Abstract
Background:
Ischemic stroke poses a major threat to human beings, and a prompt intravenous thrombolytic management remains the gold standard protocol for stroke sufferers. Although the role of thrombolytic therapy (r-tPA) for ischemic stroke patients and those with underlying impaired renal function has been advocated as effective treating strategy, there is still a lack of investigation as to finding out baseline important variables that are capable of early outcome prediction.
Objectives:
In this project, we hypothesize that the change of clinical chronic kidney disease (CKD) staging (delta stage = CKD stage after 3-month follow-up – CKD stage at admission) could serve as a crucial predictor of the prognosis of patients.
Design:
This is a cohort longitudinal retrospective study.
Sources and Methods:
A total of 765 cerebral artery ischemic stroke patients with impaired renal function were recruited and followed up for 1 year. Among them, 116 had received the thrombolytic treatment (r-tPA) after being evaluated at the triage in the emergency department and the rest had not (non-r-tPA). Propensity-matching was applied to compare the mortality between the r-tPA and non-r-tPA groups. Multiple logistic regression (LR) and decision tree (DT) algorithm were used to identify important prediction factors for mortality as well as the improvement in neurological function.
Results:
The 1-year mortality rates for r-tPA and non-r-tPA groups were 32.8% and 44.4%, respectively. The propensity-matched odds ratio of mortality for the r-tPA group compared with the non-r-tPA group is 0.469, with p = 0.003. Logistic regressions suggest that age, Hct, diabetes mellitus type 2, coronary artery disease, and delta stage are important factors for mortality for the non-r-tPA group, whereas age, diabetes mellitus type 2, chronic heart failure, hospital day, and delta stage are important factors for the r-tPA group. On the usage of antihypertensive drugs, ACEI/ARB was not associated with mortality (p = 0.198), whereas the diuretic was, with odds ratio at 1.619 (p = 0.025), indicating higher mortality after administration. Both LR and DT analyses indicate that delta stage is the most important predictor. For the r-tPA group, patients with delta stage ⩽0 had a 24% mortality, while that for delta stage >0 the mortality is 75%. For non-r-tPA patients, the corresponding mortalities were 30.9 and 66.3, respectively. Delta stage is also useful for predicting patients’ improvement of neurological function, assessed by NIHSS, mRS, and Barthel Index. The areas under the curve for the three assessments are 0.83, 0.835, and 0.663, respectively.
Conclusion:
Large-artery ischemic stroke patients who received thrombolytic treatment had significantly lower mortality, even when presenting underlying impaired renal function. The change of CKD staging (delta stage) is capable of acting as a powerful clinical baseline surrogate for both r-tPA and non-r-tPA patients in terms of early outcome prediction. Long-term use of diuretics could be potentially harmful to this group of patients. Moreover, delta stage correlates well with clinical long-term neurological functionality assessment (NIHSS, mRS, and Barthel index), which is helpful in aiding urgent clinical decision-making
Keywords
Introduction
Ischemic stroke represents the third leading cause of morbidity and mortality worldwide and it frequently occurs also in Taiwan. 1 Although there are several subtypes of stroke, middle cerebral artery infarction poses the most life-threatening sub-type of stroke and thus requires imminent management. For the first week after the index event, the cerebral infarct core as well as the penumbra regions might cause cellular edematous change of expansion of the brain parenchyma, leading to a cerebral herniation. Thus, careful observation and prompt treatment of thrombolytic therapy are the gold standard according to the American Heart Association/American Stroke Association guidelines (AHA/ASA). 1 Reviewing the thrombolytic guideline of inclusion and exclusion criteria, the proper National Institutes of Health Stroke Scale (NIHSS) scores for the potential candidates are between 4 and 25 points. In addition, those with poor body function are excluded from the thrombolytic treatment [recombinant tissue plasminogen activator (r-tPA)].2,3
Renal failure is not only a leading cause of morbidity and mortality worldwide, but also a potent risk factor for stroke. 4 In particular, the risk of stroke in the end-stage renal disease (ESRD) population may be as high as 30-fold compared to the general population and is the third most common cause of death. Case fatality rates are also higher reaching almost 90%. 5 Power et al. 6 studied the stroke risk in hemodialysis patients and found that acute stroke had overall 1-year mortality of 24% and was significantly even worse in patients with hemorrhagic events (39%).
