Abstract
Cardiovascular disease (CVD) still poses a significant risk for morbidity and mortality in patients with chronic obstructive pulmonary disease (COPD). For a long time, among functional parameters, only the forced expiratory volume in 1 s (FEV1) has been considered as predictive of cardiovascular (CV) mortality especially in elderly patients in fact, there is evidence that reductions in lung function indices can increase the risk of ischaemic heart diseases and cerebrovascular diseases, independently from other risk factors. Now, there is considerable evidence suggesting that hypoxemia, systemic inflammation, oxidative stress and hyperinflation may lead to an early sub-clinical CV involvement in patients affected by COPD. Ageing in itself impacts specific aspects of the CV system, including reduced beta-adrenergic responsiveness, increased vagal tone and myocardial and vascular stiffness, endothelial dysfunction, diminished arterial baroreflex and compromised diastolic function. The complex involved interactions include ageing mechanisms as well as multiple known and unknown (e.g. genetic) risk factors. CVDs are leading causes of mortality in individuals with impaired lung function and the two entities commonly coexist with poor outcomes in patients experiencing both conditions. However, the precise mechanisms responsible for this association remain largely unknown. In this narrative review, we summarize current knowledge regarding the co-occurrence of COPD and CVD focusing on the shared biological pathways and biological mechanisms involved in these conditions.
Keywords
Introduction
Chronic obstructive pulmonary disease (COPD) is characterized by partially reversible airflow obstruction affecting large and small airways, lung parenchyma and the pulmonary vasculature. 1 It is the most common chronic respiratory disease with a global prevalence that exceeds 10% of the adult population and has merged as a leading cause of worldwide mortality and morbidity. 2 Recently, a substantial body of evidence pointed out that COPD is not solely a lung disease but also a systemic condition, particularly impacting the cardiovascular (CV) system, contributing to atherosclerosis and promoting diseases such as myocardial infarction and arrhythmias, stroke and systemic arteriopathies.3,4 The high prevalence of cardiovascular diseases (CVDs) among COPD patients can be partly attributed to well-known risk factors such as smoking, sedentary lifestyle and poor diet. However, studies have revealed that impaired lung function is associated with CV risk even after adjusting for known CV risk factors including age, sex, smoke, cholesterol and CV-related mortality ranks highest among causes of death in COPD patients. 5 Research studies6,7 have emphasized the importance of a reduced FEV1 as an independent risk factor for CV mortality, showing that among the lower and the higher quintile of FEV1, the risk of death for CV cause is increased approximately by 75% in both women and men. Moreover, more recently, an observational study investigating the relationship between accelerated FEV1 decline and risk of CVD outcomes including mortality in COPD patients observed that frequent and severe acute exacerbations of COPD (AECOPDs) were associated with risk of CVD outcomes and mortality. 8 These increased morbidity and mortality may represent the ‘tip of the iceberg’ of unknown early CV alterations in COPD patients. Hence, there is compelling evidence claiming for an early CV risk stratification of COPD patients in the daily clinical practice to provide an appropriate diagnosis and management; nevertheless, specialists often focus on their field while an evaluation of global CV risk would be more effective to prevent a detrimental impact. Additionally, the underdiagnosis and under-treatment of CVD in COPD patients may also depend on the lack of guidelines for the management of comorbidities in these patients. The present review provides a synthesis of current knowledge on the co-occurrence of COPD and CVD focusing on the shared biological pathways and biological mechanisms involved in the development of these conditions.
COPD and CVD co-occurrence: prevalence and comorbidities
Cardiovascular comorbidities are commonly associated with COPD sharing similar risk factors and pathophysiological mechanisms, and frequently occur in the same individual as multimorbid disease making the differential diagnosis difficult. Although the prevalence of CVD in patients with COPD is highly variable throughout studies, depending mainly on the selection of study population (cohorts recruited from primary or secondary care), disease severity, number of comorbidities, methodology and adjustment for confounding factors, the majority of evidence describe an increased prevalence of CVD in COPD patients relative to general age-matched population. A systematic review and meta-analysis of observational studies by Chen et al. 9 showed a two-fold increase in the odds of having any CVD including coronary artery disease (CAD), cardiac dysrhythmia, heart failure (HF) and pulmonary and peripheral vascular disease (PVD) in people with COPD compared to the general population relative to COPD-free patients (odds ratio (OR) = 2.46; 96% confidence interval (CI): 2.02–3.00). 10 Interestingly, it is important to point out that the increase in the prevalence of CVD in COPD patients occurs even after adjustment for age, smoking habit and other shared risk factors. An increased risk of acute events including myocardial infarction and stroke in patients with COPD is now well established,11,12 while studies investigating single risk factors, that is, the age-specific relative risk for CVDs in COPD, have demonstrated an inverse relationship between the relative risk of developing CVD and increasing age in patients with COPD. 13 Moreover, an increased CV risk has also been associated with a more rapid rate of decline in lung function (FEV1) in COPD patients.14,15 This is of great importance since CV mortality is greater in mild to moderate COPD patients. 16 Recent epidemiological studies conducted in selected groups of patients presenting in secondary care with advanced COPD disease suggest that probably, the prevalence of CVD in mild disease is still underestimated and needs to be recognized. 17 On the other hand, observational studies have highlighted also the reverse condition finding that COPD is commonly present in patients with different CVDs. In HF patients, for example, the prevalence of COPD varies between 13% and 39%. It has been observed a prevalence of 30% of COPD among patients with ischaemic heart disease (IHD). 18 One study reported a prevalence of COPD between 13% and 39% in patients with atrial fibrillation (AF). 19 The Global Initiative has highlighted coexisting COPD and CVD for Chronic Obstructive Lung Disease (GOLD) as being highly important since CVD is associated with a range of unfavourable outcomes and is responsible for reduced quality of life. Mortality for cardiac causes in COPD patients is highly dependent on the study population and may vary between 12% and 60%. 20 Studies have shown a proportion of 40% of deaths in COPD patients with a CV history. 21 When considering CVD as a prognostic factor for negative outcomes in patients with COPD, it has been reported that coexisting CVDs have consistently been linked to higher rates of hospitalization, in-hospital mortality and 30 days re-admission, as well as increased length of stay in COPD patients 22 and that COPD patients have worse prognosis when suffering for an acute cardiac event, or stroke. 23 Moreover, it has also been suggested that the presence of CVD prolongs the course of COPD exacerbations, worsens physical inactivity and is associated with low scores for quality-of-life measures. 24
Heart failure
Given that the diagnosis of HF in COPD patients may be challenging especially in emergency settings, 25 it has been estimated that the prevalence of HF in COPD in the European population is common, ranging from 7% to 42%, significantly higher compared to the non-COPD population, and it is increased in hospitalized COPD patients. 4 The reason for this high prevalence has been debated and attributed to risk factors shared by the two conditions, that is, the contribution of tobacco smoking, arterial hypertension, ageing, dyslipidaemia, an unhealthy lifestyle and the chronic inflammatory burden COPD-associated. Hypoxemia, lung hyperinflation and systemic inflammation may increase the risk. Furthermore, the prevalence of HF in patients hospitalized for COPD exacerbations is approximately 30%, reaching almost 75% in those receiving mechanical ventilation. 26 Large epidemiological studies report that COPD patients with concomitant HF are more prone to hospitalizations and at an increased risk of hospital readmission, and have a poor prognosis in terms of acute exacerbations with a worse short and long-term prognosis. 27 Conversely, the prevalence of COPD in patients with HF lies in the range of 13.0%–39.0%, 28 and it has been observed that the presence of COPD may increase the prevalence of comorbidities such as arrhythmias, renal dysfunction, diabetes mellitus, higher heart rate and systolic hypertension that trigger HF decompensation and worsens the prognosis of patients hospitalized with HF. 29 One explanation may be that patients with COPD and concomitant HF seem to remain less likely to receive adequate HF treatment with, that is, beta-blockers. 30 Recent research aimed to propose a care bundle, based on clinical guidelines, to guide clinicians on the best practices for the identification and management of COPD patients with concomitant HF with a focus on the best practices to distinguish COPD and HF, especially in emergency care settings. 31
Ischemic heart disease and coronary artery disease
IHD is one of the most frequent cardiac comorbidities among patients with COPD, its frequency in stable disease seems to range between 7% and 33% reaching almost 20% in patients admitted to hospital for exacerbations. 32 Furthermore, patients with co-existing COPD-IHD have a two-fold increased chance of being hospitalized, while IHD is one of the leading causes of death for this group of patients.33,34 A considerable portion of patients (10%–17%) with a confirmed diagnosis of COPD have experienced an acute myocardial infarction, and COPD roughly doubles the risk of myocardial infarction, a clinical outcome associated with a worse prognosis; hence, all these patients should have IHD taken into consideration and treated. 35 Moreover, recent research has reported that COPD patients with clinical manifestations of IHD have significantly decreased exercise capacity, more severe dyspnoea, a decreased body mass index, more significant airflow obstruction and a worse quality of life. 24 Among 20.0%–60.0% of COPD patients suffer from CAD, although the data may vary across different studies 36 while about 8% are diagnosed with peripheral artery disease which is a condition similar to CAD, characterized by atherosclerotic plaques leading to occlusion of the arteries of the lower limbs, associated with a clinically significant decline in functional ability and health status. 37 As expected, the prevalence of CAD in COPD patients increases with the severity of airway obstruction. 32 In a large cohort of COPD patients, a systematic analysis of the Global Burden of Disease Study conducted in 2017 found that the most common cause of early mortality in COPD was CAD, accounting for more than 1 million deaths worldwide. 38 For this reason, the challenges to implementing tools to accurately assess CV risk in COPD patients conducted the development of specific risk calculators to screen IHD by algorithms predicting the presence of CAD in COPD patients. 39 This may be of great importance since certain COPD centrilobular emphysema-phenotype patients seem to be more prone to develop coronary calcifications suggesting a role of emphysema and hyperinflation in the development of atherosclerosis.40,41
Arrhythmia
It has been shown an elevated rate of the prevalence of arrhythmia in patients with stable COPD ranging between 5% and 15% and reaching 20% and 30% in patients with more severe lung function impairment 19 as assessed by two systematic analyses.9,36 AF represents the most frequently supraventricular sustained arrhythmia in COPD, with an estimated prevalence among stable COPD patients of between 4.7% and 15.0%, reaching rates ranging from 20.0% to 30.0% in patients with very severe disease. 42 During hospitalization for COPD exacerbation, it has been observed an increased rate of arrhythmia rose to 27%, while AF was observed in one-fifth of the patients 32 and it is more frequently associated with a low PaCO2 (partial pressure of carbon dioxide) and a higher pulmonary artery systolic pressure. 43 Some studies found increased mortality rates among patients hospitalized for COPD exacerbations and the coexistence of AF. 44 Furthermore, COPD patients with AF have a worse health-related quality of life (assessed by HRQoL). It has been suggested a bidirectional interlink between the two entities, as COPD exacerbations that may trigger AF, while AF per se is also a risk factor for COPD exacerbations. 45 On the other hand, the presence of COPD seems to increase the risk of hospital admission due to AF. 46
Systemic hypertension and stroke
Systemic hypertension is often present in COPD patients, and, even though it does not directly affect the progression of COPD, it represents the main vascular risk factor for the development of CVD. 47 Consequently, in COPD patients, systemic hypertension can induce the onset of HF and IHD, aggravating the prognosis of COPD. 48 The reported prevalence in COPD patients ranges between 28.5% and 64.7%. 49 Therefore, all patients with combined COPD-systemic hypertension need a proactive assessment to find a treatment strategy for both diseases and eventually mitigate the increased CV risk.
