Abstract
Background:
Nonacid gastroesophageal reflux-induced cough (GERC) remains understudied, with limited research on effective treatment options. Recently, neuromodulators such as gabapentin and baclofen have shown promise in managing nonacid GERC.
Objectives:
This study aimed to identify factors associated with response to neuromodulator therapy in nonacid GERC.
Study design:
A retrospective study.
Methods:
We analyzed medical records of patients diagnosed with nonacid GERC who received gabapentin or baclofen as an add-on therapy enrolled between December 2019 and January 2024. Retrospective analysis of general information, cough-related questionnaires, MII-pH parameters, and other assessments was conducted to establish a regression analysis model for identifying multiple factors associated with neuromodulator response.
Results:
In this retrospective cohort study, data from 184 patients were analyzed, with 106 (57.6%) classified as responders and 78 (42.4%) as nonresponders. Clinical factors significantly associated with neuromodulator efficacy included gender (OR = 4.324, p = 0.027), age (OR = 0.803, p = 0.002), and exposure to cough-aggravating factors (OR = 6.345, p < 0.001). Furthermore, multiple regression analysis further identified specific Hull Airway Reflux Questionnaire (HARQ) items—“Cough with certain foods” (OR = 2.523, p = 0.034), “Cough with eating” (OR = 4.445, p < 0.001), and “Cough brought on by singing or speaking” (OR = 5.003, p = 0.007)—as significant predictors. Additionally, Medication Adherence Questionnaire (MAQ) items such as “Forgetfulness” (OR = 0.257, p = 0.005) and “Stopping medication when “feeling better” (OR = 0.787, p = 0.017) were also identified as significant predictors of treatment response.
Conclusion:
Neuromodulators can relieve nonacid GERC in patients unresponsive to standard anti-reflux therapy. Factors such as male gender, younger age, less exposure to cough irritants, and higher HARQ and lower MAQ scores can effectively predict the efficacy of neuromodulators.
Plain language summary
Why was the study done? Nonacid gastroesophageal reflux-induced cough (GERC) remains a topic of discussion, with limited research on effective treatment options. Recently, neuromodulators such as gabapentin and baclofen have shown promise in managing nonacid GERC. This study aimed to identify factors associated with response to neuromodulator therapy (specifically baclofen and gabapentin) in nonacid GERC. What did the researchers do? This study reviewed the medical records of 184 patients treated with baclofen and gabapentin from 2019 to 2024. Patients were categorized into two groups: those who experienced relief from their cough (“responders”) and those who did not (“non-responders”). Statistical analyses were then applied to various factors to identify potential predictors of a patient’s likelihood to benefit from neuromodulators. What did the researchers find? The study found that younger patients, men, and those with less exposure to cough-aggravating factors (like certain odors or irritants) were more likely to respond positively to treatment. Additional predictors included whether a patient’s cough was triggered by specific factors, such as eating, particular foods, or activities like talking or singing. Forgetfulness and stopping medication when feeling better were associated with a lower likelihood of treatment success. Overall, these findings suggest that neuromodulators may be effective for individuals with nonacid GERC, and certain factors can help predict who might benefit the most from this treatment.
