Abstract
Tofacitinib was the first Janus kinase inhibitor to be approved for the treatment of rheumatoid arthritis (RA), and there is a large body of data to inform the efficacy and safety of this drug for patients at different places in their treatment journeys and with diverse demographics and characteristics. Here, we summarize tofacitinib clinical efficacy and safety data from some clinical trials, post hoc analyses, and real-world studies, which provide evidence of the efficacy of tofacitinib in treating patients with RA at various stages of their treatment journeys, and with differentiating baseline characteristics, such as age, gender, race, and body mass index. In addition, we review the safety data available from different patient subpopulations in the tofacitinib clinical development program, real-world data, and findings from the ORAL Surveillance post-marketing safety study that included patients aged ⩾50 years with pre-existing cardiovascular risk factors. The available efficacy and safety data in these subpopulations can enable better discussions between clinicians and patients to guide informed decision-making and individualized patient care.
Introduction
Rheumatoid arthritis (RA) is a systemic, chronic inflammatory disease that causes synovitis and is associated with increased risk of comorbidities and higher mortality, compared to the general population.1,2 Treatment options for RA have expanded beyond conventional synthetic disease-modifying antirheumatic drugs (csDMARDs), and aiming for remission, or at least low disease activity (LDA), per the ‘treat-to-target’ guidelines has become more achievable.3,4 Among the treatment options available to patients with RA are oral small molecule Janus kinase (JAK) inhibitors. Tofacitinib was the first JAK inhibitor to be approved for the treatment of RA, followed by baricitinib, upadacitinib, and filgotinib. As tofacitinib was first approved for RA in 2012, a large body of data has amassed from clinical trials, post hoc analyses, and real-world studies, which provides information to guide the appropriate use of this medication across different clinical scenarios.5–13 However, the volume of these data may also make it difficult for a clinician to quickly access the specific information they need for a particular clinical scenario. The goal of this article is to summarize the most pertinent tofacitinib efficacy and safety information based on commonly occurring clinical scenarios.
How to use this article
Table 1 provides a guide for our readers; each tofacitinib developmental study has a study number, alongside a study name used to refer to the studies more easily. The article is then divided into summaries of efficacy and safety information by patient characteristics and specific clinical scenarios.
Phase III, IIIb/IV, and LTE studies of tofacitinib in RA.

BID, twice daily; csDMARD, conventional synthetic disease-modifying antirheumatic drug; DMARD, disease-modifying antirheumatic drug; IR, inadequate response; LTE, long-term extension; MTX, methotrexate; N, total number of patients randomized and treated in the study; NI, non-inferiority; OL, open-label; PBO, placebo; PC, placebo-controlled; Q2W, every 2 weeks; QD, once daily; QW, once weekly; RA, rheumatoid arthritis; RDB, randomized, double-blind; SC, subcutaneous; TNFi, tumor necrosis factor inhibitor.
ClinicalTrials.gov identifiers: NCT01039688 (ORAL Start); NCT00814307 (ORAL Solo); NCT00856544 (ORAL Sync); NCT00853385 (ORAL Standard); NCT00847613 (ORAL Scan); NCT00960440 (ORAL Step); NCT02187055 (ORAL Strategy); NCT02831855 (ORAL Shift); NCT02092467 (ORAL Surveillance); NCT00413699 (ORAL Sequel); NCT00661661 (A3921041).
Participants receiving PBO advanced to tofacitinib 5 or 10 mg BID in a blinded manner at 3 months.
Participants receiving PBO advanced to tofacitinib 5 or 10 mg BID in a blinded manner at 3 months (non-responders) or 6 months (all remaining participants receiving PBO).
Concomitant csDMARD use was permitted. Patients could increase or decrease tofacitinib dose at the discretion of the investigator.
Use of tofacitinib based on a patient’s baseline characteristics
Tofacitinib use in seropositive versus seronegative RA
Elevated levels of rheumatoid factor (RF) and/or anti-cyclic citrullinated peptide (CCP) antibodies are seen in many, but not all, patients with a diagnosis of RA.19,20 Seropositivity for these antibodies may indicate greater disease severity and a higher risk of disease progression21,22 and has been shown to influence response to treatment for some medications. 23 A post hoc analysis of data pooled from ORAL Solo, Sync, Standard, Scan, and Step illustrated that tofacitinib significantly improved American College of Rheumatology (ACR) 20/50/70 responses, Disease Activity Score in 28 joints, erythrocyte sedimentation rate (ESR) LDA rates, and patient-reported outcomes (PROs), regardless of the patient’s serologic status. 24 However, anti-CCP-positive patients were more likely to achieve remission and LDA, and also had numerically greater improvement in physical functioning, regardless of RF status. 24 Another post hoc analysis of ORAL Scan and Start showed that tofacitinib generally reduced radiographic progression in patients with RA versus placebo (ORAL Scan) and methotrexate (MTX; ORAL Start), regardless of serologic status, but the impact on radiographic progression was larger in seropositive patients (either anti-CCP-positive or RF-positive). 25 Rates of adverse events (AEs) and discontinuations due to AEs were similar across serotype subgroups. 24
Tofacitinib use in RA based on sex, race, and geography
