Abstract
Hydrogen is a promising renewable energy source due to its versatility and low environmental impact. However, its high flammability, fast flame speeds, high diffusivity, and explosion risks necessitate a comprehensive safety review, which is highly demanded in the literature. This article systematically examines hydrogen explosion incidents, highlighting the need for robust safety measures. Key cases such as the Norway ammonia plant incident and the Gangneung disaster emphasise the importance of understanding hydrogen combustion modes and ignition conditions. The explosion behaviour of hydrogen is discussed in detail. The review categorises hydrogen storage and discusses its advancements and safety challenges, with a primary focus on gaseous storage. Advanced storage technologies, material selection, and strategies to prevent explosions and hydrogen embrittlement are explored. Hazard prediction methods, such as numerical modelling techniques are discussed. Additionally, international safety standards for hydrogen storage are reviewed. The study identifies key challenges and emphasises the need for further research to enhance storage safety.
Introduction
As dependence on fossil fuels has increased, it has caused to various problems, notably environmental pollution, global warming, and future energy scarcity. As global energy demands continue to increase, there is also an urgent need to reduce greenhouse gas (GHG) emissions. This requires an immediate shift toward sustainable, low-carbon energy solutions. Central to this change is decarbonisation, accomplished by adopting clean, renewable energy sources such as wind, solar, tidal, wave, biomass, and other emerging alternatives. This approach is crucial in tackling the significant challenges of climate change and moving toward a more sustainable energy future (Ahn et al., 2018). Hydrogen is identified as a promising energy alternative because of its ability to act as an energy source and energy carrier, zero-emission combustion, capability for long-term storage, ability to be produced from other renewables, transportability and versatility in applications (Abdin et al., 2020; Farias et al., 2022). Therefore, hydrogen is becoming one of the most promising energy alternatives and is gaining more attention for its potential as a clean energy source (Li et al., 2008).
Hydrogen has several promising qualities. It can be used as fuel, an energy storage medium, and a clean and versatile energy carrier. It also does not face Carnot efficiency limits, produces minimal emissions, and has the potential to meet diverse energy needs (Ishaq et al., 2022). Specifically, the possibility of integrating carbon capture and storage (CCS) in hydrogen energy storage (HES) plays an essential role in the transition towards decarbonisation (Wu et al., 2022). The demand for hydrogen further increases due to innovative approaches such as power-to-gas technologies, which use intermittent renewable energy sources to produce hydrogen via electrolysis (Ishaq et al., 2022). In addition, hydrogen has the rare ability to reverse its state from water back to the gaseous state, while hydrocarbons are destroyed (Momirlan and Veziroglu, 2005). Moreover, hydrogen fuel cell vehicles are gaining attention because of zero GHG emissions (only water as a by-product), low storage space requirement, and short refuelling time. These advantages have pushed automotive makers like Toyota and BMW to invest heavily in hydrogen technologies (IEA, 2021). In addition, the war in Ukraine has indirectly boosted the development of a sustainable hydrogen energy carrier due to the rise in oil prices (IEA, 2022).
According to the International Energy Agency (IEA), the annual production of hydrogen is projected to exceed 30 million metric tonnes by 2030 (IEA, 2022). Therefore, it is essential to align the development of hydrogen infrastructure with this target. This alignment involves categorising the infrastructure into five main areas: hydrogen production, storage, transportation, distribution, and utilisation. Each category encompasses the relevant structural facilities and equipment necessary for the effective management and deployment of hydrogen resources (Li et al., 2008; Vorontsov and Smirniotis, 2023). To meet the rising demand for hydrogen energy, the design and development of safe hydrogen infrastructure is becoming an increasingly important topic. Before the public can accept hydrogen technologies, it must be shown that hydrogen is as safe, reliable, and convenient to use as fossil fuels. Thus, a well-developed hydrogen infrastructure is essential for the broad utilisation of hydrogen, allowing it to be stored, transported and distributed to end users.
As a highly flammable gas, hydrogen can burn over a wide concentration range in air, with a flammability limit of 4.0 % to 75.0 mol % of hydrogen (Cheikhravat et al., 2012). Its low ignition temperature (Astbury and Hawksworth, 2007), small ignition energy (Najjar, 2013), wide explosion limit (Voevodsky and Soloukhin, 1965), fast combustion speed (Duan et al., 2022), and colourless and odourless nature (Mouli-Castillo et al., 2021) make hydrogen a hazardous substance. In addition, hydrogen has invisible flames (making it hard to detect) and can cause two modes of explosions, namely deflagration (weak) and detonation (strong). Moreover, hydrogen combustion can quickly transition from deflagration to detonation (DDT) (Ng and Lee, 2008; Sánchez and Williams, 2014). Also, hydrogen combustion has a high laminar burning velocity (Makarov et al., 2009), and can lead to boiling liquid expanding vapour explosion (BLEVE) [36]. Furthermore, being the lightest gas, its buoyancy can cause it to accumulate in confined spaces, potentially leading to vapour cloud explosions even in the open air (Rigas and Sklavounos, 2005). Considering two main gas explosion modes, the subsonic flame speed corresponds to deflagration (under weak ignition sources) while the supersonic flame speed represents detonation (under strong ignition sources) in gas explosions (Schmidtchen, 2009; Winterbone and Turan, 2015). However, when an adequate amount of obstacles and turbulence is present, lean hydrogen mixtures can transition from deflagration combustion to detonation, where the consequences can be catastrophic (Xiao et al., 2018). The transition from deflagration to hydrogen detonation is understood to some extent and has been studied in confined spaces like tunnels. However, this transition is less understood in unconfined explosions and larger confined spaces, such as enclosures. The exact conditions that cause the transition in these larger spaces, where hydrogen leaks might occur, are not yet fully known (Kotchourko, 2022; Luebcke et al., 1995). This can be critical in hydrogen storage facilities where leakage, formation of combustible gas, and ignition can lead to catastrophic damage under either explosion mode. This makes the utilisation of hydrogen more challenging and demanding, where appropriate safety design guidelines are required in the hydrogen storage infrastructure design.
Hydrogen storage is crucial for the entire hydrogen infrastructure because it plays a role in every part of the hydrogen supply chain, including production, storage, transportation, and utilisation. Existing standards cover some aspects of the design, installation, handling, storage, and transportation of hydrogen. Specifically, gaseous hydrogen plays a crucial role in existing hydrogen-based applications, being the most widely used storage method due to its economic, technological, and practical advantages (Usman, 2022). Despite successful facilities such as the Port Kembla hydrogen plant, no accepted industry standard exists to design storage facilities, especially for gaseous hydrogen, against accidental hydrogen explosions. Current standards for hydrogen storage and handling are inadequate, often referencing natural gas standards. For instance, the ISO/TR 15,916 report (Limited, 2021), adopted by Standards Australia, outlines safety concerns but lacks guidance on explosion hazard characterisation and design infrastructure to mitigate accidental hydrogen explosion consequences. Therefore, there is clearly a lack of specific standards and guidelines for characterising explosion hazards and designing safe storage infrastructure to prevent potentially deadly explosions (Pique et al., 2017; Tools, 2023). The European Commission and countries like the USA, China and Australia are already developing plans to invest billions of dollars in the development of the hydrogen industry (COAG, 2019; EC, 2023; Institute, 2023). Thus, there is an urgent need to implement safe hydrogen storage infrastructure and understand it by characterising its explosion properties under various conditions.
Recent literature reviews on hydrogen safety can be found in the works by (Abohamzeh et al., 2021b), Najjar (Hu et al., 2023; Li et al., 2022a; Najjar, 2013; Wei et al., 2022) where they address various aspects of hydrogen utilisation, safety challenges, storage and transportation issues, and bibliometric analyses of hydrogen safety research. However, there is a lack of discussion on the explosion characteristics of hydrogen, design standards, current and future storage infrastructure designs, and associated future challenges. Therefore, this manuscript aims to encapsulate the explosion characteristics of hydrogen, the research efforts made in these areas over the past few decades, showcasing the progress and notable achievements in mitigating hydrogen explosion risks and improving storage infrastructure, along with discussing future directions and challenges. First, this paper comprehensively discusses past incidents involving hydrogen energy. Next, this provides a detailed discussion of hydrogen’s characteristics compared to chemical explosives. Then, this review explores current infrastructure designs and existing standards for hydrogen storage. This also offers insights into future designs incorporating advanced materials and strategies to mitigate potential hazards, such as explosions. Collectively, this review provides valuable insights, ranging from the historical context to the future prospects of hydrogen storage infrastructure.
Past incidents of hydrogen explosions
The Hydrogen Incident Reporting Database (HIRD), French Database Analysis, Research, and Information on Accidents (ARIA), EU’s Major Accident Reporting System (eMARS), the Institution of Chemical Engineers (IChemE) UK accident database, and the Japanese Relational Information System for Chemical Accidents Database (RISCAD) offer comprehensive insights into hazardous incidents related to hydrogen (Mirza et al., 2011; Wen et al., 2022). The HIAD database offers a global overview of 700+ hydrogen incidents, compiled from publications, technical news, and other sources (Wen et al., 2022). Those incidents can be classified based on their applications, including events in hydrogen production, storage, transportation, distribution, fuelling stations, chemical/petrochemical industries, commercial uses, laboratory, and R&D facilities (Wen et al., 2022). Focusing on global hydrogen incidents reported up to 2025, nearly half of them were related to explosions, while 31% attributed to fires and only 7.2% were categorised as near misses, as depicted in Figure 1 (a). In addition, 75% of all hydrogen-related incidents directly occurred in hydrogen systems, while the remaining incidents occurred in other systems that indirectly involved hydrogen. In terms of injuries and fatalities, 83% were a result of hydrogen explosion incidents (Cristina Galassi et al., 2012). Consequently, preventing hydrogen explosions is crucial for establishing hydrogen as a safe energy source. Notably, the highest number of incidents occurred between 2000 and 2010, most likely due to increased adoption of hydrogen technologies but inadequate experience in safe hydrogen utilisation, as shown in Figure 1 (b). (a) Nature of consequences of past incidents (b) Number of hydrogen-related incidents (HIAD, 2025).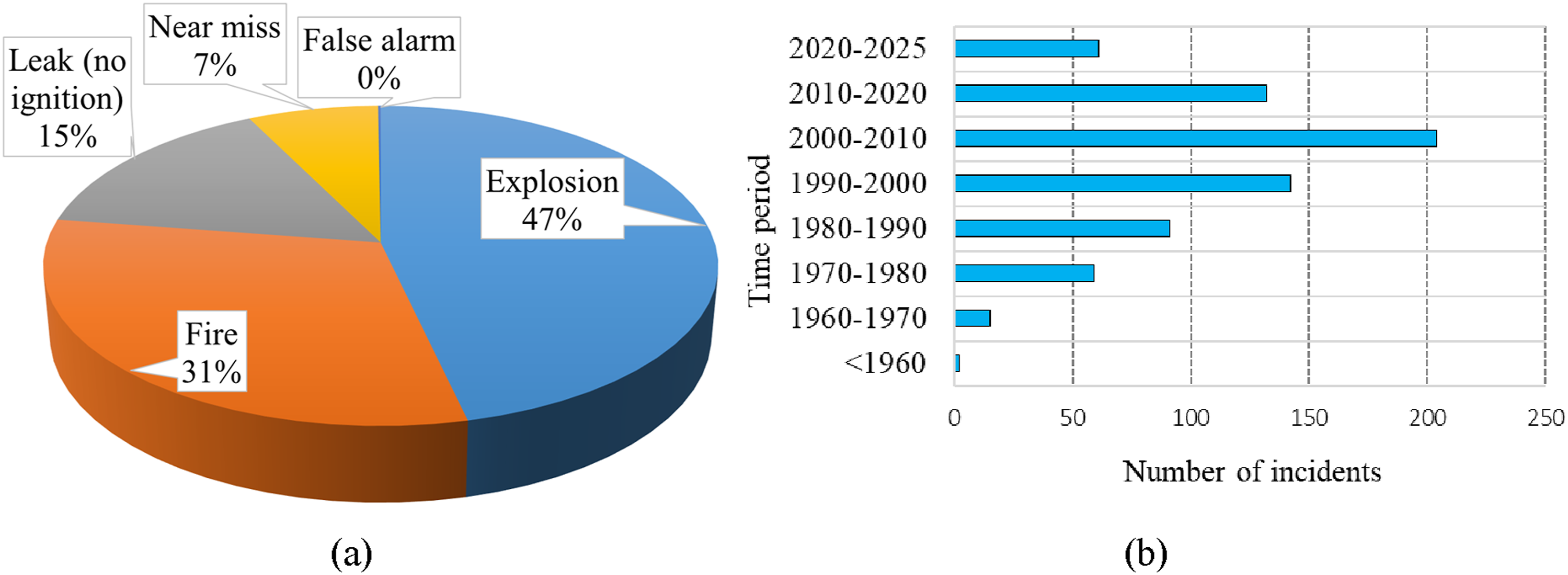
A graphical representation of key hydrogen-related incidents is presented in Figure 2. Also, Table 1 presents a summary of key hydrogen incidents in the past. It aims to give a comparative overview of the scale of a hydrogen explosion incident. Several major hydrogen storage incidents highlight the risks of hydrogen storage systems worldwide. In 2007, a rupture disk failure in Muskingum, Ohio, USA (Figure 2 (c)) caused hydrogen leakage and ignition, resulting in one death, 10 injuries, and extensive damage (WHA, 2022). A hydrogen tank explosion occurred in 2019 in Gangneung, South Korea (Figure 2 (a)), causing two deaths, six injuries, and severe damage, triggered by a static spark igniting oxygen-contaminated gas due to equipment failures (Viktoria Bohacikova, 2024). Recently, in 2023, a storage tank explosion during testing in Lebring, Austria (Figure 2(b)) caused major infrastructure disruptions and a large evacuation zone, with shockwaves felt 3 km away, due to an unknown ignition source (FCW, 2023). These events emphasise the urgent need for strict safety design, maintenance, and monitoring in hydrogen storage. In addition, as illustrated in Table 1 and Figure 2(a), a major hydrogen-air gas explosion occurred at an ammonia plant in Norway in 1985 due to a failed water pump, where a blown gasket allowed hydrogen to leak into a confined building space (Mjaavatten and Bjerketvedt, 2005). The pump, supplying water at 30 bar to an outdoor wash tower for CO and CO2 removal, failed, causing backflow and hydrogen release. An estimated 10-20 kg of hydrogen was accumulated and was likely ignited by a hot bearing. The resulting deflagration escalated to detonation due to confinement and pressure buildup, causing massive structural damage, glass breakage up to 700 m away, and two fatalities. The root causes were a combination of operational mistakes, technical failures, and poor design. Table 1 and Figure 2 also present cases of hydrogen-related explosions caused by indirect sources, such as unexpected chemical reactions, which can pose greater risks. These hydrogen-air combustion incidents can be violently ignited, resulting in detonation, as observed in the Fukushima incident and the ammonia plant incident in Norway. On the other hand, the gas cloud may catch fire through deflagration, as occurred in the Kjørbo station incident. The magnitude of the damage clearly depends on the mode of combustion as observed from the incidents. Examining past hydrogen-related explosion incidents not only highlights the catastrophic consequences of inadequate safety measures but also emphasises the urgent need for a deep understanding of hydrogen’s combustion behaviour and ignition conditions, which is thoroughly discussed in the next section. Representative images highlighting the severity of key hydrogen explosion incidents. (a) Hydrogen storage tank explosion-South Korea (2019) (Viktoria Bohacikova, 2024) (b) Massive hydrogen tank explosion-Austria (2023) (FCW, 2023) (c) Power Plant Incident-US (2007) (WHA, 2022) (d) Hydrogen Fuelling Station Incident-Norway (2019) (2019) (e) Air Product Transit Facility Incident-US (2019) (Panel, 2021) (f) Hydrogen Carrying Truck Incident-US (2023) (Central, 2023) (g) Ammonia Plant Incident-Norway (1985) (Mjaavatten and Bjerketvedt, 2005) (h) Tosco Avon Refinery incident-US (1997) (Mirza et al., 2011) (i) BP Refinery-US (2005) (Johnson, 2006 ) (j) Callide C Coal Power Station Incident-Australia (2022) (Ludlow, 2021) (k) Chemical Plant Incident-France (1988) (IMPEL, 2011) (l) Fukushima Daiichi Nuclear Power Plant Incident-Japan (2011) (Pacchioli, 2013). Notable past hydrogen releases and explosion events.

