Abstract
Background:
Erythrodermic psoriasis (EP) remains challenging to manage because it is rare and has complex complications. Although acitretin is recommended as an appropriate choice for EP, there is a lack of large-scale evidence.
Objectives:
This study aims to assess the efficacy and safety of acitretin as systemic monotherapy in EP patients.
Design:
We retrospectively analyzed data from patients with EP who received at least 3 months of acitretin as systemic monotherapy during hospitalization and out-patient follow-up from January 2005 to May 2021 at the Peking Union Medical College Hospital, China.
Methods:
The efficacy was clinically evaluated after 1, 2, 4, and 12 weeks of treatment, which was classified as a good response (>75% of lesions cleared), partial response (50%–75% cleared), moderate response (25–50% cleared), or no response (<25% cleared). Safety was assessed on the basis of physical examination results and significant changes in laboratory examination results after 12 weeks of treatment.
Results:
Overall, 81 patients (79.0% men; mean age, 47.9 years) were included. The acitretin dose ranged from 20 to 60 mg/day (0.3 to 0.8 mg/kg/day). The rates of good, partial, and moderate responses were 0.0%, 2.5%, and 42.0% at 1 week; 3.7%, 34.6%, and 61.7% at 2 weeks; 29.6%, 58.0%, and 12.4% at 4 weeks; and 85.2%, 13.6%, and 1.2% at 12 weeks after treatment initiation, respectively. EP patients transformed from psoriasis vulgaris showed a higher good/partial response rate compared with that of EP patients that developed from pustular or articular psoriasis (44.6% vs. 14.3%, p = 0.035). Patients with concurrent infection showed a lower rate of good/partial response compared with that of those without concurrent infection (16.7% vs. 44.4%, p = 0.049). Adverse effects were seen in 45 (55.6%) patients in 12 weeks, and dyslipidemia (n = 31; 38.3%), xerosis (n = 24; 29.6%), and elevated liver enzymes (n = 6; 7.4%) were most commonly reported. Twenty-three patients were followed up for over 3 years, and six (26.1%) patients had EP recurrence.
Conclusions:
Acitretin as a systemic monotherapy showed satisfactory effectiveness for EP, especially in patients developed from psoriasis vulgaris and without infection.
Introduction
Erythrodermic psoriasis (EP) is a rare but severe subtype of psoriasis, with an estimated prevalence of 1% to 2.25% of psoriasis patients.1,2 This form of psoriasis is characterized by lesions over 90% of the body surface area, causing significant morbidity and an increased risk of mortality. 1 There are several treatment options, but EP remains a therapeutic challenge due to a lack of evidence-based data to reach a consensus on a standard therapeutic schedule. Acitretin, due to its comparatively late onset of effect, is considered a first-line treatment by the National Psoriasis Foundation medical board for stable patients of EP. 2
The function of retinoids is to govern keratinocyte proliferation and differentiation, affect sebaceous gland activity, and control local inflammation. 3 These biological effects are achieved through binding to retinoic acid receptors (RARs) and/or retinoic X receptors (RXRs), which regulate gene expression. 4 Skin desquamation, xerosis, pruritus, hair loss, and cheilitis are commonly reported side effects. 5
The evidence on the effectiveness of retinoids in EP is conflicting. In 12 EP patients treated with 25–35 mg/day acitretin, clinical remission or substantial improvement of EP was seen in 10 patients. 6 Polat and Sereflican noted 50 mg/day of acitretin to be beneficial in treating a patient with concomitant EP and elephantiasis nostras verrucosa. 7 However, another EP patient who received acitretin with the same dose did not reach significant clinical remission, which might be attributed to the overweight of this patient. 8
Current evidence suggests that acitretin may be effective for EP patients, but data are still limited. In this study, we retrospectively analyze present the efficacy and safety using acitretin as systemic monotherapy in EP patients from Peking Union Medical College Hospital in China.