As with other studies in RA,26,27 the majority of patients with RA in the tofacitinib clinical development program were female; 28 however, tofacitinib has been shown to be efficacious regardless of biological sex. A post hoc analysis of ORAL Scan, Standard, and Sync specifically compared the outcomes of patients based on documented biologic sex, and indicated that male patients were generally more likely to reach remission, had slightly greater reduction in disease activity from baseline, and had greater improvement in disability index and functional assessments. 29 No consistent differences in safety findings were reported between males and females. 29 The observation that female patients have more residual symptoms has been reported with other advanced therapies and is poorly understood. 30
In another post hoc analysis of 15 phase II/III/IIIb/IV studies that evaluated the impact of race on safety and efficacy, patients in the tofacitinib clinical development program self-identified as White (n = 4145), Black (n = 213), Asian (n = 1348), or ‘Other’ (n = 649). 31 In general, patients responded to tofacitinib regardless of self-identified race, although numerically higher placebo responses were observed in Black patients compared with patients of other races, a finding that warrants further exploration. 31 Safety findings for tofacitinib were generally consistent across racial/treatment groups, except for higher rates of infections, herpes zoster (HZ), and hepatic events in Asian patients, who were mainly from Japan and Korea. 31 In a post hoc analysis of the upadacitinib RA phase III clinical program, upadacitinib was efficacious regardless of race; however, efficacy was generally lower in Black or African American patients versus patients who identified as White, Asian, or ‘Other’. Although safety was comparable across these groups, a higher overall AE rate was observed among non-White patients, while higher rates of specific AEs of special interest were noted in Asian (serious infections and HZ) and Black or African American patients [creatine phosphokinase elevations and adjudicated venous thromboembolisms (VTEs)]. 32
The consistency of tofacitinib efficacy and safety across racial groups in clinical studies are generally supported by real-world studies in patients with RA across geographic regions. For instance, in the Taiwan XTRA registry, the long-term (5 years) efficacy and safety of tofacitinib were shown to be comparable with tumor necrosis factor inhibitors (TNFi). 33 In addition, tofacitinib was effective and well-tolerated in patients enrolled in the Turkish HURBIO database, and in patients from the St. Gallen and Aarau hospitals in Switzerland.34,35 Tofacitinib was also shown to have comparable effectiveness and safety profiles to baricitinib in a real-world study in Japan, where HZ was the most common AE with both treatments. 36 Safety profiles of tofacitinib in real-world Latin American settings appeared to be generally consistent with those observed in a post hoc analysis of Latin American patients who received tofacitinib in the RA clinical program.37,38
Tofacitinib use in patients with pain and fatigue
Tofacitinib has been associated with rapid analgesic effects across multiple pain measures. 39 In ORAL Solo, where an interactive voice response system (IVRS) was used, patients receiving tofacitinib reported a reduction in their pain level in their IVRS daily diary 3 days from the start of therapy, and the difference between patients receiving tofacitinib versus placebo was statistically significant by the first post-baseline assessment at 2 weeks. 40 Supplementing this clinical trial data with recent real-world evidence, the majority (60%) of patients with RA receiving tofacitinib in clinical practice felt that their pain was reduced by half within 1 month of starting the therapy. 41 This decrease in pain level correlated with a reduction in disease activity measures. 41 Rapid pain relief has also been reported with other JAK inhibitors.42,43 In addition to targeting inflammation, JAK inhibitors may have a direct impact on the neurological pathways mediating pain; however, the exact mechanism through which this could happen has not been clearly described.
Patients with RA view fatigue as one of the most pervasive symptoms of their disease. 44 Fatigue was a pre-specified PRO in all the tofacitinib phase III studies, with the Functional Assessment of Chronic Illness Therapy-Fatigue (FACIT-F) scoring system used as an objective measure to compare baseline with Month 1 (first assessment of fatigue after therapy initiation), and time points through the remainder of each study. 45 Patients treated with tofacitinib were shown to have significant improvement in fatigue at Month 1 compared with MTX monotherapy (ORAL Start) 46 or placebo (ORAL Sync, Scan, and Standard).47–49 In ORAL Strategy, patients treated with tofacitinib either as monotherapy or in combination with MTX had similar improvements in fatigue from Month 3; improvements for patients receiving tofacitinib in combination with MTX were significantly greater at Month 6 than for those receiving adalimumab in combination with MTX. 50 Similarly, results from the SELECT-COMPARE phase III trial of upadacitinib in patients with RA with an inadequate response to MTX showed that patients treated with upadacitinib and background csDMARDs had significantly greater least squares mean changes from baseline in pain and fatigue at Week 12 versus those receiving adalimumab and background csDMARDs. 43
Tofacitinib use in a patient with depression and anxiety
A post hoc analysis of five phase III studies and one phase IIIb/IV study suggested that patients with signs of anxiety and depression had efficacy outcomes comparable with those of patients who did not have a past medical history of these diagnoses. Interestingly, the proportion of tofacitinib-treated patients who identified as having probable co-morbid major depressive disorder/anxiety (based on 36-item Short Form Health Survey [SF-36] mental component summary [MCS] score ⩽38) decreased from baseline with tofacitinib therapy (60% reduction after 6 months). 51 This analysis was limited by the use of the SF-36 MCS score to identify probable rather than confirmed major depressive disorder/anxiety. Further research using a gold-standard psychiatric interview is required to validate the use of SF-36 MCS score ⩽38.
