Abstract
Growth factors were introduced to increase predictability in periodontal regeneration and have since been widely applied in dentistry. This narrative review article highlights histological and latest findings of recombinant human platelet-derived growth factor-BB (rhPDGF-BB) and recombinant human fibroblast growth factor-2 (rhFGF-2) for periodontal regeneration. rhPDGF-BB enhances the proliferation and chemotaxis of periodontal ligament and alveolar bone cells. The optimal dose for rhPDGF-BB, in combination with beta-tricalcium phosphate, is 0.3 mg/ml. It is approved in the United States, Canada, and Taiwan for use in periodontal regeneration and treatment of gingival recession. rhFGF-2 promotes periodontal wound healing through mitogenic and angiogenic effects on mesenchymal cells in the periodontal ligament. It is approved in Japan at an optimal dose of 0.3% for periodontal regeneration in intrabony defects. Both recombinant growth factors show histological evidence of new bone, cementum, and periodontal ligament. Clinical studies demonstrate improved clinical attachment levels and defect resolution for treating intrabony and furcation periodontal defects. Presented clinical cases and consensus reports may serve as a reference for clinicians. rhPDGF-BB and rhFGF-2 are safe and effective biologics that can be applied to improve the outcomes of periodontal regeneration.
Plain language summary
Growth factors are unique proteins introduced to make gum and bone regeneration more predictable in dental treatments. They are now widely used in dentistry. This review focuses on two specific growth factors: recombinant human platelet-derived growth factor-BB (rhPDGF-BB) and recombinant human fibroblast growth factor-2 (rhFGF-2). Key Points: - rhPDGF-BB helps gum and bone cells grow and move to the damaged area. The best dose is 0.3 mg/mL, usually combined with a material called beta-tricalcium phosphate (β-TCP). It’s approved in the US, Canada, and Taiwan for regenerating tissue and treating gum recession. - rhFGF-2 aids in healing gum wounds by encouraging cell growth and new blood vessel formation. It’s approved in Japan at a dose of 0.3% for treating deep gum and bone defects. Both growth factors have been shown to help form new bone, cementum (the surface layer of the tooth root), and periodontal ligament (the tissue that helps hold the teeth in place). Clinical studies have found that they improve the connection between the tooth and gum and help resolve deep defects. Case studies and expert reports provide valuable guidance for dentists. Both rhPDGF-BB and rhFGF-2 are safe and effective for enhancing gum and bone regeneration in dental treatments.
Background
A rise in education and awareness of oral health-related quality of life among patients has resulted in tooth retention. 1 It is becoming evident in the aging population that patients are investing time and money in saving teeth to ensure a better quality of life. 2 Tooth retention promotes essential functions such as eating and speaking. In addition, it encompasses a psychological component such as self-confidence and the ability to socialize and work. A few studies have investigated whether hopeless teeth can be retained due to a compromised periodontal status and how this would impact adjacent teeth. Machtei and Hirsch 3 have found that hopeless teeth treated with periodontal therapy can be maintained long-term with no detrimental effect on adjacent proximal teeth. One of the most important factors in tooth retention is regular professional dental hygiene therapy and effective oral hygiene practice. 4 Levin and Halperin-Sternfeld 5 conducted a systematic review of the long-term survival rate between dental implants and compromised natural teeth. It was found that adequately treated and maintained teeth have comparable survival rates to dental implants. Therefore, the decision to replace teeth with dental implants should be made carefully and with proper treatment planning. Several treatment options are available to retain teeth, such as scaling and root planing, splinting, antiseptics, antibiotics, occlusal therapy, dental laser, and surgical therapy, including periodontal regeneration.