Materials and methods
Patients and setting
This was a retrospective study from Peking Union Medical College Hospital in China. We conduct a retrospective analysis of retrospectively analyzed data on adult inpatients with EP from January 2005 to May 2021. EP patients who initiated and received at least 3 months of acitretin as systemic monotherapy during hospitalization and out-patient follow-up were included. Inclusion criteria were age ⩾18 years and generalized erythema with bran-like scaling involving over 90% of body surface area. The diagnosis was confirmed by a definite history of psoriasis or typical psoriatic lesion on examination. In addition, skin histopathological examinations were performed to exclude other causes of erythroderma if necessary. Patients were excluded if there was a concurrent diagnosis or history of another erythroderma cause (e.g. drug-induced dermatosis, eczema, atopic dermatosis, and eosinophilic dermatosis). Patients with incomplete medical data due to department transfer or sudden discharge requested by the patient were also excluded.
Data collection
Data were derived from medical records and anonymized, including demographic features (age, sex, and weight), clinical characteristics (history of psoriasis, disease duration, and family history of psoriasis), systematic symptoms (fever, lower limb edema, and superficial lymphadenopathy), laboratory results (routine blood tests, biochemical tests [liver function, renal function, and serum lipids], erythrocyte sedimentation rate [ESR], high-sensitivity C-reactive protein [hsCRP]) of venous blood samples after an overnight fast for at least 8 h. Disease severity of EP was evaluated according to the method proposed by our team. 9 A moderate-to-severe EP patient exhibits at least two of the three following characteristics while a mild EP patient exhibits less than two characteristics: (1) body temperature higher than 37.3°C upon admission; (2) swelling and exudation of more than half of the skin lesion or lower extremity edema; and (3) superficial lymphadenopathy. Dose, therapeutic response, therapy duration, and adverse events (AEs) related to acitretin treatment were assessed.
Study assessments
The assessment of efficacy was conducted 1, 2, 4, and 12 weeks following the initiation of therapy. If the patient was discharged at a certain time cutoff, the efficacy was evaluated using the data from out-patient follow-up. Efficacy was retrospectively classified as a good response (>75% of lesion clearance), partial response (50–75% clearance), moderate response (25–50% clearance), or no response (<25% clearance) according to the written and/or photographic records. The time interval from the first to the last medication was regarded as treatment duration. Safety was assessed according to laboratory examination and patients’ subjective feelings at 3 months after treatment initiation.
Statistical analysis
The mean ± standard deviation (SD) for continuous variables and the frequency (%) for categorical variables are presented as summary descriptive statistics. The t-test or the Mann–Whitney U test was used to compare continuous variables between groups. For categorical variables, the Chi-square or Fisher’s exact test was used. SPSS v.25 (IBM Corp., Armonk, NY, USA) was used for all statistical analyses with a two-sided 5% level of significance.
Results
Baseline cohort characteristics
Overall, 81 patients (64 men; mean age, 47.9 years) were included. Thirty-four (42.0%) patients were classified as moderate-to-severe EP according to the evaluation criterion proposed by our team. 9 All patients had a positive history of other psoriasis types. Sixty-seven (82.7%) patients had a history of psoriasis vulgaris and 14 (17.3%) had a history of pustular psoriasis and/or articular psoriasis. Eleven (13.6%) patients had a recurrence. Sixty-three (77.8%) had an identifiable trigger factor. Rapid withdrawal or a sudden change of medication was the most common factor, followed by the use of traditional Chinese prescription and infection. Eighteen (22.2%) presented with concurrent infection during their hospital stay, and four patients had a dual infection. Respiratory infection (10 patients, 12.3%) was the most common, followed by skin infection (eight patients, 9.9%), bloodstream infection (three patients, 3.7%), and urinary infection (one patient, 1.2%). Baseline cohort characteristics including demographic, clinical, and laboratory features are demonstrated in Table 1.
Baseline cohort characteristics.

Alb, albumin; ESR, erythrocyte sedimentation rate; Hgb, hemoglobin; hsCRP, high-sensitive C-reactive protein; SD, standard deviation; WBC, white blood cells.
The hospitalization duration of 81 patients ranged from 5 to 48 days, with an average of (22.4 ± 10.1) days. One patient was hospitalized for only 5 days, and was collected efficacy data at 1, 2, 4, and 12 weeks in the out-patient department. Eighteen patients collected 1-week efficacy data during hospitalization and efficacy data at 2 weeks, 4 and 12 weeks during out-patient follow-up. Thirty-eight patients were collected efficacy data at 1 and 2 weeks during hospitalization, and efficacy data at 4 and 12 weeks during out-patient follow-up. Twenty-four patients collected efficacy data at 1, 2, and 4 weeks during hospitalization, and efficacy data at 12 weeks during out-patient follow-up.