The explosion behaviour of hydrogen
Gaseous hydrogen systems are associated with hazards including high-pressure release, jet fire, flash fire, fireball, vapour cloud explosion, deflagration, detonation, as well as long-term material issues like hydrogen embrittlement (Rigas and Sklavounos, 2005; Wei et al., 2022; Yang et al., 2021). These hazards often arise from the formation of combustible hydrogen-air clouds, which can result from leaks in gaseous or liquid hydrogen storage systems due to equipment failures such as pipe leaks, valve malfunctions, gasket failures, or rupture of storage tanks (Hu et al., 2025a; Rigas and Sklavounos, 2005). Such mixtures can explode in different modes depending on factors such as the fuel-oxidiser ratio, ignition strength, initial temperature and pressure, confinement, uniformity, obstacles, and gas purity (Liberman, 2010; Ng et al., 2007; Xia et al., 2023; Zhang et al., 2016).
Explosive substances can generally be grouped as illustrated in Figure 3. Substances that contain both fuel and oxidiser together in one form are called explosives and can be divided into high explosives and low explosives. High explosives can be further categorised into primary and secondary. Primary explosives are highly sensitive compounds that can be detonated by heat, impact, friction, flame or electric spark. They detonate directly and produce a shock that can be used to initiate less sensitive secondary explosives such as TNT (Matyáš and Pachman, 2013). High explosives typically exhibit direct detonation while low explosives undergo deflagration (Macias, 2009). Under certain conditions, other combustible substances can also act as explosives. For instance, Hydrogen is non-explosive by itself and becomes explosive when it mixes with air or oxygen to form a combustible mixture. A hydrogen-air mixture must have the right fuel-to-air ratio to ignite (Ramamurthi, 2021). Hydrogen is a unique substance that has a flammability range between 4%-75% (v/v) and 4%-94% (v/v) with air and oxygen, respectively (Ring et al., 2008). Combustion reactions start in such a mixture when the minimum ignition energy is supplied at a sufficient rate by an ignition kernel, or when the mixture reaches autoignition conditions. To initiate combustion in a hydrogen-air mixture, the minimum ignition energy (MIE) required is 0.018 mJ (Yang et al., 2021). This energy must generate a hot ignition kernel larger than the quenching distance to sustain combustion (Ramamurthi, 2021). In addition, ignition energy content must be deposited at a sufficiently high rate. Considering the ignition energy, volume of the ignition kernel, and duration of ignition energy release that govern the ignition process, a parameter known as power density can be introduced to represent the strength of the ignition source. It is described in equation (1), where Classification of explosive substances.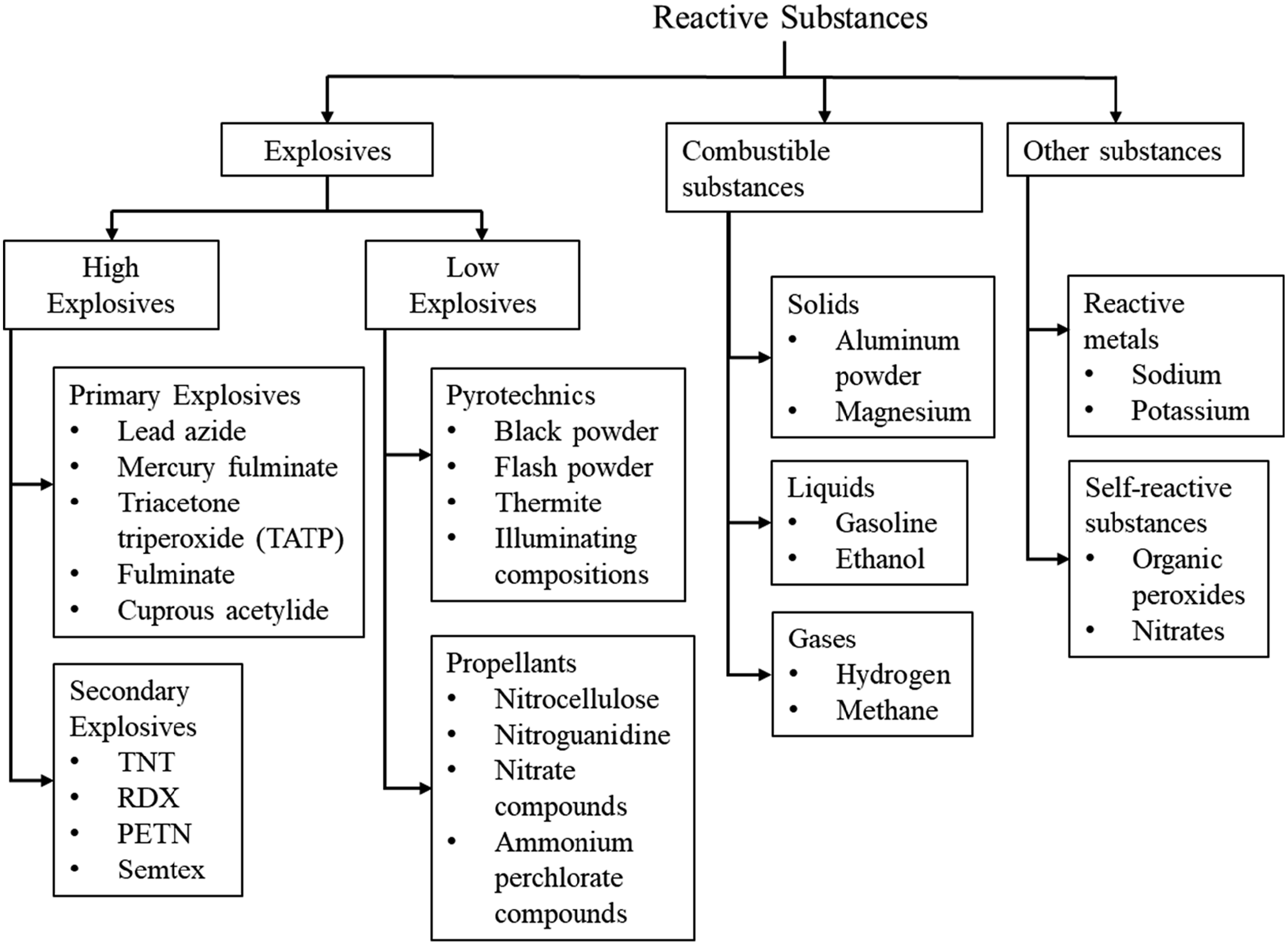
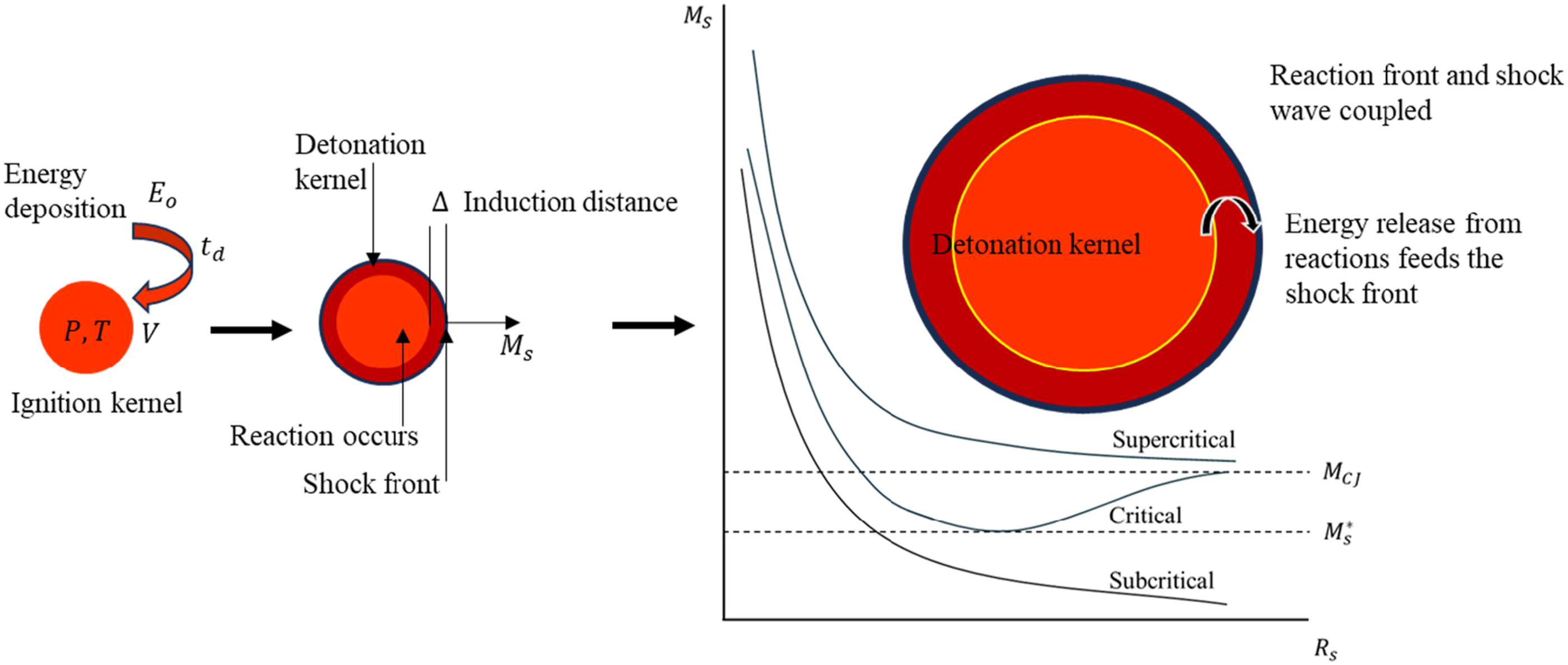
Detonation requires significantly more energy than ignition and occurs only within specific concentration ranges, known as detonation limits, which are narrower than the flammability limits. For instance, hydrogen–air mixtures typically detonate within a concentration range of about 18% to 60% hydrogen, though large-scale detonations have been reported with concentrations as low as 11% (Borisov and Loban’, 1977). Detonation is a complex and energetic process compared to deflagration. The igniter must create a volume of strongly shocked gases (a detonation kernel) that releases enough chemical energy to sustain the shock wave. The detonation kernel requires much higher energy density and should allow the shock wave to sustain through the fuel-air mixture, reaching a steady Chapman-Jouguet (CJ) velocity as described in Figure 4. It represents the deposition of ignition energy ( Formation of detonation kernel and three regimes of detonation initiation.
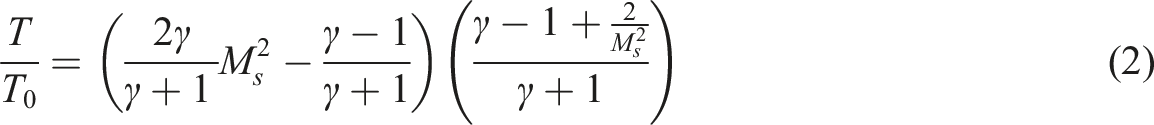
Basic set of hydrogen chain reactions.

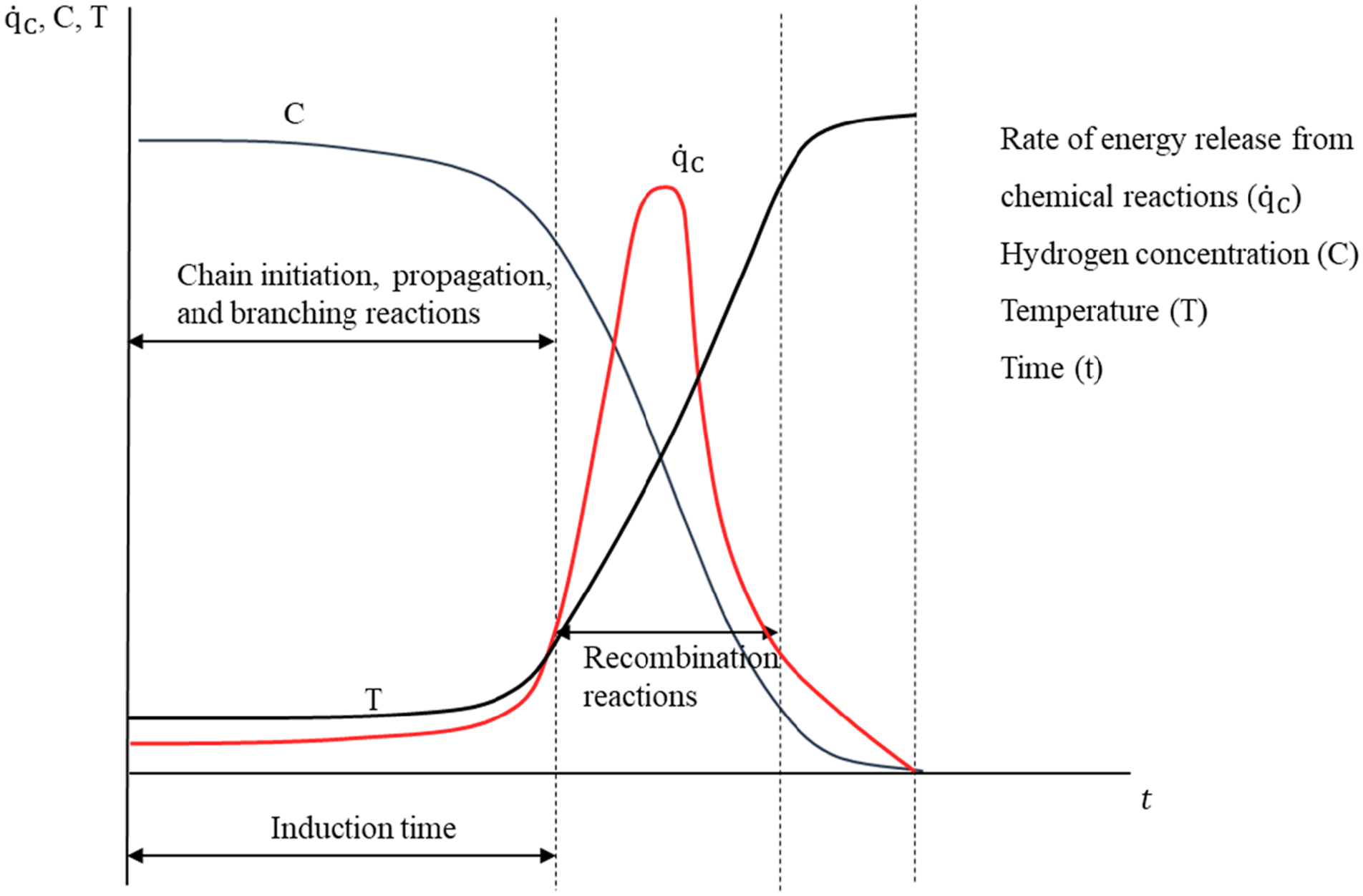
Process of hydrogen mixture reaction.