The concept of periodontal regeneration involves the replacement of destroyed tissue on a previously diseased root surface by the reformation of cementum, periodontal ligament, and alveolar bone. 6 Many factors must be considered when utilizing periodontal regeneration in clinical scenarios. For example, oral hygiene, smoking, systemic conditions, tooth mobility, defect shape, and probing depth must be carefully assessed to identify whether periodontal regenerative therapy can be successfully applied. 7 The indications and contraindications for periodontal regeneration have remained consistent throughout the years, but the surgical approaches have changed. The armamentarium has shifted from using autogenous bone grafts 8 to bone allografts9,10 to guided tissue membranes11–13 to growth-factor infused bone grafts.14–16
Growth factors were introduced to increase predictability in periodontal regeneration and have since been widely used. The biological rationale for using growth factors in regeneration comes from the ability to enhance and accelerate the natural healing processes by promoting cellular activities critical for tissue repair and regeneration. In Europe, enamel matrix derivative (EMD) is commonly applied in regenerative therapy. 17 This biologic is composed of amelogenins and carries the potential for cementogenesis, which can enhance regeneration outcomes. 18 Currently, two recombinant growth factors are approved for clinical usage in periodontal regeneration. Recombinant human platelet-derived growth factor-BB (rhPDGF-BB), in combination with beta-tricalcium phosphate, obtained United States Food and Drug Administration approval in 2005. 19 rhPDGF-BB was first demonstrated in periodontal regeneration in an in vitro study. 20 rhPDGF-BB enhances the proliferation and chemotaxis of periodontal ligament and alveolar bone cells.21,22 The outcomes of periodontal regeneration are, therefore, enhanced and can be maintained long-term. 23 The Ministry of Health, Labor and Welfare in Japan approved recombinant human fibroblast growth factor-2 (rhFGF-2) in 2016. 24 The regeneration of periodontal tissues using rhFGF-2 has been demonstrated in clinical studies. This growth factor promotes mitogenic and angiogenic effects on mesenchymal cells in the periodontal ligament.25–27 The efficacy of rhPDGF-BB and rhFGF-2 in combination with various biomaterials, including allografts, xenografts, and alloplasts, will be explored here. This narrative review article highlights historical and recent findings of two available recombinant growth factors for periodontal regeneration. Clinical cases presented provide practical insights into the use of biologics outside of controlled clinical trials, demonstrating their effectiveness and challenges in a typical private practice environment.
Recombinant human platelet-derived growth factor-BB
Animal studies
PDGF was demonstrated as a potent stimulator of DNA synthesis in in vitro and in vivo studies using fetal rat calvaria and osteoblast-like cells derived from adult human bone explants, respectively.28,29 The first evidence of periodontal regeneration using platelet-derived growth factor was demonstrated by Lynch and coworkers in a beagle dog model.30,31 The biologic agent was applied in a purified form (1 µg) to the root surfaces of teeth following open flap debridement. An assessment of local bone metabolism and histologic evaluation of block biopsies revealed increased metabolic activity and formation of the periodontal attachment apparatus (cementum, periodontal ligament, and alveolar bone). Another group investigated the use of platelet-derived growth factor (5 µg/10 µ of 0.05 M acetic acid/tooth) in combination with an expanded polytetrafluoroethylene barrier for periodontal regeneration in Class III furcation defects.21,22 Results from histologic and histomorphometric analyses also revealed the formation of the periodontal attachment apparatus in beagle dogs. 22 A split-mouth dog study comparing rhPDGF-BB + beta-tricalcium phosphate (β-TCP) and rhPDGF-BB + equine particulate bone graft in 2-wall intrabony defects found comparable clinical improvements and histologic evidence of periodontal regeneration. 32 The findings from these studies prompted further investigation and the need for human randomized controlled trials.