COPD is a recognized risk factor for stroke. Stroke prevalence in stable COPD patients does not generally exceed 10% but can reach approximately 20% in hospitalized patients, especially those hospitalized with COPD exacerbation; moreover, the diagnosis of COPD increases the risk of having a stroke. 11
Comorbid COPD-OSA
Finally, the combined presence of COPD and obstructive sleep apnoea (COPD-OSA overlap syndrome) has been described as an important comorbidity in CVD. 50 OSA is a frequent condition in patients with CV diseases and sometimes a co-presence of COPD in these patients may be observed; the overlap syndrome is more prevalent in moderate–severe COPD compared to mild COPD patients. 51 Comorbid COPD-OSA may have an important clinical impact on patients due to worse sleep quality, 52 an increases risk of exacerbations and medical costs. 53 It has been observed an association between COPD-OSA overlap syndrome and an increased risk of hypertension and peripheral arterial disease compared to patients with OSA alone, suggesting an increased global CV risk. Among the pathogenetic mechanisms, it has been suggested the contribution of an increased hypoxemic burden, especially nocturnal hypoxia 54 but also endothelial dysfunction, systemic inflammation and an increased sympathetic nerve activation have been observed. 55
Biological mechanisms of CVD in COPD: shared risk factors and pathogenesis
COPD and CVD share some common risk factors and tobacco smoking is recognized to be one of the most important factors contributing to the development of COPD but also promoting atherosclerosis, including CAD and coronary plaque instability, in this way contributing to the complex interlink between the two different comorbidities. 56 The endothelial dysfunction is typical in COPD patients and it is associated with reactive oxygen and nitrogen species, cytokines and chemokines, that contribute to the progression of both lung and CVDs. 57 The complex mechanism linking the contribution of smoking to CVD is still not fully explained, it has been hypothesized a systemic inflammatory response is a possible cause of a series of events leading to airflow obstruction with the remodelling of small airways, lung parenchyma destruction and progressive loss of normal mechanisms of repair and defence, 58 but also a tobacco smoke effect on endothelial dysfunction with vessels damage and arterial stiffness, hypoxia and oxidative stress, the development of a hypercoagulative state promoting the overlap between COPD and CV consequences. Recent studies have shown that smoking cessation is associated with a significant reduction of CVD, hence the relevance of a primary CVD preventive strategy for smokers. 59 Moreover, COPD represents a heterogeneous condition and it has been argued that specific COPD phenotypes may be associated with an increased CV risk and mortality. Some authors have suggested tracking CV risk stratifying individuals with a COPD subtype classification scheme including CT and pulmonary function and identifying individuals with greater risk for mortality. Their research highlighted that individuals can belong to an airway-predominant disease (APD) and an emphysema-predominant disease with a unique association to spirometric patterns, different imaging characteristics, biomarkers and mortality. Specifically, they found that those in the APD group are at greater risk for CV mortality. 60 The opportunity to identify high-risk COPD subtypes characterized by the underlying severity of disease processes may provide targets for interventions to reduce pulmonary function decline and mortality.
COPD phenotypes and CV risk: the genetic hypothesis
The study of genetic determinants of COPD-related phenotypes is a promising approach to understand COPD heterogeneity, and the proliferation of genome-wide association studies (GWAS) together with the accessibility of whole-genome sequencing has led to a substantial increase in the understanding of genetic variants playing a role in COPD susceptibility and COPD-related phenotypes. 61 Emphysema, airway wall thickening and expiratory gas trapping assessed by chest CT may reflect different pathophysiologic processes leading to airflow obstruction and thus may be important endotypes of COPD. 62 Chest CT phenotypes are an especially promising assessment to understand COPD heterogeneity.63,64 Data from COPD Gene (Genetic Epidemiology of COPD), a multicenter, longitudinal study of 10,000 current and former cigarette smokers showed over 20 genetic loci convincingly associated with COPD, and additional loci demonstrating association with COPD-related phenotypes such as emphysema, chronic bronchitis and hypoxemia. 65 A subsequent GWAS in 35,735 cases and 222,076 controls including data from the UK Biobank and previous studies from the International COPD Genetics Consortium identified 82 loci (of which 47 were previously described) in association with either COPD or population-based measures of lung function, 13 new loci were associated with lung function. The authors, using gene expression and regulation data, identified functional enrichment of COPD risk loci in lung tissue, smooth muscle and different lung cell types. The authors also examined all genome-wide significant loci looking for trait-associated variants. COPD is well known to have many comorbidities, such as CAD. The researchers found, at individual loci, four genetic variants associated with CAD (near CFDP1, DMWD, STN1 and TNS1) giving evidence of shared risk factors between COPD and CV disease. 66 More recently, some authors, in the largest study assessing lung-function phenotypes using GWAS and integrated genomics, reported pathways that have not been previously implicated in COPD, that is, the elastic fibre formation, PI3K-Akt signalling, integrin pathways, hypertrophic cardiomyopathy and dilated cardiomyopathy pathways. 67 A recent important issue from COPD genetic studies is to translate the effect of genetics on clinical risk. GWAS in COPD has identified risk variants with lower odds ratios, although combining multiple genome-wide significant variants, and, applying approaches that can leverage all variants and combine them into a single score per individual, there is an improvement in risk prediction. These genetic risk scores, named polygenic risk scores (PRS), are also associated with important COPD characteristics, such as quantitative computed tomography emphysema and airway features raising the possibility that PRS may predict the overall risk and reflect the risk of specific traits of COPD. 68 The reported findings in COPD genetics may have clinical and translational implications, that is, high genetic-risk individuals identified by common variant aggregation could be targeted for counselling on avoidance of harmful occupational exposures and smoking; or could be identified for clinical trials. 69 New insights from phenome-wide association studies, which explore the association between a given genetic variant and multiple phenotypes, may provide further insight into the contribution of genetic variants to specific mechanisms underlying lung function and a wide range of other phenotypes (via specific pathways). 70
Systemic inflammation
Many studies, during this time, have provided evidence of a link between COPD and systemic inflammation.71,72 As a consequence, a pro-inflammatory state has been suggested as a possible mechanism that links COPD to an increased risk for CVD. 5 Indeed, cigarette smoke can trigger the activation of the systemic inflammatory cascade in susceptible individuals. 73 Although some subjects may experience reduced inflammation after quitting smoking, in many cases, inflammation persists and becomes self-perpetuating. 74 A combination of reduced FEV1 and active smoking status exerts an impact on the levels of C-reactive protein (CRP), a widely used marker of systemic inflammation. 75 CRP, recognized as a significant risk indicator for CVD, 76 exhibits elevated levels even in stable COPD patients and may play a crucial role in the advancement of atherosclerosis in this population. Atherosclerosis is acknowledged as a chronic inflammatory disease of the arterial wall, induced by both traditional and non-traditional risk factors, and mediated through inflammatory and immune responses. The presence of a substantial inflammatory component is well established in atherosclerosis. 77 CRP upregulates the production of pro-inflammatory cytokines and tissue factors in monocytes, promotes LDL uptake in macrophages and directly triggers the expression of adhesion molecules in endothelial cells (ECs). 78 In addition to this, CPR may also deposit directly in the arterial wall during the atherogenic process, interacting with other inflammatory mediators to create foam cells, which serve as the building blocks of the atherogenic plaques. 79 On the other hand, serum fibrinogen may also promote atherosclerosis, by increasing blood viscosity and acting as a cofactor for platelets (PLT) aggregation. 80 Data also suggest a potential role of acute lung inflammation in triggering acute cardiac events by influencing the coagulation cascade through increased plasma fibrinogen and IL-6 levels, as observed in COPD exacerbations 81 (Figure 1). A potential role of endothelial dysfunction with consequent PLT activation as a mechanism of thrombotic risk in COPD patients has also been suggested. Several studies have underlined the role of endothelium dysfunction and PLT activation as significant players in the development and progression of COPD. Indeed, endothelium plays a crucial role in vascular homeostasis and impairment, due to the inflammation process enhanced by smoking.82,83 Chronic inflammation and endothelial dysfunction have been proven to drive PLT activity. Platelets are crucial for haemostasis and, once activated by a number of different factors, can mediate endothelium adhesion, and the rolling and activation of other circulating cells, such as neutrophils, responsible for tissue damage during the inflammatory process, 84 especially during exacerbations. 85 Consequently, in COPD patients, chronic inflammation does not impact only lung parenchyma but potentially involves all systems, including the endothelium of blood vessels. Impaired endothelium has several consequences, such as reduced vasodilation capacity, enhanced blood coagulation and increased PLT activation resulting in a higher risk of thrombosis in COPD patients that might explain the higher percentage of CV death in such patients. Endothelium dysfunction and PLT activation may be potential targets of therapy in patients with COPD aiming to reduce their risk of CV events.