Tofacitinib use in RA by weight and body mass index
Pooled analyses of data from the tofacitinib clinical development studies have demonstrated consistent efficacy of tofacitinib in patients with RA, regardless of their weight (Figure 1) or body mass index (BMI) at baseline.52–54 Although the improvement in disease activity score from baseline is dampened as the patient’s BMI increases, 53 this is a trend also seen with other advanced therapies such as TNFi.55,56

Probability ratios for achieving ACR20 response with tofacitinib plus MTX versus MTX alone in subpopulations of patients with RA stratified by (a) demographics and (b) disease characteristics. 52
Tofacitinib use in older patients and those with comorbidities
Note 1: The recommended dosage for RA is tofacitinib 5 mg twice daily (BID) or modified-release 11 mg once daily (QD).57,58
The impact of age on the efficacy of tofacitinib has been investigated in two major data sets. The first is the pooled data from the phase III (ORAL Step, Sync, Scan, Solo and Standard) and long-term extension (LTE; ORAL Sequel and A3921041) trials of tofacitinib in patients with RA, which showed consistent effects of tofacitinib in improving disease activity and disability measures regardless of age (⩾65 years versus <65 years). 59 However, patients aged ⩾65 years in the LTEs had numerically higher incidence rates (IRs) of serious AEs and discontinuations, and higher rates of serious infections, than those aged <65 years. 59 Subsequent analysis of tofacitinib data from clinical trials (pooled phase II/III/IIIb/IV studies) has also confirmed a higher risk of serious infections in older versus younger patients. 60 Similarly, in an integrated safety analysis of upadacitinib clinical trials across RA, psoriatic arthritis, ankylosing spondylitis, and atopic dermatitis, higher rates of major adverse cardiovascular events (MACE), VTEs, malignancies, and serious infections were observed in patients aged ⩾65 years receiving upadacitinib, adalimumab, and MTX. 61 A higher IR for serious infections in older patients was also observed with tofacitinib and biologic disease-modifying antirheumatic drugs (bDMARDs) in US RA registry data, highlighting the need for careful monitoring of older patients. 60 The increased risk for adverse outcomes of therapy in older patients with RA is an established phenomenon that has been observed regardless of treatment.59,62
A key data set where the impact of age may be observed is from ORAL Surveillance (NCT02092467). The ORAL Surveillance post-authorization study was conducted per the directive of the US Food and Drug Administration owing to observed increases in serum lipids and some malignancies in the tofacitinib RA clinical development studies. The study enrolled >4300 patients with active RA despite MTX treatment and, to enrich for cardiovascular (CV) risk, patients had to be aged ⩾50 years and have one or more additional CV risk factors. 16 The patients had a median age of 60 years (range 50–88 years), and approximately one-third were aged ⩾65 years. For reference, patients in the tofacitinib RA clinical program had a median age of 53 years (range 18–86 years), and 16% were aged ⩾ 65 years. 63 Other than their age, key points to note about the patients in ORAL Surveillance are that half of the patients were current or ex-smokers (compared with approximately one-third across the RA program) 63 ; the majority (66%) had hypertension; 17% had diabetes; and 11% had history of coronary artery disease. 16
Although this was primarily a safety trial with an open-label design, it also provided data on the efficacy of tofacitinib 5 and 10 mg BID, compared with an active control (TNFi) in this relatively older population with comorbidities. 16 Across multiple efficacy endpoints, the three treatment arms (tofacitinib 5 mg BID, tofacitinib 10 mg BID, and TNFi) showed comparable efficacy. The first post-enrollment visit was at Month 2, then from Month 3 onwards patients were seen every 3 months while in the trial, and the data show improvement in composite disease activity measures and PROs at Month 2 through to study completion at around 5 years (Figure 2). 16 During the trial, 3.7% of patients who received tofacitinib 5 mg BID, and 1.8% of patients who received tofacitinib 10 mg BID, withdrew from treatment due to insufficient clinical response, compared with 5.4% of patients who received a TNFi. 16 By the end of the study, 8.0% of patients who received tofacitinib 5 mg BID, and 7.5% who received tofacitinib 10 mg BID, had permanently discontinued background MTX, compared with 4.9% for TNFi.
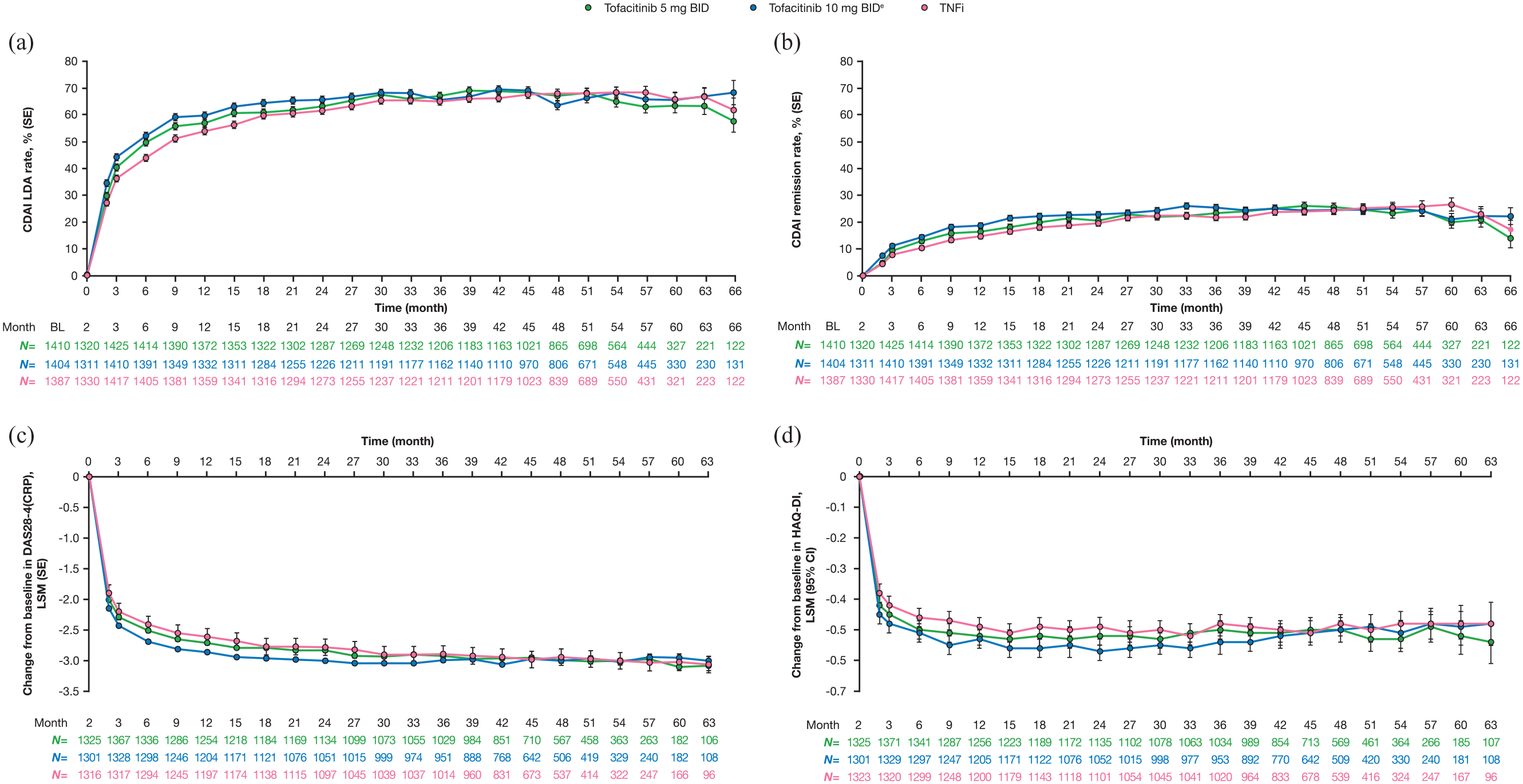
Proportion of patients achieving (a) CDAI LDAa and (b) CDAI remissionb, and change from baseline in (c) DAS28-4(CRP)c and (d) HAQ-DId over time in ORAL Surveillance.