Human studies
A combination of rhPDGF-BB and β-TCP, a synthetic scaffold matrix, in the treatment of infrabony defects, has been investigated in a large multicenter randomized controlled trial. 19 The defect had to include a probing depth of at least 7 mm and a vertical bone defect depth of 4 mm or more with at least one intact bony wall. The treatment arms included β-TCP + 0.3 mg/ml rhPDGF-BB, β-TCP + 1.0 mg/ml rhPDGF-BB, and β-TCP + buffer. An early acceleration of clinical attachment level gain, reduced gingival recession, greater bone gain, and percent defect fill in groups treated with rhPDGF-BB could be seen at 6 months. A follow-up to the initial trial found these results stable over 36 months. 23 rhPDGF-BB with β-TCP demonstrated clinical and radiographic improvements that can be maintained long-term. Nevins et al. 33 tested the application of rhPDGF-BB (0.5, 1.0, or 5.0 mg/ml) with bone allograft in intrabony and Class II furcation defects. This study found improved probing depths, clinical attachment gain, radiographic bone fill, and histologic evidence of periodontal regeneration. rhPDGF-BB in combination with xenografts has also been investigated for periodontal regeneration in intrabony defects. A study comparing rhPDGF-BB (0.3 mg/ml) + β-TCP and rhPDGF-BB + equine-derived bone matrix found similar improvements in clinical and radiographic outcomes for periodontal regeneration in infrabony defects. 34 Several human studies exist to demonstrate the safety and efficacy of rhPDGF-BB in combination with various bone graft materials for periodontal regeneration.19,23,33,34
Clinical cases