Detonation can occur not only through direct initiation but also indirectly via a process known as Deflagration-to-Detonation Transition (DDT). This process as illustrated in Figure 6 involves key parameters such as the ignition energy deposited ( Process of deflagration-to-detonation transition (DDT).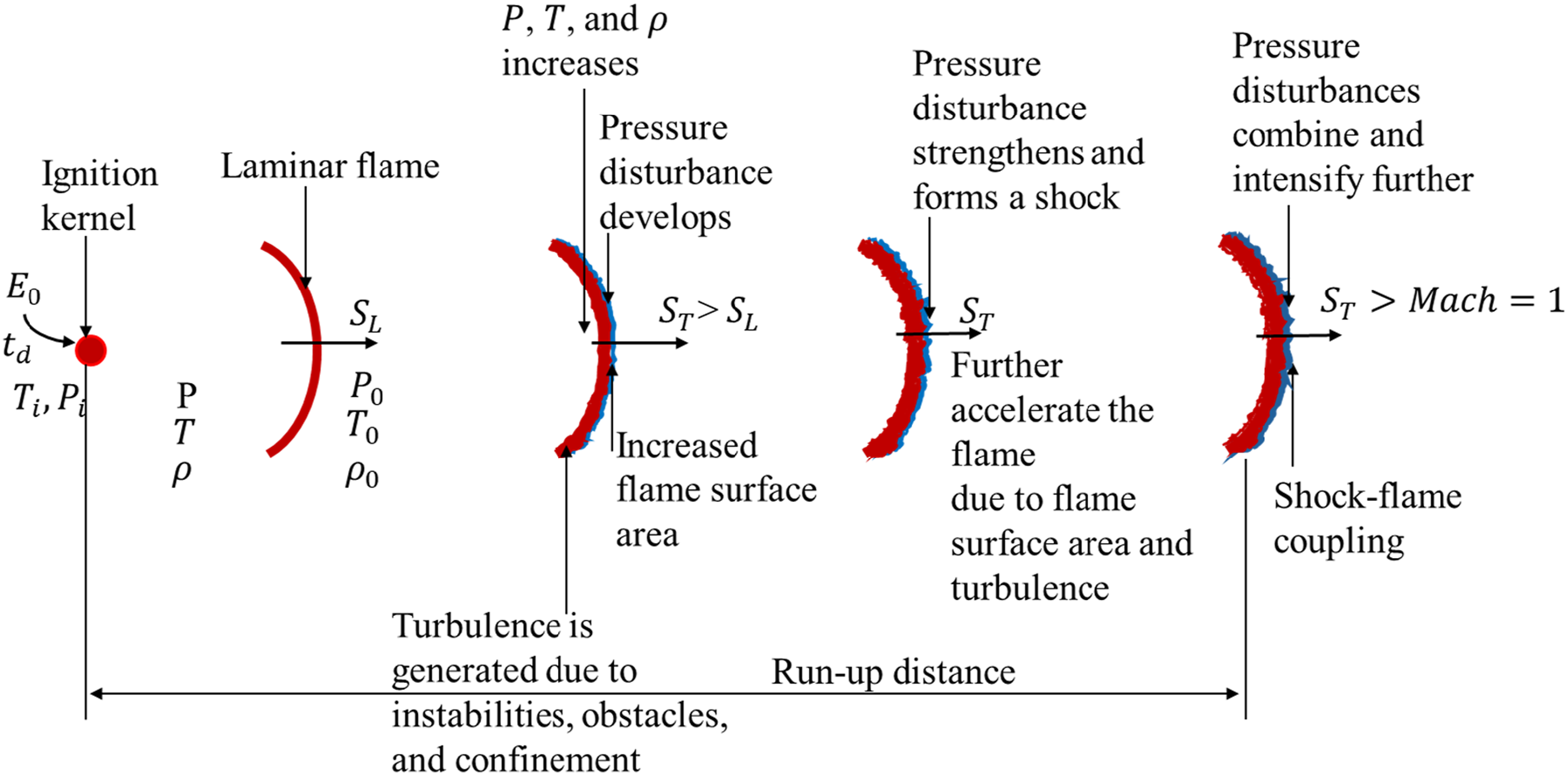
Gaseous hydrogen storage techniques and potential hazards
Gaseous hydrogen storage techniques
The hydrogen economy encompasses the production, transportation, storage, distribution, and utilisation of hydrogen as a clean energy carrier. However, these processes involve significant safety challenges due to factors such as high-pressure containers, confined spaces, long-term storage, human error, and the potential for container rupture or leakage. These issues can lead to the formation of combustible hydrogen-air mixtures that may auto-ignite and cause explosions (deflagrations or detonations) under certain conditions. As discussed in Section The Explosion Behaviour of Hydrogen, this highlights the importance of implementing preventive safety measures. Therefore, it is essential to understand the different types of available storage methods, the specific storage conditions, potential points of failure, and the risk of leakage and explosion.
Hydrogen storage methods are mainly categorised into physical-based and material-based hydrogen storage approaches based on the underlying storage technology. Physical-based storage refers to methods of storing hydrogen in either gaseous or liquid form without creating substantial chemical bonds with other materials. It relies on the physical properties of hydrogen, typically by compressing it into high-pressure tanks or cooling it to extremely low temperatures for cryogenic liquid storage (Le et al., 2024). On the other hand, material-based storage creates chemical bonds between hydrogen and different materials. It uses special materials that can reversibly store hydrogen through either adsorption or absorption (Ren et al., 2017; Stetson et al., 2016). Material-based hydrogen storage technologies comprise chemisorption and physisorption, which represent two distinct methods for hydrogen storage (Moradi and Groth, 2019). Each of these storage technologies further branches into subcategories, as illustrated in Figure 7. Additionally, Table 3 summarises the advantages, disadvantages, and engineering applications of each storage method for better understanding. Classification of hydrogen storage. Advantages, disadvantages and engineering applications of each hydrogen storage method.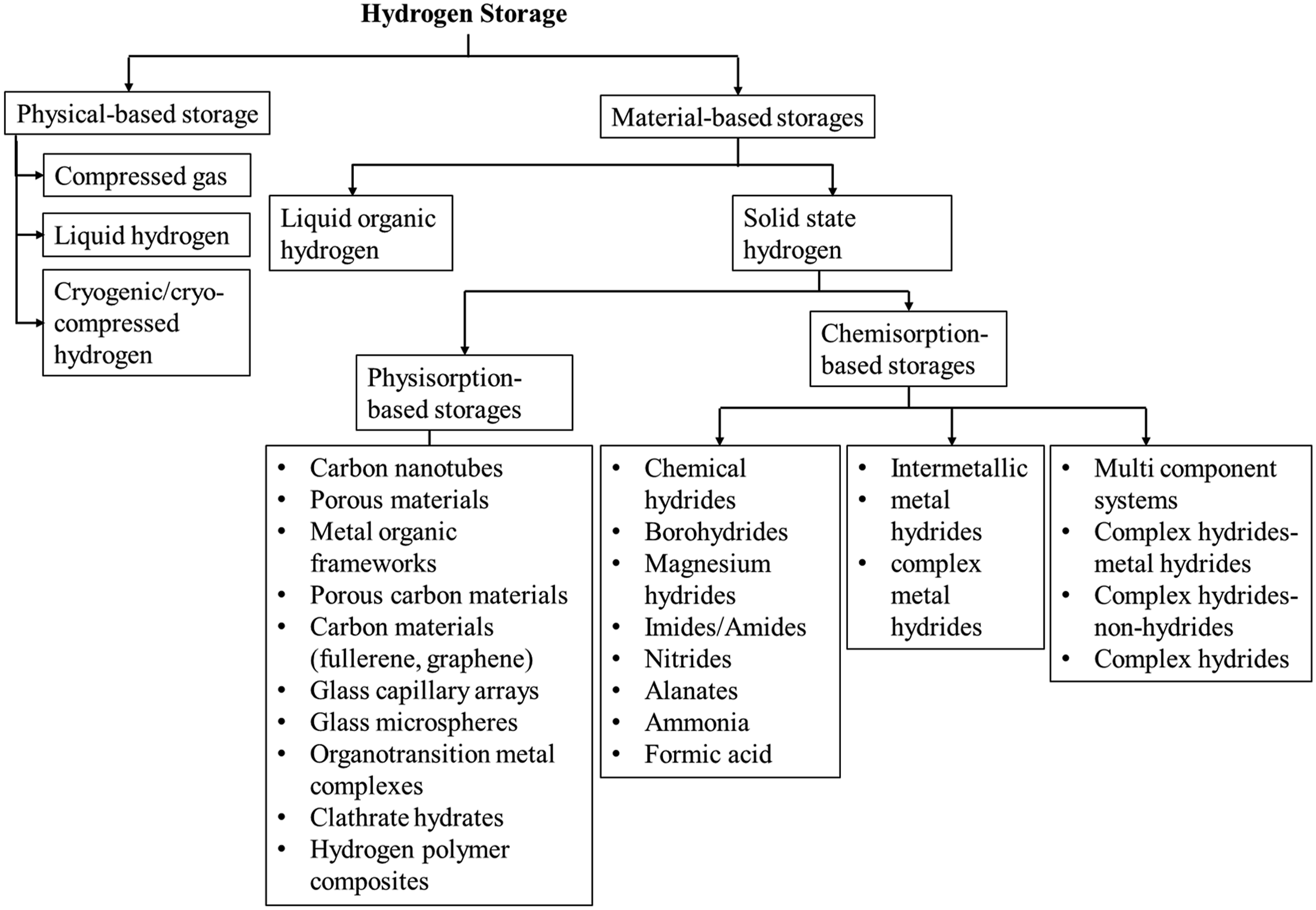
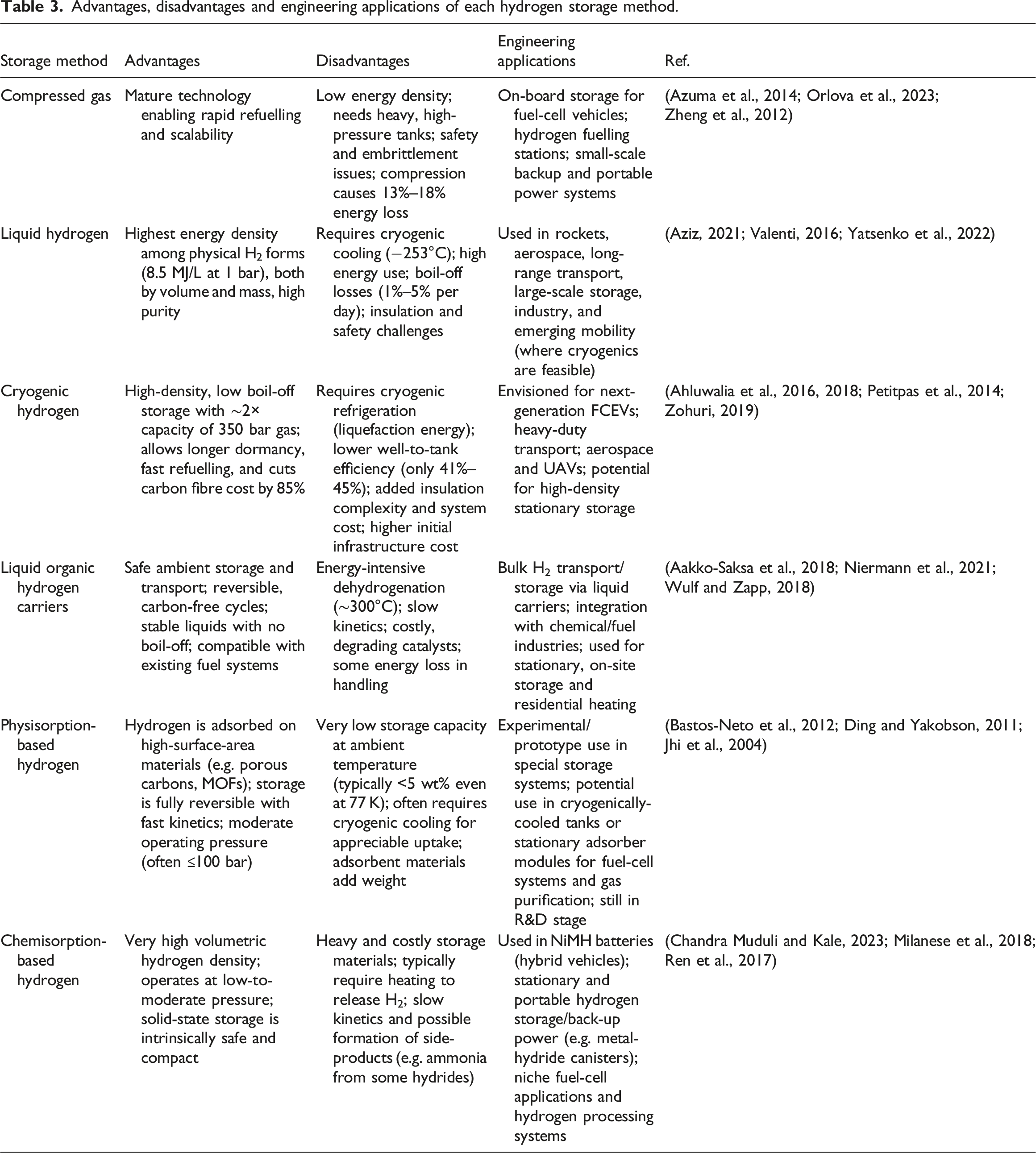
Among these different hydrogen storage technologies, gaseous hydrogen storage plays a vital role due to its economic advantage, infrastructure simplicity, widespread use, efficiency, cost-effectiveness, design flexibility, and minimal environmental impact, while still posing notable safety concerns (Risco-Bravo et al., 2024). Economically, hydrogen liquefaction consumes 30% to 40% of the lower heating value (LHV) of hydrogen, whereas the energy required for compressing hydrogen to pressures of 35 MPa to 70 MPa is approximately 5% to 20% of LHV. Additionally, releasing hydrogen from compressed gas storage requires no energy input, making gaseous storage more cost-effective. High-pressure gaseous hydrogen (HPGH2) storage is favoured for its technical simplicity, fast filling and release rates, and well-developed infrastructure, making it the most widely used method for hydrogen storage across a broad range of applications (Zheng et al., 2012).