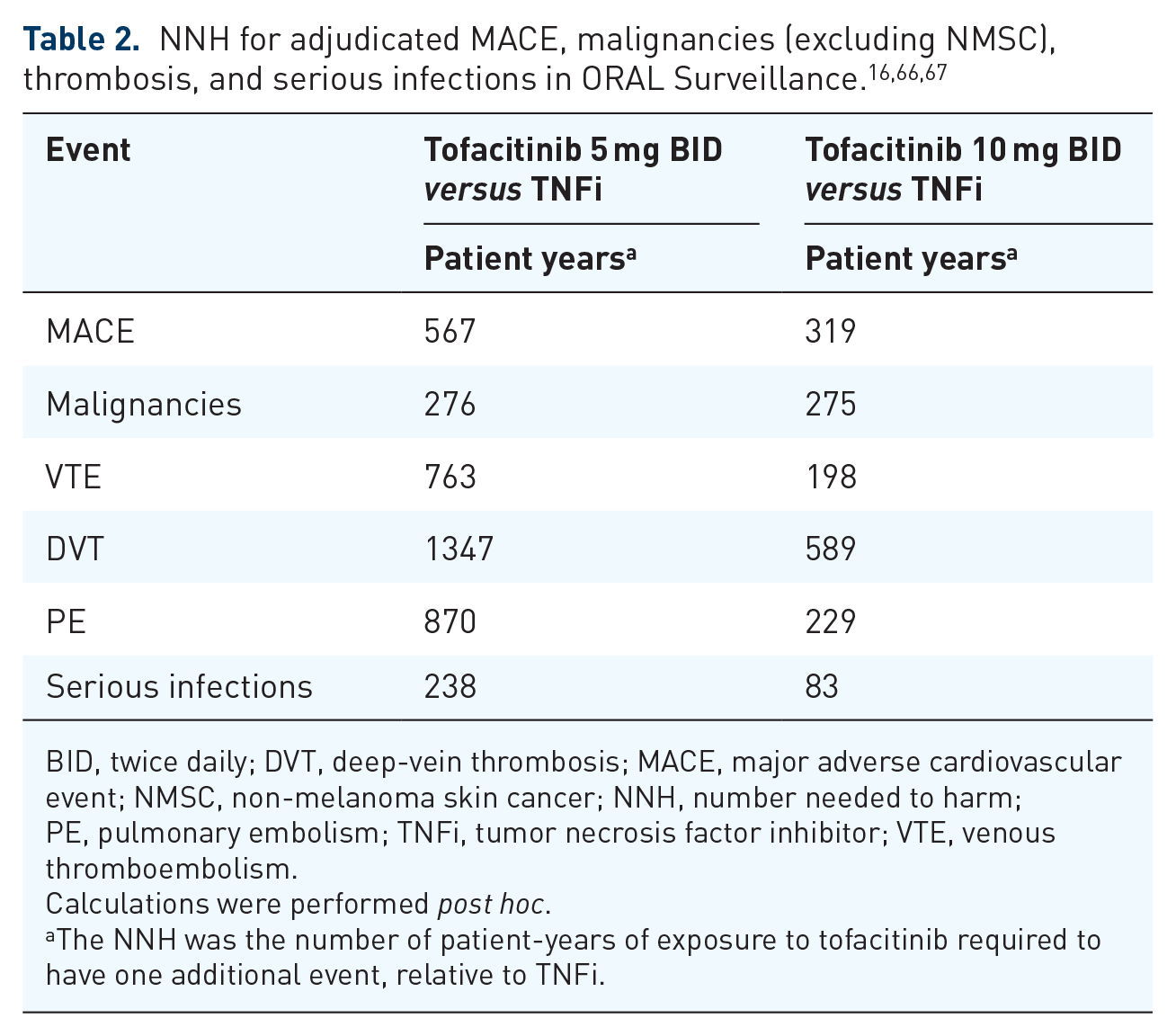
ORAL Surveillance also provided important information regarding the safety of tofacitinib versus TNFi in this CV risk-enriched older patient population. The hypothesis of ORAL Surveillance was based on the non-inferiority of tofacitinib versus TNFi for two co-primary endpoints: MACE, and malignancies excluding non-melanoma skin cancers (NMSC); at study conclusion, non-inferiority criteria were not met. The hazard ratio (confidence interval [CI]) for MACE on tofacitinib (5 and 10 mg BID combined) compared with TNFi was 1.33 (0.91–1.94), and for malignancies excluding NMSC it was 1.48 (1.04–2.09). 16 Post hoc analyses of the data revealed that age ⩾65 years was a significant risk factor in this study for MACE and malignancies excluding NMSC across treatment groups.64,65 In addition to the findings on the co-primary endpoints, patients on tofacitinib were noted to have a higher risk than those on TNFi for other AEs of special interest. Details are presented in the ORAL Surveillance primary manuscript. 16 Table 2 provides a summary of numbers needed to harm (NNH) for AEs of special interest in ORAL Surveillance. The NNH is a derived statistic that tells us the number of patient-years of exposure to tofacitinib required to have one additional event relative to TNFi. ORAL Surveillance was a safety study of tofacitinib versus an active control but without a placebo arm. It can therefore not be determined whether the difference seen in the risk of these AEs in ORAL Surveillance is due to TNFi reducing risk, tofacitinib increasing risk, or a combination thereof, in patients with RA.
BID, twice daily; DVT, deep-vein thrombosis; MACE, major adverse cardiovascular event; NMSC, non-melanoma skin cancer; NNH, number needed to harm; PE, pulmonary embolism; TNFi, tumor necrosis factor inhibitor; VTE, venous thromboembolism.