Introduction
Gastroesophageal reflux-induced chronic cough (GERC) is a subtype of gastroesophageal reflux disease (GERD) characterized by a predominant cough symptom and is considered a common cause of chronic cough.1–3 According to several studies, GERC accounts for 10%–40% of patients with chronic cough. 4 GERC can be classified into two main subtypes based on its pH value: acid and nonacid GERC. While acid reflux is a well-established cause of GERC, research has primarily focused on its diagnosis and treatment. Nonacid reflux, however, also plays a significant role in GERC, yet it remains understudied compared to acid reflux. This lack of data on nonacid reflux represents a critical gap in our understanding of nonacid GERC. Cough due to nonacid reflux (nonacid GERC) can be defined as a reflux-induced cough where the reflux episodes do not have acidic characteristics (pH ⩾ 4) but yet still cause cough symptoms. Based on the MII-pH results, nonacid reflux can be classified into two categories: weakly acidic reflux with a pH between 4 and 7 and alkaline reflux with a pH greater than 7.2,5
Two primary mechanisms link reflux to cough: (1) micro or macroaspiration of refluxate into the esophagus and airways, and (2) a vagally mediated reflex mechanism known as the esophago-tracheobronchial reflex, which triggers airway reactions without aspiration. 6 Recent studies suggest that in patients with reflux-induced cough resistant to standard anti-reflux therapy, underlying mechanisms may include incomplete acid suppression, nonacid reflux, transient lower esophageal sphincter relaxations, esophageal hypersensitivity, and esophageal dysmotility. Additionally, enhanced cough reflex sensitivity in nonacid GERC may result from sensory nerve stimulation in the esophagus, leading to mast cell activation and tachykinin release in the airways.7,8 Microaspiration of refluxate can further stimulate mechanosensitive and chemosensitive cough receptors in the laryngopharynx and lower airways, potentially exacerbating cough symptoms. Moreover, aspiration-induced airway inflammation may contribute to cough reflex hypersensitivity by modulating afferent neurons responsible for detecting tussive stimuli, thereby amplifying cough.8–10
The typical symptoms of reflux disease, such as heartburn and regurgitation, are present in less than 60% of nonacid GERC patients, 11 which can lead to an underestimation of the diagnostic accuracy of nonacid GERC. Currently, the most effective approach to diagnosing GERC is multichannel intraluminal impedance-pH monitoring (MII-pH). This method can detect all types of reflux, identify the characteristics of refluxate, and establish a temporal association between acid or nonacid reflux and cough. 12 The first-line treatment for GERC is proton pump inhibitors (PPIs) alone or combined with prokinetic medicines, 1 which can alleviate the cough in most patients. However, a significant percentage of patients with nonacid GERC do not respond to the standard anti-reflux treatment, which consists of at least an 8-week course of PPI medications, 13 leading to the need for alternative medical treatments for this condition.
Recently, neuromodulators such as baclofen and gabapentin have been identified as promising treatment options for GERC. Previous studies in our department have shown that 57% of patients with refractory GERC responded well to baclofen treatment.13,14 Baclofen has shown its antitussive efficacy in these patients by decreasing both acid and weakly acid reflux and the number of reflux episodes and reflux-related symptoms.15,16 On the other hand, gabapentin helps reduce cough in 75% of patients with GERC 17 and is helpful for refractory cough of unknown causes. 18 Studies in our department also demonstrated that, as an add-on to acid suppression therapy, both gabapentin and baclofen, with a similar therapeutic success rate, can treat GERC. 19 However, neurological side effects, including sedation, severe dizziness, and drowsiness, may lead patients to discontinue therapy, limiting clinical application and thus requiring cautious use of these medications.
Therefore, this study aimed to identify the potential factors affecting the efficacy of gabapentin and baclofen in patients with nonacid GERC to provide information for clinical decision-making and improve treatment outcomes. Retrospective analysis of the clinical data of these patients was conducted to identify factors associated with the therapeutic response to these neuromodulators.
Materials and methods
Patients
Patients with GERC treated in our department from December 2019 through January 2024 were included in this study. After a detailed medical history inquiry, physical examination, and capsaicin cough sensitivity test, all patients underwent chest X-ray and CT scan, lung function test, histamine bronchial provocation test, induced sputum cytological examination, and MII-pH monitoring to identify the causes of chronic cough according to a step-by-step protocol described previously. 20 The diagnosis of nonacid GERC was based on previous studies,2,5,11,21 the Lyon consensus, 22 and Chinese cough guidelines. 23 Patients were diagnosed with nonacid GERC if they met the following criteria: (a) Cough lasting for ⩾8 weeks, with or without regurgitation or heartburn; (b) results of MII-pH monitoring satisfied (1) and (2) simultaneously,2,11,12,16 where (1) was an AET of<6% and (2) was a symptom association probability (SAP) for nonacid reflux ⩾95% and/or an SI for nonacid reflux ⩾50% and/or nonacid reflux > 80; and (c) cough completely resolved or significantly improved (cough symptom score decreased by> 50%) in response to stepwise anti-reflux therapy.