Treatment-related outcomes
Maximal acitretin dosing ranged from 20 to 60 mg/day and from 0.3 to 0.8 mg/kg/day. Thirty-five patients (43.2%) received 40 mg daily. The treatment duration was 152.3 days (SD 138.3; range 92–916 days).
Effectiveness
One week after initiating acitretin therapy, no (0.0%) patients showed a good response, 2 (2.5%) patients showed a partial response, 34 (42.0%) patients showed a moderate response, and 45 (55.5%) patients showed no response. At 2 weeks, 3 (3.7%) patients showed a rapid good response, 28 (34.6%) patients showed a partial response, and 50 (61.7%) patients showed a moderate response. At 4 weeks, 24 (29.6%) patients reached a good response, 47 (58.0%) patients reached a partial response, and 10 (12.4%) patients showed a moderate response. At 12 weeks, 69 (85.2%) patients reached a good response, 11 (13.6%) patients reached a partial response, and one (1.2%) patient showed a moderate response. The details are shown in Figure 1.
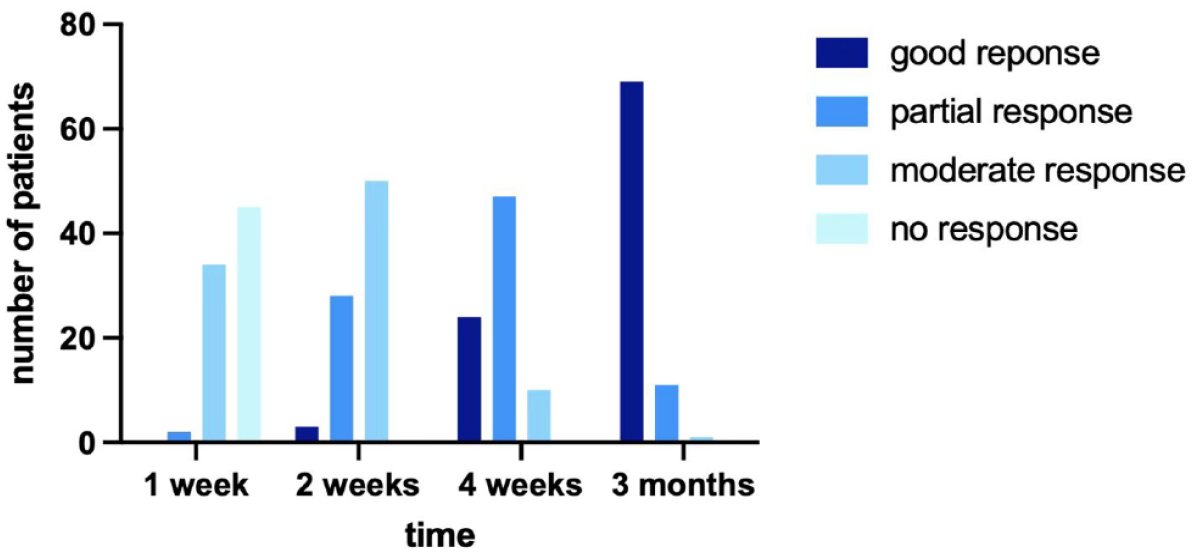
Improvement of skin lesions of the 81 studied patients at different timelines.
Safety profile
Forty-five (55.6%) patients experienced AEs, and the most commonly reported AEs were dyslipidemia (n = 31; 38.3%), xerosis (n = 24; 29.6%), and liver enzyme-level elevation (n = 6; 7.4%). The majority of AEs were mild to moderate and could be controlled by additional medications. No treatment was withdrawn because of intolerable AEs. Among the 31 patients who presented with dyslipidemia, five patients had a history of hyperlipidemia before admission to the hospital and showed an increase in lipid levels after acitretin therapy. All patients were given diet and exercise advice, and ten patients received lipid-lowing agents (LLAs). Five patients showed normal blood lipid levels when discharged. The details are shown in Table 2.
Treatment dosage, AEs, and follow-up.