Calculations were performed post hoc.
The NNH was the number of patient-years of exposure to tofacitinib required to have one additional event, relative to TNFi.
Supplementing the data from ORAL Surveillance are several post-marketing real-world studies that use data from registries and claims data; real-world data include patient populations that reflect those seen in clinical practice and are valuable in supplementing data from clinical trials. A study using the US CorEvitas RA Registry (formerly Corrona), which evaluated 5-year AE IRs between patients initiated on tofacitinib and those initiated on bDMARDs, showed that IRs for serious infections, MACE, VTEs, malignancies, NMSC, and deaths were comparable for tofacitinib and bDMARDs at 5 years after treatment initiation. 13 The CorEvitas study looked at all patients in the registry with a diagnosis of RA, and did not specifically separate out older patients or those with CV risk. In another study from US claims-based data, called STAR-RA, in which 102,263 patients were identified and of whom 12,852 initiated tofacitinib, there was no increased risk of CV outcomes (myocardial infarction or stroke) or malignancies (excluding NMSC) with tofacitinib versus TNFi when assessing the overall RA cohort; however, risk of MACE and malignancies (excluding NMSC) was numerically increased with tofacitinib relative to TNFi when they evaluated a CV risk-enriched population by mimicking the ORAL Surveillance inclusion criteria (aged ⩾ 50 years and with ⩾1 baseline CV risk factor).68,69 Conversely, analysis from the German RABBIT register did not show an increased risk of MACE with JAK inhibitors compared with TNFi, in both the overall RA population and a high CV risk cohort. 70
Tofacitinib use in RA based on smoking status
Smoking status (current smoker or ex-smoker compared with those who had never smoked) had not been found to have an effect on efficacy outcomes, 71 even when looking at the impact of smoking on efficacy in older (aged ⩾ 65 years) and younger (aged < 65 years) patients [post hoc analysis of phase III (ORAL Step, Sync, Scan, Solo, and Standard) and LTE studies (ORAL Sequel and A3921041)]. 59 However, as may be expected, smoking has been found to have a negative impact on the risk of AEs. In ORAL Surveillance, patients who were current or past smokers had a significantly higher risk for MACE and malignancies excluding NMSC compared with those who had never smoked across treatment groups.65,72 Similarly, in an analysis of the upadacitinib clinical program, smoking was shown to be a risk factor for MACE in patients with RA receiving upadacitinib. 73
Appropriate use of tofacitinib at different stages in a patient’s treatment journey
Note 2: Treatment guidelines regarding the appropriate use of tofacitinib in different lines of therapy are evolving; here, we only present the data for clinical consideration.
Impact of RA disease duration and inflammatory markers on tofacitinib efficacy
In three post hoc analyses investigating the impact of disease duration on tofacitinib efficacy, patients who were bDMARD-naïve, or early in their RA diagnosis (<1 year), had greater clinical responses to tofacitinib, compared with biologic-experienced patients or those with longer duration of disease, respectively.11,12,74 Those with higher levels of inflammation at baseline, as measured by C-reactive protein (CRP), have been shown to have a numerically higher response to tofacitinib (particularly those who had failed a TNFi previously); the effect of baseline ESR on efficacy outcomes was less clear. 75 No meaningful differences in the safety profile of tofacitinib were reported in these post hoc analyses of patients with early versus established RA (ORAL Start) or based on baseline CRP alone (nine phase II/III studies).11,75 While serious infections occurred more frequently with tofacitinib in patients with established versus early RA, the differences were not considered to be clinically meaningful. 11
Use of tofacitinib as first-line therapy for RA
The use of tofacitinib is not approved in MTX-naïve patients. In ORAL Start (MTX-naïve patients), tofacitinib monotherapy was superior to MTX monotherapy across disease activity measures and the pain and fatigue PROs (Figure 3), compared with MTX monotherapy.5,46,76 Furthermore, patients experienced more rapid improvement in physical function while receiving tofacitinib than MTX,5,46 and tofacitinib monotherapy inhibited the progression of structural damage more effectively than MTX monotherapy (Figure 3).5,76 The rates of AEs, serious AEs, and discontinuations due to AEs were similar between the two groups. 5

Tofacitinib in patients with RA who are already on MTX or chronic steroids