Inclusion criteria were as follows: (I) Age ⩾18 years old; (II) patients diagnosed with nonacid GERC; (III) patients who received baclofen or gabapentin as an add-on to standard anti-reflux therapy; (IV) Patients able to understand and complete the cough-related questionnaires and other relevant questionnaires such as the MAQ. Exclusion criteria were: (1) Incomplete medical records; (2) patients lost to follow-up; (3) current smokers or ex-smokers of ⩽2 years and those who had the other concomitant causes of chronic cough were excluded; and (4) participants with a history of acute upper respiratory tract infection during the last 2 months prior to the recruitment.
Study design
Data were collected from medical records of patients evaluated for chronic cough and diagnosed with nonacid GERC. Relevant information, including general patient characteristics, cough-related questionnaires, capsaicin cough sensitivity, cough symptom scores, GerdQ scores, MII-pH monitoring parameters, and other assessments, was extracted and analyzed.
Laboratory investigations
Lung function and histamine bronchial provocation tests were conducted per American Thoracic Society 24 and Chinese Respiratory Society 25 guidelines using an Aerosol Provocation System and MasterScreen Diffusion. Induced sputum cytology followed methods in.8,26 Capsaicin cough sensitivity was tested as described by Fujimura et al. 27 with European Respiratory Society modifications. 28 The cough threshold was the lowest capsaicin concentration inducing ⩾2 (C2) or ⩾5 (C5) coughs. MII-pH testing was conducted as previously described. 12 A combined MII-pH catheter with six impedance sensors and a pH electrode was transnasally inserted into the esophagus, positioned at specific intervals above the lower esophageal sphincter. Data were recorded by a portable logger at 50 Hz and analyzed using specialized software to identify and characterize reflux episodes, clearance times, and esophageal exposure to volume and acid. Reflux was categorized by impedance as liquid, gas, or mixed, and by pH as acid (pH < 4) or nonacid (pH ⩾ 4), with nonacid further divided into weakly acidic (pH 4–7) and weakly alkaline (pH > 7). The DeMeester score provided a global measure of acid exposure. The temporal association between reflux and cough was evaluated using an SAP. Prior to MII-pH, the Gastroesophageal Reflux Disease Questionnaire (GerdQ) was used to assess upper gastrointestinal symptoms. 29
Therapeutic regimen
After treatment failure with Standard anti-reflux therapy (omeprazole 20 mg twice daily plus mosapride 10 mg thrice daily), these patients were treated with neuromodulators. All the patients followed a gradual dose escalation schedule at the start of treatment and a 3-week dose reduction schedule at the end of treatment. The dose of baclofen (Novartis, Beijing, China) was 10 mg three times daily on the initial 3 days and then was increased by an extra 10 mg every 3 days until a maximum of 60 mg/day (20 mg, three times daily) was reached or the patients could not tolerate the side effects. Similarly, gabapentin (Hengrui, Jiangsu, China) was given at an initial dose of 100 mg, three times daily, with a 300 mg increase daily every 3 days until a maximal dose of 900 mg a day (300 mg, three times) or the occurrence of intolerant side effects. The patients were instructed in a 3-week dose reduction schedule, resulting in baclofen or gabapentin cessation after the cough disappeared completely or improved (reduction in cough symptom ⩾ 50%).
Relevant questionnaire assessment
The Hull Airway Reflux Questionnaire (HARQ), developed by Morice et al., 30 is a 14-item self-assessment tool that evaluates cough triggers, exacerbating factors, and the severity of related symptoms such as postnasal drip, acid reflux, and heartburn. Patients rate the frequency of each symptom over the past month using a six-point scale (0–5), resulting in a total score ranging from 0 to 70. Higher HARQ scores indicate increased cough sensitivity.
The GerdQ includes six questions related to reflux symptoms. 31 Patients are asked to recall how often they have experienced each symptom over the past week and respond on a scale of 0–3 points based on severity. The total score ranges from 0 to 18 points, with higher scores indicating a higher likelihood of gastroesophageal reflux disease.