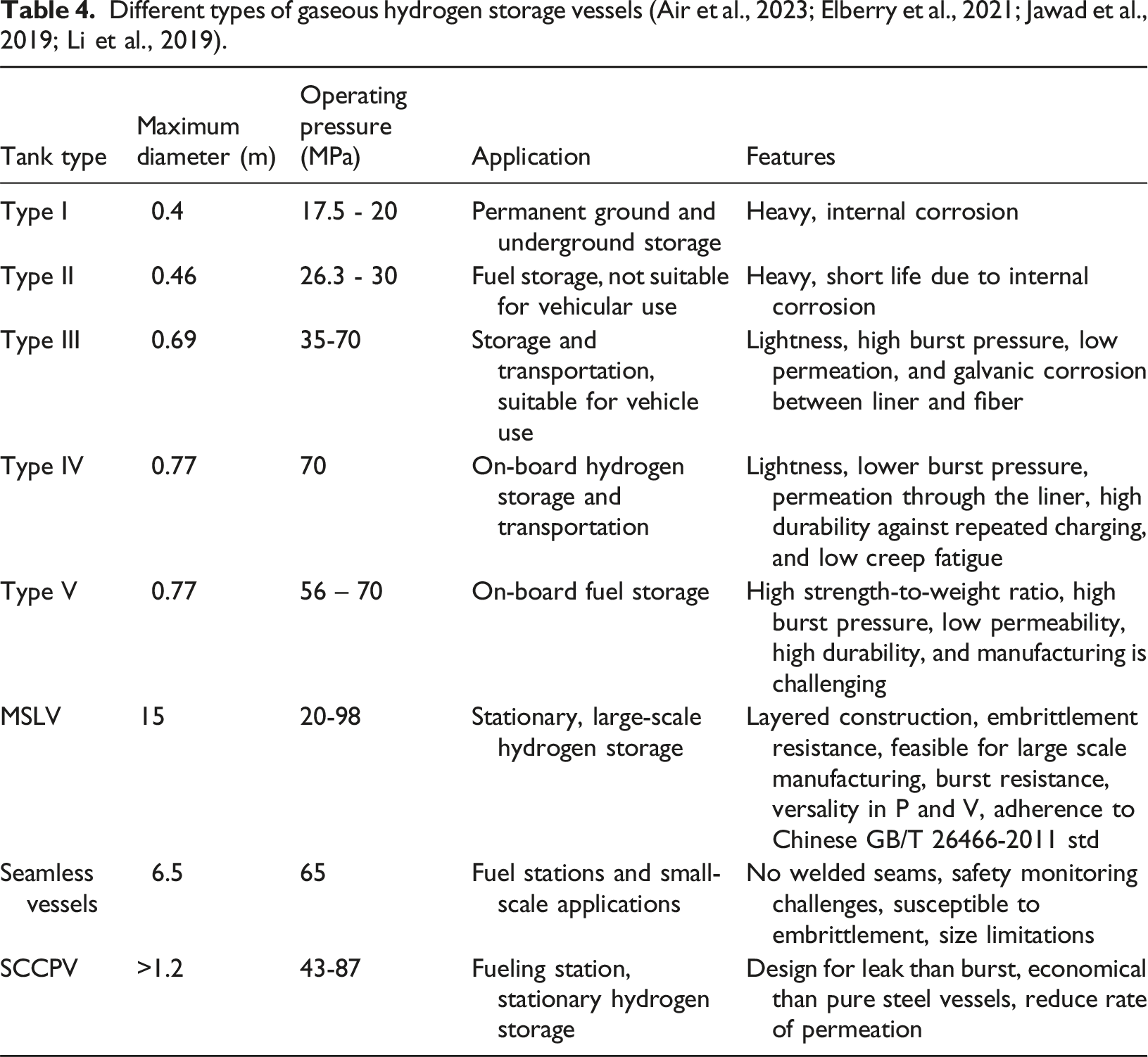
There are five basic types of gaseous hydrogen storage vessels known as Type I, II, III, IV and V (Air et al., 2023), and their constructions are illustrated in Figure 8. Type I pressure vessels are considered conventional and the least expensive among them, typically made from steel. It is the heaviest vessel type due to its metal construction. The ease of fabrication also makes it economically more viable in fixed, stationary storage scenarios. Type I vessels can withstand pressures of up to 50 MPa. Type II storage vessels are constructed using steel and feature a composite wrap. While they are more expensive than Type I vessels, they offer the advantage of being relatively lightweight. Type III vessels, on the other hand, are composed of a full composite wrap, which provides structural rigidity and robustness to withstand the CGH2. These vessels contain a metal liner made of aluminium that helps prevent hydrogen embrittlement, a problem commonly observed in Type I and Type II vessels. They are also notably lighter than Type II but incur additional manufacturing costs in comparison. Type IV vessels are fully composite with high tensile strength and include a polymer liner, which mitigates hydrogen embrittlement and allows Type IV vessels to withstand pressures up to 100 MPa (Bouhala et al., 2024). The ability to withstand high pressures means that these vessels have higher energy density. This attribute, coupled with their lightweight nature of construction and overall structural robustness, means that Type IV vessels are suitable for use in H2 vehicles. Type IV vessels are currently adopted in examples such as the Toyota Mirai and Honda Clarity, where the nominal working pressure is 70 MPa (Air et al., 2023). Additionally, there are liner-less, fully composite vessels known as Type V, which offer further weight reduction and compatibility with higher pressures. Advanced manufacturing methods, particularly Automated Fibre Placement (AFP), can improve Composite Overwrapped Pressure Vessels (COPV), and CPV typically stands for Composite Pressure Vessel designs, offering higher accuracy and lower wastage compared to traditional filament winding. Despite the promise of Type V and AFP technology, engineering challenges remain before they can replace Type IV tanks (Air et al., 2023; Okonkwo et al., 2023). Table 4 further describes characteristics (maximum diameter achieved, operating pressure, applications, and features) of Type I to Type V vessels, multifunctional steel layered vessels (MSLV), seamless vessels, and steel-concrete composite pressure vessels (SCCPV). Basic construction of type I, II, III, IV and V vessels. Different types of gaseous hydrogen storage vessels (Air et al., 2023; Elberry et al., 2021; Jawad et al., 2019; Li et al., 2019).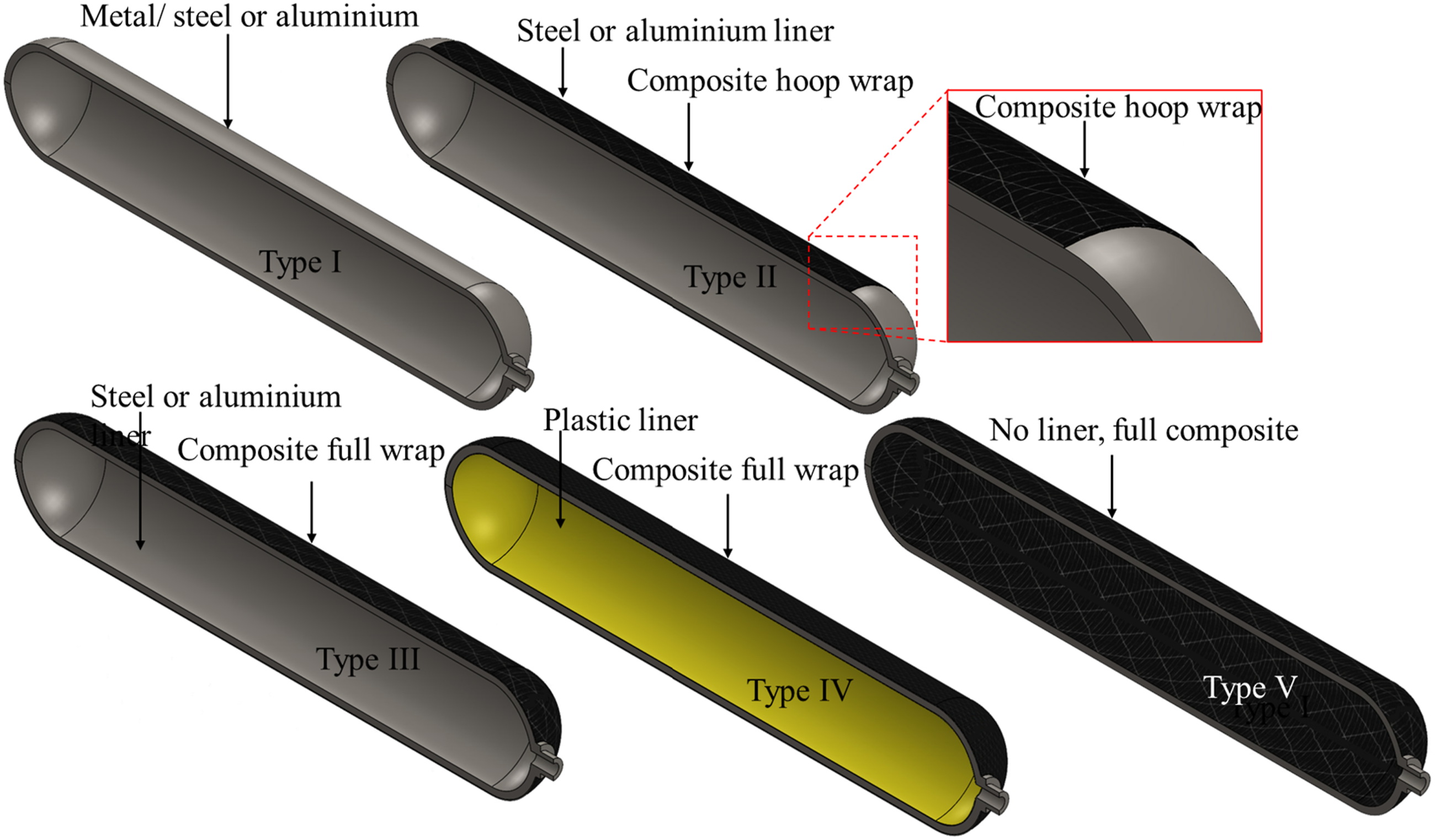
The Multifunctional Steel Layered Vessel (MSLV) has been adopted for long-term stationary storage in industrial applications. There are some variants to the MSLV design, but typically, it is constructed from a flat steel ribbon-wound cylinder, double-layered hemispherical heads, reinforcing rings, support structures, and nozzles as displayed in Figure 9. In addition, MSLVs are designed with many safety features that help prevent leaks and the potential for explosions. Their main advantage lies in their ability to store large volumes of hydrogen at high pressure, resulting in a higher storage density. MSLVs are still subjected to extensive research that focuses on the testing and evaluation of materials, such as austenitic steel, for compatibility with hydrogen and storage conditions (Elberry et al., 2021). Organisations like Sandia National Laboratories, Sumitomo Metal Industries, Kobe Steel, Japan Steel Works, and the National Institute of Advanced Industrial Science and Technology (AIST) are actively involved in research on developing MSLVs. Notably, the world’s first 77 MPa MSLV with a volume of 2.5 m3 is operational in a demonstration hydrogen refuelling station in China. Additionally, storage vessels for hydrogen and compressed natural gas blends (HCNG) with volumes of 10 m3 and 15 m3 have been constructed for the world’s largest HCNG refuelling station in Shanxi province, China (Zheng et al., 2012). Steel ribbon wound vessel, 1 - outer shell, 2 - inner shell, 3 – hemispherical head, 4 – nozzle, 5 – reinforcing ring (Du et al., 2023).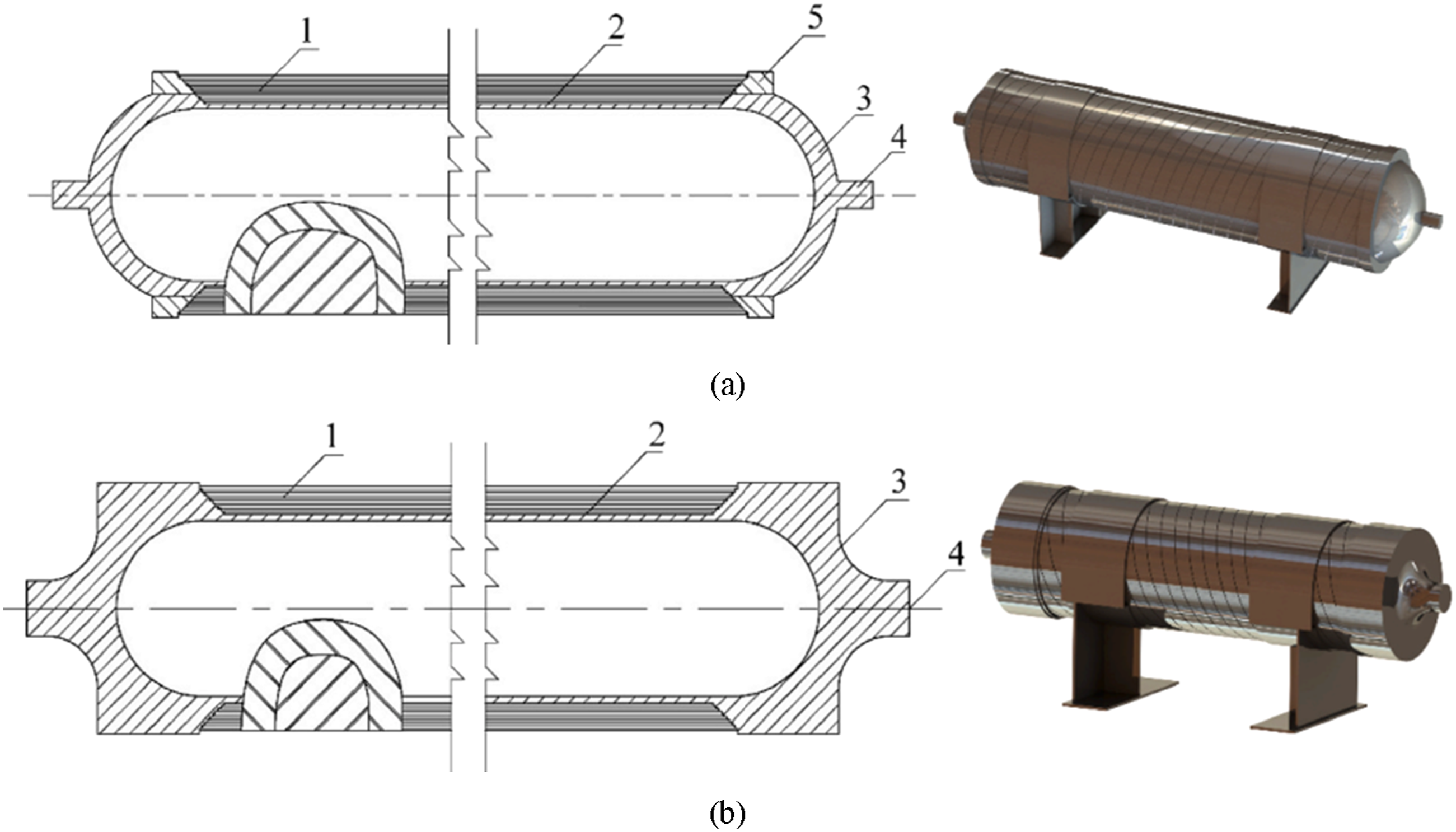
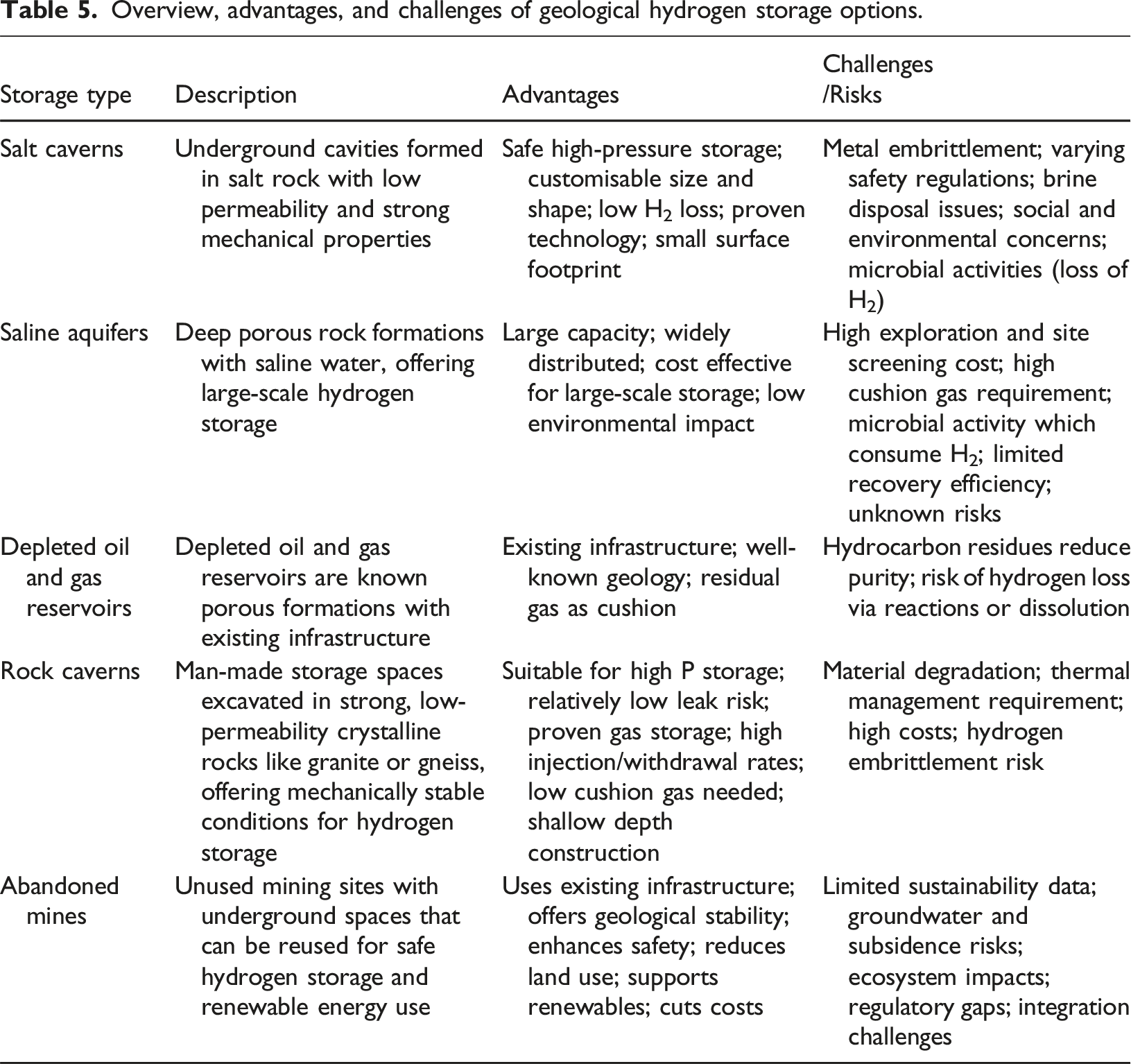
Overview, advantages, and challenges of geological hydrogen storage options.
