Abstract
Background:
With rising obesity rates worldwide, clinical trials focused on identifying effective treatments are increasing. While guidelines exist for pharmaceutical drugs targeting obesity, there are none for herbal medicine clinical trials for anti-obesity. Both industries refer to the same guidelines for clinical trials.
Objectives:
This scoping review aimed to gather information from herbal medicine anti-obesity randomised controlled trials (RCTs), analyse the methodologies and assess their alignment with international guidelines.
Eligibility criteria:
This review included RCTs of participants of all ages with obesity utilising herbal medicine with any comparators and focusing on various outcome measures.
Sources of evidence: Only published journal articles were included.
Charting methods:
Articles were extracted from MEDLINE, CENTRAL and EMBASE using predetermined keywords. Relevant data, such as the study characteristics, types of herbal interventions and controls, treatment durations, outcome measures and safety monitoring methods were recorded in a table format for comparative analysis.
Results:
We included 99 RCTs that showed participant sample sizes ranging from 8 to 182, ages 18 to 80 years and body mass indexes (BMIs) between 25 and 49.9 kg/m2. Herbal interventions used single herbs (n = 57) and mixtures (n = 42), given for 14 days to 56 weeks. Studies implementing diet modifications include restricted calorie diets (n = 35), food-portion controlled diets (n = 7) and fixed calorie diets (n = 7). Of the 28 studies implementing exercise, most were of moderate intensity (n = 22). All studies collected BMI and weight as primary outcomes. Body fat composition was measured in over 50% of studies using a body analyser (n = 57). Waist, hip and abdominal circumferences were infrequently measured. Radiological tools used include dual-energy X-ray absorptiometry (n = 16), computed tomography scans (n = 10) and ultrasound (n = 2). Safety monitoring methods were reported in most studies (n = 76).
Conclusion:
In conclusion, almost 50% of the studies adhered to international pharmaceutical clinical trial guidelines, addressing dietary, lifestyle, physical activity and cardiovascular risk factors. Nonetheless, more herbal anti-obesity studies need to consider the assessment of weight maintenance.
Plain language summary
The global rise in obesity has increased the focus on clinical trials to identify effective treatments. While guidelines such as the USFDA Guidance for Industry Developing Products for Weight Management (2007) and the EMA Guideline on Clinical Evaluation of Medicinal Products Used in Weight Management (2017) exist, they have not been updated recently, and specific guidelines for herbal medicine are lacking. Consequently, both industries often refer to the same guidelines. Our review aimed to gather and analyse data from randomised controlled trials (RCTs) involving herbal medicine for anti-obesity treatment. We examined how these trials are conducted, their adherence to international guidelines, and identified specific challenges and limitations. The review included 99 studies, with sample sizes ranging from 8 to 182, ages 18 to 80 years, and BMIs between 25 and 49.9 kg/m². Nearly 50% of the RCTs adhered to international pharmaceutical guidelines, but significant gaps remain, particularly regarding study duration, lifestyle modifications and safety monitoring. These findings highlight the need for specific guidelines for herbal medicine trials.
Introduction
Globally, obesity is a rising health crisis. Obesity is one of the most prevalent non-communicable diseases, causing a multitude of health challenges. In the United States of America (USA), the prevalence of obesity has escalated from 30.5% in 2000 to 41.9% in 2020. 1 An increase in obesity prevalence is also seen locally in Malaysia. The National Health and Morbidity Survey (NHMS) reports of 2015 and 2019 estimated that the prevalence of overweight adults was 30.0% and 30.4%, whereas obesity prevalence was 17.7% and 19.7%, respectively. 2 Increased risk of many serious diseases, including cardiovascular diseases, stroke, diabetes mellitus, hypertension, dyslipidaemia, chronic kidney disease, respiratory dysfunction, osteoarthritis and certain cancers are associated with the growing obesity epidemic and sedentary lifestyle. 3 Obesity has also been identified as an independent risk factor for severe COVID-19, intensive care unit admissions and deaths.4,5
Overweight and obesity are influenced by a combination of risk factors, including individual dietary, exercise, behavioural and lifestyle choices, in addition to individual genetics, environment and metabolic risk factors. 6 A review by Swinburn et al. 7 reported that poor dietary habits and inadequate physical activity are contributing factors to the development of obesity. Dietary fibre (non-starch polysaccharides) and physical exercise were found to be substantially protective against obesity, while sedentary lifestyles and high intakes of high-energy food which are deficient in micronutrients were risk factors for obesity. 7 Another review discussed that sedentary lifestyle, in addition to decreasing energy expenditure, is also associated with an increased risk of cardiometabolic diseases, independent of physical activity levels. 8 A questionnaire-based study by Junne et al. 9 found that specific obesity-related psychological and social factors, such as dissatisfaction with body figure and social insecurity, can lead to stress. Prolonged stress affects metabolism, causes negative emotions and hormonal imbalances, which can then promote binge eating, a heightened appetite and cravings for unhealthy foods. 9
In response to the escalating prevalence of obesity, there has been a surge in clinical trials aiming to identify effective interventions. These trials utilise a wide range of approaches to address the complex nature of obesity. For instance, there are anti-obesity trials with behavioural approaches,10–12 pharmacological approaches,13–15 dietary approaches,16–18 and others. In anti-obesity human trials, a comprehensive approach is usually recommended. This begins with a thorough history taking, anthropometry measurements, physical examination and necessary laboratory tests. This initial assessment is critical for evaluating the severity of obesity and related health issues. Following that, setting realistic weight loss goals is crucial. The main principle of obesity treatment is lifestyle modification which includes adopting a hypocaloric or normocaloric diet, increasing physical activity and in some cases, undergoing cognitive behavioural therapy. Non-invasive treatment methods that could be beneficial include pharmacotherapy, vaccines, gut microbiota modulation and gene therapy. That said, obesity management could differ among individuals, and a tailored approach needs to be adopted. 19
Historically, pharmacological management of obesity has been associated notoriously with serious adverse effects. The infamous anti-obesity drug, Sibutramine, which was withdrawn in 2010, was associated with an increased risk of heart attacks and strokes in patients with cardiovascular disease history. 20 The same goes for several other banned anti-obesity drugs such as ephedra, 21 rimonabant and dinitrophenol, which have been associated with negative cardiovascular, pulmonary and metabolic effects. 22