The Leicester Cough Questionnaire (LCQ) is a 19-item, self-administered quality-of-life questionnaire for chronic cough. It comprises three domains: physical (eight items), psychological (seven items), and social (four items). 32 The total severity score ranges from 3 to 21, with lower scores indicating more significant impairment of health status due to cough.
The MAQ, developed by Morisky et al., 33 is a widely used four-item self-report tool for assessing medication adherence. 33 Respondents answer “yes” or “no,” scored as 0 or 1. The total score ranges from 0 to 4, evaluating unintentional nonadherence (Items 1–2) and purposeful nonadherence (Items 3–4).
In addition to using HARQ to assess cough triggers and exacerbation factors, information about exposure to cough-aggravating factors was gathered by asking patients an open-ended question during follow-up: “Is there any factor that aggravates your cough?” Respondents answered “yes” or “no,” and their responses were scored as 1 (yes) or 0 (no). If the patient answered “yes,” specific cough triggers were also recorded.
Treatment outcome assessment
Cough severity was assessed using the Chinese version of Hsu et al.’s cough symptom scores, 34 rating daytime and nighttime cough on a 0–5 scale. Therapeutic success (responders) was defined as cough control or a ⩾50% reduction in symptom score after baclofen or gabapentin treatment. Unsuccessful outcomes (nonresponders) included therapy failure, worsening, or no improvement. Additionally, reductions of ⩾50% in symptom scores, GerdQ, HARQ, and LCQ scores were considered successful outcomes, following established guidelines and previous studies.1,13,23,35
Statistical analysis
The normally distributed data are expressed as the mean ± standard deviation (SD), while skewed data are expressed as the median (interquartile range). After logarithmic transformation, C2 and C5 were expressed as the geometric mean ± SD. Kolmogorov–Smirnov (KS) test was performed to evaluate the normal distribution of the quantitative variables. To test the differences between nonparametric variable means in the study groups, the Mann–Whitney U test and independent t test were performed. Chi-square and Fisher’s exact tests were used for qualitative variables. Spearman and Pearson’s tests were used for the correlation analysis. A univariate regression analysis was performed to screen significant variables. Then, multiple logistic regression analysis was performed to evaluate the effects of different factors and predict the therapeutic efficacy of neuromodulators. In addition, Receiver operating characteristic (ROC) curve analysis was performed to evaluate the model equation. SPSS 25.0 software (SPSS, Chicago, IL, USA) was applied for statistical calculation. A 5% probability of a type I error (two tailed) and a power of 80% were considered in the analysis.
Results
General information about the patients
Out of 503 patients diagnosed with GERC during the study, 184 (36.58%) were eligible for inclusion in the study. The clinical characteristics of these patients are presented in Table 1. The exclusions consisted of 10 patients with incomplete records, 249 with acid GERC, and 41 with dual or triple etiologies (refer to Figure 1). The participants comprised 96 females (52.7%) and 88 males (47.8%) with an average age of 48 years (ranging from 18 to 81 years). All patients received neuromodulator treatment, with 90 (48.9%) assigned to the baclofen group and 94 (51.1%) to the gabapentin group. Of these, 106 patients (57.6%) responded to the treatment, while 78 patients (42.4%) did not. Responders had a different gender distribution (p = 0.027), were younger (45.84 vs 51.42 years, p < 0.001), had higher HARQ scores (28.00 vs 25.85, p < 0.001), and lower MAQ scores (1.00 vs 3.00, p < 0.001) compared to nonresponders. No significant differences were observed in BMI, cough duration, symptom scores, capsaicin thresholds, MII-pH, or lung function parameters.
Demographic and clinical characteristics of responders and nonresponders to neuromodulator treatment for nonacid GERC.

Data are presented as mean ± SD, median (interquartile range), or (n, %) unless otherwise indicated.
p < 0.05.