Periodontal regeneration of an infrabony defect. (a) Preoperative intraoral view. (b) Preoperative periapical radiograph. (c) Exposure of defect following buccal full-thickness flap elevation and scaling and root planing. (d) Mix of allograft and rhPDGF-BB placed at defect. (e and f) 3-Year clinical and radiographic follow-up. (g and h) 5-Year clinical and radiographic follow-up. (i and j) 10-Year clinical and radiographic follow-up, revealing a stable result.
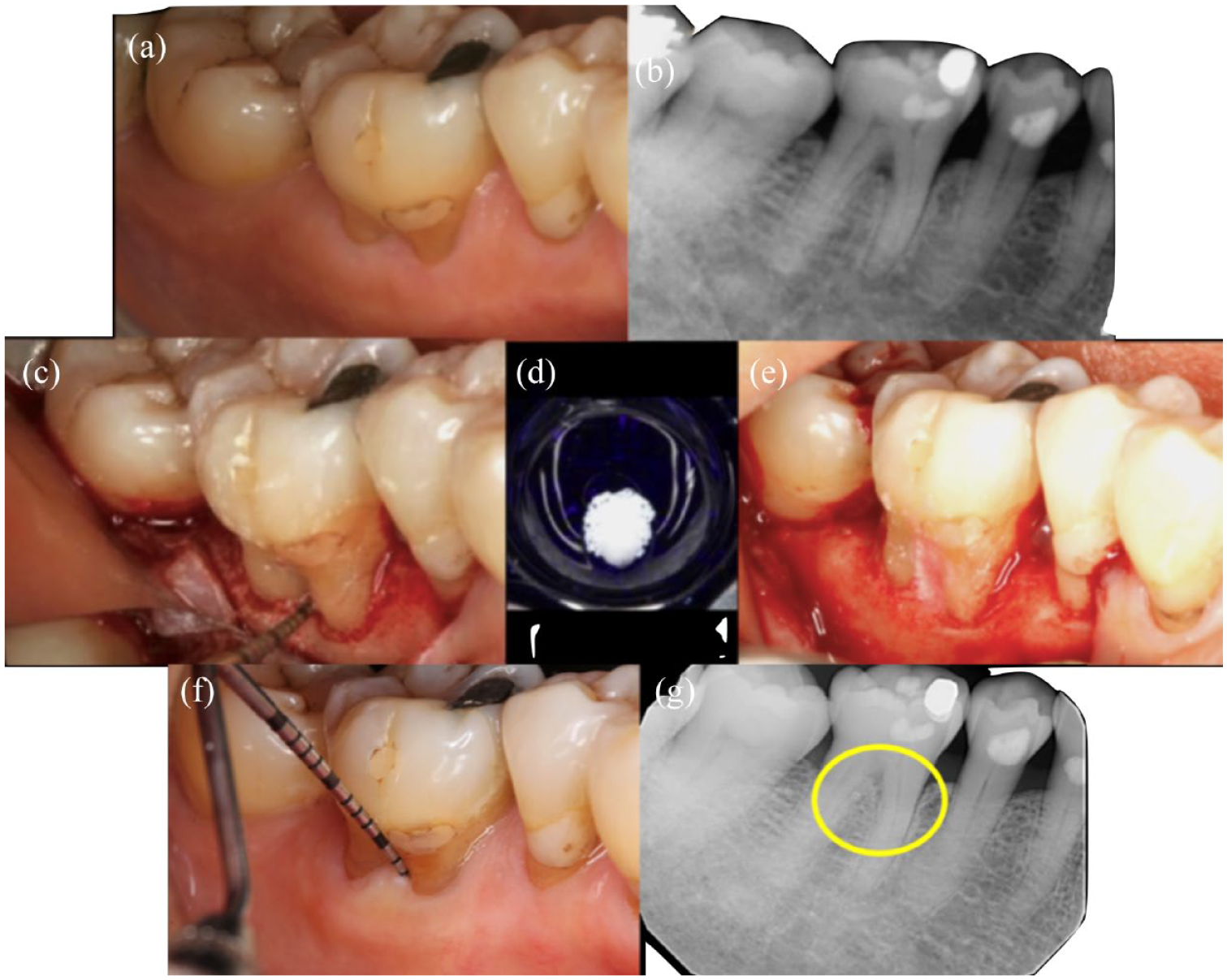
Periodontal regeneration of a furcation defect. (a) Preoperative intraoral view. (b) Preoperative periapical radiograph. (c) Exposure of defect following buccal full-thickness flap elevation and scaling and root planing. (d) Xenograft soaked in rhPDGF-BB. (e) Application of biomaterials at the defect. (f and g) 11-Year clinical and radiographic follow-up, revealing the stable result.
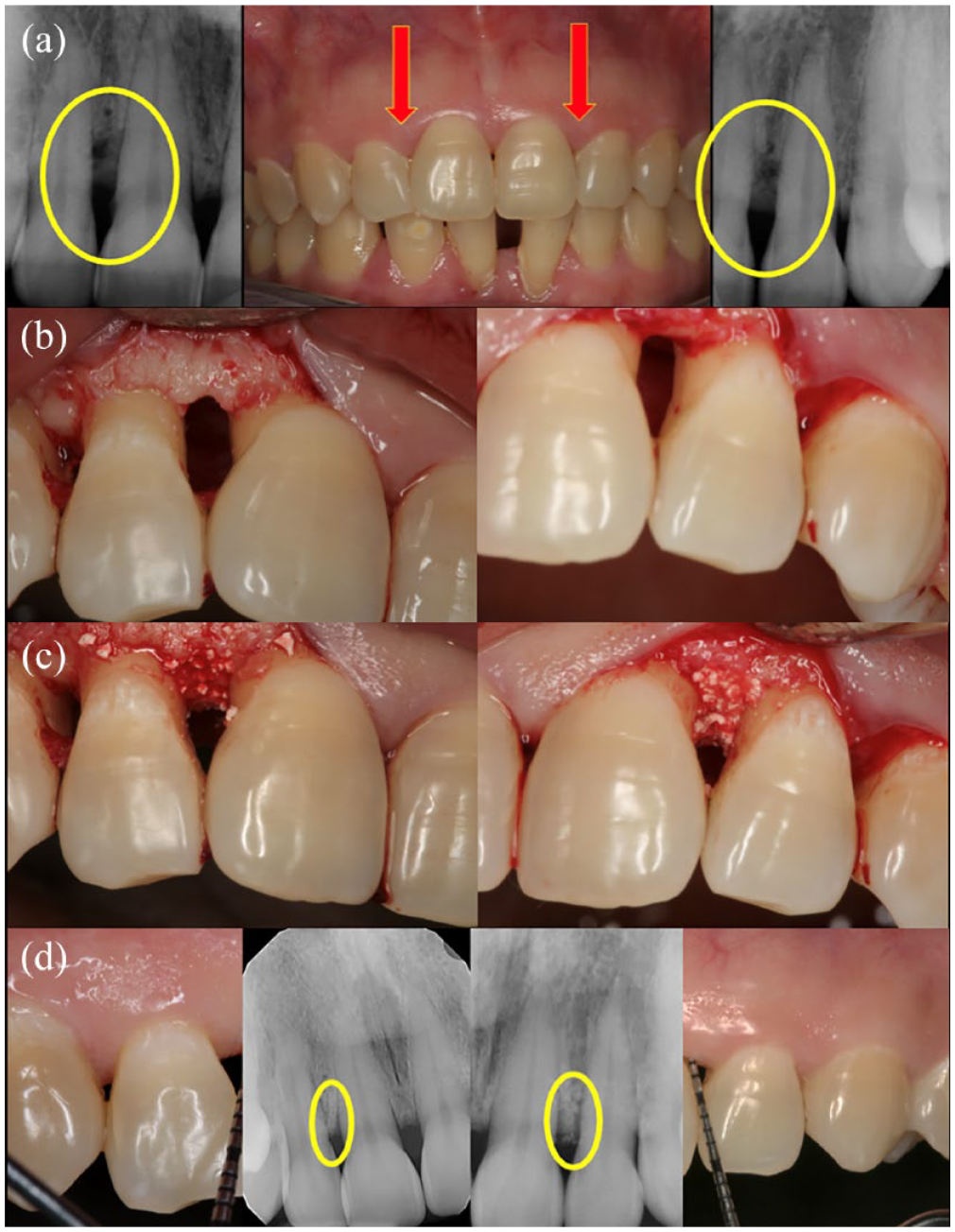
Periodontal regeneration of bilateral infrabony defects. (a) Preoperative intraoral and radiographic view. (b) Exposure of defect following buccal full-thickness flap elevation and scaling and root planing. (c) Application of alloplast and rhPDGF-BB at defect. (d) 10-Year clinical and radiographic follow-up, revealing the stable result.
Consensus reports
A recent systematic review and meta-analysis on biologics for treating periodontal infrabony defects was published as part of the American Academy of Periodontology Best Evidence Consensus. 35 It was found that combination therapy, biologic and bone graft, was superior to monotherapy, bone graft alone. Adding barrier membranes did not enhance outcomes in the presence of a biologic. Superior clinical outcomes were achieved when a biologic was combined with allogenic or xenogenic bone grafts. rhPDGF-BB showed more significant clinical attachment gain, pocket depth reduction, and radiographic bone gain than other biologic agents. In another Best