Concerned with the risk of harm associated with pharmacological anti-obesity drugs, there is growing interest in developing herbal and complementary medicines with the perception that natural remedies could potentially offer a safer alternative, though this remains to be proven. Herbal medicine has been used for centuries to prevent and treat various diseases, and many studies have reported their efficacies in obesity treatment. 23 Herbal medicine is defined by the World Health Organization (WHO) as ‘herbs, herbal materials, herbal preparations and finished herbal products, that contain parts of plants, other plant materials or combinations thereof as active ingredients’. 24 Among the potential anti-obesity benefits that may be offered by herbal medicine include mechanisms like thermogenesis, inhibiting pancreatic lipase activity, reducing food intake and increasing lipolysis. 25 Secondary metabolites commonly found in medicinal plants, such as flavonoids and saponins, can trigger physiological changes (i.e. upregulating glutathione and superoxide dismutase production and inhibiting cell growth), offering potential therapeutic benefits for metabolic disorders and obesity treatment. 26
Medicinal products (conventional and herbal medicine) with weight loss claims should be tested rigorously in clinical trials to prove their efficacy and safety in humans. The European Medicines Agency (EMA) 27 and United States Food and Drug Administration (US FDA) 28 have laid out guidelines for the industries to conduct clinical trials for medicinal products on obesity, outlining some recommended requirements to facilitate drug development. These guidelines are used universally for all interventions. Despite the standard recommendations for obesity trials, it is common for individual clinical trials to be conducted uniquely as there is no one-size-fits-all methodology. Anti-obesity clinical trials often have the additional consideration of incorporating adjunct therapies such as lifestyle modifications. A long study duration with additional follow-ups conducted at least 6 months after treatment cessation is also often recommended to reflect good and sustainable efficacy. These requirements can be challenging to be met by both investigators and participants of clinical trials, consequently leading to high drop-out rates, or loss to follow-up. 29
Although there are some similarities between herbal medicine and conventional medicine trials, there are also additional requirements. For example, in herbal medicine interventional trials, the quality of the herbal interventions is an important consideration.30,31 In addition, compared to conventional medicines or synthetic small molecules, which often have specific pharmacological targets, herbal medicine, rich in phytoconstituents, is often reported to have pleiotropic effects, modulating multiple pathways simultaneously. 32 In some cases, the principles of treatment of obesity may differ between traditional medicine philosophies and modern medicine approaches. For instance, traditional Chinese medicine addresses diseases in an integrated manner, targeting multiple interactions in a biological system, and is personalised according to the individual. Modern medicine, on the other hand, comprises well-defined chemical compounds that are designed to target specific molecular pathways or biological processes, with their direct effects well-understood and predictable. 33 In view of the known differences between herbal medicine and conventional medicine, given that current anti-obesity guidelines for clinical trials are catered generally to conventional drug development, there is a significant research gap in understanding how herbal medicine trials have been conducted. Therefore, we conducted this scoping review to collate literature of clinical trials investigating the anti-obesity effects of herbal medicines to gain insights towards the landscape of methodologies used in such trials and explore the associated challenges as well as limitations unique to herbal trials.
Methods
This review was conducted according to the York Framework of scoping studies by Arksey and O’Malley, 34 refined by Levac et al. 35 This framework serves as a guide for a standardised and systematic approach in conducting scoping studies to address new or broad research questions of complex or heterogeneous nature. All five stages of the scoping review, namely, (1) identification of research question(s), (2) identification of relevant studies, (3) selection of studies, (4) data charting and (5) collation, summarisation and reporting of findings, were undertaken. 36 We followed the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines to ensure relevant items for the scoping review were included. The PRISMA Scoping Review checklist is provided inSupplemental Material 1.
Identification of research questions
This review was conducted based on the primary research question ‘What are the methodologies used for herbal medicine randomised controlled trials (RCTs) for the management of obesity?’
Secondary research questions include the following:
What are the commonly collected measurements to assess anti-obesity effects of herbal medicines in RCTs and how were they measured?
What are the common laboratory tests carried out to assess the efficacy of intervention in reducing obesity?
What adjunct therapies are commonly used in the trials?
How was the safety monitoring carried out?
Were herbal trials performed differently from those as recommended in guidelines for interventional anti-obesity RCTs? If so, how are they different?
Identification of relevant studies
Inclusion and exclusion criteria
Only English-language publications were included. There was no year limit on publications. Review papers and book sections were excluded. All searches were performed and matched by two independent investigators. Search results were managed using bibliographic software (EndNote X9), and duplicates were removed. The following population, intervention, comparison, outcome and study design (PICOS) framework was used to identify relevant articles:
Participants
Participants included individuals of all ages who are obese or overweight, with or without comorbidities. Overweight was defined as a body mass index (BMI) ranging from 25 to <30 kg/m2, while obesity was defined as BMI ⩾30 kg/m2. 37
Interventions
Herbal medicine is defined by the WHO 24 as the main intervention. This review included herbal medicines in the form of a single herb or herbal mixture in any formulation.
Comparators
Studies with any comparators were included.
Outcome
The outcomes of our review focused on parameters of methodologies, mainly:
anthropometric measurements (e.g. BMI, waist and hip circumference and body fat composition)
objective measurement tools of efficacy (e.g. weighing scales, measuring tapes, bioelectrical impedance analysis, dual-energy X-ray absorptiometry (DEXA))
subjective measurement tools of efficacy (e.g. questionnaires to evaluate the participant’s quality of life, stress response, eating habits and other factors related to obesity)
laboratory parameters (e.g. biochemical or hormone tests related to obesity)
behavioural interventions (e.g. diet regimen and physical activities)
multi-disciplinary team approach (e.g. involvement of dietitian or allied healthcare professionals)
use of concomitant herbs, drugs or supplements
parameters for safety monitoring (e.g. clinical examination, clinical bedside tests such as vital signs measurement and electrocardiography, biochemical tests and adverse event self-reporting).
Study design
RCTs were included.
Search strategy
A systematic search was conducted by two independent investigators for published literature with predetermined keywords. Three electronic databases (MEDLINE via PubMed, CENTRAL via Cochrane Library, Embase via Ovid) were searched from their inception until March 2023. The search was constructed around the keyword search terms ‘obesity or overweight’, ‘weight loss’ and ‘herbal medicine’. These were adapted for each database as necessary. The complete search strategy for the databases is shown in Supplemental Material 2.
