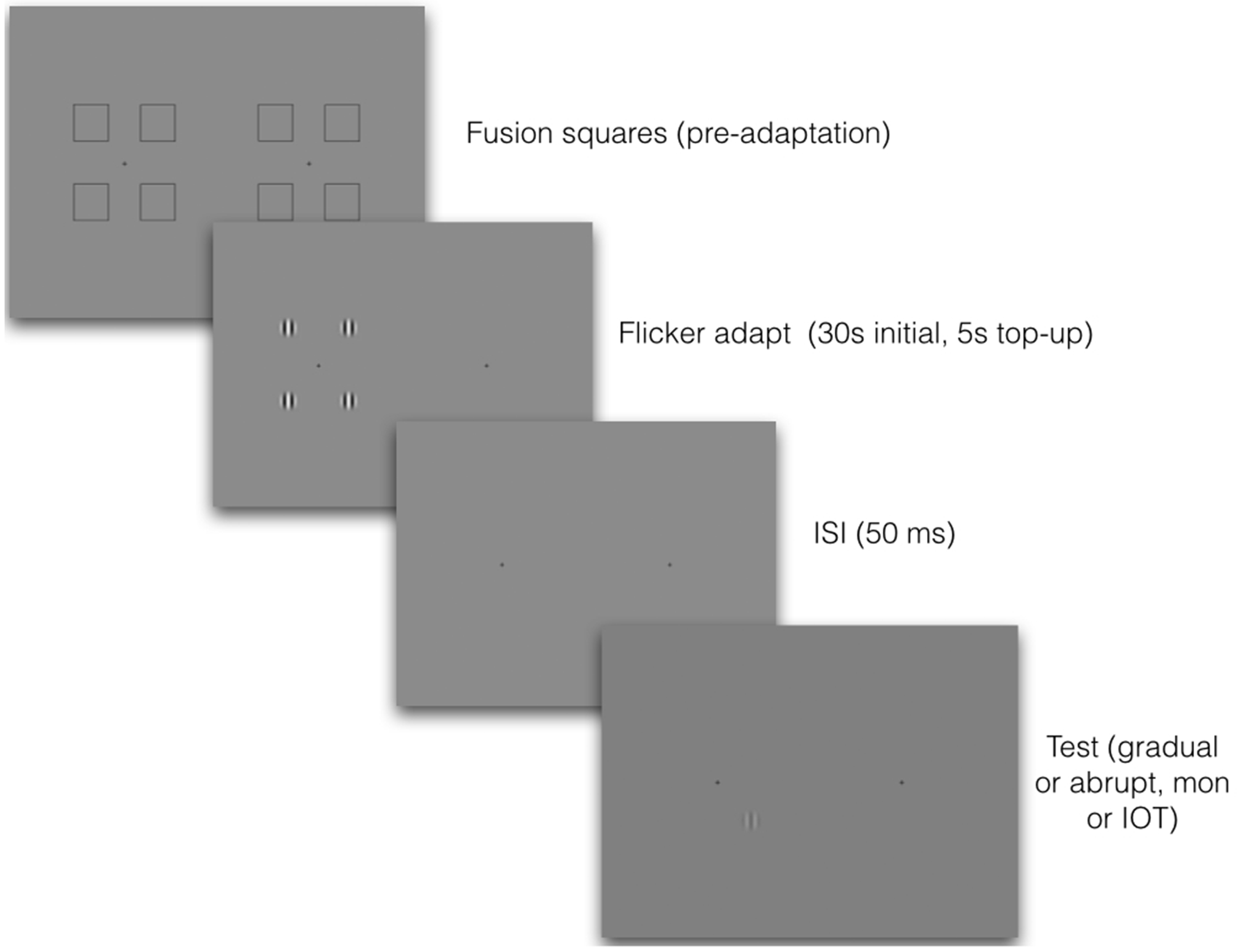
Each adaptor appeared in a circular aperture whose diameter subtended 2° of visual angle and was edge-blurred by a cosine ramp that transitioned from minimum to maximum over 0.78° of visual angle.