Evidence Consensus statement, Avila Ortiz et al. 36 discussed clinical practice recommendations for applying biologics. A biologic agent may provide additional benefits to conventional therapy, especially in more challenging cases. It may be considered for use in patients with poor wound healing, less predictable defects, limited surgical access, aesthetics, and/or a history of failed therapy. The clinician should also assess local and systemic factors at the patient level to ensure optimal case selection for biologics.
Recombinant human fibroblast growth factor-2
Animal studies
In 1974, the fibroblast growth factor was isolated from cow pituitary glands. 37 In in vitro and in vivo studies, it was found that FGF-2 promotes cell proliferation and inhibits chondrocytic differentiation.38–40 Furthermore, the biologic agent was found to promote revascularization and facilitate bone fracture healing by stimulating mesenchymal stem cell growth and biochemical functions.41,42 rhFGF-2 was first evaluated in periodontal regeneration therapy for treating furcation class II defects in animal models, including beagle dogs and monkeys.26,27 Compared to the control groups, the treatment groups using rhFGF-2 alone (0.3%) showed significant periodontal regeneration for 3-wall infrabony defect, consisting of new cementum, periodontal ligament, and alveolar bone. 43 Similar results were seen in an artificially created 2-wall infrabony defect in a beagle dog model. 44 Animal studies using combination therapy, rhFGF-2 plus membrane or bone graft, demonstrated superior outcomes of regeneration in comparison to monotherapy.45–48 These results stimulated interest and the need for human randomized controlled trials before the widespread application of the biological agent.