Selection of studies
The articles from each database were then imported into the Endnote (Version X9, Clarivate Analytics, Philadelphia, 2013) reference manager for deduplication. After deduplication, the remaining articles were transferred to an Excel document for title and abstract screening. This screening was conducted by five pairs of investigators independently and cross-checked afterwards. In cases of disagreements, a third investigator was consulted. Studies were selected based on the inclusion and exclusion criteria with reference to the research questions and PICOS elements mentioned. Studies that appeared to meet the inclusion criteria subsequently undergo screening of the full text. Full-text article screening was conducted by four pairs of investigators independently and cross-checked afterwards. Following full-text screening, two investigators assessed the overall studies included again before proceeding with data extraction.
Data charting
Data extraction was performed by four pairs of investigators. The characteristics of each study were extracted, including study details (author, year of publication, country, study design, trial registry number, ethics approval, study phase, primary and secondary objectives, sample size, drop-out rate, power of study and informed consent), participants (age, sex, inclusion criteria, exclusion criteria, recruitment sites or intervention delivery setting), intervention (type, dose, frequency, duration, route of administration, voucher specimen, qualitative or quantitative tests and any use of concomitant herbs, drugs or supplement), behavioural interventions (diet regimen and physical activities) and outcome measurements (primary and secondary parameters, physical measurements, radiological estimation measurements, biochemical measurements, questionnaires, other methods for determining obesity parameters). We also extracted data on the involvement of dieticians or allied healthcare professionals, compliance monitoring methods and safety monitoring (reporting of adverse events or other related investigations).
Collation, summarisation and reporting of findings
We performed a descriptive numerical analysis using the extracted data details. Subsequently, a comparative analysis was undertaken to assess the methodologies employed in the included studies. This comparison was based on established obesity clinical trial guidelines, specifically the USFDA Guidance for Industry Developing Products for Weight Management (2007) 28 and the EMA Guideline on Clinical Evaluation of Medicinal Products Used in Weight Management (2016). 27
Results
Study selection
A total of 1367 records were identified from online databases. Prior to the abstract screening, 105 duplicate records were removed, resulting in 1262 studies being screened. Of these, 114 full-text studies were assessed for eligibility, and 99 studies were included for analysis. Our primary focus is on the methodologies of the trials rather than their findings. The study selection process is presented in the PRISMA flowchart shown in Figure 1.

PRISMA flowchart of included studies.
Characteristics of included studies
Study characteristics
The study designs of the included articles were all RCTs of herbal-related interventions for obesity. The sample size included in the study ranged from 8 to 182 participants and were mainly from the age range of 18–80 years. There were no participants aged younger than 18 years in the studies found. The BMI range of all the studies was from 25 to 49.9 kg/m2. The participants were either healthy patients with obesity (n = 81 studies) or with comorbidities (n = 18 studies) including hyperlipidaemia, metabolic syndrome, type 2 diabetes mellitus and schizophrenia. The types of intervention used were all herbal medicine as defined by the WHO. 24 The recruitment locations for different studies varied and included virtual recruitment through social media platforms or marketing (n = 13), as well as recruitment sites such as medical and research centres (n = 46), public health clinics (n = 3) and other public facilities (n = 3). Some studies did not mention their recruitment sites (n = 34). Additionally, studies were categorised based on whether they were conducted at a single centre or involved multiple centres. This diversity in recruitment strategies and study settings contributes to the overall understanding of the recruitment landscape in the reviewed studies. In several studies (n = 6), a run-in period was implemented prior to study commencement. The duration of the run-in was from 3 days to 2 weeks. A summary table of the characteristics of the included studies is shown inSupplemental Material 3.
Countries included RCTs
The USA had the highest number of herbal medicine anti-obesity RCTs (n = 18), followed by Korea (n = 12), Iran (n = 11), India (n = 9), Japan (n = 6), Italy (n = 5), Cameroon (n = 4), Australia (n = 4), Brazil (n = 3), Taiwan (n = 3), China (n = 3), Pakistan (n = 2), Germany (n = 2), Canada (n = 2), Poland (n = 2) and France (n = 2). The remaining countries, namely Mexico, Thailand, Romania, Spain, Denmark, Iraq, Russia, Armenia, Syria, Netherlands and Argentina, had one study each. Figure 2 shows the worldwide distribution of anti-obesity RCTs involving herbal medicines.

Geographical demographics of included RCTs.
Herbal intervention, daily dosage, duration
We categorised the herbal interventions into single herbs (n = 57) and mixtures (n = 42). The treatment duration varies from as short as 14 days to as long as 56 weeks. The most studied herbs in obesity RCTs are Ephedra sinica as a mixture (n = 9), Camellia sinensis as a single herb (n = 5) and in mixture (n = 4). Only one study utilised an injectable form for administering the herbal medicine, whereas the others (n = 96) used oral formulations in various forms such as tablets, capsules, gel, powder, granules, decoction, oral spray, syrup, seeds, cereal, confection or served as a beverage. Two studies did not provide information about the type of formulation used. Details of the herbal interventions included RCTs were presented in Table 1.
Herbal intervention details of included RCTs.

CBFI, combination of bioactive food ingredients; GA, gallic acid; RCTs, randomised controlled trials; XJXGF, Xin-Ju-Xiao-Gao-Fang; 5-HTP, 5-hydroxytryptophan.
Lifestyle modifications
For adjunct treatments, we categorized the included studies into the following groups: (1) without lifestyle modifications, (2) diet only, (3) exercise only and (4) both diet and exercise implementation. The distribution of studies across these categories is shown in Figure 3. The majority of studies incorporated dietary modifications, with restricted calorie diet being the most frequently implemented (n = 35), followed by a food-portion controlled diet (n = 7) and fixed calorie diet (n = 7). Additionally, 23 studies adopted a multidisciplinary approach involving a dietitian, nutritionist, or exercise physiologist.43,58,59,62–65,73,74,77,86,93,102–105,107,112,117,122,123,126,128 Among studies incorporating exercise, two included RCTs used light intensity, 22 used moderate intensity, and four studies used a combination of moderate to vigorous intensity. Detailed information on lifestyle modifications in the included RCTs is shown in Table 2.