AET, acid exposure time; BMI, basal mass index; FEV1, forced expiratory volume in 1 sec; FVC, forced vital capacity; GerdQ, gastroesophageal reflux disease questionnaire; HARQ, Hull airway reflux questionnaire; IQR, interquartile range; LCQ, Leicester Cough Questionnaire; MAQ, Medication adherence questionnaire; MII-pH, Multichannel intraluminal impedance; SAP, the symptom association probability; SD, standard deviation.

Flow diagram of patient selection.
Difference in HARQ scores between responders and nonresponders
Based on the above results, we observed a significant difference in the total HARQ scores between responders and nonresponders. Therefore, we conducted comparative analyses of each individual HARQ item score between the two groups. Responders rated higher HARQ scores than nonresponders, especially in items including “Excess mucus in the throat or drip down the back of your nose (3.00 vs 2.05, p = 0.033),” “cough with certain foods (3.00 vs 2.00, p = 0.010),” “cough with eating (4.00 vs 3.00, p < 0.001),” and “cough brought on by singing or speaking (5.00 vs 3.00, p = 0.002).” However, the scores of other variables were comparable between the two groups (Table 2).
Comparison of HARQ measures between responders and nonresponders.

Data are presented as Medians (25–75 IQR).
p < 0.05.
HARQ, Hull airway reflux questionnaire.
Difference in MAQ scores between responders and nonresponders
Responders had significantly lower total MAQ scores (1.00 vs 3.00, p < 0.001). For unintentional nonadherence, responders showed better adherence regarding forgetfulness (1.00 vs 2.00, p = 0.004) and carelessness (3.00 vs 2.00, p = 0.018). Among purposeful nonadherence items, stopping medication when feeling better was significantly lower in responders (1.79 vs 2.00, p = 0.003), while stopping when feeling worse showed no significant difference (p = 0.209; Table 3).
Comparison of MAQ measures between responders and nonresponders.

Data are presented as Medians (25–75 IQR).
p < 0.05.
MAQ, Medication Adherence Questionnaire.
Factors associated with the therapeutic efficacy of neuromodulators
Variables with significant differences between responders and nonresponders were selected for univariate logistic analysis. Multivariate logistic regression identified gender (OR = 4.324, p = 0.027), age (OR = 0.803, p = 0.002), and exposure to cough-aggravating factors (OR = 6.345, p < 0.001) as independent predictors of neuromodulator efficacy (Figure 2). Significant HARQ items included—“cough with certain foods” (OR = 2.523, p = 0.034), “cough with eating” (OR = 4.445, p < 0.001), and “cough brought on by singing or speaking” (OR = 5.003, p = 0.007. MAQ items such as “forgetfulness”(OR = 0.257, p = 0.005) and “stopping medication when feeling better” (OR = 0.787, p = 0.017) were also independent predictors (Table 4). The final model, Logit(p) = 2.563 + 0.115X₁ − 0.220X₂ − 0.786X₃ − 0.134X₄ − 0.150X₅ − 0.213X₆ − 1.359X₇ − 0.239X₈, explained 79.5% of the variance (R² = 0.795) and demonstrated high predictive accuracy (F = 232.12, p < 0.001). The Hosmer–Lemeshow test confirmed good fit (χ² = 5.286, p = 0.727), and the Durbin–Watson statistic was 1.975, indicating no autocorrelation.

Forest plot of clinical factors associated with neuromodulator treatment response in nonacid GERC patients: overall, factors such as male gender, younger age, and specific HARQ items related to cough triggers were significantly associated with a positive response to neuromodulator therapy, while poor medication adherence and female gender negatively impacted outcomes.
Simple and multiple regression analyses of clinical factors affecting neuromodulator treatment response.