A recent study has shown that outcomes following r-tPA and endovascular therapy do not differ between chronic kidney disease (CKD) and non-CKD patients and concludes that both therapies should be offered to eligible patients, regardless of their renal function. 7 Another recent meta-analysis concluded that the risk of spontaneous intracranial hemorrhage is higher in patients with CKD undergoing r-tPA. 8 A post hoc analysis of the ENCHANTED study, which also echoed the possibility of the spontaneous intracranial hemorrhage, also showed that CKD is associated with increased mortality but not disability or bleeding tendency in thrombolysis-eligible and treated acute ischemic stroke patients. 9 However, in clinical practice, the authors have encountered several middle cerebral artery infarction patients with underlying poor renal function. This could be considered a clinical dilemma, as first-line neurologists are those meant to evaluate patients and to provide explanations to their families, and in these circumstances, it would occur without a proper support of information and clinical evidence. Upon reviewing the current published literature,1–3 although the effectiveness of administration of intravenous thrombolytic agent (r-tPA) on this group of patients has been documented as fair and will not increase the long-term mortality rate, there still exists a clinical gap as to finding out baseline important variables that are capable of early predicting patients’ long-term outcomes.
The primary aim of the current investigation is thus to define the effectiveness and outcome of thrombolytic therapy for this group of patients. For r-tPA receivers and non-r-tPA receivers, the authors intend to identify the most important clinical predictors to forecast the patients’ long-term functional outcomes. In addition, for the clinical question, the research intends to determine whether the antihypertensive drugs could have a long-term impact on patients. Finally, once the important parameters have been found, we will consider the clinical outcome assessment tools [modified Rankin Scale (mRS), Barthel Index, and NIHSS] of their recordings, to correlate the usefulness and accuracy with the baseline important variables. In this project, we hypothesize that the change of clinical CKD staging as delta stage (3-month follow-up CKD staging – admission CKD staging) could serve as a crucial altering point in terms of long-term outcome prediction. Through this project, the authors hope to provide first-line clinicians with important information that can be crucial both in the therapy decision-making process and in the moment in which they will be required to create a dialogue with the patient and their families.
Materials and methods
Patient selection
Based on the clinical experience of the hospital that the 1-year mortality of ischemic stroke patients with impaired renal function was nearly 40% and the ratio of r-tPA to non-r-tPA treatment was about 1:5, the study was designed to recruit 130 r-tPA and 650 non-r-tPA patients, respectively, to reach a desired power of 0.8 and a maximal type I error rate of 0.05 if odds ratio (OR) between the treatment groups is >1.7. In this investigation, the authors retrospectively studied the data of 765 middle cerebral artery ischemic stroke patients with impaired renal function from the medical records of Changhua Christian Hospital (CCH) between January 2001 and January 2018. The cases were collected following specific inclusion and exclusion criteria. All the patients were enrolled through triage of emergency department (ED) and evaluated by the stroke neurologist to be considered a thrombolytic treatment candidate. After the benefits and harms were weighed, a thrombolytic treatment (rt-PA) was prescribed if the condition allowed and if the patient was within the golden time of 4.5 h of stroke onset. The participants were subsequently admitted to the intensive care unit and neurological ward for advanced treatment and discharged for outpatient follow-ups for up to more than 1 year. The ultimate number of 765 stroke patients was triaged and collected at the ED, with 116 patients receiving intravenous thrombolytic therapy, while 649 patients were not suitable for the treatment (Figure 1).

Study profile. r-tPA and mortality prediction.
