Abstract
Transjugular intrahepatic portosystemic shunt (TIPS) is a life-saving procedure for patients with severe portal hypertension and persistent variceal bleeding. Stent fracture is a rare and severe complication; however, its cause and mechanisms remain poorly defined. This case helps understand the factors contributing to its occurrence, complications, and subsequent poor outcomes. A 63-year-old male was presented with ruptured bare stent after a TIPS procedure. The upper edge of the bare stent was ruptured, and its fraction subsequently migrated to the entrance of the right atrium. Meanwhile, a mural thrombus was formed in the inferior vena cava. A surgery for the removal of free fracture was planned for preventing the form of pulmonary embolism. Before the surgery, the fracture was shifted to the right inferior pulmonary artery. Therefore, the surgery was canceled for further evaluation. Then, hematemesis suddenly occurred with a high possibility of variceal bleeding and/or gastric ulcer bleeding. Despite comprehensive treatments, the patient symptoms were still worsened with the development of chest tightness, shortness of breath, severe hypoxia, and heart failure. Finally, the patient succumbed to systemic multiorgan failure and death. Taken together, a ruptured unstable stent should be removed as early as the patient is hemodynamically stable, as it is difficult to balance between hemostasis therapy and anticoagulation treatment in patients with liver-cirrhosis-related severe portal hypertension. Physicians should be on high alert of the potential complications of bare stent rapture after TIPS.
Plain language summary
Unstable stent rupture is a life-threatening complication of TIPS and severely complicates the treatment of gastric ulcer bleeding. Early removal of the ruptured stent is necessary to prevent further complications.
Keywords
Core tips
Unstable stent rupture is a life-threatening complication of TIPS and severely complicates the treatment of gastric ulcer bleeding. Early removal of the ruptured stent is necessary to prevent further complications.
Introduction
Transjugular intrahepatic portosystemic shunt (TIPS) is a well-established procedure in the treatment of patients with decompensated portal hypertension as it establishes a blood flow channel from the portal vein to systemic circulation. TIPS can effectively improve complications of portal hypertension, including variceal hemorrhage and refractory ascites. 1 Although TIPS implantation is widely accepted in clinical practice, severe complications like stent fracture may occur. Previous studies have indicated that stent fracture can migrate to various locations, such as chordae tendineae of the tricuspid valve, the inferior vena cava, the pulmonary arterial system, the right ventricle, and even multiplaces (inferior vena cava, the right atrium, and the right ventricle).2–6 Overall, these cases indicated that stent ruptures generally have a good prognosis after removal.2–6
Here, we present a rare case of a bare stent fracture migrated to the pulmonary artery after TIPS, which increased the risk of death due to the difficult balance between anticoagulation and hemostasis when gastric ulcer bleeding occurred. Despite active medical interventions, the patient’s condition deteriorated rapidly and ultimately succumbed to respiratory and circulatory failures.
Case description
A 63-year-old man was admitted to our hospital, complaining of chronic fatigue (>2 years) and melena for 6 h.
Upon physical examination at admission, his body temperature was 37.1°C, heart rate 75 beats/min, respiratory rate 17 breaths/min, and blood pressure 105/55 mmHg. An abdominal examination indicated an active bowel sound during auscultation and a soft sensation while performing palpation without any muscle guarding or rebounding pain. The remainder of the physical examination was unremarkable, excepting for abdominal four surgical scars.
Past medical history
Three gastroscopic variceal vein treatments with endoscopic injection of gastric varices tissue glue and endoscopic variceal sclerosing agent; radiofrequency ablation for hepatocellular carcinoma, in 2 years.
The patient was diagnosed as acute upper gastrointestinal bleeding, gastric ulcer, hepatitis B cirrhosis, liver schistosomiasis, severe esophageal gastric varices with red signs (gastroesophageal varices 3 type), portal vein thrombosis, superior mesenteric vein thrombosis, and portal hypertensive gastropathy were identified.
Upon admission, the patient was given intravenous somatostatin to reduce splanchnic and portal vein flow, inhibit the secretion of gastric acid and pepsin, and help to stop bleeding at mucosal lesions. However, portal vein pressure was measured at the high level of 28 mmHg. Therefore, angiography showed portal vein widening and gastric coronary vein tortuosity. Considering the high risk of recurrent bleeding, TIPS was performed, and its date was defined as day 1 of postprocedure. A self-expanding metallic bare stent (Boston Scientific 8 mm × 10 cm, USA) and a stent graft (Gore 8 mm × 5 cm, USA) were implanted for the intrahepatic shunting. The proximal end of the stents was located on the hepatic vein branch, and the distal end was located on the portal vein main stem. The portal vein pressure was reduced to 18 mmHg after TIPS.