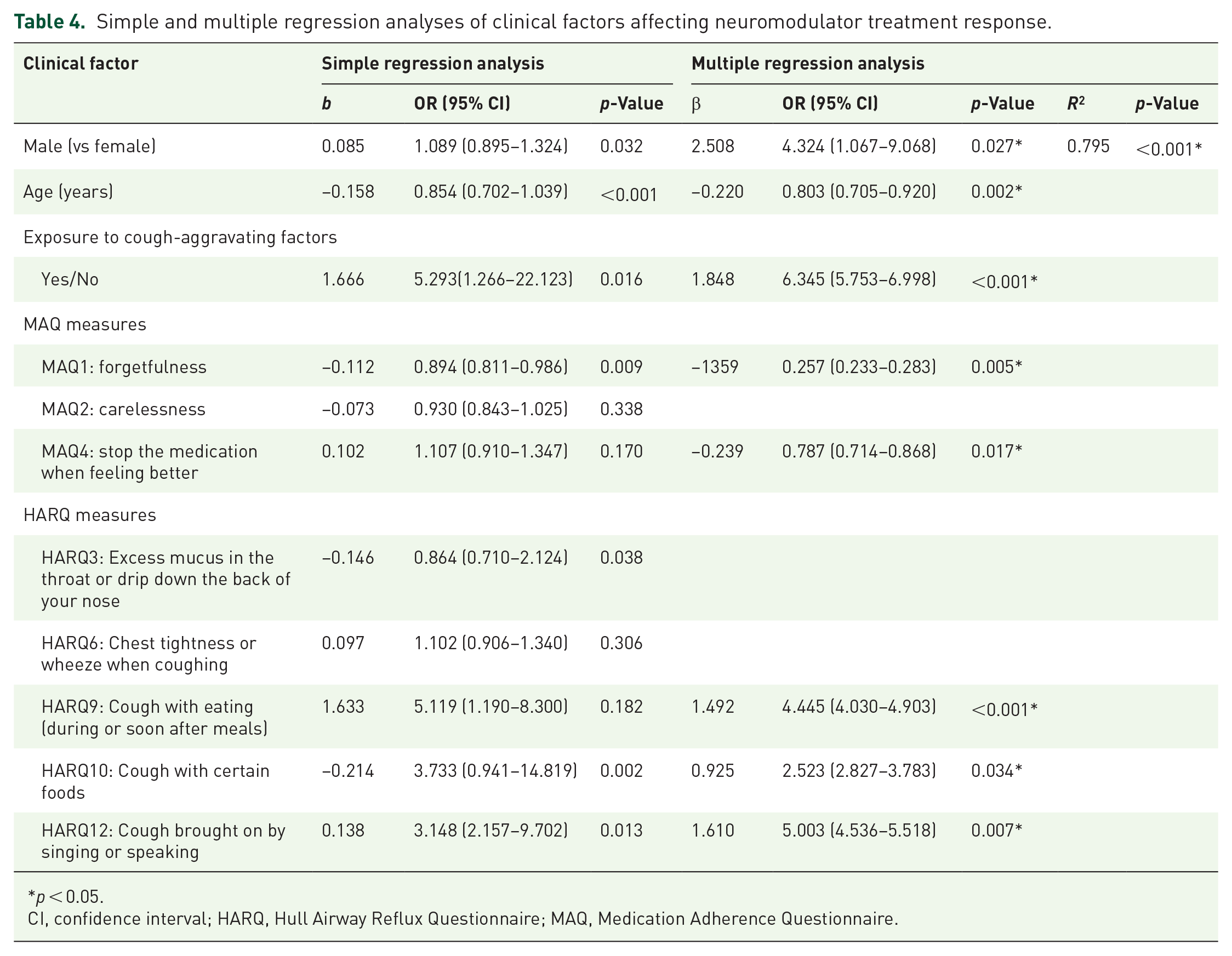
p < 0.05.
CI, confidence interval; HARQ, Hull Airway Reflux Questionnaire; MAQ, Medication Adherence Questionnaire.
When using a cutoff point of p ⩾ 0.6425, the logistic regression model effectively distinguished between responders and nonresponders to the treatment. This cutoff point was validated through ROC analysis, which demonstrated that the model could identify responders from nonresponders with 82.10% sensitivity and 63% specificity. The area under the curve (AUC) was 0.820, with a 95% confidence interval (CI) of 0.770–0.871 and a p-value of <0.001. This AUC value indicates a high discriminative capacity, with an 84.5% probability that, in any given pair consisting of one responder and one nonresponder, the model will correctly identify the responder. Therefore, the logistic regression model exhibits strong discriminative efficacy in differentiating between responders and nonresponders within the study population (Figure 3).