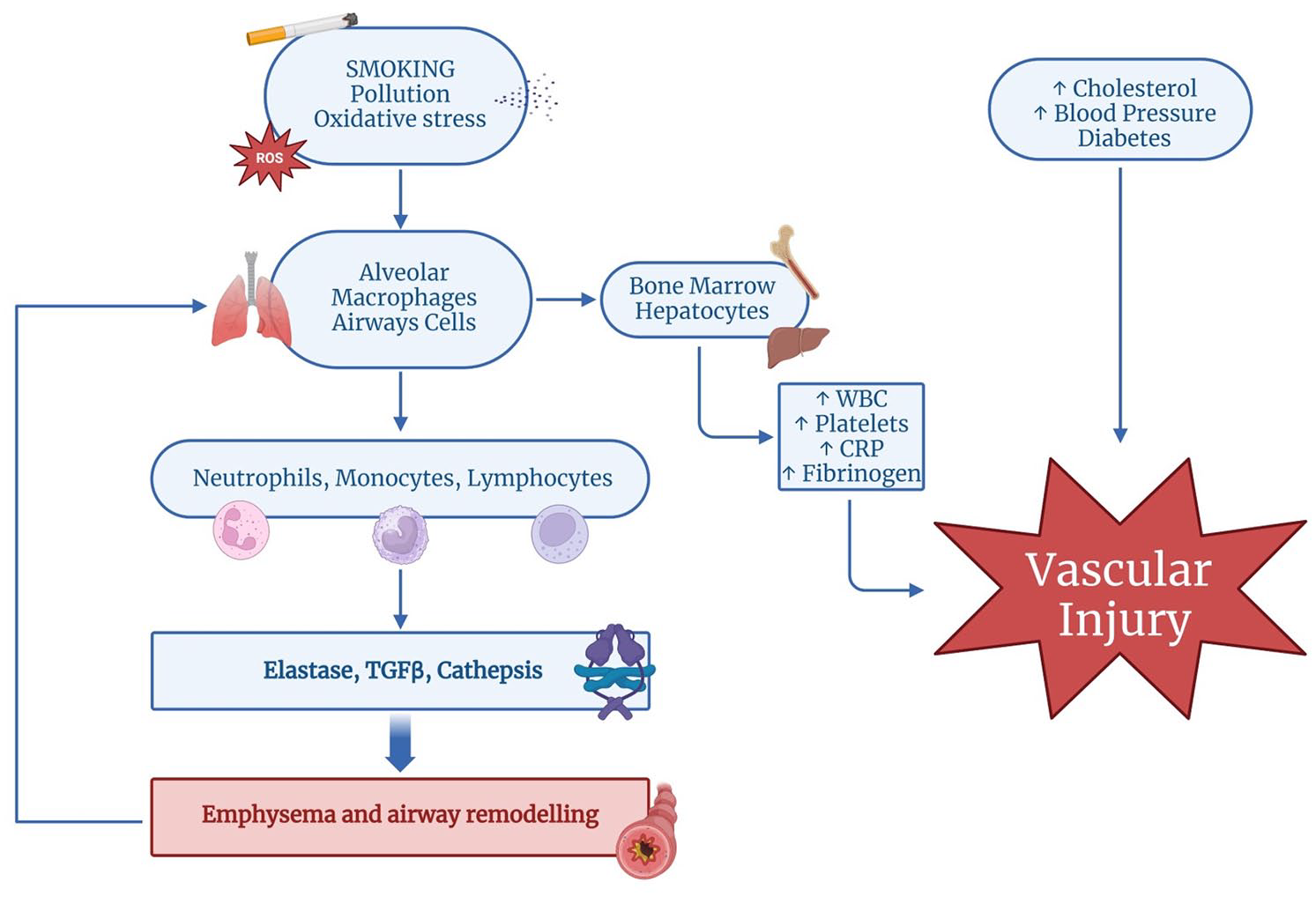
Risk factors and pathophysiological mechanisms leading to COPD and linking COPD to CVDs. Increased lung oxidative stress in COPD may derive from exogenous oxidants (cigarette smoke, air pollution) and endogenous oxidants (ROS, superoxide anions, hydrogen peroxide, mitochondrial oxidants, peroxynitrite, myeloperoxidase, xanthine oxidase). Oxidative stress drives COPD through several mechanisms, including the activation of alveolar macrophages (derived from circulating monocytes) and bronchial epithelial cells which secrete a variety of signalling molecules, inflammatory proteins and release chemokines that recruit and activate cytotoxic T lymphocytes contributing to alveolar cell apoptosis and the development of emphysema. Also, the release of elastolytic enzymes, including MMPs and cathepsins, causes elastolysis and contributes to emphysema together with cytotoxic T lymphocytes. The release of TGF-β can induce fibrosis and remodelling of the small airways. The sustained systemic inflammatory response in COPD, moreover, especially during exacerbations, upregulates (via bone marrow and hepatocytes) the production of pro-inflammatory cytokines, cells and tissue factors (CRP, fibrinogen, platelets, WBC) promoting atherosclerosis and endothelial disease. These factors in conjunction with traditional risk factors, such as hypertension, hypercholesterolemia and diabetes, may lead to vascular injury and CVD.