Due to portal vein thrombosis, the patient was given rivaroxaban tablets after TIPS. On day 28 of postprocedure date, a postoperative computed tomography (CT) scan suggested that the bare stent was intact with a slight angle at the place where it entered the inferior vena cava [Figure 1(a)].
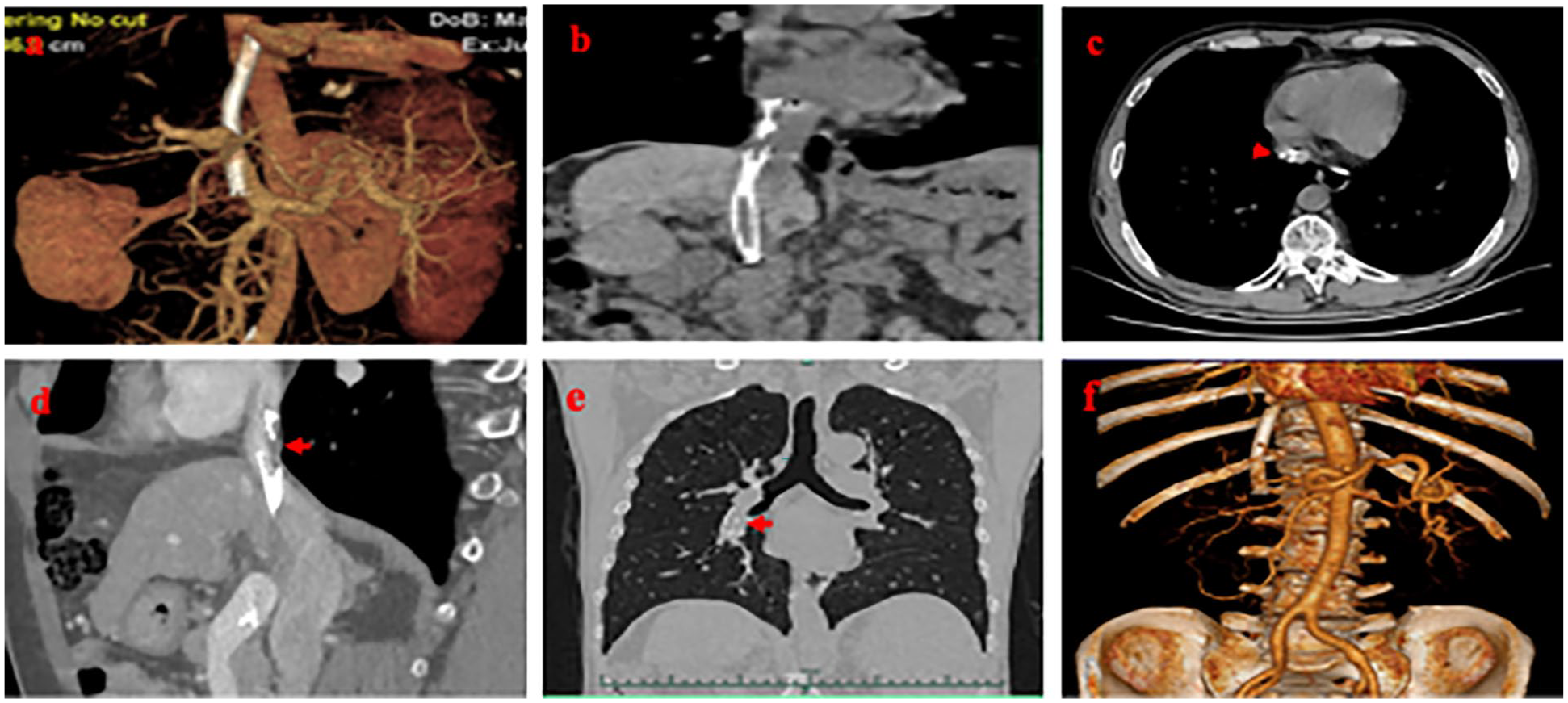
(a) The reconstructed CT image showed that the stent was continuous, and a slight included angle was visible at the place where the stent entered the inferior vena cava on day 28 of postprocedure date. (b) The upper end of the bare stent was broken and free, which was located at the entrance of right atrium on day 236 of postprocedure date. (c) Inferior vena cava mural thrombus could be seen between intrahepatic part and free part of rapture stent as shown by red arrow on day 237 of postprocedure date. (d) No obvious progress of mural thrombus was indicated as shown by red arrow on day 250 of postprocedure date. (e) Chest CT imaging showed that the free part of raptured stent was moved into the right lower pulmonary artery as shown by red arrow on day 265 of postprocedure date. (f) CT enhancement imaging of Affiliated First Hospital of Medical School of Zhejiang University showed no change in the position of intrahepatic stent, smooth blood circulation, and no residual free part in the right atrium on day 265 of postprocedure date.