Potential hazards in gaseous hydrogen storage systems
In gaseous hydrogen storage systems, primary hazards include high-pressure vessel rupture or gas cloud explosions, which may occur under varying environmental conditions (e.g., open spaces, fully confined spaces, vented enclosures, process plants, or urban areas) (Hu et al., 2023). In contrast, liquid hydrogen (LH2) storage systems face distinct hazards such as Boiling Liquid Expanding Vapour Explosion (BLEVE), Rapid Phase Transition (RPT) explosions, detonations from accumulated LH2 and solidified air, pressure relief failures, and vapour cloud explosions due to evaporated LH2 (Aziz, 2021; Wang et al., 2024). However, material-based storage systems have different safety risks. They are less likely to explode because the hydrogen is chemically stored in solid materials. But, some of the materials can ignite easily when exposed to air (pyrophoric risk) and may be toxic if powders are inhaled (Klopčič et al., 2023).
High-pressure vessel rupture mechanisms and deflagration risks
Among the most critical hazards in gaseous hydrogen storage systems is pressure vessel burst (PVB), also referred to as high-pressure vessel rupture. It often leads to a deflagration mode of explosion. This occurs when a storage tank catastrophically fails due to mechanical overstress, thermal loading, or material degradation, leading to rapid hydrogen release and potential ignition (Hu et al., 2023). Different types of storage systems exhibit various fracture mechanisms and leak behaviours. For high-pressure gas cylinders, fractures typically result from material fatigue or external impacts, leading to rapid gas release due to the high internal pressures, as the process illustrated in Figure 10. It shows the Carbon Fibre Reinforced Polymer (CFRP) tank delamination, high-pressure hydrogen jet release, instantaneous ignition and explosion. In addition, hydrogen embrittlement, which happens when hydrogen dissolves in steel, causes stress concentrations that exceed the strength limit of the steel and form tiny cracks (white spots). Hydrogen’s high diffusivity, low viscosity and light weight enable it to leak through subtle cracks (Messaoudani et al., 2016). Commercial challenges include optimising manufacturing processes, increasing service pressure, reducing costs, and developing a materials property database to understand failure mechanisms (Filippov and Yaroslavtsev, 2021; Mironov et al., 2015; Zheng et al., 2012). Spontaneous ignition of a hydrogen vessel during an accident.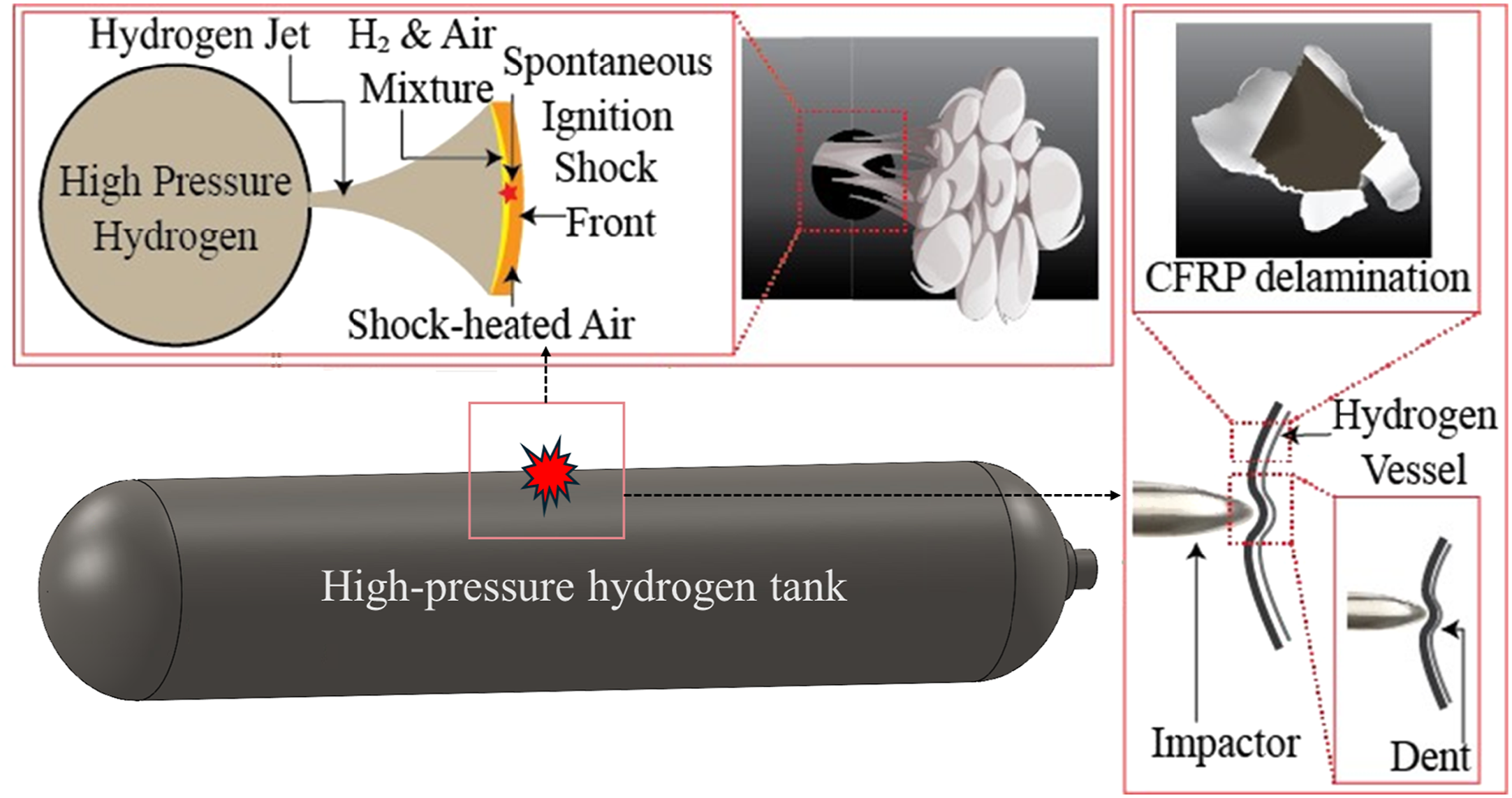
Hydrogen leakage behaviour and dispersion patterns in unconfined and partially confined spaces
The behaviour of a high-pressure hydrogen gas leakage depends on the location of the leak (during production, storage, transportation, or use) and the surrounding environment. After a leak, hydrogen gas mainly spreads because it is lighter than air (buoyant plume). When hydrogen leaks from the side of a tank, it spreads slowly sideways and slows down over time. A top leak spreads upward much faster, but the spread rate decreases over time. More hydrogen builds up near the ceiling if the leak is far from a side vent. However, if the leak is from the floor, how far it is from the bottom vent doesn’t affect how quickly hydrogen reaches the top (Xu and Zhang, 2008). Under good ventilation, a small amount of hydrogen builds up, while hydrogen can gather in layers and gradually create a dangerous mixture that could explode (Yang et al., 2024). In a typical garage, an upward hydrogen leak can lead to over 29% concentration at 2.6 m height. Hydrogen forms layers, and its concentration changes with height. The closer the leak is to the ground, the more the hazardous area shifts downward (Pitts et al., 2012). Figure 11 shows the behaviour of hydrogen gas after a leakage from a storage. Leak patterns and concentration distribution from different H2 leakage incidents: (a), (b), (c) (Hall et al., 2017; Wang et al., 2020; Zhou et al., 2023a), (d), (e) (Li et al., 2022b, 2024b; Zhou et al., 2023b). (f), (g), (h) (Zhang and Shang, 2024).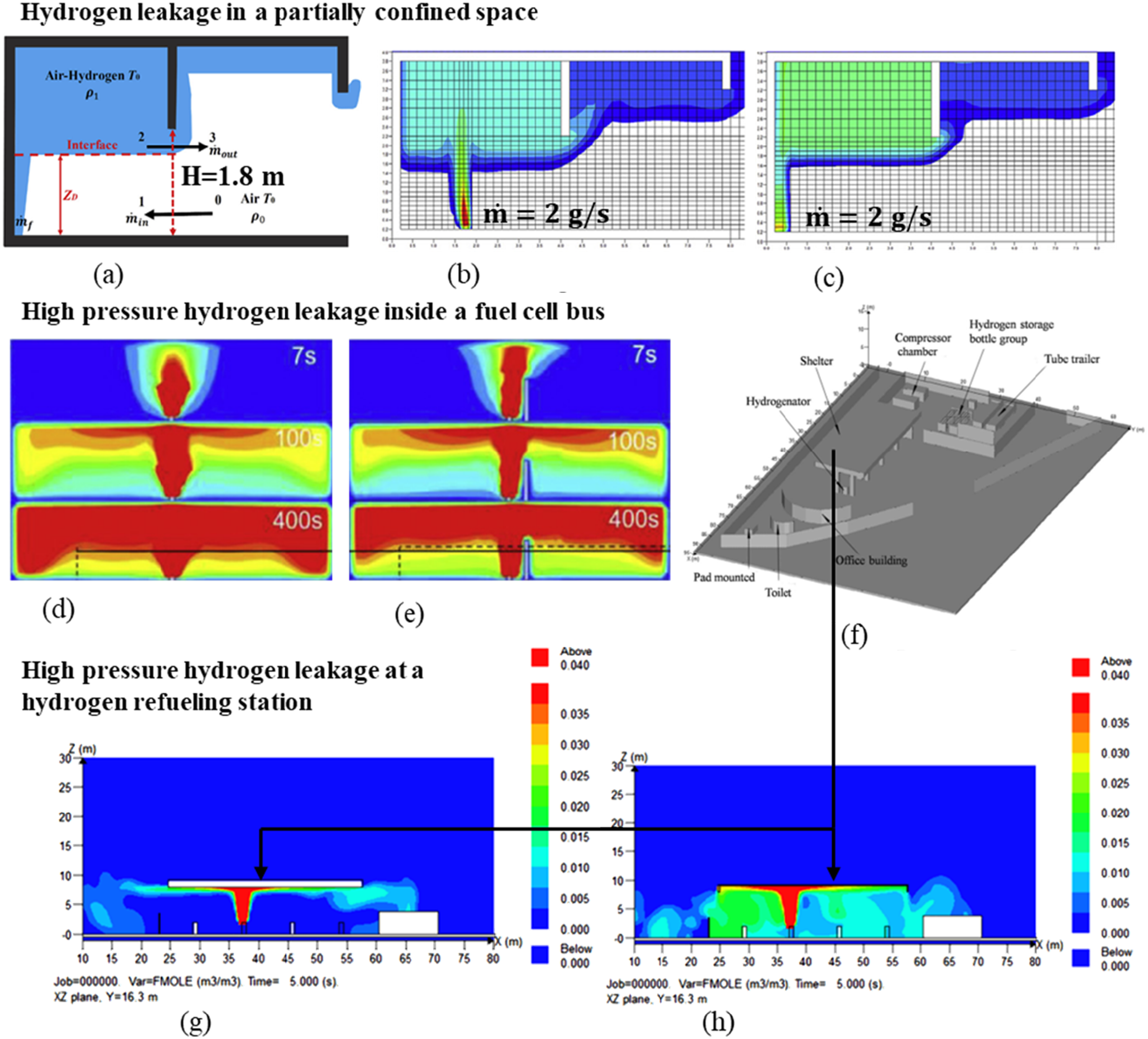
After hydrogen leaks from a storage tank, particularly in confined spaces like garages, the behaviour of the gas can lead to significant hazards. A catastrophic tank failure can cause explosive damage, while continuous leakage from minor cracks typically results in a gradual release of hydrogen. When a high-pressure hydrogen leak (hydrogen jet) occurs from a storage tank, it creates a directed hydrogen flow along the path of the leak. In such cases, it forms a supersonic jet that is unstable and hard to control, potentially igniting and causing explosions. High-pressure storage, large volume requirements, material limitations, self-ignition risks, significant energy storage needs, and transportation difficulties further complicate its use (Agll et al., 2016; Elberry et al., 2021; Jafari Raad et al., 2022; Moradi and Groth, 2019). The buoyancy effect acts on the leaking gas when the momentum of the gas jet reduces and forms a combustible mixture at the top of the confined space (Yang et al., 2021). The experimental study by Han et al. examined hydrogen jets released through small openings (0.5 mm, 0.7 mm, and 1.0 mm in diameter) at high pressures of up to 40 MPa, revealing key aspects of flame behaviour and concentration changes under rapid ignition at the leak point. The flame remained straight along the jet’s centreline even when pressure decreased up to 1 MPa, indicating negligible buoyancy effects and demonstrating that the jet’s momentum overpowers gravitational forces. The hydrogen concentration along the jet decreases gradually with distance from the leak, influenced by factors such as crosswind effects and viscous heating, which can alter the dispersion pattern and affect the ignition potential of the released hydrogen. For example, the concentration has decreased from 10% at 1 m to 1% at 7 m from the leak point for a 0.5 mm diameter fractured hole, following a hyperbolic decay pattern (Han et al., 2014). To give a sensation of high-pressure gas jet leakage and jet flame, Figure 12 (a) illustrates gas leak scenarios from illuminated gas release images for a range of pressures for a fracture diameter as small as 0.7 mm. Figure 12 (Han et al., 2014), and (b) shows the jet flame behaviour from 15 MPa to 43.5 MPa of pressure jet release and flame from 1.06 mm to 0.64 mm orifices (Hall et al., 2017), respectively. (a) Gas leak scenarios captured from illuminated gas release images at various pressures for a fracture diameter of 0.7 mm (Han et al., 2014), (b) Jet flame behaviour from pressure releases at 15 MPa and 43.5 MPa through orifices of 1.06 mm and 0.64 mm (Hall et al., 2017).