More recently, the role of exosomes has been described as promising biomarkers and therapeutic tools for chronic respiratory disorders. Exosomes are nano-sized vesicles released by almost all cell types, with a central role as mediators of intercellular communication as they seem to play a pivotal role in inflammatory processes especially in conditions with dysregulated inflammatory processes like COPD. 86
Overall, there is compelling scientific evidence for a sustained systemic inflammatory response in COPD increased especially with disease severity 87 and further amplified during exacerbations. 88 Statins have shown benefits to both CVD and COPD, with a probable effect on the inflammation in the lungs and in the atheromatous plaques. 89 Data on inhaled corticosteroids (ICS), in contrast, are not unequivocal, as previous data showed that they did not exert a significant effect on CV mortality in COPD. 90
Oxidative stress
COPD is a lung disease, but as it is well known, it is better defined as a complex of symptoms and comorbidities that extend far beyond simple lung disease, most of which are related to CVDs such as hypertension, advanced atherosclerosis and CAD. Even today, the link between CVD, vascular dysfunction and COPD is not fully understood. The new hypothesis for this strong correlation between the various systems seems to be found at the basis of the physiopathology of these diseases, their inflammatory nature, which is closely related to oxidative stress, defined as an alteration of the pro/antioxidant balance, dangerous for the life of the cells due to excessive production of highly reactive oxygen species (ROS) and reactive nitrogen species (RNS). 91 It is estimated that oxidative stress is associated with more than one hundred pathological conditions, including arterial hypertension, atherosclerosis, Alzheimer’s disease, diabetes mellitus, rheumatoid arthritis, COPD and asthma. This biochemical reaction of our body is born from the imbalance between oxidation-inducing phenomena such as exposure to cigarette smoke, air pollutants, bacterial or viral infections, protease/antiprotease imbalance, allergic and inflammatory reactions and antioxidant enzymes such as glutathione peroxidase, small thiol molecules such as N-acetyl-L-cysteine and carbocisteine, activators of the Nrf2-regulated antioxidant defence system (sulforaphane), as well as vitamins such as C, E and D. 92 ROS are highly reactive species that contain oxygen and include O2 (superoxide anion), OH (hydroxyl) and H2O2 (hydrogen peroxide), which can be converted into radicals via the Fenton reaction. Physiologically, ROS levels are controlled with antioxidant enzymes such as superoxide dismutase, catalase and glutathione peroxidase. 93 Cigarette smoke is a dangerous mixture of many free radicals and ROS that has two phases: the tar phase and the gas phase. During the tar phase, there is the production of the quinone/hydroquinone radical Q/QH2, which can reduce oxygen with the production of the superoxide anion O2, which can lead to the generation of H2O2. The main reactant is OH, as it is capable of damaging all types of macromolecules during collision. 94 Hydroxyl radicals can be generated through the Fenton reaction involving H2O2 and ferrous iron or cuprous copper, constituting dangerous reactions between metal and redox homeostasis. Indeed, being particulate pollutants rich in iron and increasing oxidative stress, by damaging Fenton’s balance, increases the production of free radicals in the lungs. The most reactive and perhaps most dangerous is the superoxide anion, which can damage all kinds of macromolecules during collision. H2O2 mainly participates in two-electron redox chemistry, predominantly with sulphur in the cell, but in some cases may also participate in one-electron chemistry with transition metals. It can therefore be considered a signalling molecule at low concentrations but also a harmful agent at high concentrations. It therefore has a complex role with overlapping mechanisms of transduction and destruction. The gaseous phase of the smoke then has even more reactive molecules than the tar phase. In this phase, it is possible to reach a concentration of 10 × 15 organic and inorganic radicals for puff. Nitric oxide (NO−) reacts quickly with O2 and forms peroxynitrite (ONOO−). Peroxynitrite is an RNS involved in many physiological and pathological processes; in fact, it has excellent oxidation and nitration capacities, leading to damage to DNA and proteins. Other reagents that undergo the action of nitric oxide are organic lipid peroxyl radicals (ROO−), with which they form alkyl peroxynitrites (ROONO) cytotoxic species. 92 Inhalation exposure can trigger an inflammatory response in the airways and alveoli that leads to disease in genetically predisposed individuals. The process is believed to be mediated by increased protease activity and decreased antiprotease activity. Lung proteases, such as neutrophil elastase and matrix metalloproteinases, degrade elastin and connective tissue in the normal process of tissue repair. 95 Their activity is normally balanced by antiproteases, such as alpha-1-antitrypsin, an inhibitor of secretory leukoproteinases produced by airway epithelium, elastin and a tissue inhibitor of matrix metalloproteinases. In patients with COPD, activated neutrophils and other inflammatory cells release proteases as part of the inflammatory process; their activity exceeds that of the antiproteases, and the result is tissue destruction and overproduction of mucus. 96 Activation of macrophages and neutrophils also causes the accumulation of free radicals, superoxide anion and hydrogen peroxide, which inhibit antiproteases and cause bronchoconstriction, mucosal oedema and mucus hypersecretion. The oxidative damage induced by neutrophils, the release of profibrotic neuropeptides (e.g. bombesin) and decreased vascular endothelial growth factor levels contribute to the apoptotic destruction of lung parenchyma. 97 Inflammation in COPD increases as the severity of the disease increases, and in severe (advanced) cases of the disease, the inflammation does not fully resolve despite smoking cessation. The increase in inflammation is directly proportional to an increase in oxidative stress markers in the air spaces, sputum, breath, lungs and blood of COPD patients. 98 In the lungs, there is an increase in the sequestration of neutrophils in the microvascular vessels, oxidative inactivation of antiproteases and surfactants, hypersecretion of mucus, membrane lipid peroxidation, alterations of mitochondrial respiration, alveolar epithelial damage, remodelling of the extracellular matrix (ECM) and finally apoptosis.
Expanding the clinical gaze, the ROS present in the heart have a crucial role in cellular homeostasis by modulating proliferation, differentiation and excitation–contraction coupling. Among the various pathophysiological consequences of an increase in ROS, if the CV system is affected, it can result in multiple disorders. In fact, some superoxides contribute to endothelial dysfunction of the vascular tissue by reducing the availability of NO; moreover, increased secretion of cytokines and therefore inflammation of the vascular tissue can occur. The adhesion molecules then act on the wall of the endothelial blood vessels and are the main cause of the formation of atherosclerotic plaque. 99 The increased oxidative stress, both local and systemic, has been associated with IHD and studies have demonstrated that ROS are involved in the development of atherosclerosis and inflammatory responses, 100 even though, at present, there is little direct evidence that increased oxidative stress in COPD contributes to an increased risk for IHD. 101
Therefore, an effective and broad-spectrum antioxidant therapy with sufficiently high characteristics of bioavailability and potency is necessary to control the oxidative but also systemic and localized inflammatory processes that occur in the pathogenesis of COPD.
Hypoxia and hypoxemia
Hypoxemia is a condition characterized by low levels of oxygen in the blood due to impaired lung function. In a healthy individual, lungs efficiently exchange oxygen from the air into the bloodstream by concurrently removing carbon dioxide thus consistently maintaining normal PO2 and PCO2 levels. Conversely, in COPD patients, the chronic inflammation of the bronchi leads to airways narrowing with progressive reduction of the outflow which, in turn, determines a progressive air trapping and a reduction of carbon dioxide elimination. Both heart and brain require a continuous and efficient supply of oxygen and nutrients to meet their metabolic needs and every reduction in O2 delivery leads to their functional impairment. 102 Oxygen is indeed of paramount importance in the formation of nitric oxide (NO), which contributes to the control of the vascular tone and, consequently, to the blood supply to the tissues. 103 Oxygen has also a central role in the production of ROS which may induce irreversible damage to cells. 104 Particularly, it is the contractile function that is hampered when oxygen delivery is lacking, and cell apoptosis and necrosis are direct consequences of contractile derangement. To counteract this condition an immediate adaptation, consisting of coronary vasodilation, develops. Hypoxia-inducible factors (HIFs) sense hypoxia to regulate the expressions of a series of downstream genes expression, which participate in multiple processes including cell metabolism, cell growth/death, cell proliferation, glycolysis, immune response, microbe infection, tumorigenesis and metastasis. 105 In the presence of hypoxia, protective mechanisms undergo modulation through the activation of the transcription factor hypoxia-induced factor 1 (HIF-1), which orchestrates adaptive responses to low oxygen conditions. HIF-1 transcriptional factor is a heterodimer consisting of the α-subunit, which includes HIF-1α, HIF-2α and HIF-3α isoforms, and the HIF-β subunit. 106 In normoxic conditions, the HIF-α is maintained at low levels. Its subunits are not stabilized and do not modulate their target genes because the subunits are continuously degraded by the enzyme prolyl hydroxylases (PHDs). 107 In fact, in the condition of physiological oxygen levels (normoxia), HIF-PHDs hydroxylate proline residues on HIF-α subunits leading to their destabilization by promoting ubiquitination by the von-Hippel Lindau ubiquitin ligase and subsequent proteasomal degradation. HIF-α transactivation is also repressed in an O2-dependent way due to asparaginyl hydroxylation by the factor-inhibiting HIF. 108 When oxygen delivery is reduced such as during ischaemia or in a hypoxic environment, the degradation of HIF-alfa is reduced. HIF-alpha protein accumulates and translocates to the nucleus by forming a complex with HIF-beta. The HIF complex interacts with hypoxia response elements situated in the promoter regions of target genes, leading to the enhancement of their expression. These are activated as a protective response to the inadequate oxygen supply to the central and peripheral tissues. Some of the key roles of HIFs in CVD include angiogenesis, erythropoiesis, glycolysis and cellular survival and apoptosis as shown in Table 1.
HIF-1 mechanisms and adaptive responses to low oxygen conditions.

EPO, erythropoietin; HIF-1, hypoxia-induced factor 1; VEGF, vascular endothelial growth factor.
Although the activation of HIFs can have some benefits in the short term by supporting tissue survival and repair, prolonged activation in chronic CVDs may also lead to pathological changes. For example, excessive angiogenesis can lead to abnormal blood vessel growth and promote the development of atherosclerosis and retinopathy. Consequently, gaining insight into the functions of HIFs and their downstream targets is crucial in the quest for targeted therapies that can effectively regulate their activity in CVD, leading to enhanced patient outcomes.