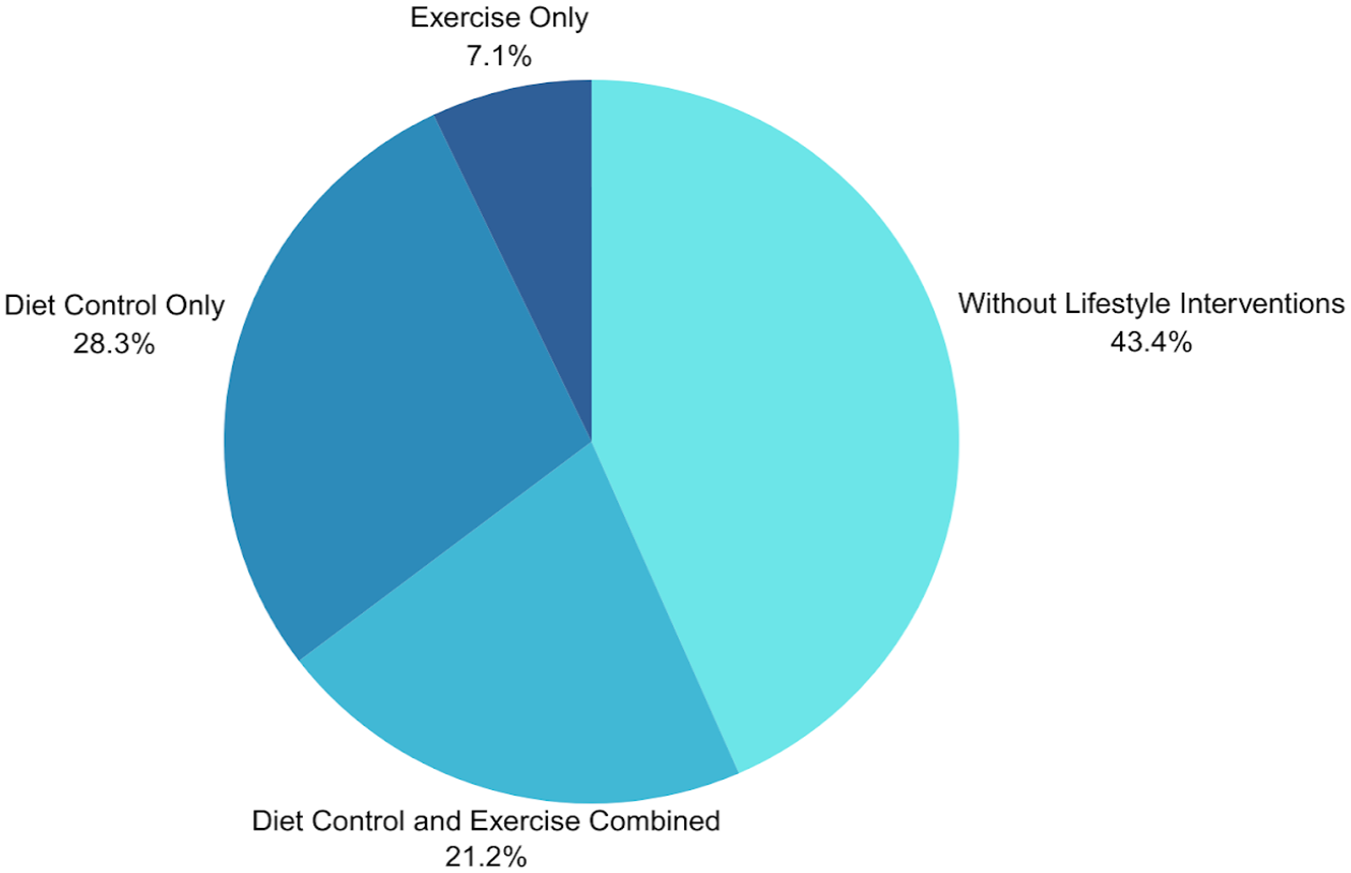
Lifestyle modifications in included studies.
Diet, exercise and combination adjunctive interventions.

Measurement methods for obesity-related parameters
The measurement methods utilised in the included RCTs consisted of physical body measurements, radiological tools, laboratory tests and others. These measurements are used to assess the changes in patient’s metabolic profile. All included RCTs (n = 99) utilised both BMI and weight as their main outcome measurement. Over 50% of the studies measured body fat composition using a body analyser (n = 57). Other anthropometry measurements such as waist circumference, hip circumference and abdominal circumference were not routinely measured.
As for the radiological tools used to assess obesity-related parameters, DEXA (n = 16), Computed Tomography (CT scans) (n = 10) and ultrasound (n = 2) were used. The utilisation of radiological measurements, including DEXA, CT scans and ultrasound, in the evaluation of obesity-related parameters provide more detailed information about the distribution and composition of body fat, as well as other related factors.
Majority 87% (n = 86) of the studies utilised laboratory test for biosampling including blood and urine. These laboratory assessments include the lipid profile which is the most commonly used biochemical test to be conducted during anti-obesity study. Apart from that, the studies utilised hormone tests such as insulin, adiponectin, thyroid hormone, testosterone, cortisol and leptin; urinalysis; inflammatory markers such as C-reactive protein, interleukin 6 and plasma fibrinogen; oxidative stress markers such as lipid peroxidation levels, plasma antioxidant capacity, glutathione; and nutrition profile such as vitamin panels and glycoalbumin.
The methods of measurement of obesity-related parameters are shown in Table 3.
Methods of measurement of obesity-related parameters.