Ignition mechanisms and combustion risks in pressurised hydrogen systems
Common ignition mechanisms relevant to hydrogen storage.
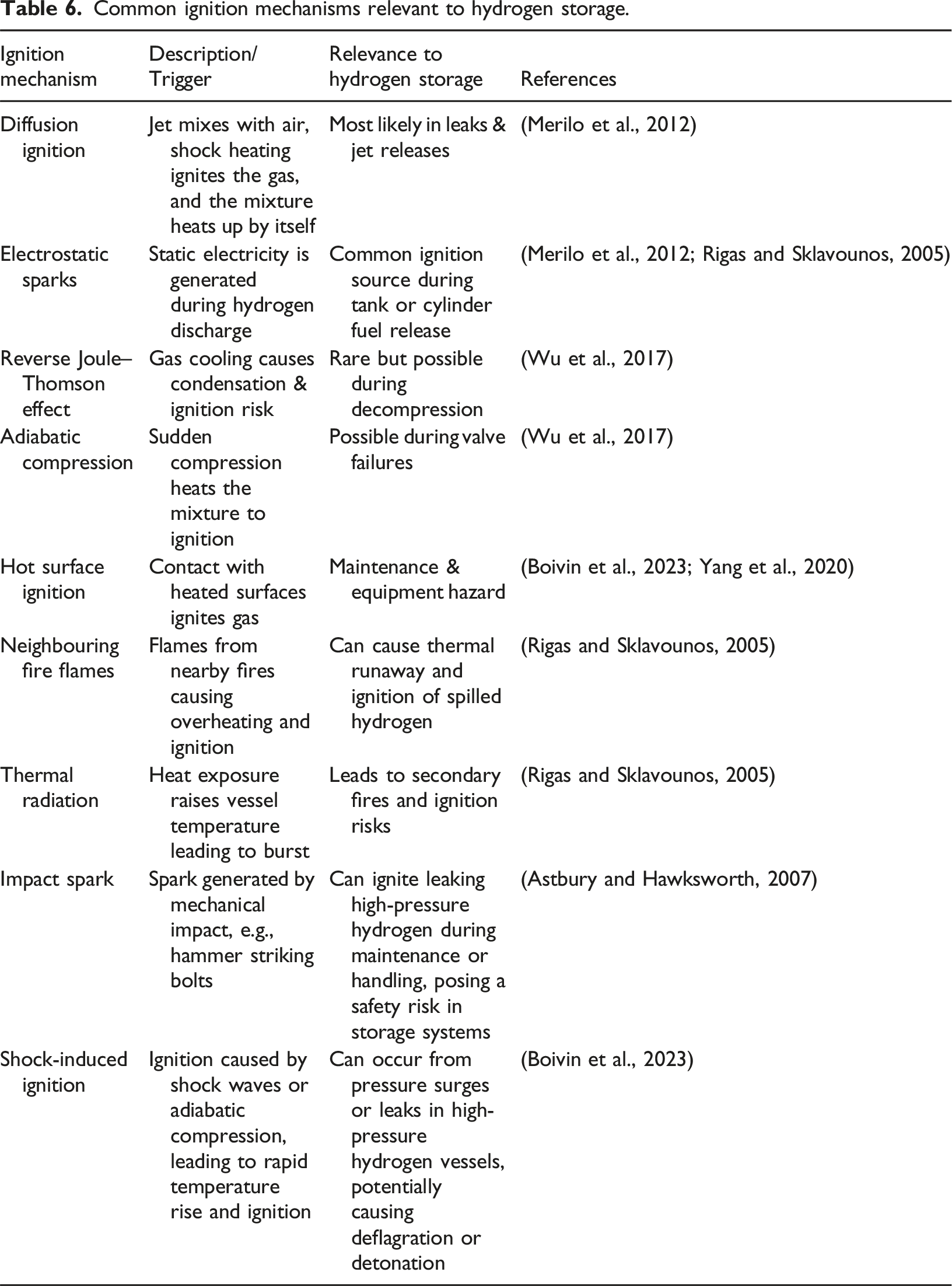
If fuel concentrations are within appropriate limits, and an ignition source is present, combustion can initiate, flame propagates, accelerates and might eventually lead to detonation according to the DDT process described in Section The Explosion Behaviour of Hydrogen. The mode of incident is mainly determined by the environment (confined or unconfined), leakage type (sudden or steady), and ignition characteristics (rapid or delayed). When a gradual leakage forms a gas cloud, it can cause an explosion or fireball under delayed ignition, or it may freely expand and diffuse, temporarily raising the ambient temperature without reaching autoignition conditions. The risk of explosion is higher in a confined environment due to the delayed diffusion under limited ventilation, the formation of high gas concentrations, the possibility of localised high temperatures, and the serious risk of property damage and casualties (Aftab et al., 2022). Obstacles are a significant factor influencing the development of overpressure in hydrogen explosions. The explosion intensity is particularly affected by the shape of the obstacles, their blockage ratio, and spatial arrangement. Obstacles can also amplify reflected blast overpressure, increase flame velocity, and alter flame propagation direction. Therefore, hydrogen storage systems should be designed to minimise the presence or impact of obstacles that could contribute to undesirable explosion effects (Ahad et al., 2023).
Experimental insights into hydrogen explosion pressures in unconfined, confined, and vented environments
Research has been conducted to understand the potential pressure levels and damage resulting from confined, semi-confined, and unconfined hydrogen gas explosions. For instance, Nozu et al. (Nozu et al., 2005) conducted an experimental study on the deflagration and detonation of 5.27 m3of hydrogen-air and the resulting hazardous blast wave. It was observed that the deflagration of a stoichiometric hydrogen-air mixture produced 2 kPa on a wall located 5 m from the ignition position, while the detonation of the same mixture produced 180 kPa at the same distance. The ignition occurred at the centre of the tent containing the mixture, as shown in Figure 13 (a). Similarly, Zhou et al. (Zhou et al., 2023a) conducted a similar explosion study with a larger volume of 27 m3 of stoichiometric hydrogen-air mixture, which produced 6 kPa at 3 m away from the ignition source, as illustrated in Figure 13 (b). In addition, Gan et al. (Gan et al., 2025) recently investigated the deflagration, fast deflagration and detonation in their experiment of 5.09 m3 of unconfined cylindrical hydrogen-air mixture with a comprehensive comparison with other experimental studies. Their results showed distinct overpressure profiles at different distances. At 2 m from the ignition source (with a mixture radius of 0.9 m), the recorded overpressures were approximately 7.5 kPa for deflagration, 42 kPa for fast deflagration, and 200 kPa for detonation. At 8 m, these values decreased to about 3 kPa, 15 kPa, and 38 kPa, respectively. (a) Unconfined hydrogen explosion test in a 5.27 m3 cuboidal tent (Nozu et al., 2005), (b) Unconfined hydrogen explosion test in a 27 m3 cuboidal tent (Zhou et al., 2023a).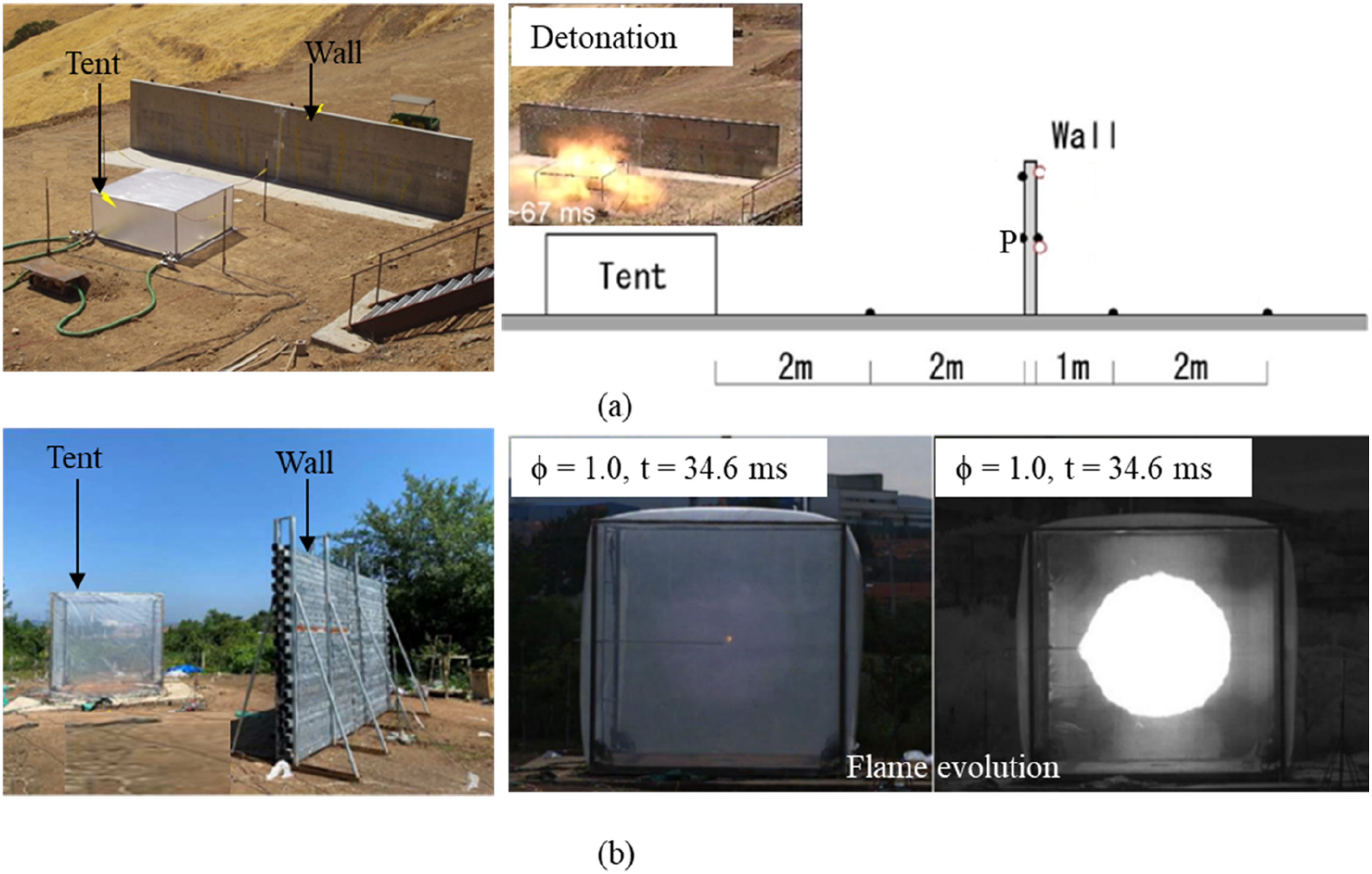
However, in confined spaces, hydrogen accumulates and produces high concentrations that can produce intense explosions. Pitts et al. (Pitts et al., 2012) conducted an experimental study investigating explosion events that can happen in residential garages with an automobile, as illustrated in Figure 14 (a). It was observed that the measured pressure at a side wall is as low as less than 1 kPa for a hydrogen concentration of 15.6 %, and it is up to 60 kPa for a higher concentration of 29.3 % under an ignition source of 80 J squib charger, which caused deflagration of the mixture. Another study has been done by Li et al. (Li et al., 2024b), highlighting the confined hydrogen explosion effects when blended with methane, as shown in Figure 14 (b). It produced 25 kPa of positive peak pressure and 32.5 kPa of negative peak at the window inside the confined space, where the space was filled with a mixture with an equivalence ratio of 1.1 and 30 % of hydrogen blended with methane. Even though these confined hydrogen explosions belong to deflagration explosion mode, the confinement significantly increases hazards to the structure and the surrounding environment due to the rapid flame propagation, high magnitude of reflected overpressure, external explosions and fragments. In addition, Shirvill et al. (Shirvill et al., 2018) investigated the effect of congestion and confining wall on an explosion of real-scale hydrogen storage explosion by considering a hydrogen-air cloud of 129 m3. It produced nearly the maximum of 27 kPa at 15 m away from the ignition point, while a 300 m3 unconfined hydrogen air deflagration test conducted by Groethe et al. (Groethe et al., 2007) produced only 17 kPa at the same distance of 15 m from the ignition location. These experimental studies highlight the danger of hydrogen leakage and ignition, which demand safe designs and techniques to mitigate accidental explosions in hydrogen storage facilities. Hydrogen–air mixture explosions in confined environments can cause significantly greater damage than unconfined cases due to the higher overpressure generated during deflagration. Table 7 presents a summary of key experimental studies on hydrogen explosions, providing additional details on the parameters beyond those discussed earlier. (a) Experimental study of hydrogen explosions in a residential garage with an automobile (Pitts et al., 2012), (b) Confined hydrogen-methane explosion test highlighting pressure effects inside a closed space (Li et al., 2024b). Key experimental studies on hydrogen explosions.