The patient experienced recurrent gastrointestinal bleeding on 7th–8th month after postprocedure date. Gastric ulcer bleeding after anticoagulation therapy was considered. Internal hemostasis therapies were given as the bleeding volume is not significant. On day 236 of postprocedure date, portal vein CT venography (CTV) revealed that the upper part of the bare stent had ruptured, and its free end located at the entrance of the right atrium [Figure 1(b)]. In addition, an inferior vena cava mural thrombus was observed between the intrahepatic part and the free part of the ruptured stent [Figure 1(c), red arrow]. Cardiac ultrasound suggested a strong stent echo in the right atrium, indicating that the broken stent measuring 2.8 cm in size had caused mild-to-moderate tricuspid valve regurgitation. Surgical treatment was recommended, but due to COVID-19-related restrictions, the surgery could not be arranged immediately.
On 8th–9th month after the postprocedure date, the free part of the ruptured stent migrated further to the entrance of right atrium with no significant changes in mural thrombus [Figure 1(d)]. After the mandatory COVID-19 restrictions were lifted, the patient planned for a surgery again to remove the free part of the broken stent from the right atrium. However, 1 day before the planned surgery, the fractured stent moved to the right lower pulmonary artery without thrombus or dyspnea [Figure 1(e)]; enhanced CT scan showed no changes in the position of the intrahepatic stent and no residual stent found in the right atrium [Figure 1(f)]. The surgery was thus canceled for further evaluation. Anticoagulation therapy was given to avoid pulmonary artery thrombosis.
On 9th month after the postprocedure date, the patient experienced a recurrent gastrointestinal bleeding and was treated with fasting, rehydration, acid suppression, and somatostatin. Blood coagulation function tests showed international normalized ratio (INR) of 1.7, fibrinogen of 1.08 g/L, activated partial prothrombin time of 45.2 s, and D-dimer of 269 mg/L. Blood test results showed hemoglobin of 73 g/L and platelets count of 49 × 109/L. On day 271 of postprocedure date, coagulation function tests showed INR of 1.6, fibrinogen of 1.08 g/L, activated partial prothrombin time of 39.5 s, and D-dimer of 372 mg/L. The liver function test showed albumin of 35.5 g/L, alanine aminotransferase of 17 U/L, aspartate aminotransferase of 37 U/L, and total bilirubin of 52.4 mmol/L.
Few days later, the patient vomited massive blood. Gastroscopic tissue glue and Sengstaken–Blakemore tube compression was applied for bleeding control. The patient suddenly experienced chest tightness, oxygen saturation dropping to below 80%, pink foam sputum, and wet rales in both lower lungs. The patient eventually developed respiratory and circulatory failure, as well as finally succumbed.
Discussion
TIPS can create an effective shunt between the portal vein and hepatic vein via the stent to relieve portal hypertension. Stent rupture is a rare but a serious complication that can cause bleeding, re-occlusion, or even death. In 2017, Chinese specialists recommended the use of GORE® VIATORR® stents (Viatorr stents) to minimize postoperative complications and improve patient quality of life as it is with diameter control to reach a targeted portal pressure gradient. 7 The characteristics of Viatorr stent are on the following: (a) Controlling the diameter; (b) lasting diameter control; (c) engineered for flexibility; and (d) single device with different sizes. 7 Viatorr stents as the standard stent for TIPS have been widely used in Europe and the USA. 8 Due to the high cost of Viatorr stents, many hospitals in China and western countries still use traditional bare stents combined with stent grafts in TIPS for limited expenses.