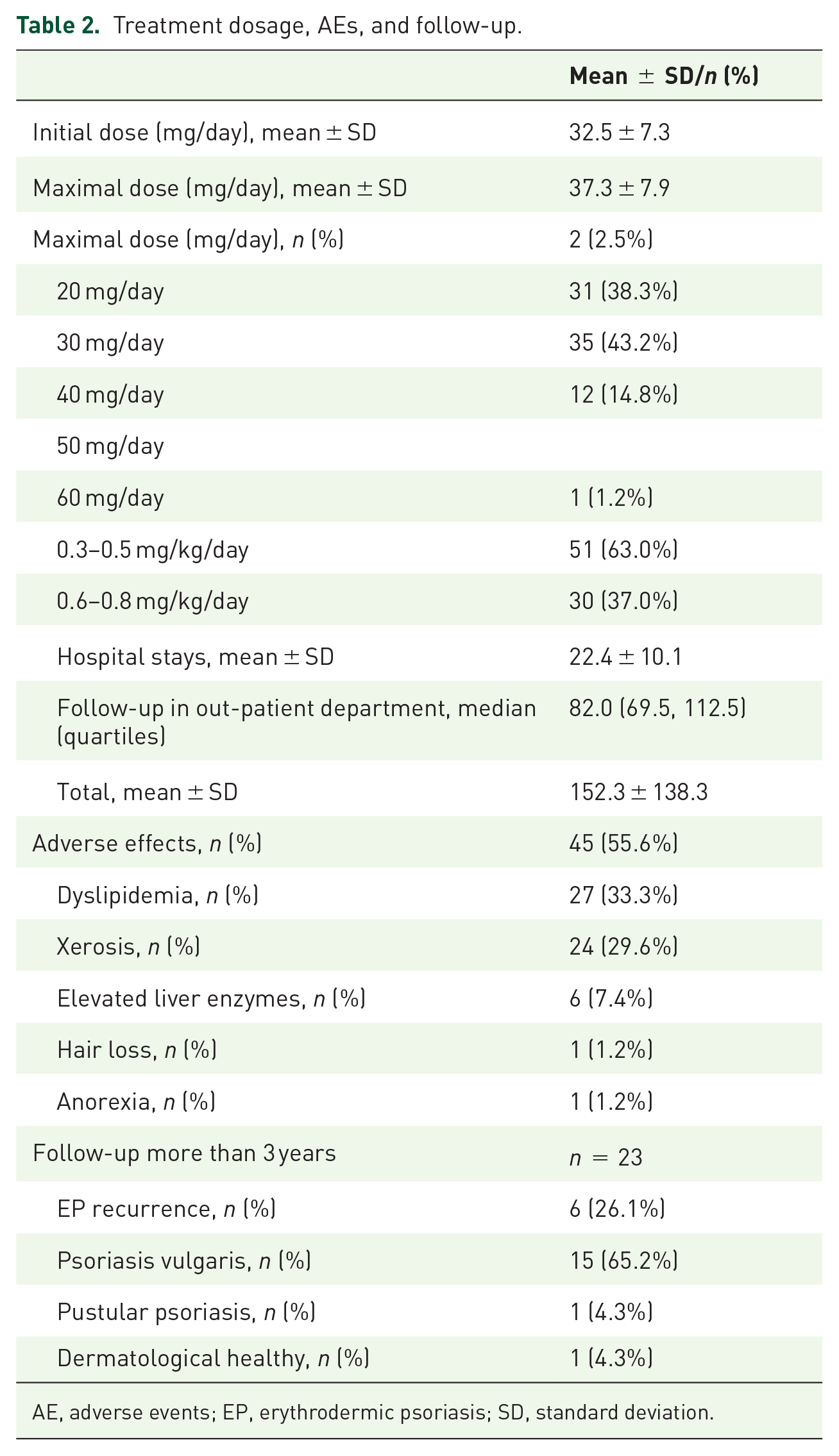
AE, adverse events; EP, erythrodermic psoriasis; SD, standard deviation.
Follow-up
Twenty-three patients were followed-up for more than 3 years. Psoriasis was present continuously in 22 (95.7%) patients, through either recurrence of EP (n = 6; 26.1%) or other psoriasis types (n = 16; 69.6%). Identifiable trigger factors included pneumonia, sudden withdrawal of acitretin, pulmonary embolism, and overwork. The details are shown in Table 2.