Chronic hypoxia can lead to significant and clinically relevant modifications, affecting both the organism’s structure and function. Two well-known examples are polyglobulia, which refers to an increase in red blood cell count, and the remodelling of pulmonary vessels. 113 Polyglobulia occurs as a compensatory mechanism to enhance oxygen-carrying capacity in response to prolonged hypoxia. 114 On the other hand, the remodelling of pulmonary vessels involves structural changes in the blood vessels of the lungs, which can occur as an adaptation to chronic hypoxia, ultimately impacting pulmonary circulation and function.
As mentioned above, pulmonary hypertension is another CVD, which is influenced by HIF modulation. In this context, HIF-1 activation induces the proliferation of pulmonary arterial smooth muscle cells, while HIF-2 activation affects ECs. These events collectively contribute to the elevation of pulmonary blood pressure. 115 Exploring therapeutic strategies that target HIF transcription inhibition in patients with pulmonary hypertension could present a promising approach for treatment (one study has demonstrated that C76, an inhibitor of HIF-2, effectively reduces pulmonary artery blood pressure in various experimental models 116 ). Furthermore, oxygen is fundamental in the formation of nitric oxide (NO), and its role in maintaining normal CV regulation and overall health has been extensively established. 117 Indeed, beyond its role in promoting blood flow and regulating blood pressure through endothelium-dependent vasodilation, endothelial NO synthase (eNOS) exhibits a plethora of vascular protective effects. These effects include but are not limited to inhibiting PLT aggregation and adhesion, promoting angiogenesis, exerting anti-inflammatory actions and inhibiting the progression of atherosclerosis. The multifaceted actions of eNOS underscore its crucial role in maintaining vascular health and homeostasis. 118 In conclusion, sustained or intermittent hypoxemia, if present, may enhance systemic inflammation, oxidative stress, foam cell production and upregulation of cellular adhesion in ECs thus contributing to the progression of CAD. 37 Chronic hypoxia induces pulmonary vascular remodelling and endothelial dysfunction contributing to pulmonary hypertension. 119 Therefore, it is prudent and highly recommendable to address and attempt to correct this consequence of COPD in routine clinical practice. Indeed, long-term oxygen therapy administered for more than 15 h/day has been demonstrated to increase survival rates in patients with chronic respiratory failure, especially those experiencing severe resting hypoxemia. 120 By doing so, we can mitigate its negative impact on both the CV system and the overall systemic health of patients. Early detection and management of hypoxia-related complications are essential to optimize patient health and quality of life (Figure 2).
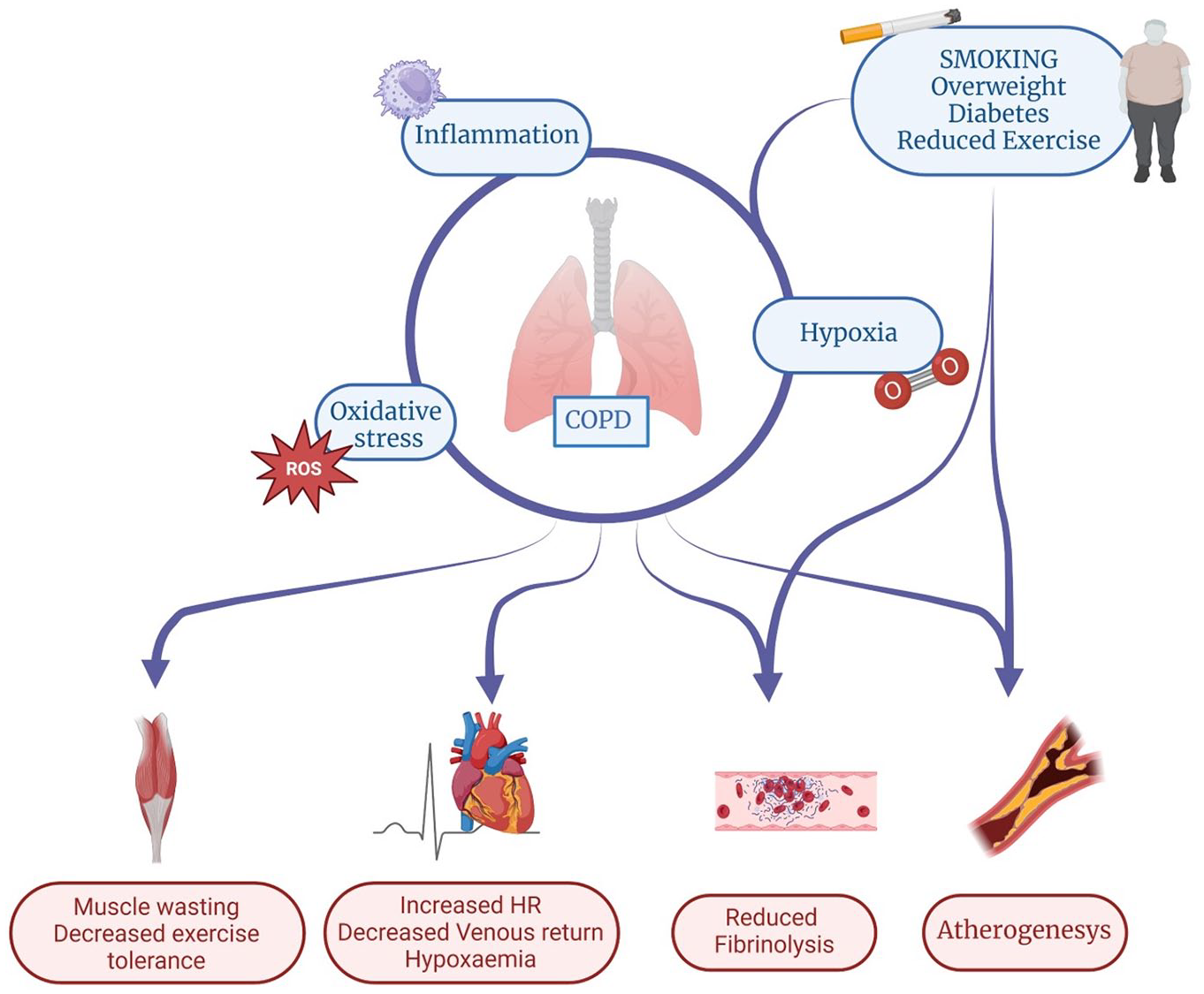
Hypothetical mechanisms increasing the risk of cardiovascular diseases in COPD. Contribution of inflammation, oxidative stress and hypoxia. COPD and CVD often share common risk factors (smoking, persistent exposure to air pollution), and individual conditions like being overweight, a lifestyle with reduced exercise, hypertension, diabetes and sleep disturbances. Once COPD has been established, the risk of CVD is increased through the effects of shared pathways, that is, an amplified systemic inflammation, hypoxia and oxidative stress leading to a major incidence of haemodynamic abnormalities including endothelial dysfunction, autonomic nerves imbalance and impaired cardiac function which may amplify the risk of developing cardiac disease and predispose to the development of CV events such as IHD, HF, stroke and arrhythmias.