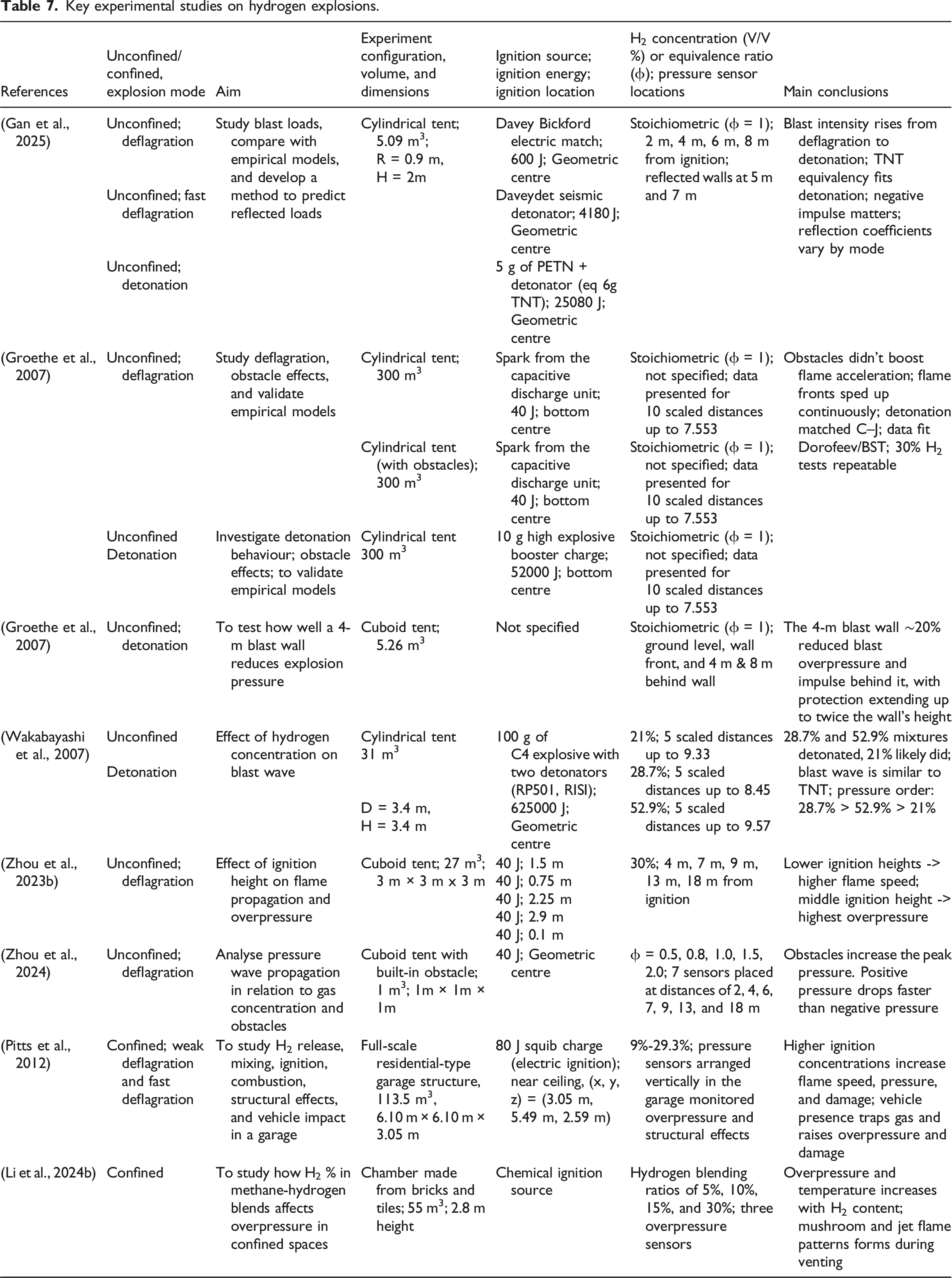
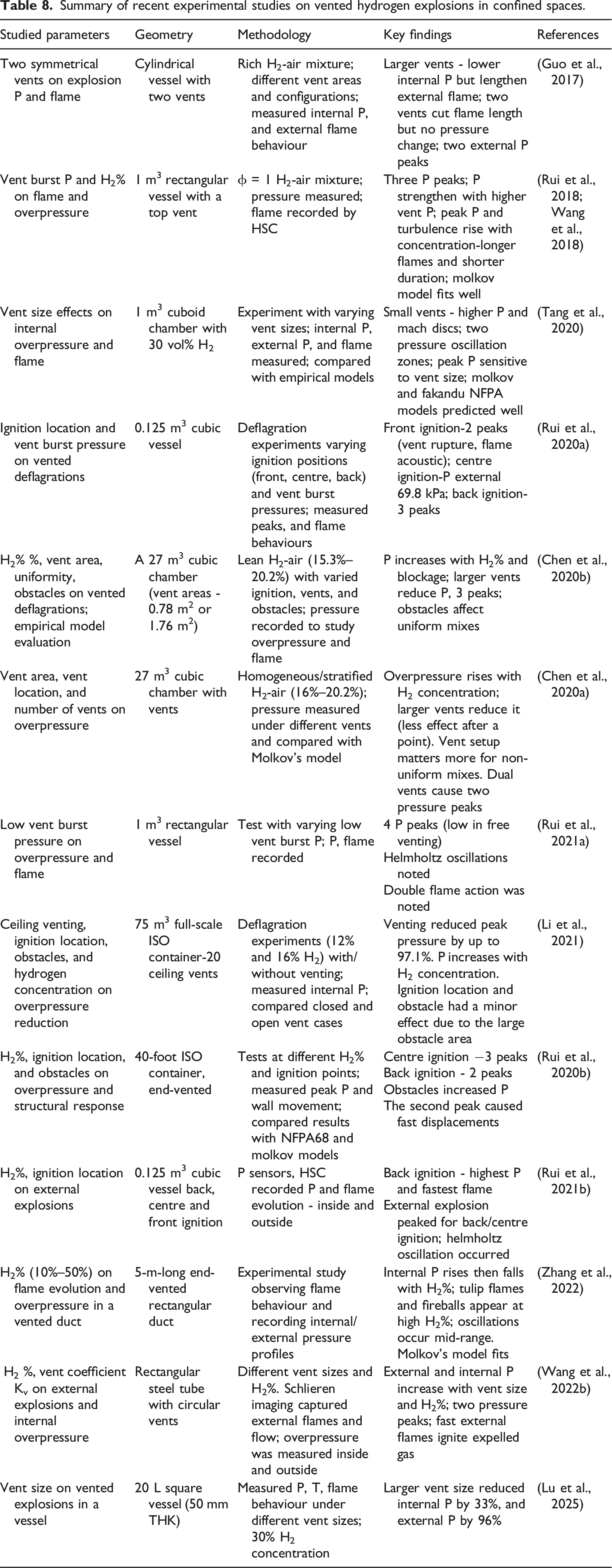
The vented hydrogen explosion has been a topic of interest because vents reduce hydrogen explosion damage in confined environments, as vented explosions generally produce lower overpressures compared to non-vented ones. For instance, the overpressure rise rate (dP/dt) is significantly influenced by venting. The presence of a vent changes flame propagation, which directly affects the rise of overpressure (Hu et al., 2025b). Explosion vents protect equipment and infrastructure from explosions by rapidly releasing the energy generated from the explosion (Zhang and Zhang, 2018). There are standards developed by the National Fire Protection Association (NFPA) for safe vented infrastructure designs against confined explosions, which correspond to hydrocarbon explosions. Due to hydrogen’s higher reactivity compared to hydrocarbons, vented hydrogen-air explosions occur more rapidly and involve more complex phenomena. A thorough understanding of these explosion characteristics is crucial for the structural safety design of vessels and buildings, as well as for ensuring the protection of individuals and property (Saffers and Molkov, 2014). Therefore, research has been conducted on analysing the effect of various venting parameters, ignition location, obstacles, burst pressure etc., on vented hydrogen explosions (Guo et al., 2015; Zhang and Zhang, 2018).
Summary of recent experimental studies on vented hydrogen explosions in confined spaces.
The safe implementation of gaseous hydrogen systems depends critically on strict compliance with continuously developing international standards. Key organisations such as the International Organisation for Standardisation (ISO), European Committee for Standardisation (CEN), National Fire Protection Association (NFPA), and European Industrial Gases Association (EIGA) have established guidelines that currently focus mainly on preventing material embrittlement (41% of existing standards) and designing storage vessels (32%). However, only 7% of standards specifically address pipeline transportation requirements, and there is insufficient guidance for properly evaluating and mitigating explosion hazards (Ordin, 1997; Yang et al., 2020). While current standards provide essential safety fundamentals, significant shortcomings remain in understanding accidental explosion behaviour and designing infrastructure to withstand such events. These limitations highlight the necessity for coordinated progress in material technologies, novel design approaches, and safety codes and standards, which will be examined in detail in Chapter 5. Encouraging developments across these research areas are now helping to resolve existing technical challenges while supporting the expansion of reliable hydrogen storage technologies.
Advancements and challenges in safe hydrogen storage
The effective storage of hydrogen is pivotal for establishing a hydrogen-based economy in the coming years. Thus, exploring potential avenues for the secure design of gaseous hydrogen storage systems is crucial. This section explores cutting-edge material technologies, strategic design approaches, codes and standards, and provides an understanding of the prominent challenges involved.
Material technologies for the safe design of hydrogen storage
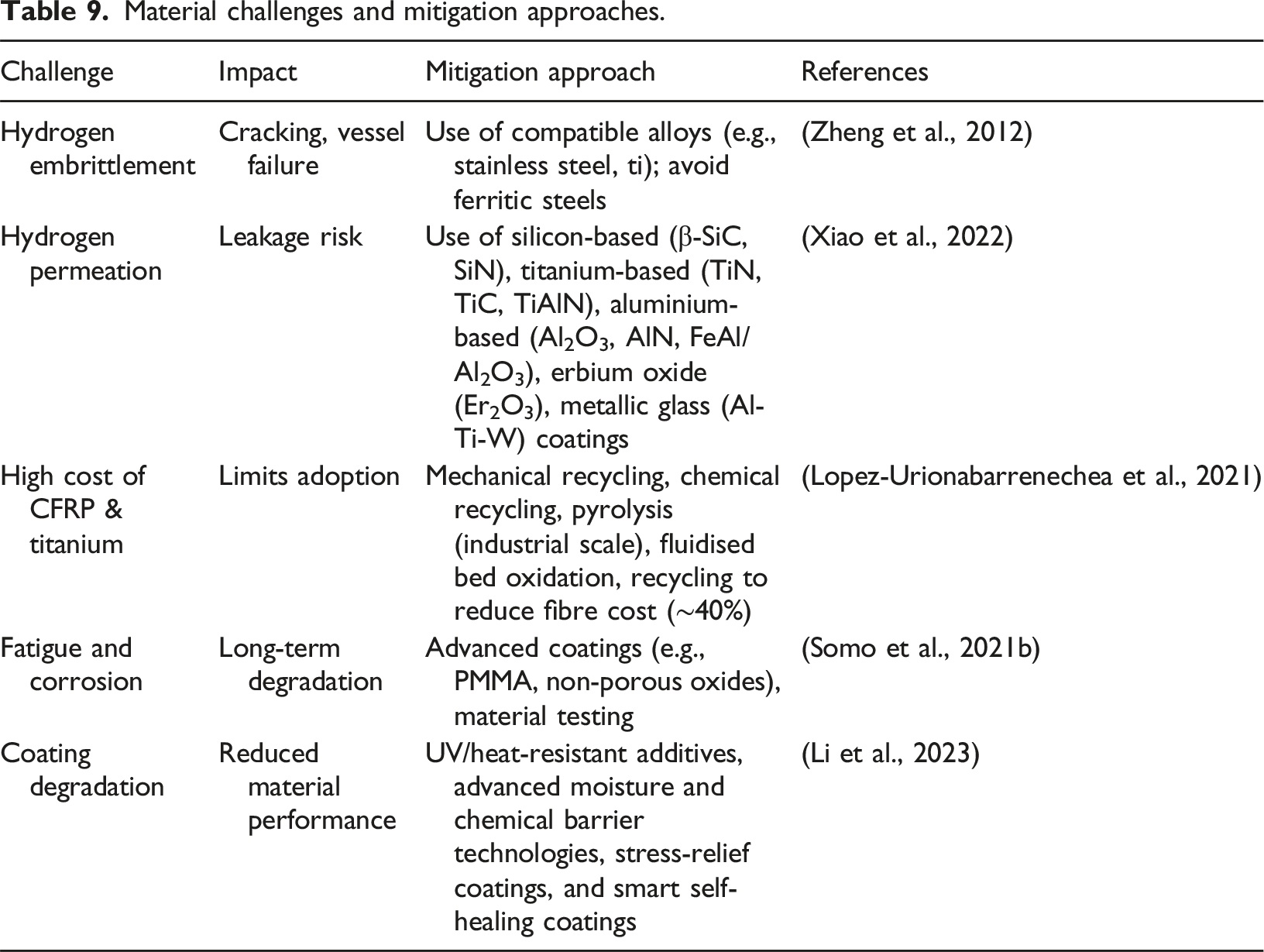
Material selection plays a critical role in the design of compressed gaseous hydrogen (CGH2) storage vessels, directly influencing their pressure limits, efficiency, and safety. For Type-I steel vessels, the maximum operating pressure is typically constrained to 50 MPa due to hydrogen embrittlement risks, which can lead to material degradation, leakage, and potential explosions (Meda et al., 2023). As storage pressures increase to improve density, materials must simultaneously withstand mechanical stresses (e.g., cyclic loading, corrosion) while maintaining lightweight properties (Okonkwo et al., 2023). This has driven the adoption of advanced materials like carbon fibre-reinforced polymers (CFRP) and titanium in Type-III/IV vessels, though their temperature sensitivity (e.g., CFRP degradation above 358K) introduces new safety trade-offs (Tashie-Lewis and Nnabuife, 2021).
Material challenges and mitigation approaches.
Exploring design strategies to mitigate explosion incidents in hydrogen storage
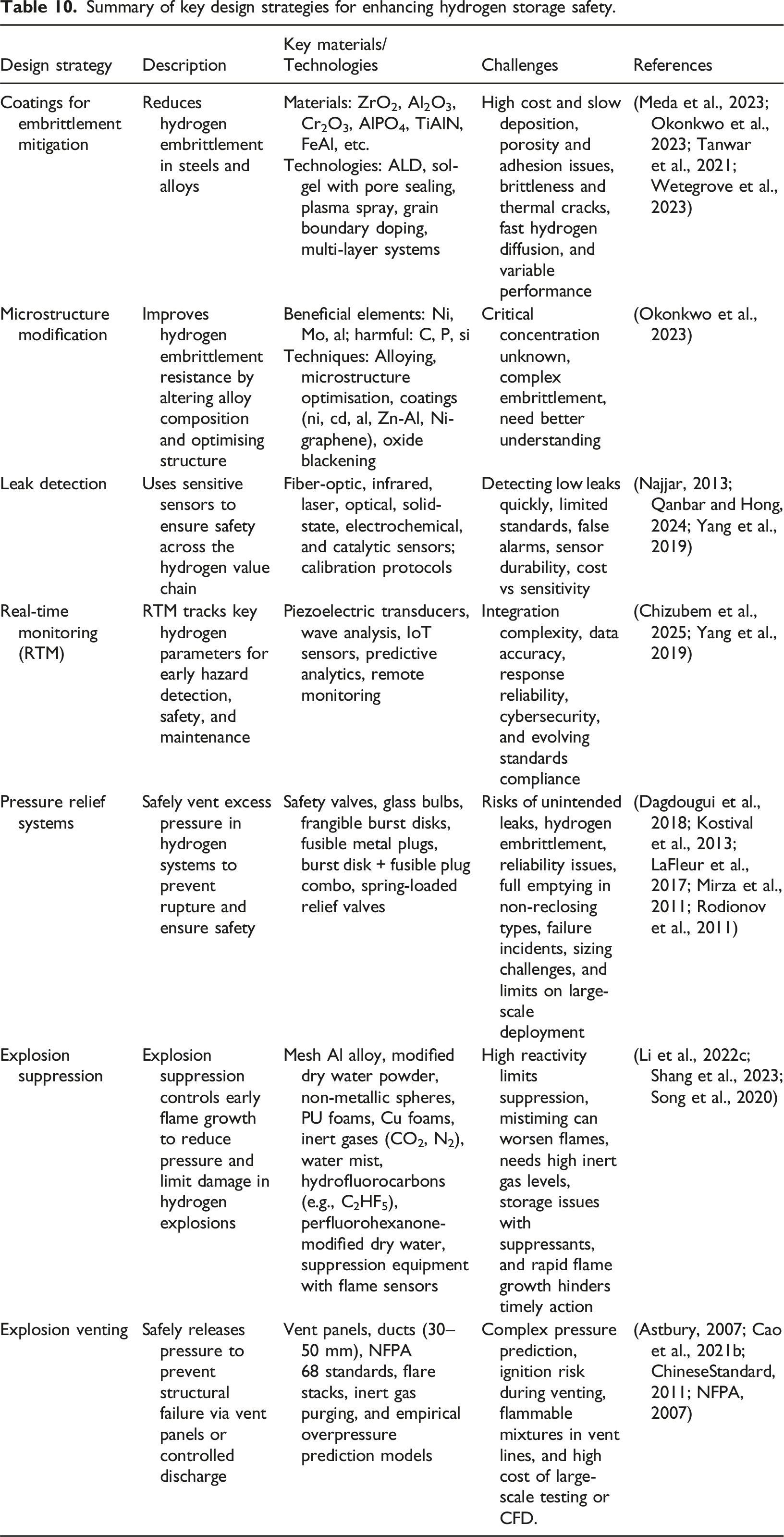
Hydrogen-related accidents have caused extensive damage and serious issues, as discussed in Section Past Incidents of Hydrogen Explosions. Most of these disasters have led to improvements in the design of hydrogen storage technologies. For instance, a fuelling station explosion incident at Sandvika, Norway, paved the way for the MSLV. The MSLV is notable for its cylindrical shell, which has three layers, and its two hemispherical heads, each composed of double layers. Each layer and head serve a specific function. This design combination reduces the chances of hydrogen embrittlement, which can potentially be catastrophic if exposed to an electrostatic charge. Also, diffusion ignition-related incidents may be prevented if a robust design is achieved. Notably, MSLV resist embrittlement due to compatible materials such as 316L stainless steel, which is used for the inner shell cladding. Additionally, integrated safety measures help prevent brittle fractures, allowing only gas leakage. Chinese standards set strict parameters for MSLVs, with a 7.5 m radius, 30 m length, and a maximum pressure of 100 MPa (Faye et al., 2022; Zheng et al., 2008). In addition to MSLVs, a steel-concrete composite pressure vessel, consisting of an inner steel vessel made of SA 724 material and an outer concrete vessel, effectively shares axial and hoop stresses. This feature enables the construction of larger vessels with thinner walls, leading to both size advantages and cost savings. The design allows for leaking before bursting, and it prevents catastrophic failures. As an example, retrofitting a 1.5 MW wind turbine tower with this approach can optimally store 940 kg of hydrogen at 11 bar pressure (Elberry et al., 2021; Zheng et al., 2008). The design strategies described in the following content will be crucial for advancing safe hydrogen storage methods.