Two clinical questions can be considered here. The first is whether being treated with MTX or corticosteroids (and the dose) impacts tofacitinib efficacy. To address this, two post hoc analyses were conducted to determine the impact of background MTX dose or concomitant corticosteroids on efficacy outcomes in tofacitinib phase III RA studies. These analyses demonstrated no impact of background concomitant MTX dose level (⩽12.5, >12.5–<17.5, or ⩾17.5 mg/week) or use of corticosteroids (⩽10 mg/day) on the efficacy of tofacitinib versus placebo.77,78 Details of studies on the efficacy of tofacitinib as monotherapy versus in combination with MTX are presented in the section ‘Tofacitinib as monotherapy’. Generally, the safety profile of patients on tofacitinib versus tofacitinib plus csDMARD was comparable. 79 However, the proportion of patients with elevated liver enzymes, as well as rates of serious infections and HZ, was numerically higher with combination therapy 79 ; the incidence of HZ increased in patients on concomitant corticosteroids. 80
The second question is whether patients on a combination of tofacitinib and MTX/chronic oral corticosteroids can be tapered off these add-on therapies while maintaining response on tofacitinib monotherapy. A post hoc analysis of pooled tofacitinib LTE data (combined doses) showed that approximately 70% of patients who had achieved Clinical Disease Activity Index (CDAI) remission at Month 3, and subsequently discontinued MTX or corticosteroids, remained in remission after 3 years. 81 This finding suggested that response to tofacitinib can be maintained regardless of MTX or corticosteroid withdrawal. ORAL Shift subsequently confirmed that MTX can be withdrawn when patients have achieved LDA. In that study, patients received modified-release tofacitinib (11 mg QD) plus MTX, and then MTX was withdrawn. If patients had already achieved CDAI LDA after 24 weeks of receiving the combination, their disease activity remained controlled without MTX. 15
Tofacitinib as an add-on therapy when RA activity is refractory to csDMARDs
A post hoc analysis of phase II/III studies has suggested that the addition of tofacitinib can improve the symptoms of RA in patients who fail MTX or csDMARD alone. 52 When tofacitinib 5 mg BID was added to MTX in ORAL Scan, Standard, and Strategy (all of which included patients with an inadequate response to MTX), patients experienced a significant reduction in RA disease activity, pain, and fatigue, as well as improved functioning and sleep (Figure 4), and had a higher likelihood of achieving remission than on csDMARD alone after 6 months of treatment.7,8,14,47,49,50,82 Similar results were observed in ORAL Sync, wherein patients with an inadequate response to previous DMARD therapy received tofacitinib in addition to csDMARDs including MTX, leflunomide, sulfasalazine, or hydroxychloroquine (Figure 4).6,48 In ORAL Scan, adding tofacitinib, compared with placebo, to MTX, also inhibited the progression of structural damage (significantly with tofacitinib 10 mg BID), and both doses of tofacitinib were associated with significantly higher rates of non-progression at Month 6 (Figure 4), regardless of the background MTX dose used.7,78,83 It is known from a post hoc analysis that patients who achieved remission early with tofacitinib had significant long-term inhibition of radiographic progression. 76 A small open-label study in which patients with RA received tofacitinib 5 mg BID added to csDMARDs captured early improvements in musculoskeletal ultrasound and demonstrated an association between early ultrasound response and 12-week clinical outcomes. 84

Another important point to note from these trials in patients with RA refractory to csDMARDs is that two of the trials (ORAL Standard and Strategy) also included a group of patients who received a TNFi (subcutaneous adalimumab every other week added to MTX).8,14 In both studies, tofacitinib 5 mg BID added to MTX yielded similar clinical improvements as adalimumab added to MTX, although it should be noted that ORAL Standard was not designed for a formal comparison between the tofacitinib and adalimumab treatment groups (Figure 4).8,14
Safety findings for tofacitinib added to MTX or other csDMARDs were generally consistent with the monotherapy studies. 79 As mentioned previously, the proportion of patients with elevated liver enzymes, as well as rates of serious infections and HZ, were numerically higher with combination therapy compared with tofacitinib alone. 79 Increased risk of HZ is a repeated safety finding across tofacitinib studies, as well as other JAK inhibitor studies in RA.26,80,86,87 This highlights the importance of vaccination to prevent HZ in patients with RA, as discussed in the section ‘Use of tofacitinib in a patient requiring vaccination’.
Tofacitinib as monotherapy