Atherosclerosis and ageing
Atherosclerosis represents a risk factor for stroke, PVD and coronary heart disease.121,122 The role of inflammation is now well established in COPD and atherosclerosis, it is related to endothelial dysfunction, increased proliferation of smooth cells and acceleration of formation, evolution and rupture of vascular plaque.123–126 In COPD patients, the activation of the angiotensin–renin system due to chronic hypoxia and the consequent endothelial dysfunction may represent a linking mechanism with CAD. 127 Also, hypoxia-related COPD represents a drive to the development of CVD in fact, the declined pulmonary function (FEV1 < 0.8) leads to an increased risk of IHD.128,129 In a previous study by Fisk et al., the authors highlighted numerous biomarkers that can be used to predict CV risk in COPD patients. In their study, 458 COPD patients were matched for age, sex and body mass index with 1657 non-COPD control. They observed that patients with COPD had an increase in aortic pulse-wave velocity, systemic inflammation as showed by elevated CRP levels, an increase in carotid artery thickness, arterial stiffness and subclinical atherosclerosis. 130 It has been suggested that arterial stiffness in COPD patients may be associated with elastin degradation in the lung and vasculature and it is usually assessed by measuring aortic pulse wave velocity. Another study showed that COPD patients have an increased risk of critical arterial lesions with worse characteristics if compared with patients without COPD. 131 In a study of 201 patients with CAD, those with COPD disease have more often coronary lesions on PCI, more lesions of the left coronary trunk and calcified plaque when compared to patients without COPD (p < 0.0001). Furthermore, some studies highlighted the correlation between COPD severity and CAD.132,133 The precise cause of elastin degradation in arterial walls is not completely known but it seems to be related to a protease/antiprotease imbalance. Some ECM metalloproteinase proteins (MMPs) are implicated in the pathogenesis of COPD and atherosclerosis and it is likely that their overexpression may explain the increased CVD risk in COPD patients. These proteins have an intrinsic enzymatic activity involved in the degradation of ECM proteins. It is known that MMPs are involved in COPD, in particular, MMP-9 is associated with emphysema, mucus hypersecretion and chronic inflammation. 134 In particular, Kraen et al. in their study, have shown that the prevalence of MMPs was different in association with plaque, in COPD patients and smoking: MMP-1 and MMP-3 were associated with both COPD and plaque, MMP-7 was primarily associated with atherosclerosis but influenced by smoking and age. MMP-12 was present above all in atherosclerosis disease but also associated with COPD patients. 134 At the same time, MMPs are implicated in the development, stabilization and rupture of plaque in atherosclerotic disease and can be considered a serum biomarker of this disease.135,136 It is of crucial importance an early identification of the two conditions in a clinical setting for proactive patient management and treatment. 137
Arterial stiffness is also a feature of ageing as shortened leukocyte telomere length has been associated with increased arterial stiffness, a predictor of coronary disease events, and, since the age of the population is progressively increasing, it is fundamental to examine the pathophysiological cellular changes in the lung and its correlation with disease. Due to age-related changes, the lung has evolved various systems to preserve homeostasis and fight harmful stimuli. The broncho-alveolar lavage, for example, contains antioxidants such as vitamins, glutathione peroxidases, catalase and surfactants that are useful to protect from injuries.138,139 Genetic background and lifestyle promote the lung changes and favours the progression of airway diseases. The chronic exposure to noxious stimuli can lead to diminishing tissue repair capacity due to cycle arrest cells, telomere shortening, DNA damage, mitochondrial dysfunction, cell senescence of ECs and reduction of cell proliferation. Some authors have described in COPD lung disorganized collagen fibres caused by an increase in collagen, fibronectin and laminin, leading to the remodelling of airways and parenchyma and enhancing susceptibility to pulmonary disease. 140 The increase in the senescent cells number in response to intrinsic and extrinsic factor is associated with amplification and progression of lung disease. 141 Moreover, COPD lung shows more senescent cells and higher levels of senescence-associated secretory phenotype proteins in response to advanced age. 142 These cells are characterized by secreting pro-inflammatory factors such as IL-6 and IL-8 contributing to the pathogenesis and progression of COPD.143,144 Ageing itself may lead to both qualitative and quantitative changes in the lung structure. For example, it has been observed a reduction of adult stem cells that decreases. These cells show deficits in differentiation capacity contributing to pulmonary diseases such as emphysema and pulmonary fibrosis, it has been observed a reduction of ciliated cell number and slower ciliary movements with worsening of mucociliary clearance, impaired differentiation of alveolar type 2 cells into alveolar type 1, altered lipid and cholesterol metabolism.145–148 Also, reduction of the density of bronchioles and increase in their diameter, loss of alveolar surface area by enlargement of alveolar and airspace size were described. 149 Finally, ageing is associated with shortening telomeres, which might be the cause of vascular and pulmonary disease for endothelial dysfunction, tissue destruction and inflammation. 150
Cardiovascular dysfunction
As has already been highlighted, COPD and CV or cerebrovascular disease frequently coexist. According to observational studies meta-analyses, patients diagnosed with COPD have a two-fold increased risk of CV disease in their lifetime. More precisely, the increased risk for COPD patients is reported for HF, hypertension, dysrhythmia, acute myocardial infarction, diabetes and stroke when compared with the general population (age and gender-adjusted). 9 In addition, evidence suggests that these patients die more often due to CVD rather than respiratory failure. 151 The relationship between COPD and CVD is bidirectional: CV disease increases the risk and frequency of COPD exacerbations and the latter has been associated with increased risk of acute CV events (Figure 3). Although currently, the underlying mechanisms are still unclear, the harmful interaction between respiratory and CV systems has widely been described. Firstly, COPD and CVD share common risk factors, such as cigarette smoking, a sedentary lifestyle and ageing. Then, inflammation, hypoxia and hyperinflation have also been studied as possible causative factors. 152 A recent systematic review and meta-analysis conducted by Müllerová et al. 153 shows a statistically significant increased risk of acute myocardial injury in the 1–3 months after a COPD exacerbation, relative risk (RR) 2.43 (95% CI = 1.40–4.20), and a statistically significant increased risk of stroke after a COPD exacerbation, RR 1.68 (95% CI = 1.19–2.38) for 1–3 months after the acute event. Kunisaki et al. performed a post hoc cohort analysis from the SUMMIT Randomized Clinical Trial which included 16,485 patients from 1368 centres in 43 countries diagnosed with moderate COPD (FEV1 50%–70% of predicted). These patients were current or former smokers and already had CVD or increased CVD risk. This study supports the notion that AECOPD increases the risk for CVD events (here composite outcomes: CV death, stroke, unstable angina, myocardial infarction, transient ischaemic attack). They reported a fourfold increased hazard ratio for CVD events in the first 30 days after AECOPD and a 10-fold increase in those who were hospitalized due to AECOPD. During AECOPD, the concentration of circulating proinflammatory biomarkers increases and then slowly decreases to baseline. This trend might explain the higher CV events risk during the first 30 days after AECOPD. 154 Also, the degradation of elastin in lungs and arteries has been considered as a possible link between COPD and CVD. Maclay et al. pointed out that COPD patients have increased skin elastin degradation compared with controls (age and smoking history adjusted) due to increased proteolytic activity. The data have been associated with arterial stiffness and emphysema severity and therefore they show a novel possible shared mechanism for the pulmonary, vascular and cutaneous features of COPD. 124 As already mentioned, ageing is a process that has consequences both in the lungs and heart. In particular, the loss of sirtuins, antiaging proteins that regulate processes such as transcription and apoptosis has been studied. 155 In bone marrow, circulating endothelial progenitor cells (EPCs) and specifically blood outgrowth EPCs are produced to ensure endothelial integrity. It has been found that EPCs from smokers and COPD patients show increased DNA double-strand breaks and cellular senescence compared to the same cells from non-smokers. These alterations correlate with reduced expression of sirtuin-1 and consequently accelerate the ageing process being inefficient in repairing endothelial damage. Therefore, they provide a link between COPD and vascular aging and its consequences (e.g. IHD). 156 The first mechanism to deepen is lung hyperinflation, the abnormal air trapping following spontaneous exhalation due to disruption of lung parenchyma and consequent loss of its natural elastic capacity. The chronic airflow limitation and emphysema causing lung hyperinflation, together with hypoxic vasoconstriction resulting in increased pulmonary vascular resistance, could directly increase the pressure in the pulmonary artery. That so-called ‘pulmonary hypertension’ usually increases right ventricular diastolic dysfunction 157 which can exist independently of a coexisting systemic or pulmonary hypertension in patients suffering from COPD, anyway. 158 Hyperinflation can also have a direct impact on the cardiac dimension, resulting in decreased diastolic filling and reduced systolic ejection fraction.159,160 Watz et al. studied the relationship between lung function and heart size and dysfunction. They found that the increasing severity of COPD according to the Global Initiative for Obstructive Lung Disease classification (GOLD) is associated with decreasing heart size. In their observational study, they found a strong association between lung hyperinflation, measured in inspiratory-to-total lung capacity ratio (IC/TLC), functional residual capacity and residual volume, with heart chamber size (echocardiographic measurements). In addition, patients with an IC/TLC ⩽0.25 had a significantly impaired left ventricular diastolic filling pattern and global right ventricular function. They also obtained functional information relating the echocardiographic measurements of cardiac function to the 6-min walk distance test: an impaired left ventricular diastolic filling pattern is independently associated with reduced exercise tolerance. 161 In this regard, Stone et al. more recently proposed a double-blind, placebo-controlled, crossover study to test if it were possible to modify the cardiac structural and functional alterations seen in stable hyperinflated COPD patients through pharmacologic lung deflation. To investigate cardiac function and morphology, they used cardiac magnetic resonance. They demonstrated the cardiac chamber size reduction in this cohort of patients and the beneficial effect on this theme obtained by the ICS/long beta-2-agonist (LABA) therapy versus placebo. 162 Concerning left ventricular dysfunction itself, it has recently been demonstrated that it has a negative impact on exercise tolerance in patients with COPD, being associated with anxiety, depression, reduced carbon monoxide (CO) diffusion and a higher prevalence of right ventricular dysfunction. 163 It seems clear that the heart and lungs stand in a vicious circle affecting each other when dysfunctional. Another well-known CV comorbidity affecting COPD patients is HF, which is much more prevalent among the ones experiencing an exacerbation and is the most important cause of death and hospitalization in these patients. Moreover, the differential diagnosis between these two entities can be challenging. 159 To help the physician in this distinction, plasma B-type Natriuretic Peptide (BNP) dosage can be useful. However, BNP can also be elevated during COPD exacerbations due to pressure or volume overload acting on the right ventricle. Although this increase usually is not as high as in severe HF, it may produce confounding results in many patients. 164 In general, the prevalence of HF among COPDs is estimated to be in the range of 10%–30%. 19 In a Canadian cohort, Curkendall et al. 10 estimated an age-adjusted risk ratio for HF of 4.5 (95% CI: 2.8–6.2). Several studies have also deepened the interrelationship between lung function and CVD.165–167 Agarwal et al. 167 conducted a longitudinal study and they also found an inverse relation between decreased FEV1 and increased incidence of HF. Silvestre et al., relaying on serial spirometry of 10.351 patients followed for 17 years, found that a rapid decline in lung function correlates with a higher incidence of subsequent CVD independently of baseline values and smoking/non-smoking status. Specifically, rapid decline in FEV1 has been associated with a higher incidence of HF, stroke and death. On the other hand, rapid decline in FVC has been associated with a higher incidence of HF and death. There was no significant difference in sex and race. 165 A recent population-based study conducted using the Julius General Practitioners’ Network, a wide registration network including over 3,70,000 enlisted individuals in the Netherlands, provides further demonstrations of the higher incidence of all major CVDs in patients suffering from COPD. 155 They estimated the incidence of IHD, AF and HF in patients with or without COPD (Table 2). The highest incidence ratio has been observed for HF with no significant difference between males and females, suggesting that COPD may interfere or reduce the well-known CV protection that (premenopausal) females normally have. 168 High IRR in young patients has been highlighted, suggesting that COPD may not only increase the risk of CVD but also accelerate its progression (due to the ‘accelerated ageing’ process observed in COPD as previously explained). 169
Age-adjusted and sex-adjusted IRR of ischaemic heart disease, atrial fibrillation and heart failure for patients with and without COPD.