All patients included in the study have met the following pre-defined inclusion and exclusion criteria: (1) aged between 20 and 90 years, with evidence of middle cerebral artery ischemic stroke at the index event and evaluated by cerebral computed tomography (CT), angiography (CTA), and CT perfusion (P-CT); (2) no observation of recurrent cerebral or other vascular events during the study period; (3) patient with no severe carotid stenosis/vertebral stenosis ever receiving carotid endarterectomy or carotid stenting; (4) completion of at least 12 months of follow-up after treatment. On the other hand, the exclusion criteria were: (1) NIHSS score <4 or >25 points at the initial evaluation; (2) patients receiving long-term anticoagulant therapy before the index event; (3) patients with cerebral bleeding based on the cerebral tomography scan; (4) patients with cerebral arteriovenous malformations or aneurysms; (5) recurrent stroke during the study period; (6) patients demonstrating systemic vascular events or recurrent cerebral vascular episodes. The impaired renal function is defined as a decline in estimated glomerular filtration rate (eGFR) of greater than 30% (eGFR <60 ml/min per 1.73 m2). The eGFR was calculated based on MDRD (modification of diet in renal disease) equation as follows:
According to the Kidney Disease: Improving Global Outcomes (KDIGO) definition and classification, in people with GFR <60 ml/min/1.73 m2 (GFR categories G3a–G5) over 3 months, CKD is confirmed10,11 (GFR 59–30 ml/min/1.73 m2: stage 3; GFR 29–15 ml/min/1.73 m2: stage 4; GFR <15 ml/min/1.73 m2: stage 4; GFR 89–60 ml/min/1.73 m2: stage 2; GFR ⩾90 ml/min/1.73 m2: stage 1 with proteinuria or hematuria). The CKD staging was assessed on admission to the ward and reassessed again at 3-month follow-up at the outpatient clinic settings. The change of staging, denoted as delta stage, was taken as the follow-up GFR stage minus the admission GFR stage. Consequently, delta stage with value ⩽0 indicates no deprivation on GFR stage.
Baseline biochemistry and neuro-radiological examinations were carried out in the emergency room and neurological ward. The comorbidities, neurological and physical examinations, and relevant previous drugs and personal history were carefully recorded during the hospitalization period. The carotid duplex examination was conducted within 3 days of initial admission to the hospital. All the parameters of the carotid ultrasound were documented. The clinical information collected was cross-checked to ensure consistency between the original (paper) medical records and the electronic information of the CCH 2000 computer-based medical record system network. The study procedures were approved by the Institutional Review Board of the CCH in Changhua, Taiwan [refer to institutional review board (IRB) certificate, CCH IRB number: 180511]. The informed consent was waived due to the nature of retrospective study and approved by the IRB committee.
Intravenous thrombolytic therapy
All patients were evaluated for adequacy of thrombolytic therapy (r-tPA) at the ED by the in-charge physician. (The window of time for the rt-PA was set at ⩽4.5 h from symptom onset.) A thorough evaluation of every single patient was carried out before the administration of r-tPA, including neuroimaging, and the patients’ stroke severity was recorded using the NIHSS scoring method. Shortly after the r-tPA, the patients’ baseline specific aspects were documented, together with the demographic information, NIHSS score, Barthel index, mRS, and blood biochemistry. The neurological outcome assessment was recorded via NIHSS score, Barthel index, and mRS upon arrival of triage of ED. The abovementioned assessment recordings were documented 3, 6, and up to 12 months in the outpatient clinic settings. For each assessment, recordings were documented by both the case manager and the clinician in charge to avoid any discrepancies. If the two recordings were too large, a third party, usually another neurologist or neuroradiologist, was invited to confirm the measures. The NIHSS score is specifically used for measuring the degree of neurological defects (with ⩽3 defined as mild and ⩾25 defined as a severe ischemic stroke episode). The mRS is used for assessing general daily life functionality and independence (ranging from 0 to 6, with ⩽2 defined as independence and 3–6 defined as having neurological defects and with dependency on other people). Similarly, the Barthel index is used for assessing general daily independence using more detailed items (ranging from 0 to 100 points, with ⩾60 points indicating favorable life mobility and ⩾95 denoting normal or near-normal life mobility).