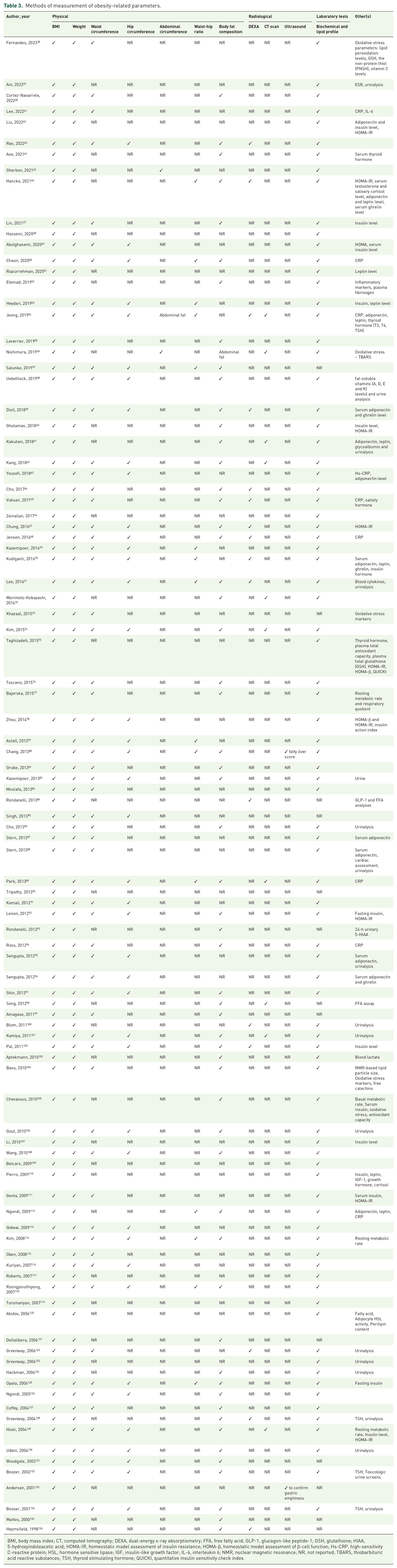
BMI, body mass index; CT, computed tomography; DEXA, dual-energy x-ray absorptiometry; FFA, free fatty acid; GLP-1, glucagon-like peptide-1; GSH, glutathione; HIAA, 5-hydroxyindoleacetic acid; HOMA-IR, homeostatic model assessment of insulin resistance; HOMA-β, homeostatic model assessment of β-cell function; Hs-CRP, high-sensitivity C-reactive protein; HSL, hormone sensitive lipase; IGF, insulin-like growth factor; IL-6, interleukin 6; NMR, nuclear magnetic resonance; NR, not reported; TBARS, thiobarbituric acid reactive substances; TSH, thyroid stimulating hormone; QUICKI, quantitative insulin sensitivity check index.
Approximately 47% (n = 46) of the studies employed questionnaires to assess participants’ dietary and nutritional intake, along with their appetite, mood and overall well-being. These include the Food Frequency Questionnaires, 3-day food records, 24-h dietary recalls, food history questionnaires, food compliance questionnaires, dietary composition analysis using specialised software, assessment of hunger and fullness, mood evaluations, quality of life (QOL) assessments, international physical activity questionnaires and metabolic equivalent scales. These tools record participants’ food intake, dietary patterns and their adherence to study guidelines. Some of the questionnaires were internationally validated such as the Korean version of Eating Attitudes Test-26, Three-Factor Eating and the Obesity and Weight-Loss Quality of Life measure. The questionnaires utilised in the studies are shown in Table 4.
Questionnaires used in the RCTs.

BDI-II, Beck Depression Inventory; BES, Binge Eating Scale; IPAQ, International Physical Activity Questionnaire; KEAT-26, Korean version of Eating Attitudes Test-26; KOQOL, Korean Obesity-related Quality of Life; MET, Metabolic Equivalent; OWLQOL, Obesity & Weight-Loss Quality of Life measure; POMS, Profile of Mood Status; QSCC, Questionnaire for the Sasang Constitution Classification; SRI, Stress Response Inventory; SRRS, Social Readjustment Rating Scale; VAS, Visual Analogue Scales; WRSM, Weight-Related Symptom Measure.
Power of study and sample size calculation
In our analysis of the included studies, it was found that 37 out of the total 99 studies achieved the final sample size based on their stated study power calculations and sample size justifications.39–42,44,59,60,64–66,75,87,100–105,117,127,135 Among these, 33 studies provided clear documentation of their power, significance level and effect size estimates. However, in four studies, the reporting on effect sizes was not sufficiently specified.99,101,108,135
Compliance monitoring methods
Nine methods of compliance monitoring were reported in the included RCTs (n = 70) categorised as follows, ‘Patient diary’ involved monitoring the pill counting, daily records of experimental tablet intake, food intake frequency records, physical activity records, ‘Email communication’ involved monitoring compliance through email in addition to site visits, ‘Regular Telephone Calls’ maintained protocol adherence through calls and messaging apps, ‘Capsule Calendar’ provided calendars for medication tracking, ‘Measuring Cup and Containers’ included measuring cups and containers for supplement measurement and compliance assessment, ‘Follow-Up Visits’ assessed compliance during scheduled visits, ‘Direct Observation’ confirmed supplement consumption through direct observation, ‘Compliance Survey’ assessed adherence during visits and ‘Feedback and Strategies’ provided feedback and strategies to enhance compliance. There were 29 studies that did not report on the compliance monitoring methods they undertook. The most used compliance monitoring method was patient diary recording (n = 28). Details of the compliance monitoring methods and related studies are shown in Table 5.
Compliance monitoring methods used in included RCTs.

RCT, randomised controlled trials.
Safety monitoring
Majority of the studies (n = 76) either mentioned in their methods section or in their methodology or suggested through their results and discussion section that some safety monitoring was conducted. A handful of studies (n = 6) did not mention safety monitoring or adverse events at all throughout the report. The safety monitoring methods include ‘Adverse Event Recording’ involving the documentation of adverse events, their severity, and relation to the intervention at follow-up visits, ‘Laboratory Tests’ including baseline and post-treatment measurements of haematological and biochemical parameters for safety assessment, ‘Other Safety-Related Parameters’ involving measurements like blood pressure, pulse rate and ECGs to assess safety and ‘Assessment of Compliance and Adverse Events’ evaluating adherence alongside adverse event monitoring, including the counting of unused study products. Details of the safety monitoring methods and related studies are shown in Table 6.
Safety monitoring methods used in the RCTs.