The internal accuracy of the logistic regression model was evaluated using the ROC curve. The AUC was found to be 0.820 (95% CI: 0.770–0.871; p < 0.001), indicating that in 82.10% of paired participants (one responder, one nonresponder), the responder scored higher. These findings suggest that the logistic regression model used in the study had a moderately good ability to differentiate between responsive and unresponsive participants.
Discussion
This retrospective database analysis aimed to identify predictive factors for the response to neuromodulator therapy, specifically gabapentin and baclofen, in patients with nonacid GERC. Our study identified several key factors associated with therapeutic efficacy, offering valuable insights for clinical decision-making and potential improvements in treatment outcomes. The findings demonstrated a significant association between several factors and treatment response, emphasizing the complex interplay of various aspects of patient health. Biological factors, including gender and age, were significant predictors of treatment response. Specifically, male gender and younger age were linked to a better response to neuromodulator treatment. Additionally, patients who responded positively to treatment exhibited lower MAQ scores, indicating good medication adherence. These patients were also exposed to fewer or no cough-aggravating factors and had higher HARQ scores, which alone might predict the therapeutic efficacy of gabapentin. A significant proportion of patients in this study demonstrated a favorable response to the prescribed baclofen or gabapentin treatment. The diagnostic approach for nonacid GERC in this study involved meticulous screening to exclude alternative causes of cough with a structured and stepwise diagnostic approach. Integral to this was the use of comprehensive objective measurements from MII-pH monitoring, essential for securing a definitive diagnosis.
In our study, responders to neuromodulators exhibited significantly higher HARQ scores, which were positively associated with the efficacy of both baclofen and gabapentin. The HARQ, developed by Morice et al., 30 assesses cough hypersensitivity, evaluating sensitivity to stimuli such as cold air, talking, eating, laughter, and odors, unlike the GerdQ. HARQ effectively predicts both acid and nonacid GERC. 36 Multiple logistic regression identified specific HARQ items—“cough with certain foods,” “cough with eating,” and “cough brought on by singing or speaking”—as independent predictors of treatment efficacy, indicating central cough hypersensitivity and airway reflux.37,38 Baclofen inhibits TLESRs to block reflux events, while gabapentin targets central hypersensitivity, 19 making these HARQ items reasonable predictors of treatment efficacy. Although HARQ cannot definitively measure central sensitization, certain items may reflect central cough hypersensitivity and airway reflux. Therefore, HARQ may assist in screening patients with nonacid GERC suitable for baclofen and gabapentin therapy, given its moderate-to-good predictive ability for therapeutic efficacy.
This study found that medication nonadherence, as assessed by the MAQ, negatively impacted the response to neuromodulator therapy. The MAQ, 33 widely employed in treatment studies, categorizes nonadherence into purposeful and unintentional. Both purposeful and unintentional nonadherence, particularly “forgetfulness” and “discontinuing medication when feeling better,” were associated with reduced therapeutic responses to baclofen and gabapentin. Responders had lower MAQ scores, indicating better adherence, whereas nonresponders had higher scores, reflecting poor adherence. Medication adherence refers to medication-taking behavior that significantly impacts patients’ health outcomes, although it is not directly related to the pharmacokinetics or pharmacodynamics of drugs. For instance, elderly patients with multiple chronic conditions and younger patients with busy lifestyles often forget to take their medications. 39 Baclofen and gabapentin require specific dose escalation and tapering schedules to optimize their effectiveness and minimize side effects. 35 However, when patients forget or fail to adhere to their treatment regimen, missing doses can result in inconsistent drug levels, thereby reducing the overall effectiveness of the medication. Additionally, prematurely discontinuing medication when symptoms subside can cause relapse or prolong symptoms. Therefore, our findings underscore the importance of considering medication nonadherence as a critical risk factor for neuromodulator efficacy.