The target stimulus was a static Gabor with equal spatial frequency to the adaptors but had variable orientation (0° or 90°) and temporal onset. Target stimuli were equal in diameter to the adaptors at 2° of visual angle. As in Experiment 1, gradually onset target stimuli were ramped on by a temporal Gaussian with a standard deviation of 200 ms and peak amplitude at 1,000 ms, while abruptly onset targets were presented within a rectangular temporal window of 300 ms duration.
To ensure that participants had sufficient stereoacuity to support interocular transfer (Mitchell & Ware, 1974; Movshon, Chambers, & Blakemore, 1972), an Optec 2500 Vision Tester capable of testing down to 20 seconds of arc was used to test participants’ depth perception. Published thresholds for normal stereoacuity range from 40 to 60 seconds of arc (Adams et al., 2008; Romano, Romano, & Puklin, 1975). A mid-range cut-off of 50 seconds of arc was chosen because substantial interocular transfer is still observed for this level of ability (Mitchell & Ware, 1974). All participants had better stereoacuity than the cut-off (M = 29, SD = 12.8).
To maximise the potential for interocular transfer, participants’ ocular dominance was determined behaviourally by a standard finger pointing measure (Coren & Kaplan, 1973). Each participant pointed to a coin glued to a wall and observed how far their finger appeared to deviate when viewing with their left eye closed compared with when their right eye was closed. The open eye that caused the least deviation was judged to be dominant. Using this method, three participants were identified as right-eye dominant and two as left-eye dominant. The adaptor was always presented to the dominant eye. Several studies have shown that this method of testing produces significantly greater magnitudes of interocular transfer compared with when the non-dominant eye is adapted (Howarth, Vorobyov, & Sengpiel, 2009; Mitchell & Ware, 1974; Mohn & Van Hof Van Duin, 1983; Movshon & Blakemore, 1973).
Procedure
Observers viewed all stimuli through a mirror stereoscope with a total reflection path of 57 cm. Prior to the adaptation period, each eye was presented with four fusion-locking squares of 4.5° of visual angle surrounding a central fixation cross, presented on a grey background of mean luminance (33.8 cd/m2). After each trial was initiated, the fusion squares disappeared and the adaptation stimuli appeared in the centre of the location of each square, which was 5.9° from the central fixation cross. See Figure 2 for a schematic illustration.
Participants adapted to counterphasing stimuli presented to either the left or right eye. In monocular conditions, the adapting stimulus was presented to the nondominant eye. To maximise the potential for interocular transfer to occur, participants had their dominant eye adapted in the interocular conditions. This meant that the target was always presented to the nondominant eye, regardless of ocular condition.
Participants pressed a key to initiate trials and were presented with four counterphasing adaptor gratings, one appearing in each aperture. Adaptors were displayed for 30 s for the initial two trials, with 5 s ‘top-ups’ for subsequent trials (Pilot testing with 60 s initial adaptation and 10 s top-ups indicated that the shorter adaptation times produced identical results). Following offset of the adaptors, the target appeared in one of the four apertures (randomized), and a spatial four-alternative forced-choice task required observers to use a standard keyboard to indicate in which aperture the target appeared. Conditions were blocked by target orientation, temporal onset of the target (gradual vs. abrupt) and tested eye (monocular vs. interocular transfer [IOT] conditions). The order of completion was counterbalanced to ameliorate practice effects.
Results
In this experiment, we presented all the target stimuli in the 4AFC paradigm at full contrast, either ramped up gradually or with abrupt onset, and measured the proportion of times the observers failed to correctly identify the location of the target grating. Participants’ subjective reports indicated that in many instances, the gradual onset targets were simply not seen, even though they were ramped up to full contrast. This is shown clearly in the data (see Figure 3); target disappearances are strongly orientation-specific, but interestingly, there was absolutely no interocular transfer of the effect. In addition, abruptly presented targets never disappeared (perhaps unsurprisingly, since they were at full contrast).