CI, confidence interval; COPD, chronic obstructive pulmonary disease; CVD, cardiovascular disease; IRR, incidence rate ratios.

The bidirectional relationship linking CVD and COPD. Among the several processes associated with COPD, systemic inflammation, hypoxaemia and hyperinflation (especially during exacerbations) may increase the risk of CV disease that, in turn, may increase the risk and frequency of COPD exacerbations. Lung hyperinflation together with hypoxic vasoconstriction, commonly observed in COPD patients, contributes to the development of pulmonary hypertension, usually associated with increased right ventricular diastolic dysfunction. Hyperinflation itself is associated with a decreased heart size, an impaired left ventricular diastolic filling pattern and a reduced right ventricular function with consequences on symptoms (increased dyspnoea and reduced exercise tolerance). Moreover, skin elastin degradation due to increased proteolytic activity, usually reported in COPD, has been associated with arterial stiffness and emphysema severity highlighting another possible shared mechanism between pulmonary and vascular system.
The clinical impact of pathogenetic mechanisms linking COPD to CV comorbidities
At present, the research of CVD mechanisms in COPD is still in progress. COPD is undoubtedly associated with a number of CV risks factors and mediators, atherosclerosis and thrombosis playing a specific role in the pathogenesis. 100 Among novel risk factors, genetic subtypes and different COPD phenotypes may have relevant clinical implications in the development of a specific individual CV risk with the need of an early assessment in those vulnerable subjects, by, that is, the use of risk-scoring tools, 170 with educational projects on smoking cessation, lifestyle interventions on nutrition and physical exercise, the recognition and treatment of shared risk factors including hypertension, diabetes and hyperlipidaemia. Among well-known risk factors, systemic inflammation, oxidative stress, hypoxia with production of ROS, abnormalities of the vascular wall including arterial stiffness, PLT activation and endothelial dysfunction with a hypercoagulable state, ageing and cellular senescence were found to be synergistically involved in the atherothrombotic process in COPD. 171
The interactions between inflammatory cells and PLT may play a key role in the pathogenesis of vascular events in COPD.172–174 In this perspective, PLT activation is an important prothrombotic manifestation of the disease related to CV events; however, an agreement on its assessment is still lacking. Recent interesting research with important clinical repercussions was done to better characterize atherosclerotic coronary features in COPD patients. Russo et al. studied atherosclerotic coronary plaque features in COPD patients presenting with acute coronary syndrome using optical coherence tomography. They concluded that COPD is an independent predictor of plaque inflammation and vulnerability. These results may suggest that a higher inflammatory state in COPD patients might enhance local coronary inflammation, promoting CAD development and plaque vulnerability. 175 On the other hand, it has been previously observed that a considerable proportion of COPD patients may suffer from cardiac abnormalities which often remain underdiagnosed, especially during the early stages of the disease, that is, subclinical left ventricular filling impairment has been reported, and is frequently found in COPD patients at the earlier stage of the disease, even in the absence of any other CV dysfunction. 176 The pathogenesis of CV dysfunction in COPD patients seems to be complex and related to multiple alterations in cardio-pulmonary homeostasis, although the pre-clinical link between respiratory and CV morbidity in COPD patients may explain the increased CV risk in these patients. In such patients, case finding for underlying disease including blood measurements (emoganalysis, BNP, glucose, lipids, ECG, echocardiography, lung function tests, imaging tools) may be useful to identify those at major risk. Among COPD-related factors associated with the increased risk of CVD and acute cardiac events, recurrent exacerbation is a central point especially in certain COPD phenotypes. The assessment of promising biomarkers like blood fibrinogen, an acute phase glycoprotein involved in blood coagulation 82 may be of pivotal importance since it has been demonstrated a direct relationship between plasma fibrinogen and the risk of mortality in COPD patients (to note that levels of fibrinogen are increased during COPD exacerbations when there is a major risk of CV and thrombotic events).85,177,178 As increasing evidence suggests that the systemic inflammatory pathway provides a common link between COPD and its comorbidities, the research and development of biomarkers may provide a greater understanding of the interlink between COPD and CVD, may have a diagnostic role for early intervention against crucial risk factors preventing the onset of COPD-CVD comorbidity and will be important in developing new therapeutic interventions to treat COPD and CV comorbidities. Increased levels of circulating inflammatory mediators are found in the peripheral blood of COPD patients and have been investigated as potential biomarkers of the disease (IL-6, TNF-α, ICAM-1, Gal-3). Galectin-3, a β-galactoside-binding lectin, is involved in various biological processes including inflammation, fibrosis and myocardial remodelling. 179 Elevated serum Gal-3 levels have been identified as a potential biomarker for COPD exacerbation, 180 additionally Gal-3 can enhance the inflammatory response by recruiting macrophages, and contributing to tissue fibrosis and remodelling, suggesting that elevated levels may reflect ongoing myocardial fibrosis and remodelling in COPD patients with CV comorbidities. 181 A recent study showed a correlation between Gal-3 levels with NT-proBNP and maximal pulse rate in COPD patients suggesting its potential role, in clinical settings, as biomarker identifying patients (COPD and pre-COPD populations) at risk for developing CV comorbidities and providing insights into the extent of myocardial remodelling/fibrosis. 182 Some inflammatory mediators (CRP, fibrinogen, IL-6, IL-8) are also elevated in patients with atherosclerosis, suggesting a state of systemic inflammation that could contribute to the development of CVD.183,184 Increased levels of fibrinogen (that can be present either in presence of pulmonary acute infections) have been associated with a higher risk of coronary events in COPD patients.20,81,185 Elevated leukocytes (in particular neutrophil infiltration) are responsible of endothelial dysfunction associated with the development of atherosclerosis, which can, in turn, lead to CVD. 186 Levels of circulating PLT-monocyte aggregates (indicating PLT activation, a CAD risk factor) are increased both in stable COPD and during exacerbations. 124 Cardiac troponin (cTnT) is a potential reliable indicator of CVD in COPD patients, it demonstrated significant correlations with multiple cardiopulmonary parameters (FEV1%, walking distance, inflammatory markers), highlighting its importance in clinical setting. Elevated levels of cTnT have been associated with COPD and acute exacerbations and may be a marker of increased risk of MI in COPD.187,188 NT-proBNP levels are significantly elevated in AECOPD, pulmonary hypertension and HF. 189 Oxidative stress generated by resistive breathing and flow limitation particularly during exercise or exacerbations in COPD patients may also play a key role in COPD-related inflammation, providing another source of potential biomarkers, although direct measurement of oxidative stress biomarkers is difficult due to their highly reactive nature. Long-term effects of oxidative stress on nuclear damage and cellular senescence have been described, and, a biomarker like Sirtuin-1 may be useful, as its reduced expression in COPD smokers correlates with vascular accelerated ageing and is linked to inefficient repairing endothelial damage, with a possible role in IHD.156,190,191 Hypoxia is another mechanism responsible of vascular remodelling and endothelial dysfunction with increased vascular resistance, that, in turn, may conduct to an augmented risk of atherosclerosis. Increased levels of thrombin–antithrombin complex and prothrombin activation fragments 1–2 were observed in COPD patients undergoing hypoxic challenge, this resulted in coagulation activation and an enhanced risk of venous thromboembolism. 192
Additionally, there is an increasing interest in whether biomarkers that reflect underlying pathophysiological processes can be used to predict the effects of therapeutic interventions, and, combining clinical information with biomarkers may improve the benefits of pharmacological treatments. Blood eosinophil counts, for example, can be used to predict the effect of ICS treatment. Some COPD patients, in fact, show elevated eosinophil levels that are associated with a better response to ICS 171 and predict the ICS effect on exacerbation prevention eventually reducing mortality for CV events, more frequent during COPD exacerbations. Moreover, eosinophils also appear to be involved in atherosclerosis, it has been shown that they are usually absent if the plaques are stable but are detected when they rupture. 193 Platelet activity also may have a role as a pharmacological biomarker since antiplatelet therapy has been shown to increase COPD patients’ survival rates probably due to a systemic antithrombotic effect 194 and that antiplatelet treatment significantly decreases the absolute risk of ischaemic events in COPD patients. 195 Elevated levels of lipids (i.e. total cholesterol, LDL, triglyceride) may be controlled by statins that also exhibit pleiotropic pharmacological actions including antioxidant, anti-inflammatory, antithrombotic and immunomodulatory effects. 196 Treatment with statins in some cases has been associated with improved survival and with a reduced risk of pulmonary hypertension development in COPD patients.197,198 The above-mentioned biomarkers are summarized and their role is described in Table 3. Some of them, as reported in the table, are suitable for a routine assessment in clinical practice.