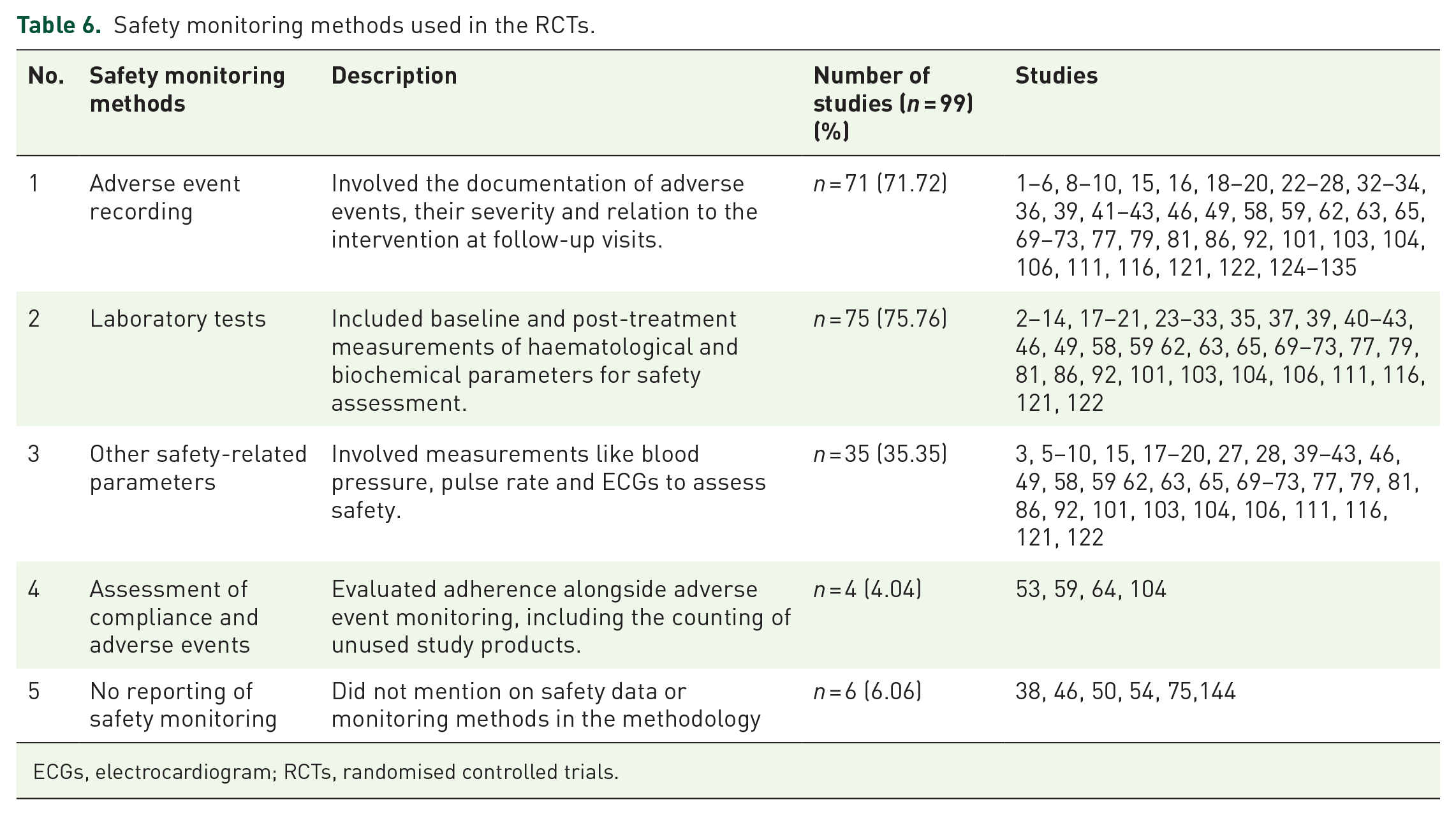
ECGs, electrocardiogram; RCTs, randomised controlled trials.
Methods of follow-up
Methods of follow-up reported in the included RCTs can be categorised into five major types based on the planned outcomes of interest that is measured at each visit. Firstly, ‘In-Person on Site’ visits involved participants physically visiting a specific location for various activities, including measurements, interviews and sample collections, often occurring at regular intervals. Secondly, ‘In Person’ visits included face-to-face interactions with participants, but the location (on-site or elsewhere) and specific activities were unspecified. Thirdly, ‘In Person at Site and Phone Calls’ encompassed study visits conducted both in person at a physical site and via phone calls, allowing for a combination of physical examinations and remote interactions. Additionally, ‘Virtual Visits’ were carried out virtually, eliminating the need for physical presence, though the activities involved were not specified. Lastly, ‘Routine Health Checkup’ visits provided participants with routine health assessments, with specific components remaining unspecified in the provided information. The most common follow up methods utilised was the ‘In-Person on Site’ (n = 74). The follow-up methods and related studies are shown in Table 7.
Follow-up methods used in the RCTs.

RCT, randomised controlled trials.
Guideline recommendations
We extracted pertinent guideline recommendations from the USFDA Guidance for Industry Developing Products for Weight Management (2007) 28 and the EMA Guideline on Clinical Evaluation of Medicinal Products Used in Weight Management (2017) 27 to align with anti-obesity clinical trial standards. The details on the guideline points with RCTs that adhere to them are shown in Table 8.
Guideline recommendations for clinical trials in weight management product development.

CVS, cardiovascular system; DEXA, dual-energy X-ray absorptiometry; DM, diabetes mellitus; ECG, electrocardiogram; QOL, quality of life; WC, waist circumference
In conducting anti-obesity trials, specific methodological aspects of both guidelines are essential to ensure compliance with regulatory standards and align with scientific best practices. Both guidelines emphasise that trials should have a minimum duration of 12 months to adequately assess the long-term effects on weight management and associated comorbidities. The primary endpoint should be body weight loss, aiming for at least a 5% reduction from baseline to demonstrate efficacy. Additionally, secondary outcomes should include improvements in comorbidities such as cardiovascular risk and diabetes, QOL and waist circumference measurements. Safety and tolerability are crucial components of these trials, requiring careful monitoring of safety outcomes, including dose adjustments and any adverse effects. Lastly, the selection of participants should focus on individuals with a baseline BMI of at least 27 kg/m², particularly those with obesity-related health risks, to ensure the study population reflects the target demographic for potential treatments.27,28
Based on the guideline aspects, the comparative analysis showed that most studies (n = 83) had a study duration of 0–6 months, while only six studies were carried out between 6 and 12 months, and just two exceeded 12 months. Majority studies had primary endpoint of weight loss (n = 80) with two of the studies also having an aim of 5% weight reduction from baseline. There were also studies that account for cardiovascular risk reduction in conjunction with the weight loss herbal intervention (n = 49). Safety monitoring was reported in most studies (n = 93).