Percentage of incorrect responses for full contrast target stimuli after adaptation to counterphasing gratings, for targets which were either 0° or 90° of relative orientation from the adapting stimuli, with either gradually onset (a) or abrupt (b) targets, tested either in the same eye as the adapting stimulus (dark grey) or in the other eye (light grey). Shaded areas ∼ 1 standard error, lines show standard deviations, and individual grey dots are individual data points. Solid lines show the means and dotted lines the medians. All five subjects completed all conditions.
In this experiment, there were significant main effects of onset type (gradual vs. abrupt), F(1, 4) = 58.82, p = .002, eye tested (monocular vs. IOT), F(1,4) = 35.36, p = .002, and relative orientation (0° vs. 90°), F(1,4) = 50.69, p = .002. There were also significant two-way interactions between onset type and eye tested, F(1,4) = 35.36, p = .004, onset type and relative orientation, F(1,4) = 68.48, p = .001, and eye tested and relative orientation, F(1,4) = 32.83, p = .005. In addition, there was a significant three-way interaction between onset type, eye tested and relative orientation, F(1,4) = 32.83, p = .005. In summary, the only case in which adapting to the flickering pattern caused disappearance of the target was in the monocular condition where the gradually presented target was parallel to the adaptor, as can be clearly seen from Figure 3.
Experiment 3: Orientation Tuning of AIB
In this experiment, we aimed to more closely examine the orientation tuning of the AIB effect seen in Figure 3, where the gradual targets show a strong adaptation effect when parallel to the adaptor (at 0°), but no adaptation at 90°. Thus, we explored the space between these orientations, since orientation tuning of the effect was not reported in the original article.
Methods
Participants, apparatus and stimuli were the same as in Experiment 2, with the difference that only gradual target stimuli were tested. The procedure differed only in that target stimuli were presented at orientations of 0°, 2.5°, 5°, 7.5°, 10°, 15° and 90° from vertical. This was tested in both monocular and IOT conditions, counterbalanced between participants. A control experiment also tested detection of the targets without the adapting stimuli.
Results
Here, it is clear that the effect is tightly orientation tuned (see Figure 4), and is well fitted by a Gaussian function with a standard deviation of 7.76° and an amplitude of 37.4%.
The orientation tuning of the percentage of error rates at full contrast. Red data points show percent of errors for stimuli presented in the same eye as the adapting stimuli; blue points show those presented in the unadapted eye, and green show the control stimuli presented without adaptors. The solid line shows a Gaussian fit to the data, fitted in ProFit Version 6.2.11, using a Leverburg-Marquardt algorithm; the fit was given two free parameters (amplitude and bandwidth), with a fixed mean of 0 and baseline of 0. Standard deviation of the fit was 7.76°, with an amplitude of 37.4%.
The fitted amplitude of the function (37.4%) is very close to the approximate percentage of target disappearances at 0°, on average, across participants, as seen in Experiment 2. We note that this is also very close to the reported percentage of incorrect responses (34%) in the original AIB paper (Motoyoshi & Hayakawa, 2010). It should be noted that the actual percentage of targets not seen in our experiment (and in Motoyoshi’s, in fact) would probably be higher, given that the 4AFC paradigm would result in a 25% guess rate, on average. It is important to note here that instances of disappearance were almost completely absent at full contrast, regardless of the relative orientation of adaptor and target. The implications of this will be covered in the Discussion section.
Discussion
The first demonstration of AIB at the 2008 Vision Sciences Society met with astonishment. The audience was shown two high-contrast Gabor patches: one with an abrupt temporal onset profile and the other a more gradual profile. Unsurprisingly, both were trivially easy to detect. A flickering Gabor stimulus followed, and the audience was instructed to fixate upon it for about 10 seconds. This adapting stimulus was then replaced with the high-contrast targets. To the audience’s audible surprise, only one patch was visible—the abrupt one. The gradual patch was completely invisible, apparently suppressed from awareness. Why had it disappeared? Was it a form of threshold elevation? What neural mechanisms might be responsible? To help understand this phenomenon, we replicated this basic effect across a range of very specific conditions. Experiment 1, showed that AIB effects are indeed profound, with gradual target detection performance plummeting to chance levels at all contrasts tested (0.06–1.0 contrast).