Our study identified a potential gender difference in the effectiveness of neuromodulators, with male patients showing a higher likelihood of better treatment outcomes compared to female patients. This difference could be attributed to several factors. Research has demonstrated that the balance of GABAB presynaptic and postsynaptic activities varies between men and women, with differences in GABAergic receptor activity and spontaneous inhibitory currents, potentially contributing to the observed disparities in gabapentin and baclofen responses. 40 Studies have also reported significant gender differences in tolerability and dropout rates among patients taking baclofen. For example, a randomized controlled trial by James et al. 41 found that women receiving higher doses of baclofen were more likely to experience side effects such as sedation and drowsiness, often leading to discontinuation or dose reduction. Gabapentin may be preferable for women due to its fewer side effects, but whether there exists a gender difference in the cough-suppressing effects of baclofen and gabapentin remains unclear and requires further investigation.
Age was identified as a significant predictor, although it alone does not directly predict the response to treatment. Younger patients exhibited significantly better responses to baclofen and gabapentin compared to older patients. This may be attributed to elderly patients facing challenges with complex medication regimens, potential drug interactions with medications for chronic conditions, and age-related changes in drug metabolism. 42 Additionally, younger patients may respond more effectively to certain medications due to factors such as better overall health, fewer comorbidities, and differences in pharmacokinetics and pharmacodynamics. Although age alone is not a strong enough predictor to identify nonacid GERC patients most likely to respond to gabapentin and baclofen or to predict therapeutic success. However, considering age alongside other factors such as medication nonadherence, polypharmacy, and overall health status can help achieve better treatment outcomes.
This study also revealed a significant association between exposure to cough-aggravating factors and treatment response. The predictive value of cough-aggravating factors in identifying subjects at risk of prolonged cough or treatment failure should be recognized. A survey demonstrated that several cough-aggravating factors, such as nighttime or early morning, weather changes, pollen season, and the common cold, were significantly correlated with prolonged cough. The study further suggested that the presence of these factors, in the absence of severe allergic conditions, may indicate underlying cough hypersensitivity. 37 Cough hypersensitivity, triggered by various stimuli, is a common feature among chronic cough patients, with different underlying causes exhibiting distinct trigger profiles. For instance, a meal-triggered cough is often indicative of GERD. In such cases, reflux-induced microaspiration or neuronal sensitization via esophageal-bronchial crosstalk is thought to be the primary mechanism underlying the cough in GERC.38,43 In the present study, nonresponders to therapy reported a higher incidence of exposure to cough-aggravating factors compared to responders. This result suggests that nonresponders may have heightened cough sensitivity or may be exposed to persistent triggers such as allergens, irritants, or environmental conditions that exacerbate their symptoms, reducing their cough’s responsiveness to therapy.
Our study reflects the interaction of multiple factors that may predict the therapeutic efficacy of baclofen and gabapentin. However, several limitations of this study must be taken into account. First, the retrospective design limits the ability to establish causality and introduces potential selection bias due to reliance on existing medical records. Second, being conducted at a single center may limit the generalizability of our findings, as our patient population and clinical practices may not reflect those in other settings. Third, the information on cough triggers in this study was recorded during follow-up, which may not accurately reflect exposure patterns during the initial stages of therapy, limiting the ability to assess the temporal relationship between trigger exposure and therapeutic response. Fourth, this study does not provide a detailed calculation or justification of the sample size, which could affect the validity and reliability of our findings. Future studies should include rigorous sample size estimation to ensure adequate statistical power. Additionally, while our logistic regression model shows promise, it has only moderate predictive ability for the therapeutic success of gabapentin and baclofen. Future research should incorporate a broader range of variables, prospective data collection, and appropriate sample size determination to better predict neuromodulator efficacy in nonacid GERC patients.
Conclusion
In conclusion, gender, age, exposure to cough triggers, MAQ, and HARQ moderately distinguished nonacid GERC patients responsive and unresponsive to baclofen and gabapentin. Some HARQ and MAQ items were equally important as independent predictors. Therefore, HARQ might be useful for identifying nonacid GERC patients who are most likely to respond favorably to baclofen and gabapentin. In addition, implementing strategies to improve medication adherence and reduce exposure to cough triggers could optimize treatment outcomes.