Factors related to efficacy
Patients who had a moderate response at 2 weeks had a significantly longer hospital stay (26.2 vs. 16.4 days, p < 0.001) compared with those who had already achieved good/partial response at 2 weeks. The type of psoriasis history was also related to a rapid response at 2 weeks. Patients transformed from pustular psoriasis or articular psoriasis had a lower good/partial response rate compared with those transformed from psoriasis vulgaris (14.3% vs. 44.6%, p = 0.035). Moreover, a comorbidity of infection seemed to be associated with a slow response. Patients with concurrent infection presented with a significantly lower rate of good/partial response at 2 weeks compared with that of patients without infection (16.7% vs. 44.4%, p = 0.049). The particulars are shown in Table 3.
Comparison between good/partial response and moderate response groups.

Alb, albumin; ESR, erythrocyte sedimentation rate; HGB, hemoglobin; hsCRP, high-sensitive C-reactive protein; WBC, white blood cells.
Good/partial response defined as ⩾ 50% clearance.
Moderate response defined as 25–50% clearance.
Statistical significance defined as p < 0.05 (χ2 test).
Factors related to the dose
Analysis of the effective acitretin dosage was conducted in 80 patients who had good or partial response at 3 months after treatment initiation. Patients with systematic symptoms were more likely to have received large acitretin doses (⩾0.6 mg/kg/day) to achieve a satisfactory response compared with those without systematic symptoms (41.3% vs. 11.8%, p = 0.024). We found that when treating EP patients transformed from pustular psoriasis, the initial dose (35.0 ± 9.3 vs. 31.9 ± 7.2 mg/day, p = 0.276) and maximum dose (38.8 ± 8.3 vs. 37.2 ± 8.0 mg/day, p = 0.276) of acitretin were tend to be higher than those of EP patients transformed from psoriasis vulgaris, but there was no statistical significance. The particulars are demonstrated in Table 4.
Comparison between regular doses and large doses group.

Alb, albumin; ESR, erythrocyte sedimentation rate; HGB, hemoglobin; hsCRP, high-sensitive C-reactive protein; SD, standard deviation; WBC, white blood cells.
Regular doses are defined as 0.3–0.5 mg/kg/day.
Large doses defined as ⩾ 0.6 mg/kg/day.
Statistical significance defined as p < 0.05 (χ2 test).
Discussion
This study assessed the efficacy and safety of the systemic monotherapy with the RAR-agonist acitretin in 81 EP patients. EP is an uncommon and severe variant of psoriasis characterized by unique histopathologic and clinical features. At present, EP is widely considered a unique subtype of psoriasis, accounting for 1–2% of all psoriasis patients,1,2 with a higher incidence rate among Asian people. 10 There are two clinical subtypes of EP.11,12 One is that it gradually develops into systemic erythroderma on the basis of previous plaque lesions of psoriasis vulgaris. Typical psoriatic plaques still remain different from the erythroderma. This EP subtype has a longer course of the disease, relatively stable condition, less systemic symptoms and a better prognosis. This subtype of EP is more likely to be psoriasis with increased severity both generically or abnormally. The other subtype is characterized by a rapid outbreak of erythema in the whole body without definite psoriasis plaque. This type of EP usually has an acute disease course and unstable condition. It is also more likely to be accompanied by systemic symptoms and abnormalities in laboratory tests. The prognosis is relatively poor compared with the first type, and it is more likely to relapse. Serious cases can endanger life.11,12 This subtype of EP tends to be a unique psoriasis subtype.
Research on the pathological mechanism of EP found that: (1) EP patients often present with elevated levels of interleukin 4 (IL-4) and interleukin 10 (IL-10), while psoriasis vulgaris patients mostly have increased levels of tumor necrosis factor α(TNF-α), interleukin 17 (IL-17), and interleukin 23 (IL-23). 13 (2) The level of immunoglobulin E (IgE) may increase in EP patients, while the levels of IgE in patients with psoriasis vulgaris are usually normal. 14 (3) The overall immune response of EP patients is more inclined to activate Th2 inflammatory response, while patients with psoriasis vulgaris are more inclined to activate Th1 inflammatory response.13,15 (4) Immunohistochemical staining analysis found the percentages of Th17 cells (CD4+ /STAT3+ cells) in CD3+ T cells were 27.1% and 14.7% in chronic psoriasis and EP, respectively. 16 In addition, IL-17 responses were demonstrated to be the main inflammatory signal shared by chronic plaque psoriasis and EP. 17 (5) EP patients have increased levels of peripheral blood adhesion factor. 18 While no similar observation is seen in patients with psoriasis vulgaris. (6) Immunohistochemical characteristics of histopathology in EP patients are closer to atopic dermatitis (AD) than psoriasis vulgaris. 16 These findings suggest that EP may have different pathological mechanisms compared to psoriasis vulgaris.