Discussion
In our review, we identified 99 studies meeting our inclusion criteria, primarily involving individuals aged 18–80 years, with BMIs between 25 and 49.9 kg/m2. Herbal interventions were diverse, comprising single herbs and mixtures, with durations spanning from 14 days to 56 weeks. Diet modifications were implemented through various approaches, including restricted calorie diets, food-portion controlled diets and fixed calorie diets. Exercise, often at moderate intensity, was commonly integrated with the interventions. BMI and weight were the primary outcomes across all studies, while body fat composition was measured in over 50% of studies using various methods such as body analysers and radiological tools. Interestingly, there were no interventional anti-obesity RCTs on herbal medicine conducted for participants aged younger than 18 years in our included studies, indicating a gap in research despite the rising cases of childhood obesity. This suggests that herbal medicine trials have not yet been globally recognised as an alternative therapy for combating obesity. However, survey studies have shown that adolescents aged 12–18 do in fact take herbal medicines for weight loss. 137 Research on the underage is challenging due to the requirement of parental consent and the risk of litigation. Hence, lifestyle changes are often advocated first before any medical intervention.
Some of the RCTs implemented a run-in period prior to the commencement of their studies. The purpose is to identify and exclude participants who may not adhere to the study protocol or have a high risk of dropping out. The run-in phase of clinical trials can double as a washout period to gradually discontinue prior medications to prevent sudden cessation effects and clear previous treatments from patients’ systems before introducing new interventions. Some of the trials integrated this approach, ensuring any lingering effects are eliminated before administering the herbal treatment.46,58,81,101,108 These strategies enhance the validity and safety of study outcomes, especially for those with multiple medication regimens. Leahey et al. 138 assessed the predictive value of behavioural run-in data, encompassing food diary completion, questionnaire responses and staff interviews, on intervention adherence, trial retention and outcomes in a behavioural weight loss trial. Using run-in information, researchers categorised participants as having high, moderate, or low adherence and then randomised those with predicted high or moderate adherence. Results indicated that predicted high adherers exhibited better intervention adherence, as evident from session attendance and record completion, and achieved more favourable outcomes, particularly in terms of weight loss. However, run-in data did not predict trial retention. This suggests that run-in assessments effectively identify participants likely to adhere to intervention protocols, bolstering behavioural efficacy trial validity.
Adherence to medication is crucial, but adherence to follow-up is equally significant and is most commonly measured by the follow-up rate. A systematic review reported that the pooled odds ratio (OR) for improved follow-up rate in the group receiving Short Message Service (SMS) reminders compared with the control group was 1.76 (95% CI (1.37, 2.26); p < 0.01), while the pooled OR for enhancing the follow-up rate in the group with telephone reminder compared to the control group was 2.09 (95% CI (1.85, 2.36); p < 0.01). Although telephone reminders demonstrated a greater probability than SMS reminders in improving the follow-up rate, the authors argued that cell phone SMS interventions could be more practical and easier to adapt to various settings. 139 Among the included studies in this review, a majority of the herbal medicine anti-obesity studies did not report on the adherence rate, but those that did (n = 30) reported up to 70%–100% adherence rate. In our study, of the five studies that utilised email communication 43 and regular telephone calls,63,66,113,123 only three reported adherence rates exceeding 87.5%, indicating a satisfactory level of adherence.63,66,113 Hence, at this point in time, the exact methods to improve adherence to herbal trials are still unclear.
Clinical counselling that emphasises dietary change stands out as a key strategy in addressing adult obesity. 140 Dietitians and nutritionists are healthcare experts specialising in weight management, play a pivotal role in delivering this form of treatment. 141 Participants who received intervention from dietitians achieved an extra weight loss of 1.03 kg (95% CI (−1.40, −0.66); p < 0.0001) and a BMI reduction of 0.43 kg/m2 (95% CI (−0.59, −0.26); p < 0.0001) compared to those who received usual care. 142 In this review, studies utilising these allied healthcare professionals (n = 23) however did not consistently report lower dropout rates overall. The drop-out rates ranged from 0 to 39%, whereby the drop-outs for seven studies ranged 0%–9%,59,72,86,92,104,121,125 six studies ranged 10%–19%,58,74,102,103,111,122 seven studies ranged 20%–29%,43,63,65,77,101,106,127 and three studies ranged 30%–39%.62,64,116
In accordance with the USFDA (Guidance for Industry Developing Products for Weight Management 2007) and EMA (EMA Guideline on Clinical Evaluation of Medicinal Products Used in Weight Management 2017) guidelines, patient selection often considers a lower baseline BMI, especially for individuals with weight-related risk factors (e.g. ⩾27 kg/m2). While interventions may contribute to improvements in cardiovascular health, they cannot be attributed to reducing blood pressure, glucose levels, heart rate, etc., apart from the targeted claim of weight loss. The guidelines recommend a study duration of 12 months to comprehensively assess the impact on weight development and obesity-related comorbidities. Our study conducted a comparative analysis between methodologies employed in herbal RCTs and the guidelines for developing products for weight management. This comparison allows an assessment of how the methods prescribed in clinical trial guidelines align with those implemented in real-life studies. Notably, the primary efficacy endpoint in most studies is body weight loss, with limited consideration for weight maintenance over 6 months to1 year, as suggested by the guidelines. Few studies reported on certain secondary efficacy endpoints, such as sleep apnoea, joint pain, urinary incontinence, impaired fertility, depression, anxiety and functional limitations. One study included patients with schizophrenia. 41 In adding a comparative context with guidelines, both the USFDA and EMA recommend anthropometric measurements, with BMI and weight being the most utilised. Although the guidelines advise the implementation of diet and physical activity in anti-obesity studies, only 21 studies incorporated these lifestyle modifications alongside herbal interventions. A significant portion of the included studies (n = 43) did not implement lifestyle modifications, requiring participants to maintain their current lifestyle. This may be due to the apparent difficulty in administering and monitoring compliance with lifestyle interventions alongside herbal interventions. Conducting anti-obesity RCTs poses challenges, as most of these included studies planned for participants to maintain their preexisting lifestyle to avoid additional confounding factors influencing body weight. This is contrary to the guidelines that recommend the implementation of lifestyle modifications alongside the tested interventions. We were unable to extract three suggestions outlined in the guidelines, including ‘meaningful dose reduction/withdrawal from concomitant medication’, ‘selection of patients who have failed at least one trial of weight-reducing diet’, and the ‘inclusion of various demographic, ethnic and racial groups where obesity prevalence is high’. These data were not reported in the trial publications across the included studies.