Human studies
A multicenter randomized controlled trial was performed to treat 2- and 3-wall infrabony defects using a placebo and rhFGF-2 concentrations of 0.03%, 0.1%, or 0.3%. 49 The results showed a significant difference in the rate of linear increase in bone height between the placebo (23.92%) and 0.3% rhFGF-2 (58.62%) groups at 36 weeks. In a similar study, various concentrations of rhFGF-2 outperformed the placebo group in terms of the percentage of bone fill at 36 weeks. 50 The peak percentage of bone fill was observed in the 0.3% rhFGF-2 group. Furthermore, 0.3% rhFGF-2 was superior to EMD and flap surgery in linear alveolar bone gain in infrabony defects. 24 The combination of rhFGF-2 and bone substitutes has also been investigated. A randomized controlled trial assessed the treatment of infrabony defects with at least 1-wall using β-TCP alone, β-TCP + 0.1% rhFGF-2, β-TCP + 0.3% rhFGF-2, and β-TCP + 0.4% rhFGF-2. 16 The two higher concentration groups were associated with greater clinical attachment gain and percentage of bone fill. Seshima et al. 51 investigated using 0.3% rhFGF-2 with deproteinized bovine bone material versus rhFGF-2 alone in infrabony defects over 4 years and found similar improvements in clinical and radiographic outcomes. These studies also demonstrated the safety and efficacy of rhFGF-2 for use in periodontal regeneration.
Clinical cases

Periodontal regeneration of an infrabony defect. (a) Preoperative intraoral view. (b) Preoperative periapical radiograph. (c) Exposure of defect following full-thickness flap elevation and scaling and root planing. (d) Application of autograft and rhFGF-2. (e and f) 3-Year clinical and radiographic follow-up.

Periodontal regeneration of an infrabony defect. (a) Preoperative intraoral view. (b) Preoperative periapical radiograph. (c) Exposure of defect following buccal full-thickness flap elevation and scaling and root planing. (d) Application of rhFGF-2. (e) Application of xenograft. (f and g) 2.5-Year clinical and radiographic follow-up.

Periodontal regeneration of an infrabony defect. (a) Preoperative intraoral view. (b) Preoperative periapical radiograph. (c) Exposure of defect following buccal full-thickness flap elevation and scaling and root planing. (d) Mix of xenograft and rhFGF-2 placed at the defect. (e and f) 2.5-Year clinical and radiographic follow-up.
Consensus reports
A systematic review and meta-analysis were conducted to evaluate the use of rhFGF-2 in periodontal regeneration. 52 The carrier for rhFGF-2 was used alone as the control group. It was found that the percentage of bone formation in human subjects treated with 0.3% rhFGF-2 was 22.37% higher than the control group, reaching statistical significance. The linear bone growth was also 1.13 mm higher, and the mean clinical attachment level gain was slightly better (0.27 mm). In another systematic review, Pouliou et al. 53 reported that the radiographic bone fill percentage using 0.3% rhFGF-2 and a bone substitute reached 74.6%. However, no additional benefit was seen regarding probing depth and attachment levels compared to rhFGF-2 alone or negative controls. rhFGF-2 may provide additional benefits in challenging periodontal regeneration cases when used alone or with a bone substitute. A couple of studies investigated factors that influence long-term outcomes of periodontal regeneration using rhFGF-2.54,55 It was found that the depth of the radiographic bone defect, radiographic bone defect angle, the involvement of most posterior teeth, and the presence of diabetes mellitus had the greatest impact on outcomes.