The potential risk of morbidity and mortality associated with the condition requires better management for these patients. Due to its rarity and complexity, EP is often difficult to manage, and there is a dearth of high-quality studies assessing treatment alternatives. EP patients are treated on the basis of disease severity and the patient’s comorbidities. Expert consensus is that conventional systemic treatments should include cyclosporin A (CsA), methotrexate (MTX), and acitretin. 19 Studies have demonstrated that CsA monotherapy shows effects more quickly than the other two conservative drugs, which could help 27–50% of patients to achieve 70% lesion clearance between baseline and after 3 weeks to 6 months.20 –22 The CsA dose ranged from 3.5 to 5 mg/kg/day.20,21 Reported adverse events related to CsA administration included hypertension, cerebrovascular disorder, gastrointestinal upset, headache, and fatigue.20 –22 For MTX, two retrospective studies found that 88.2% of patients achieved 75% clearance among 17 EP patients.23,24 The dosage of MTX varied from 7.5 to 15 mg/week, with an average of 10 mg/week.23,24 However, time to response or relapse was not recorded.
In the last several years, there have been more alternatives for treating EP, including tumor necrosis factor (TNF)-α inhibitors (etanercept, infliximab, and adalimumab), interleukin (IL)-17/IL-17 receptor inhibitors (secukinumab, ixekizumab, and brodalumab), IL-12/23 inhibitors (ustekinumab), IL-23 inhibitors (guselkumab), and other biological treatments (alefacept and efalizumab).19,22,25,26 These agents have shown potential in managing erythrodermic psoriasis. TNF was the first cytokine targeted for EP therapy. 25 A clinical trial conducted in 2006 found that 60% (6/10) of EP patients treated with etanercept 25 mg twice a week achieved a PASI75 (PASI score has decreased 75% from baseline) within 24 weeks. 27 Poulalhon et al. 28 demonstrated that 60% of patients with EP (3/5) receiving infliximab (5 mg/kg at weeks 0, 2, 6, and every 8 weeks) achieved PASI75 by 14 weeks, and 40% (2/5) achieved PASI90. IL-17 was implicated as a central cytokine in EP pathogenesis. A retrospective study indicated that 76.9% (10/13) EP patients achieved PASI90 in 4 weeks without relapse (52 weeks follow-up) using secukinumab (300 mg subcutaneously at weeks 0, 1, 2, and 3 and every 4 weeks). 29 An open-label study that enrolled eight EP patients reported that all patients receiving ixekizumab reached PASI 75 after 12 weeks. 30 Drugs targeting IL-23 were shown to be applicable in EP patients. Over 60% of the 22 EP patients reached PASI75 after ustekinumab administration (45 or 90 mg subcutaneously for patients weighing <100 or >100 kg, respectively, at weeks 0 and 4 and every 12 weeks) for 16 weeks. 31 A phase 3 open-label study demonstrated robust efficacy in 90.9% (10/11) of EP patients within 16 weeks. 32 Common adverse events of biologics agents include pruritus, infusion reaction, arthralgias, pulmonary infection, urinary tract infection, cutaneous infections, lymphoma, and immunoallergic shock. 22 The details are summarized in Table 5.
Summary of clinical studies on treatment of EP.

EP, erythrodermic psoriasis; TNF-α, tumor necrosis factor α; IL, interleukin.