In a phase IIb dose-ranging study in DMARD-IR patients, tofacitinib monotherapy (5 or 10 mg BID) achieved rapid and significant improvement versus placebo, as indicated by a higher proportion of patients achieving ACR20 responses at Month 3, while improvement with adalimumab monotherapy was not statistically significant in comparison with placebo. 88 In ORAL Strategy, tofacitinib plus MTX was non-inferior to adalimumab plus MTX. Tofacitinib monotherapy was not shown to be non-inferior to either combination. 14 These results suggest that in patients with an inadequate response to MTX, the addition of tofacitinib or adalimumab is equally efficacious, and the addition of tofacitinib to MTX is preferable to switching to tofacitinib monotherapy, although there was no difference in the remission rate (secondary endpoint) between groups.
Tofacitinib for biologic-experienced or JAK inhibitor-experienced (failed efficacy or intolerance) patients with RA
One challenging clinical scenario is the treatment of patients with RA who have already failed one or several bDMARDs. Approximately one-third of the patients in ORAL Step had tried and failed ⩾2 prior TNFi; 10 despite this, the treatment of TNFi-IR patients in this study with tofacitinib plus MTX led to improved disease activity and PROs versus placebo by Month 3 (Figure 5). The safety profile of tofacitinib in this population was generally consistent with that reported in other studies.10,28
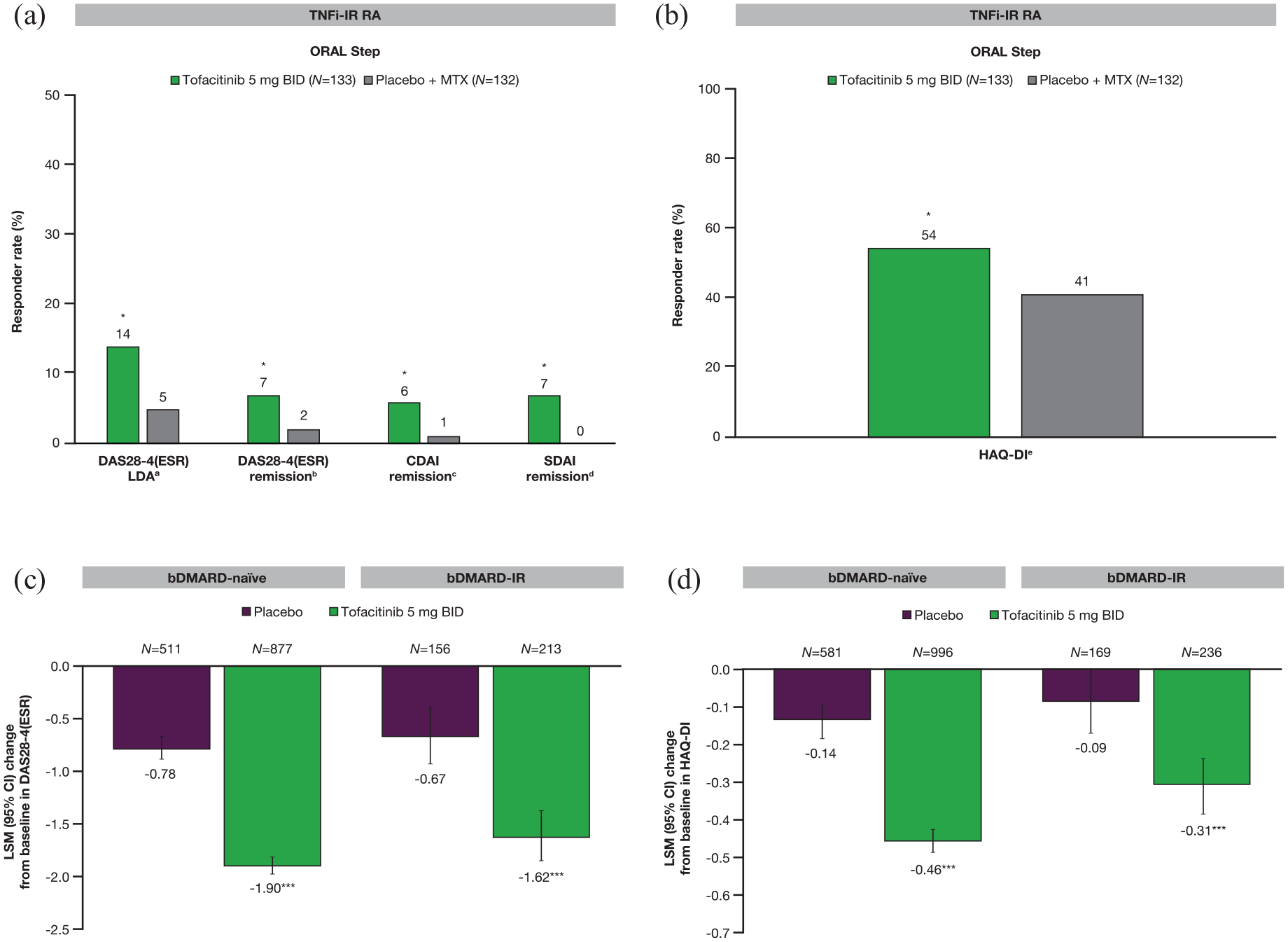
Effects of tofacitinib in combination with csDMARD on (a) disease activity and (b) patient-reported outcomes at Month 3 in a phase III study of patients with TNFi-IR RA,10,82 and change from baseline in (c) DAS28-4(ESR) and (d) HAQ-DI at Month 3 in phase II/III trials of bDMARD-naïve and bDMARD-IR patients (adapted by permission from BMJ publishing Group Limited [Charles-Schoeman C, et al. Ann Rheum Dis 2016]). 74
Tofacitinib has shown efficacy (reduced signs and symptoms, and improved PROs) in patients who had previously failed bDMARDs. This was demonstrated in a post hoc analysis of phase II/III studies, including patients with prior inadequate response to ⩾2 TNFi and other bDMARDs (abatacept, rituximab, or tocilizumab). 74
Registry data in patients who failed a first JAK inhibitor suggest that switching to a second JAK inhibitor seems to lend similar efficacy as switching to a bDMARD, 89 and higher rates of drug retention than switching to a TNFi. 90 At this time, there are limited data regarding patients who have failed another JAK inhibitor and then started treatment with tofacitinib.
In general, we know that patients who have failed one or more advanced therapies tend to have more treatment-refractory RA. 91 While patients with RA have shown improvement with tofacitinib regardless of whether or not they have been on a previous biologic, the data suggest that the biologic-naïve patients had a greater response to tofacitinib than bDMARD-experienced patients (Figure 5). 74 Suboptimal treatment compliance among patients who have already failed multiple therapies has been shown to also negatively impact outcomes.92,93
Tofacitinib in RA when treatment is interrupted and resumed
Temporary discontinuation of tofacitinib treatment for surgery, vaccination, or other reasons may become necessary. In a substudy of ORAL Sequel, patients discontinued tofacitinib to receive pneumococcal and influenza vaccines, and then re-initiated treatment at the same dose 2 weeks later. 94 Clinically meaningful worsening of disease activity measures was observed during the two-week interruption period. 94 One month after re-initiation of treatment, patients had returned to their baseline disease control status, both in terms of disease activity measures and PROs. 94 These data suggest that efficacy could be re-established with tofacitinib after temporary treatment interruption. 94
Use of tofacitinib in a patient requiring vaccination
The impact of tofacitinib on vaccine efficacy has been studied for T-cell-dependent (tetanus, influenza, and 13-valent pneumococcal vaccine) and T-cell-independent (23-valent pneumococcal vaccine) vaccines in phase II/III studies. Patients newly starting tofacitinib, particularly those receiving concomitant MTX, had diminished responsiveness to the 23 valent pneumococcal vaccine, but not to the influenza vaccine. Temporarily discontinuing tofacitinib for 2 weeks following vaccination had minimal effect on the responses to either vaccine. 95
Similarly, a phase II study on the use of live attenuated zoster vaccination (Zostavax®), given 2–3 weeks prior to initiating tofacitinib treatment, found that tofacitinib did not impact vaccine response. 96 However, in this study, one patient without prior exposure to varicella zoster virus (VZV) developed vaccine disseminated virus, and clinical judgment should be used to preferably allow a longer interruption of tofacitinib when administering live vaccines. 96 Recombinant zoster vaccine (Shingrix®) is an important alternative to the live vaccine for immunocompromised patients, and data from a real-world study support its use in patients with RA; disease flares and AEs reported after vaccination were mild and self-limiting and did not require a change in RA therapy. 97 Clinical guidance on long-term strategies for the vaccination of patients with RA receiving JAK inhibitors is still evolving.