Summary of key design strategies for enhancing hydrogen storage safety.
Hazard and risk analysis is important because it helps develop design strategies and anticipate potential accidents. To achieve this, various risk analysis techniques are used, including Hazard and Operability Study (HAZOP), Job Safety Analysis (JSA), Safety Integrity Level Analysis (SIL), Human Reliability Assessment (HRA), Quantitative Risk Assessment (QRA), Failure Mode and Effects Analysis (FMEA), Fault Tree Analysis (FTA), Event Tree Analysis (ETA), Bowtie Analysis, Layers of Protection Analysis (LOPA), What-If analysis, scenario analysis, consequences analysis, and risk matrix analysis. These methods are used to identify and manage potential risks in stationary high-pressure hydrogen storage vessels (Ade et al., 2020; Correa-Jullian and Groth, 2022; Duan et al., 2016; Gye et al., 2019; Moradi and Groth, 2019; Rigas and Sklavounos, 2005; Sánchez-Squella et al., 2022; Xu et al., 2009). Material testing approaches such as fracture roughness test, Charpy impact test, fatigue crack propagation test, fatigue test, and slow strain rate tensile test are also essential for selecting suitable materials, and designing pressurised hydrogen storage vessels ensuring their durability, safety, and resistance to hydrogen-induced damage (Zheng et al., 2012).
In addition to these risk assessment and material testing techniques, identifying ignition sources related to hydrogen releases is crucial. For example, the Major Hazard Incident Database Service (MHIDAS) recorded 81 hydrogen leakage incidents, but ignition sources were identified in only about 15% of these cases. The identified sources included impacts, direct flames, hot surfaces, electric discharges, and friction sparks (Astbury and Hawksworth, 2007). Techniques such as Electrostatic Hazard Analysis, Flame Detection, and spark detection devices help to identify potential ignition sources (Cassutt et al., 1962; Shen-jian et al., 2021). To prevent accidental ignition sources, key design strategies include proper equipment inspection and maintenance practices, adequate ventilation, gas leak detection sensors, and providing training and awareness sessions. Flame detectors quickly identify flames and trigger alarms or automatic responses to prevent explosions, while flame arrestors stop flames from spreading. A flame arrestor usually has perforated plates, layered screens, and slots within a housing to absorb heat and suppress flames (Nolan, 2019; Sutton, 2015). By suppressing flames during their initial growth phase, the risk of explosion is considerably reduced (Shang et al., 2023). In addition, Tzimas (Tzimas et al., 2003) discussed important safety strategies and techniques to mitigate hydrogen-related incidents. Finally, implementing passive fire protection, backup systems, emergency plans, safety training, and the design strategies mentioned above can ensure highly secure hydrogen storage systems.
An active approach under a preventive design strategy is the use of numerical modelling techniques to simulate potential accidental hazards. CFD is a powerful numerical simulation tool that accurately predicts hydrogen dispersion behaviour, fire, and explosion, and supports the development of robust design strategies (Edelia et al., 2018). CFD has grown in popularity and is widely applied in the modern design of hydrogen systems, thanks to advancements in computational resources that enable real-world applications (Tolias et al., 2019). These simulations can assess critical parameters such as generated pressure, flame temperature, flame speed, shock wave speed, shock wave phase durations, heat flux, impulse, safety distances, and dispersion patterns from CFD models to support the development of safe design techniques (Abohamzeh et al., 2021a). CFD tools such as ANSYS FLUENT, FLACS, COM3D, REACTFLOW, AutoReaGas, OpenFOAM, CAST3M, etc., have been used in many research areas of hydrogen release, dispersion, ignition, jet fire, venting of gases, deflagration, and detonation scenarios (Cao et al., 2021a; Tolias et al., 2019). The accuracy of the CFD model heavily depends on combustion models and turbulence models (Tolias et al., 2019). Researchers used combustion models such as the eddy dissipation concept, beta flame model, flame speed model, gradient method, detailed kinetics, and premixed combustion models to simulate the gas explosion (Makarov et al., 2009; Tolias et al., 2014, 2019). Also, commonly used turbulence models for gas explosion include standard k − ε, SST k − ω, renormalisation group large eddy simulations (RNG LES), eddy viscosity large eddy simulations (LES), direct numerical simulations (DNS) (Kim and Kim, 2020; Makarov et al., 2009; Tolias et al., 2019). Previous numerical studies on gas explosions have focused on simplified combustion models without considering detailed chemical kinetics and interaction between turbulence and chemistry (Groethe et al., 2007; Makarov et al., 2009; Xia et al., 2023). In response, the authors developed a novel CFD model that tackles the combustion problem by integrating detailed chemical kinetics through a coupled CFD and chemical kinetics solver (Senarathna et al., 2025). In addition, an efficient turbulence modelling approach known as the zonal LES approach was employed to accurately capture turbulence-chemistry interactions. This technique uses a 24-reaction mechanism to simulate hydrogen explosions and has been specifically applied to an unconfined explosion, for which laboratory experiments have also been conducted. It was concluded that using the technique combined with detailed chemical kinetics provides an efficient solution with accurate results. CFD analysis and FEA techniques are used together to produce solutions for fluid-structure interaction (FSI) problems. It can be effectively utilised under different conditions, including high pressure, dynamic loads, impacts, and elevated temperatures (Wurster, 2016). For example, studies done by Chen et al. (Chen et al., 2023) analysed the effect of hydrogen-air detonation on structures made of aluminium tubes and concrete walls from the coupling of LS-DYNA CESE solver and the Finite Element Method (FEM). Another study done by Wang et al. (Wang et al., 2024) used numerical simulations (FLACS and ANSYS) to investigate the effects of structural stiffness and BLEVE wave duration on reflected BLEVE pressure, proposing prediction charts for accurate BLEVE load and effect predictions on structures. More importantly, experimental studies and validated CFD models pave the way for developing prediction models, such as semi-empirical equations or ML models, which are more practical from a design engineer’s perspective.
As discussed in this section, various strategies exist to prevent accidental explosions, including different vessel configurations to avoid embrittlement and leaks, advanced coating technologies, material microstructure modifications, leak detection systems, ignition prevention systems, real-time monitoring, safety valves, venting systems, explosion suppression equipment, risk assessment tools, flame detectors, flame arrestors, developing strategies using CFD and FEA simulations. Despite the advancements in design strategies, several challenges identified in mitigating explosion incidents in hydrogen storage. One of the significant challenges is the complexity of simulating and predicting hydrogen explosions, which requires advanced numerical models and experimental validation. Development of predictive models, either semi-empirical or machine learning (ML) models, can achieve reasonable reliability and efficiency (Makarov et al., 2009). Recent studies have demonstrated that artificial neural networks (ANNs) trained on CFD-generated hydrogen-air explosion data can provide accurate and near-instantaneous predictions of explosion loads, while also offering interpretability of the key contributing factors (Hu et al., 2024). Another major challenge is scaling up design strategies for larger hydrogen storage systems without compromising safety or efficiency. Furthermore, the development of cost-effective and reliable explosion suppression equipment, venting systems, and risk assessment tools remains a significant challenge. Additionally, the integration of multiple design strategies, such as real-time monitoring, safety valves, and flame detectors, into a single system can be a complex task. Preventing ignition sources in complex industrial environments is also difficult, especially under high-pressure and temperature conditions (Yang et al., 2021). Addressing these challenges is crucial to ensure the safe and efficient design of hydrogen storage systems. Research studies have been conducted by utilising previously discussed methods to produce design guidelines, codes and standards. The following section illustrates such existing codes and standards developed to prevent hazards in hydrogen systems.
Codes and standards for preventing explosions in hydrogen energy systems
Codes and standards are crucial for ensuring consistent design and providing guidelines for the design, manufacturing, and operation of hydrogen systems, including storage and transportation. Codes provide specific rules to follow when establishing facilities, detailing the types of apparatus and equipment allowed within those facilities. They serve as societal agreements that carry the weight of law, ensuring that everyone adheres to the same standards. This promotes safety, reliability, and uniformity across all hydrogen-related facilities and operations. Standards, on the other hand, are voluntary guidelines that provide best practices and technical specifications for consistent quality and performance; both codes and standards for hydrogen-related applications are still in development, with standards serving as essential instructions or regulations for designing and implementing systems (Moretto and Quong, 2022). The United States Department of Energy (DOE) has issued certain guidelines for working with hydrogen equipment and facilities (San Marchi et al., 2017). The Hydrogen Tools database (Tools, 2023, 2024) offers access to more than 400 documents encompassing codes and standards relevant to hydrogen, spanning areas like hydrogen infrastructure, hydrogen fuel cell applications, hydrogen system design, and system testing. Among these, around 350 documents have already been published and have been issued by various esteemed organisations including the Society of Automotive Engineers (SAE), American Institute of Aeronautics and Astronautics (AIAA), American Society of Mechanical Engineers (ASME), Compressed Gas Association (CGA), CSA America, National Fire Protection Association (NFPA), Institute of Electrical and Electronics Engineers (IEEE), Underwriters Laboratories (UL), International Electrotechnical Commission (IEC), CEN/CENELEC, China - Hydrogen Standards, Standards Council of Canada, Korean Standards Association, and numerous others (Tools, 2023).
Codes and standards for safe hydrogen storage and transportation (Source: H2 Tools Portal, EERE).

While codes and standards offer strategies for mitigating various scenarios, case studies provide insights based on specific incidents. For instance, to counteract the potential for catastrophic failures caused by extremely high storage pressure in hydrogen storage tanks at filling stations, it has been recommended to implement cooling with a sufficient coolant flow rate. Additional suggestions include defining minimum distances between compressors and accessible areas for personnel. At fuel stations, the establishment of hydrogen leak detection systems serves as a safety measure to reduce the risk of fire and explosions (Pan et al., 2016; Xu et al., 2009). Similarly, risk mitigation measures have been introduced for stationary high-pressure hydrogen storage vessels. Recommendations include the selection of appropriate materials, structural enhancements, real-time monitoring, and proper facility management to prevent incidents (Xu et al., 2009). The process of mitigating incidents begins with the identification of risks. Therefore, conducting risk analysis, as mentioned in Section (design strategies), for each scenario related to hydrogen storage becomes pivotal in avoiding undesired explosions and accidental events. For instance, Squella et al. (Sánchez-Squella et al., 2022) studied risks linked with hydrogen fuel cell vehicles within underground mining operations, utilising the HAZOP technique. The study yielded insights into deviations, root causes, consequences, and recommended actions for each identified node. In certain cases, risk assessment methodologies like QRA have offered perceptions for decision-making in hydrogen systems, defining dimensions for secure perimeters, recognising hazards, predicting potential consequences, and assessing the likelihood of dangers (Moradi and Groth, 2019).
Considering all aspects of gaseous hydrogen storage, numerous barriers exist across economic, technical, political, environmental, and social dimensions. Economically, there are high investment costs, limited profitability, and a lack of financial incentives. Technologically, challenges include low energy conversion efficiency, immature technology, and safety concerns. Politically, there is inadequate policy support and unclear regulations. Environmentally, there are potential risks and negative impacts. Socially, there is a lack of public acceptance and awareness, conflicts, and a shortage of skilled workers. Addressing these multifaceted challenges is essential for the successful advancement and widespread adoption of hydrogen storage technologies.
Conclusions
This review highlights the hazards of hydrogen, particularly in storage systems, and discusses current methods, technological advancements, and future challenges in safe hydrogen storage. Hydrogen’s potential as a clean energy source makes it a key player in global decarbonisation efforts. However, its flammable nature and explosion risks, like deflagration-to-detonation transitions, require careful attention. As hydrogen storage infrastructure grows, designing systems that can prevent accidents is crucial. Addressing these safety challenges is essential for advancing the hydrogen economy and building public trust.
With the increasing use of hydrogen, accidental explosions across industries highlight the need to understand hydrogen’s behaviour in different environments to prevent failures. Hydrogen explosions can occur as deflagrations, detonations, or jet fires, depending on factors like mixture ratio, volume, confinement, and ignition source. Research highlights the importance of studying these explosion characteristics, as combustion can develop from mild deflagrations to severe detonations with dangerous shock waves. Safer hydrogen storage infrastructure is being developed, with gaseous hydrogen storage methods like Type I to Type V vessels, MSLV, seamless vessels, etc. Ongoing research focuses on preventing leaks and explosions while maintaining high storage density. The behaviour of high-pressure hydrogen gas leaks depends on the location and surrounding environment, with even minor leaks posing risks of ignition and explosions. Confined hydrogen explosions generate higher pressures and more dangerous blast waves than unconfined ones, though unconfined blasts still produce fatal pressures. Venting systems and careful material selection are critical for improving safety. Research focuses on high-strength materials, addressing issues such as hydrogen embrittlement and permeation risks, alongside design strategies like multilayered vessels and real-time monitoring. Ongoing advancements in guidelines and standards should aim to close knowledge gaps and enhance safety in hydrogen storage systems. Despite advancements in design strategies and safety regulations for hydrogen storage, several challenges remain. There is a notable gap in accurately simulating and predicting hydrogen explosions due to the complexity of combustion interactions and the limitations of current numerical models. The development of cost-effective explosion suppression technologies and reliable monitoring systems also requires further research and innovation.
Future research should focus on improving the accuracy of predictive models for hydrogen explosions, especially by incorporating advanced experimental and CFD techniques. Along with this, developing risk assessment tools, machine learning models, semi-empirical models, codes, and the development of codes and standards will be crucial for hydrogen safety. Advancements in explosion suppression technologies and real-time monitoring systems are crucial to enhancing safety in hydrogen storage systems. There is also a need for cost-effective materials and innovative designs that prevent leaks, embrittlement, and autoignition. Lastly, public education and training initiatives should be expanded to build awareness and ensure the safe handling and storage of hydrogen. Hydrogen holds immense potential as a key energy source for a sustainable future, but its safe storage and handling remain critical challenges. By addressing these technical, economic, and regulatory barriers, hydrogen could become a key aspect in the global transition to cleaner energy systems.
Footnotes
Acknowledgement
The authors wish to acknowledge the scholarship provided by the Australian Research Council (ARC), Discovery Project Grant (DP230101133), to proceed with the research in the context of hydrogen safety. Providing all the facilities, including the scholarship, the University of New South Wales (UNSW), Canberra, is gratefully acknowledged.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