Discussion
In animal and human studies, rhPDGF-BB and rhFGF-2 have been demonstrated to increase the predictability of periodontal regeneration. An improvement in clinical attachment level, probing depth, and radiographic defect fill is evident. In addition, both growth factors show histologic evidence of new bone, cementum, and periodontal ligament. A summary of indications, contraindications, and other considerations are presented in Table 1. rhPDGF-BB and rhFGF-2 are safe and effective biologics that should be considered in appropriate clinical scenarios. An assessment of patient-, tooth-, and surgical-related factors must be completed to determine the prognosis of regenerative therapy. 7 A patient who demonstrates optimal oral hygiene and is a nonsmoker may have greater chances of success.56,57 The morphology and anatomy of the periodontal defect are also important.58,59 A deep and narrow defect with thick, soft tissue is most favorable for regeneration. 7 The evaluation of defect angle on periodontal regeneration therapy indicates that a radiographic angle of ⩽25 degrees leads to greater attachment than defects with angles of ⩾37 degrees. 60
Summary of clinical knowledge and considerations for rhPDGF-BB and rhFGF-2.

rhPDGF-BB, recombinant human platelet-derived growth factor-BB; rhFGF-2, recombinant human fibroblast growth factor-2.
Regarding surgical-related factors, primary wound closure, stability, minimally invasive, and passive flap tension are associated with improved outcomes. 7 Increasing evidence now supports the use of minimally invasive approaches. This approach is particularly effective when used with biologics, which are frequently combined with bone grafts. Cortellini and Tonetti 7 have conducted numerous clinical studies using a minimally invasive surgical approach, finding improvements in clinical attachment levels of ⩾4 mm, radiographic bone fill, reduced patient morbidity, and minimal gingival recession. A thorough understanding of these factors and the application of growth factors will help ensure the success of periodontal regeneration.
As demonstrated in the clinical cases, combining a biologic and bone graft yields successful treatment outcomes. An uncontained defect and/or extended flap design necessitates combination therapy. 7 Many studies and consensus reports have concluded that combination therapy produces superior clinical outcomes than monotherapy.35,53 The type of bone graft may differ in osteogenic, osteoconductive, and osteoinductive properties. When combined with a biologic, the bone graft also acts as a natural scaffold or matrix, supporting the biologic’s function in promoting new bone formation. 61 The European Federation of Periodontology S3 level clinical practice guidelines recommend that the choice of biomaterial or possible combinations should be based on defect configuration. 62
Limited evidence directly compares the clinical considerations and outcomes of rhPDGF-BB and rhFGF-2. A systematic review and meta-analysis by Khoshkam et al. 63 evaluated the effects of these biologics on intrabony defects compared to a carrier alone. The analysis found that rhPDGF-BB showed superior linear defect fill and clinical attachment level, while rhFGF-2 demonstrated greater linear defect fill. In this review article, the clinical knowledge of rhPDGF-BB and rhFGF-2 is presented side by side (Table 1). Future research should include preclinical studies directly comparing the outcomes of these biologics in periodontal regeneration.
Post-operative care and maintenance following periodontal regeneration are equally important. Patients should avoid chewing and practicing oral hygiene during the first few weeks in the surgical area. A systemic antibiotic and 0.12% chlorhexidine mouthwash may be prescribed to control bacterial contamination. Removal of sutures can occur as early as 10 days. 64 A 3-month recall system should be in place to allow for the supragingival prophylaxis. Subgingival instrumentation should be avoided for at least 6 months to maximize the opportunity for regeneration. Cortellini and Tonetti 7 reported that 96% of teeth with severe infrabony defects treated with regenerative therapy can be maintained for up to 15 years when a supportive periodontal-care program is in place. Therefore, patient concordance and oral hygiene become vital in the long-term success of periodontal regeneration.
The use of rhPDGF-BB and rhFGF-2 may also be applied in other periodontal procedures, such as sinus lifts and tooth replantation. Short-term studies are available, but long-term clinical data is lacking.65–67 Future research may include randomized controlled trials investigating the stability of outcomes with and without growth factors.
Conclusion
rhPDGF-BB and rhFGF-2 are safe and effective biologics that can be applied to improve radiographic and clinical outcomes of periodontal regeneration. The advantages of recombinant growth factors should be especially considered in the presence of challenging local and systemic factors. Future research may explore the long-term benefits of using rhPDGF-BB and rhFGF-2 in other periodontal procedures, including sinus lifts and tooth replantation.