Recent guidance on the COVID-19 vaccination recommends interruption of therapy with JAK inhibitors, owing to concerns about the inhibition of interferons as a result of JAK inhibition. 98 Data from clinical studies of tofacitinib demonstrate small and variable changes in immune cell subsets during therapy. 99 As previously described above, from an analysis investigating changes in disease activity after only 2 weeks of treatment interruption, patients had steadily increasing disease activity levels, consistent with a clinically meaningful change, as well as increases in CRP. 94 This finding was not surprising given the short half-life of tofacitinib. Importantly, however, both patients in the group who continued tofacitinib therapy and those who interrupted therapy for influenza vaccination reached satisfactory immune responses (66.3% versus 63.7%, respectively). 95 Therefore, in the shared decision-making process, prescribers should consider both the benefits and potential risks of therapy interruption in the context of vaccination. 100 Evidence with regard to the management of immunosuppressants in the context of COVID-19 vaccination is still evolving. Available data from a population-based study of 315,101 US adults with COVID-19 demonstrate an increased risk of severe COVID-19 for patients with RA relative to a comparator cohort with COVID-19, but do not show a further increase in risk with use of JAK inhibitors, including tofacitinib. 101
Long-term use of tofacitinib
Data on the long-term effectiveness of tofacitinib are available from ORAL Surveillance (see section ‘Tofacitinib use in older patients and those with comorbidities’), LTE, and real-world studies.13,17,18 More than 4000 patients (>16,000 patient-years of exposure) who received tofacitinib as monotherapy or combination therapy in phase I to III clinical development trials participated in a global, open-label LTE study (ORAL Sequel), which showed sustained efficacy of tofacitinib for up to 8 years. 17 Median drug effectiveness was 5 years in the pooled analysis of data from the tofacitinib LTE studies. 102 The evidence for sustained efficacy in the clinical trials is supported by global post-marketing data 103 and real-world data from registries.104–107 One real-world study suggested higher drug retention for tofacitinib than for a TNFi, particularly when the TNFi was administered as monotherapy. 107 Patients with seronegative disease, diabetes, or hypertension were more likely to discontinue tofacitinib in the LTE studies. 102 A possible explanation is that patients with comorbidities might have experienced a higher frequency of AEs. Patient preferences regarding the frequency of therapy administration may also influence compliance and treatment outcomes; a real-world study from a US claims–based analysis demonstrated an adherence benefit for the modified release formulation of tofacitinib (11 mg QD), with comparable clinical outcomes as tofacitinib 5 mg BID. 105
Long-term safety data for tofacitinib can be obtained from the open-label LTE studies that had data for up to 9.5 years of treatment,17,18 as well as integrated safety summary (ISS) analyses, which combined the phase I/II/III/IIIb/IV and LTE trials with >7000 patients followed for up to 10.5 years.28,63 Long-term safety data were consistent with the safety profile established in the individual studies in the tofacitinib RA program. Long-term safety data in a CV risk-enriched older patient population can be gleaned from ORAL Surveillance (see ‘Tofacitinib use in older patients and those with comorbidities’). 16
Other safety data for tofacitinib in patients with RA
The most common side effects experienced by patients with RA receiving tofacitinib as monotherapy or in combination with MTX include upper respiratory tract infection, nasopharyngitis, urinary tract infection, bronchitis, HZ, diarrhea, headache, and hypertension.16,28,57 The most frequent serious AEs observed in patients with RA receiving tofacitinib are infections [8.2% of patients in tofacitinib RA ISS; IR (95% CI) 2.5 (2.3–2.7) patients with events/100 patient-years], most commonly pneumonia and HZ. 28 Similar results were observed in ORAL Surveillance, with serious infections reported in 9.7% [IR (95% CI) 2.9 (2.4–3.4) patients with events/100 patient-years] and 11.6% [IR (95% CI) 3.6 (3.1–4.2) patients with events/100 patient-years] of patients receiving tofacitinib 5 and 10 mg BID, respectively. 16 While the safety profile of tofacitinib is generally similar when given as monotherapy or in combination with csDMARDs, liver enzyme elevations are more frequent with combination therapy and serious infections and HZ appear to be less frequent with tofacitinib monotherapy (without csDMARDs or corticosteroids).67,79,80 For HZ in particular, post hoc analysis shows that the concomitant use of csDMARD and/or corticosteroids is associated with substantially higher IRs compared with tofacitinib monotherapy, and corticosteroid use has been identified as a significant risk factor for developing HZ while receiving tofacitinib. 80 For information on laboratory monitoring, local prescribing information should be consulted.
AEs of special interest: HZ
The elevated risk of HZ observed with tofacitinib108,109 is also seen with other JAK inhibitors 109 and is believed to be target related.110,111 Most cases of HZ reported in the tofacitinib RA ISS were monodermatomal and non-serious, although cases of multidermatomal (5.5% of patients with HZ) and disseminated (1.0%), including ocular HZ (0.1%), were reported. 28 In patients who received live attenuated zoster vaccination (Zostavax®) prior to tofacitinib treatment in a phase IIIb/IV study (N = 144), the VZV reactivation rate was 2.1%, compared with 1.5% for non-vaccinated patients (N = 616), with overall milder manifestations. 112 The efficacy of the live attenuated vaccine is 70% in patients aged 50–59 years and only 51% in those aged >60 years, 113 while the recombinant vaccine (Shingrix®) provides efficacy of >90% in older patients.114,115 A study on the use of recombinant zoster vaccine in patients with ulcerative colitis receiving tofacitinib is planned.
Conclusion
Tofacitinib was the first JAK inhibitor to be approved for the treatment of RA and has been in clinical use in different countries for a decade. An extensive evidence base on the efficacy, effectiveness, and safety of tofacitinib across different clinical scenarios and patient populations has accumulated from the clinical development program, post-marketing studies, and real-world use. Although not all the literature could be discussed in this narrative review, these data demonstrate the benefits of tofacitinib for patients with RA across populations. While the efficacy of tofacitinib appears generally consistent across patients with different demographic and disease characteristics, there is evidence that some patient groups might derive particular benefit (e.g. seropositive disease) and that some patients (e.g. older patients and those with CV risk factors, particularly smoking) are at greater risk of serious AEs while receiving tofacitinib, compared to TNFi. While we appreciate that everyday clinical scenarios can be complex, the summary of the available safety and efficacy data provided here is intended to help rheumatologists advise appropriate patients regarding treatment with tofacitinib, and, in conjunction, assess the risk/benefit profile of this medication.
Footnotes
Acknowledgements
This review was sponsored by Pfizer. New data presented are from studies sponsored by Pfizer. Medical writing support, under the direction of the authors, was provided by Kirsteen Munn, PhD, CMC Connect, a division of IPG Health Medical Communications, and was funded by Pfizer, New York, NY, USA, in accordance with Good Publication Practice (GPP 2022) guidelines (Ann Intern Med 2022; 175: 1298–1304). We thank Jose Rivas and David Gold, of Pfizer Inc, for their review and input.