In our review, 93 out of 99 studies reported safety monitoring of the participants, revealing a strong awareness of this crucial aspect. However, we would expect a 100% reporting rate for such a critical aspect. Safety reporting is a mandatory requirement in RCTs, particularly in pharmaceutical trials, as emphasised by the CONSORT checklist (2010). 30 However, in herbal medicine or natural product clinical trials, the emphasis on safety reporting is sometimes overlooked in comparison to efficacy reporting. This discrepancy may stem partly from regulatory differences, such as those observed in Malaysia’s regulations concerning herbal and traditional medicines. Marketed products in this category may face less stringent approval processes compared to pharmaceuticals, potentially posing challenges in monitoring safety and quality. Limited consumer awareness of the risks associated with herbal medicine further compounds this issue. 143 Fortunately, there is growing recognition of the importance of safety reporting in herbal interventions. Specific clinical trial guidelines, such as the CONSORT Extension for Chinese Medicine Formulas 2017 31 and the Guideline for Herbal Medicine Research 2023, 144 underscore the importance of prioritising safety in herbal medicine clinical research.
This scoping review has certain limitations that should be considered when interpreting its findings. Only studies published in English were included due to resource constraints. We did not exclude pilot studies or those with less than 6 months of follow-up, a decision made to ensure a comprehensive overview of the available literature. Due to the inadequate reporting of the sexes analysed in the included trials, we also could not conclude the numbers of recruited genders. Lastly, our assessment of included studies is based on reported information in published literature only. There may be instances where quality indicators were addressed in a trial but not reported, influenced by factors such as word count restrictions, editorial decisions and investigators’ perceptions of importance. We utilised clinical study guidelines for weight management products, as there are currently no standard international guidelines for conducting RCTs specifically focusing on herbal medicine in anti-obesity interventions. These guidelines are based on drafts from the USFDA and the EMA, last updated in 2007 and 2017, respectively. Although not recently updated, these guidelines serve as a foundational reference for comparing the methods commonly employed in weight management trials. This review did not assess the included RCTs against the CONSORT guidelines, as the primary scope was to evaluate the methodology in relation to the two regulatory guidelines. However, future studies can consider including an assessment based on CONSORT guidelines to provide a more comprehensive evaluation of the included RCTs.
Conducting RCTs for anti-obesity interventions is challenging, given the multitude of factors to consider. These include various parameters such as weight loss measures, evidence of fat reduction, sustained weight maintenance, lowered cardiovascular risks, among others. Additionally, the ideal requirement for long-term clinical trials to establish both efficacy and sustainability adds to the complexity. Despite the difficulties, developing effective weight-loss products can bring significant benefits, particularly in marketing, if the trials show positive results across various indicators. Companies that can manage these challenges may set themselves apart by creating reliable, evidence-based products, which is something not commonly achieved due to the inherent complexity of conducting such comprehensive trials.
In conclusion, approximately half of the RCTs assessing the anti-obesity efficacy of herbal medicine interventions adhere to international guidelines, incorporating considerations for diet control, lifestyle, physical activity and the assessment of cardiovascular risk factors. In addition to efficacy, adherence to recommendations for safety monitoring and reporting should also be emphasised. There is a need for more herbal anti-obesity RCTs to evaluate weight maintenance among participants.
Supplemental Material
sj-docx-1-taj-10.1177_20406223241310206 – Supplemental material for Methodological insights into intervention and outcomes in randomised controlled trials of herbal medicine for obesity: a scoping review
Supplemental material, sj-docx-1-taj-10.1177_20406223241310206 for Methodological insights into intervention and outcomes in randomised controlled trials of herbal medicine for obesity: a scoping review by Janice Sue Wen Chan, Mei Siu Lau, Siti Hajar Muhamad Rosli, Siti Khadijah Mustapha Kamal, Wan Nurul Nadia Wan Seman, Nurul Hidayah Ali, Terence Yew Chin Tan, Ida Farah Ahmad, Puspawathy Krishnan, Marisa Khatijah Borhan, Ami Fazlin Syed Mohamed and Xin Yi Lim in Therapeutic Advances in Chronic Disease
Supplemental Material
sj-docx-2-taj-10.1177_20406223241310206 – Supplemental material for Methodological insights into intervention and outcomes in randomised controlled trials of herbal medicine for obesity: a scoping review
Supplemental material, sj-docx-2-taj-10.1177_20406223241310206 for Methodological insights into intervention and outcomes in randomised controlled trials of herbal medicine for obesity: a scoping review by Janice Sue Wen Chan, Mei Siu Lau, Siti Hajar Muhamad Rosli, Siti Khadijah Mustapha Kamal, Wan Nurul Nadia Wan Seman, Nurul Hidayah Ali, Terence Yew Chin Tan, Ida Farah Ahmad, Puspawathy Krishnan, Marisa Khatijah Borhan, Ami Fazlin Syed Mohamed and Xin Yi Lim in Therapeutic Advances in Chronic Disease
Supplemental Material
sj-docx-3-taj-10.1177_20406223241310206 – Supplemental material for Methodological insights into intervention and outcomes in randomised controlled trials of herbal medicine for obesity: a scoping review
Supplemental material, sj-docx-3-taj-10.1177_20406223241310206 for Methodological insights into intervention and outcomes in randomised controlled trials of herbal medicine for obesity: a scoping review by Janice Sue Wen Chan, Mei Siu Lau, Siti Hajar Muhamad Rosli, Siti Khadijah Mustapha Kamal, Wan Nurul Nadia Wan Seman, Nurul Hidayah Ali, Terence Yew Chin Tan, Ida Farah Ahmad, Puspawathy Krishnan, Marisa Khatijah Borhan, Ami Fazlin Syed Mohamed and Xin Yi Lim in Therapeutic Advances in Chronic Disease
Footnotes
Acknowledgements
The authors gratefully acknowledge the Director General of Health Malaysia, Deputy Director General of Health Malaysia (Research and Technical Support), Director of Institute for Medical Research and the Head of Herbal Medicine Research Centre for their authorisation and permission for the publication of this article.
Declarations
ORCID iDs
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