Compared with biological agents, acitretin is inexpensive and convenient to administer orally. In addition, patients taking acitretin are not at increased risk of infection or tumor development. Koo reported a sequential therapy for EP in which patients begin treatment with 25 mg/day of acitretin, which is subsequently raised by 10 to 25 mg every 2 to 4 weeks until reaching the highest tolerated dosage. 40 So far, reliable data on the use of acitretin for EP are sparse and inconsistent. Ten of 12 patients treated with etretinate 20 to 60 mg/day by 2 to 11 months in a case series study reported favorable outcomes. 33 A patient with EP achieved almost complete clearance after acitretin treatment of 50 mg/day for 1 month. 7 Conversely, Rosinska et al. 41 observed that only 40% of patients treated with etretinate presented with satisfactory outcomes by 1 to 4 months. A retrospective study of two individuals with EP found that neither had a favorable response after three months of therapy with acitretin. 20 A 4-month trial of acitretin at a dose of 50 mg/day in one EP patient did not show any clinical improvement. 8 Our study showed that 29.6% (24 of 81) of patients achieved a good response, and 58.0% (47 of 81) of patients had a partial response by 4 weeks. The best efficacy was observed at 12 weeks after treatment, with 85.2% (69 of 81) of patients showing a good response.
The relationship between clinical factors (source of EP, concurrent infection, and hsCRP level increase) and quality of the response (good/partial and moderate response) to acitretin treatment at 2 weeks was observed. A respiratory infection is considered to be a triggering factor of EP, 11 and thus, it may exacerbate the disease. A skin infection is considered to be a consequence of the normal skin barrier breaking down, and its occurrence indicates the disease severity. In addition, the high inflammation status indicated by of the increase in hsCRP levels can account for the weaker response to treatment.
Our study showed that patients with EP that developed from psoriasis vulgaris seemed to have a better response compared with that those with EP that transformed from pustular psoriasis or articular psoriasis. This may be caused by the difference in intensity and the extent of immune activation in different psoriasis types. Psoriasis vulgaris accounts for approximately 90% of all psoriasis cases with a typical chronic disease course, and the inflammation status of this type seems to be mild, lasting, and limited to the skin.42 –44 Pustular psoriasis, on the other hand, is characterized by acute and progressing disease course and unique manifestation of widespread redness and subcorneal pustules. 45 Thus, the inflammation status of pustular psoriasis seems to be more acute and intense compared with that of psoriasis vulgaris. In addition, patients with articular psoriasis present with a wide range of inflammation involving cartilage and bone.46 –48 Our results indicate that the response to acitretin may be associated with the intensity and range of inflammation in patients.
No statistically significant difference was seen in the quality of the response or the dose between primary and relapsed EP patients, indicating that relapsed patients could undergo the same therapy schedule as primary patients. However, relapsed patients accounted for half of the patients (four of eight) who had an EP recurrence during the 3-year follow-up. The underlying reason could be the low compliance with standard maintenance treatment, preference for folk prescriptions, and other unidentified causes.
The maximal dosage was correlated with the presentation of systematic manifestations, including fever, lower extremity edema, and superficial lymphadenopathy. Patients with at least one of the three above-mentioned symptoms were more likely to have a large dosage (⩾0.6 mg/kg/day). Systematic manifestation synchronizes with the intensity of disease inflammation. This condition indicates that a larger dosage of acitretin may be preferred when treating EP patients with systematic symptoms.
The main weakness of our research is associated with the retrospective study design. The drug response was mainly estimated on the basis of written and/or photographic records. It would be more accurate if the PASI was recorded for every patient. Also, the calculation of the sample size selected was not performed in this study. Besides, patients in our study used two brands of acitretin (Fangxi and Neotigason). The choice of the brand was based on the patient’s economic condition and personal wishes. Since 2014, all patients used Fangxi, because there have been no sales of Neotigason in Chinese mainland. In addition, the adverse effects might be underestimated because they were retrospectively obtained using medical records. In the future, as EP transformed from pustular psoriasis seems to be a unique subtype, we hope to carry out a prospective study to compare between EP transformed from pustular psoriasis and psoriasis vulgaris with longer follow-up.
Conclusion
In conclusion, according to our best knowledge, we presented the largest cohort of EP patients treated with acitretin as a systemic monotherapy. According to our analysis, acitretin is a promising agent against EP, particularly in patients with EP that developed from psoriasis vulgaris, without infection and with normal hsCRP levels. Acitretin also appears to be well-tolerated. The dose is related to body weight and presentation of systematic symptoms. Larger prospective controlled studies with an extended follow-up are needed for further confirmation.