Stent fracture after TIPS is not uncommon (Table 1). In a case reported by Ding et al., 2 the ruptured stent did not form a specific angle. It is believed that the proximal free part of the bare stent is affected by factors such as respiratory motion and cardiac heartbeat. In the overlapping part of the stent, sustained force is generated at its proximal end, which can directly cause the stent to fracture. Stent fracture occurs for multiple reasons, including the design and materials of the stents (stents made of nickel titanium alloy with a self-expanding feature and a bare stent with an open hole design have higher risk to fracture). 3 High-strength bending is a potential risk factor for stent fracture.2,3 Using double stents to replace Viatorr stents may increase the risk of fracture. As of now, Viatorr stents have not been reported to fracture.
Previous reports on stent fracture after TIPS procedure.

BMS, bare metal stent; TIPS, transjugular intrahepatic portosystemic shunt.
Post-TIPS abnormalities may result from the many causes, including (a) iatrogenic factors, such as damage caused by guidewires or balloon catheters during surgery or improper operation during release, and improper selection of the hepatic vein or portal vein puncture sites; (b) factors related to underlying diseases, such as twisting of the hepatic vessels, increased blood flow in the shunt, and reginal narrowing of the blood vessels leading to high pressure or high velocity; (c) behavioral factors, such as the impact of respiratory movements of the diaphragm or the heart beating causing fatigue damage to the stent; and (d) biological factors, such as inflammatory reactions or thrombus formation in adjacent tissues affecting the integrity and stability of the stent.
Changes in the shape of the stent may lead to the multiple consequences: (a) Stent fracture and related consequences: This may be caused by iatrogenic damage, metal fatigue, corrosion from inflammatory reactions, or stress concentration at the bending point caused by shape distortion. The broken end of the stent may detach, migrate, and cause embolism, leading to organ infarct or even death. (b) Stent displacement: The stent may become twisted and unstable due to positional deviation during release or improper selection of the hepatic vein puncture site, causing its displacement with external traction or compression and occlusion. (c) Stent compression: The stent may become deformed due to compression from adjacent organs or tissues (such as the liver, diaphragm, stomach, or heart), causing shunt stenosis or occlusion.
In this case, the patient had no obvious vascular distortion during TIPS preoperative evaluation. The operation was performed with standard procedures, and no obvious iatrogenic damage was occurred. The stent was smoothly deployed to the desired position. No obvious shunt distortion was observed after the surgery, and the shunt remained unobstructed during follow-up for nearly 6 months. Therefore, it is possible that the stent fracture was caused by fatigue damage resulting from the movement of the diaphragm or the beating of the heart.
Although gastrointestinal bleeding may have been the direct cause of death, stent dysfunction may have triggered the recurrence of gastrointestinal bleeding. First, anticoagulant therapy was recommended to avoid thrombus for the broken end of the stent entered the pulmonary artery, which can trigger gastric ulcer bleeding. Second, the irregular fracture of the stent may make the wall of the inferior vena cava more vulnerable to forming a thrombus at the broken end of the stent. The thrombus may reduce the shunting effect of the stent and increase the portal vein pressure, thus contributing to the occurrence of variceal rupture and bleeding. In addition, the instability of the fractured stent makes endoscopic hemostasis more difficult under general anesthesia. Finally, the death of this patient may have also been contributed by the alternating use of anticoagulant and hemostatic treatments, massive blood loss, coagulation dysfunction, and multiple organ dysfunction. Therefore, in case of stent fracture with potential bleeding risks, it is important to balance the timing of hemostatic and anticoagulant therapies and promptly remove the broken end of the stent through surgery.
Based on our experience, we think the following are important measures to prevent and treat possible complications of stent abnormalities: (a) using the stents from qualified suppliers; (b) conducting a comprehensive vascular CTV examination before surgery to fully evaluate the hepatic vascular deformity and identify an appropriate puncture point in the liver; (c) following well-recognized techniques and standardized procedures to avoid or minimize iatrogenic stent damage; (d) monitoring hemodynamic parameters, such as portal vein pressure, flow, and resistance, to promptly identify and correct any stent abnormalities; (e) preventing stent thrombosis by anticoagulants and anti-inflammatory agents after the surgery. In case of inflammation, short-term anti-inflammatory agents may be given to reduce inflammatory reactions around the stent; (f) doing regular follow-up (once every 3–6 months) for imaging to promptly detect any stent-related complications or abnormalities. A management flowchart for stent fracture is presented in Figure 2.

Flowchart for TIPS stent fracture management.
Conclusion
In summary, classical double stents for TIPS have potential risks for the incidence of stent fracture. Regular imaging studies are necessary after TIPS to monitor the stent changes, and this can help clinicians take prompt actions when stent fracture occurs. Early removal of the free raptured stent is the appropriate approach to prevent severe complications.