Specific biomarkers associated with COPD/CVD and clinical implications.

CAD, coronary artery disease; COPD, chronic obstructive pulmonary disease; CRP, C-reactive protein; cTnT, cardiac troponin; CVD, cardiovascular disease; Gal-3, galectin-3; ICS, inhaled corticosteroid; IHD, ischaemic heart disease; IL-6, interleukin-6; MI, myocardial infraction; NT-proBNP, N-terminal pro-brain natriuretic peptide; PLT, platelet; TNF-α, tumour necrosis factor-alpha; VTE, venous thromboembolism.
Hypoxia and chronic respiratory failure may also induce hemodynamic stress increasing heart rate, impacting ventricular function and affecting renal circulation with the result of increased peripheral vasoconstriction and activation of the sympathetic nervous system, with an increased risk of CV events.199,200 The preliminary results of this complex interlink are globally giving a greater awareness, in COPD patients, of the increased risk both for CV adverse events and pulmonary deterioration, so that a ‘cardiopulmonary risk’ definition has been coined 201 to improve the current symptom-based treatment approach to a more proactive and preventive approach, reducing future risks and, hopefully, improving survival.
The reciprocal effects of treatment for COPD and CVD
The aim of COPD treatment includes the control of symptoms and strategies to reduce the risk of exacerbations which are associated with an elevated frequency of acute CV events. Among pharmacological treatments, the combination of LABA with long-acting-antimuscarinic agents (LABA/LAMA) with or without the addition of an ICS is the pivotal therapy for those patients with a favourable risk–benefit profile as recommended by International Guidelines. 1 Dual bronchodilation treatment with LAMA/LABA in a single inhaler with dose combination has been demonstrated to be able to improve quality of life in COPD patients in terms of symptom scores, rescue medication use202,203 and lung hyperinflation, reducing air trapping, hence improving exercise tolerance.204–210 The deflationary effect on resting and dynamic air trapping in COPD patients treated with an association ICS/LAMA, moreover, has been shown to have also an effect on ventricular filling and improve heart function, as reported in the CLAIM study. 211 The therapeutic rationale for treating COPD patients with the association ICS/LABA/LAMA is based on the assumption that the combination offers double bronchodilation with an anti-inflammatory effect, maintaining control of COPD exacerbations. As observed previously, corticosteroids often used in combination with beta2-agonists and anticholinergic bronchodilators in the treatment of COPD work synergistically. ICS enhances the effect of LABA’s on airway smooth muscle increasing prejunctional auto-inhibitory M2-receptor gene expression in airway smooth muscle and activating the beta2-receptor gene, with augmentation of receptor number and a reduced desensitization. They finally act as anti-inflammatory, long-acting bronchodilators and reduce hyperinflation, all mechanisms linking COPD with CVD, especially during exacerbations. On the other side, LABA primes the glucocorticoid receptor and enhances nuclear translocation. In airway smooth muscle, corticosteroids inhibit the contractile effects of acetylcholine, whereas M2-receptor antagonism increases the relaxant activity of isoproterenol. 212 The prevention of COPD exacerbations is a major goal in COPD treatment, being exacerbations associated with an important risk of CV events and mortality. Treatment with ICS/LABA/LAMA was compared with ICS/LABA and LAMA/LABA treatment in two large studies, ETHOS and IMPACT, evaluating all-cause mortality as a secondary endpoint in the COPD population analysed for exacerbation rates. In both studies, triple therapy was superior when compared to the alternative treatment in reducing exacerbations and mortality. In the ETHOS study, there was a greater reduction in mortality in the ICS/LABA/LAMA group compared to the LABA/LAMA with the evidence that the difference was due to CV events and the suggestion that the addition of ICS is associated with the reduction of mortality for the mechanisms above mentioned. It has been argued that bronchodilators may worsen existing underlying CVD or, at least, increase the risk of developing CVD 213 due to the activation of the sympathetic nervous system leading to cardiac rhythm disturbances even though the option of beta-2 selective LABA may improve this concern. ICS also may worsen cardiac arrhythmias and HF even though increasing evidence from clinical trials confirms that inhaled COPD treatments do not pose a significant CV risk in people free from CV comorbidities 214 while there may be some concern in the use of these medications in COPD patients with a history of heart disease that are more prone to have an increased risk of AF and other CVD. 215 The co-existence of comorbid COPD-CVD may complicate the management of COPD, though current recommendations state to treat CVD in people with COPD irrespective of COPD diagnosis, including the use of beta-blockers, that have shown a good safety profile of cardio-selectivity, in patients with AF or HF.216,217 Despite this, CVDs are often underdiagnosed and undertreated in COPD patients (i.e. HF is one of the most underdiagnosed comorbidities in COPD). It has been shown the underuse of beta-blockers in COPD patients with a history of myocardial infarction in spite of the strongly supported evidence of their benefits on morbidity and mortality. This may be due to the lack of guidelines in the assessment of comorbid COPD-CVD. 218 Real-world evidence has shown better outcomes in COPD patients receiving treatment with CV drugs 219 and there are suggestions indicating that the use of CV medications may improve longer-term outcomes, that is, all-cause and CVD mortality in COPD patients. 220 Antiplatelet therapy has shown to be effective and associated, in observational studies, with reduced exacerbation rates and mortality due to its role in contrasting thrombotic complications.221,222 Statins seem to have a role and to be beneficial in COPD patients with CVD as highlighted in a meta-analysis by Zhang et al. 223 including 10 trials. In conclusion, considering the prevalence of CVD and risk factors in COPD patients, the primary prevention including a risk factor modification strategy may be an important issue. Smoking cessation, regular exercise, regular CV assessment and treatment of hypertension and hypercholesterolemia should be promoted especially in those younger COPD patients with symptoms suggestive of CVD in whom an early intervention may be most effective. In terms of the management of comorbid COPD-CVD patients, there are suggestions for the use of CV medications that may improve longer-term outcomes including all-cause and CVD mortality (beta-blockers, statins, antiplatelet therapy). Finally, inhaled COPD treatments did not show a significant risk of CVD compared to their benefits and should be prescribed following current recommendations.
The relevance of early identification and management of CVD risk in COPD
Patients with COPD frequently die of respiratory and CV causes, yet the identification and management of cardiopulmonary risk still remain suboptimal. Acute exacerbations point to the progression of COPD in many patients, reducing lung function and increasing the risk of subsequent exacerbations and CV events that may lead to death. COPD should be considered an independent factor of high CV risk, and efforts should be directed to the early identification of CVD in COPD patients. Assessment of the overall CV risk is important especially in patients with severe exacerbation episodes. The combination of CV and pulmonary risks may constitute the so-called ‘cardiopulmonary risk’, the calculation of which can help in the clinical management of COPD patients. 224 Moreover, the management of cardiorespiratory diseases (prevention, early diagnosis, acute illness, treatment,216,225 rehabilitation and chronic patients) is a relevant point since it has been reported that COPD is still underdiagnosed in 60% of patients with CVD and 87% of patients with IHD. A high prevalence of undiagnosed CVD is currently observed in hospitalized COPD patients.
Conclusion
Cardiovascular events are still responsible for a large proportion of deaths among patients affected by COPD and CVD often may remain underdiagnosed and asymptomatic especially in COPD patients whose symptoms can be attributed to the underlying condition or, at least, challenging to diagnose with classical tools. There is no evidence of a common background of low-grade systemic inflammation favouring and linking the two conditions, triggered by different stimuli including cigarette smoke and pollutants. Exacerbations have an important role in COPD progression especially in certain COPD phenotypes; hence, the relevance of early identification of CV problems with new approaches and specific predictive biomarkers is needed to better stratify these patients at risk of CVE, to provide effective screening programmes and improve their treatment and prognosis. Current recommendations for the management of CVD in COPD patients are still lacking; therefore, a multidisciplinary approach with agreement between pneumologists and cardiologists may be useful to improve the management and the specific tailored therapeutic setting for these patients.