Experiments 2 and 3 were designed to measure AIB’s selectivity for orientation and its interocular transfer. Our results show very clearly that the AIB effect is tightly selective for orientation. Disappearances were common when the adaptor and target Gabors were similarly oriented, with performance improving monotonically out to approximately 20° of relative orientation, at which point performance reached ceiling (100% accuracy). That AIB should prove so specific in the orientation domain implies that it is mediated by a population of neurons with a similarly tightly tuned orientation preference. Neurons with such receptive fields are common in several early visual cortical areas, including areas V1, V2 and V3 (Boynton & Finney, 2003; Hubel & Wiesel, 1968). Exactly how early in cortical processing are the neurons which mediate AIB? Experiments 2 and 3 addressed this with an interocular transfer manipulation. While disappearances were common when adapting and testing in the same eye, adapting a single eye to flicker and presenting the target to the other eye afforded nearly perfect performance. Such a high degree of eye specificity shows that AIB is purely monocular.
From a classical view of adaptation, our finding that AIB is both orientation-tuned and purely monocular is surprising. It is now well established that threshold elevation, which occurs as a consequence of flicker adaptation, is composed of a binocular and a monocular component (Cass et al., 2012; Meese & Baker, 2011). The binocular component is orientation-tuned whilst the purely monocular component is untuned. That AIB should be both tightly orientation tuned and purely monocular distinguishes it, therefore, from classical adaptation-induced threshold elevation. It also suggests that AIB involves a subset of early cortical neurons whose receptive fields are both orientation specific and purely monocular. fMRI evidence indicates that both orientation-specific adaptation and eye of origin information is evident in early visual cortical areas, V1, V2 and V3 (Boynton & Finney, 2003; Schwarzkopf, Schindler, & Rees, 2010), although purely monocular neurons tend to be found only in V1.
One study investigating the precise relationship between neural orientation and eye preference in primate V1 (Bartfeld & Grinvald, 1992) found that orientation-selective cells are in fact located within ocular dominance columns. Given that ocular dominance columns contain neurons which are both orientation tuned and ocularly specific in their response, it seems reasonable to speculate that AIB may therefore be mediated by these cells, located close to the centre of a single ocular dominance column, such as those found in V1 (Bartfeld & Grinvald, 1992).
This conjecture that AIB occurs relatively early in the visual cortical process is supported by two visual crowding studies showing that crowding can be largely extinguished if the flanking stimuli which produce the crowding effect are suppressed from awareness using AIB (Shin & Tjan, in press; Wallis & Bex, 2011). According to Shin and Tjan (in press), this implies that AIB is likely to occur at a level of neural processing preceding visual crowding. fMRI studies indicate that crowding effects can be differentiated as early as V1 (Anderson, Dakin, Schwarzkopf, Rees, & Greenwood, 2012). If AIB does in fact precede crowding, this would point to AIB being mediated at a very early stage of visual processing.
Motoyoshi and Hayakawa’s finding that an inducing tilted surround grating rendered invisible by AIB can still induce a tilt illusion (2010) implies that the neural process(es) underlying AIB are unlikely to precede those which cause the tilt illusion—an interpretation which is difficult to resolve if AIB occurs at a very early cortical stage. An alternative interpretation, not inconsistent with an early cortical locus for AIB, is that the mechanisms responsible for AIB and the tilt illusion operate independently. Future studies are necessary to examine this hypothesis.
Motoyoshi and Hayakawa’s (2010) AIB effect is undoubtedly a striking and curious phenomenon that remains to be fully understood. In first reporting the phenomenon, the authors’ original conjecture was that AIB occurred relatively late in visual processing. The evidence presented here, however, from our orientation and interocular transfer experiments and complemented by related work on AIB and crowding, converge on the conclusion that AIB is very likely to be mediated at a very early cortical stage of visual processing.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: David Alais was funded by ARC Discovery Project DP0878371, John Cass was funded by ARC Discovery Project DP0774697, and Deborah Apthorp was funded by NHMRC Early Career Fellowship APP1054726.
Author Biographies