Statistical analysis
Statistical analyses were performed using the statistical package SPSS for Windows (Version 16.0, SPSS Inc; Chicago, IL, USA). Pre- and post-scores were compared to determine patient outcomes. The baseline clinical data are used to correlate 1-year functional outcomes (as assessed using the NIHSS score, mRS, and Barthel index) on index episode after the stroke event. Statistical comparisons between groups were conducted using Pearson’s chi-square test and Mann–Whitney U test for categorical and continuous variables, respectively. Univariate and multivariate logistic regression was employed to assess the significance of risk factors and to obtain ORs. A p value <0.050 is considered statistically significant to seek for predictive factors determining the long-term outcomes. Receiver operating characteristic (ROC) curve was applied to identify the variable with the best diagnostic power. The Youden index was used to determine the optimal cut-off value.
Propensity score matching method was conducted using matchit library in R package to reduce the unbalance. The propensity score was estimated by fitting logistic regression of r-tPA status on the above significant variables: p (tPA = 1 | gender, diuretics), and then each patient was matched with the corresponding group by a greedy algorithm.
The decision tree was processed with the Chi-square Automatic Interaction Detection (CHAID) system for detecting the interaction between variables. As CHAID can reveal the most critical risk factors in both groups, it could support clinicians’ decisions of whether patients were suitable for stenting. CHAID applied a pre-pruning strategy; a node was split only if a significant criterion was satisfied, thus preventing overfitting. CHAID was selected as the decision tree due to the criticality of interpretation and tree construction that would have helped to avoid overfitting.
Results
Table 1 shows the baseline characteristics between the intervention group (received r-tPA) and control (non-r-tPA group). Upon analyzing the baseline variables with ultimate mortality, the discharged GFR and admission GFR are significantly associated with r-tPA. The following explanatory characteristics also have relevant p values: gender, age, platelet, natrium, length of hospital stay, diabetes mellitus (DM) type 2, essential hypertension, mixed-type hyperlipidemia, coronary artery disease, chronic heart failure, underlying cancer etiology, chronic obstructive pulmonary disease (COPD), gout, use of angiotensin-converting enzyme inhibitor/angiotensin receptor blocker (ACEI/ARB), and diuretics, possible confounders that may cause the unbalanced analysis between r-tPA and non-r-tPA groups. To improve the situation, a propensity score matching method was applied, conducted using matchit library in R package to reduce the unbalance between r-tPA and non-r-tPA groups. First, the propensity score was estimated by fitting the logistic regression of r-tPA status on the above significant variables: p (r-tPA = 1 | gender, diuretics); therefore, each patient in the r-tPA group was matched with two to three non-r-tPA patients by a greedy algorithm.
Baseline characteristics between the control and intervention groups.

ACEI, angiotensin-converting enzyme inhibitor; ARB, angiotensin receptor blocker; CAD, coronary artery disease; CHF, congestive heart failure; CKD, chronic kidney disease; COPD, chronic obstructive pulmonary disease; delta GFR, discharged GFR – admission GFR; DM, diabetes mellitus; GFR, glomerular filtration rate; GFR slope, delta GFR / admission GFR; GPT/ALT, alanine aminotransferase; Gout, gouty arthritis; Hb, hemoglobin; Hct, hematocrit; WBC, white blood cells; r-tPA, recombinant tPA; tPA, tissue plasminogen activator.
p value by Mann–Whitney U test.
Table 2 shows the characteristics between r-tPA and non-r-tPA groups after propensity matching, where the variables used in propensity have become less or insignificant. (Figure 2) The propensity-matched OR of mortality for the r-tPA group compared with the non-r-tPA group is 0.469, with p = 0.003, indicating that the r-tPA group has better prognosis. To identify factors that influence mortality, separate prediction models for r-tPA and non-r-tPA groups were established by multiple regression analysis in Table 3. For the non-r-tPA group, age, hematocrit (Hct), DM type 2, coronary artery disease, and delta stage are significantly associated with mortality (p < 0.05). On the other hand, for the r-tPA group, age, DM type 2, chronic heart failure, hospital day, and delta stage are the significant factors (p < 0.05). These models elucidate how delta stage can be a powerful prediction variable for both groups.
Baseline characteristics between the control and intervention groups after propensity matching.
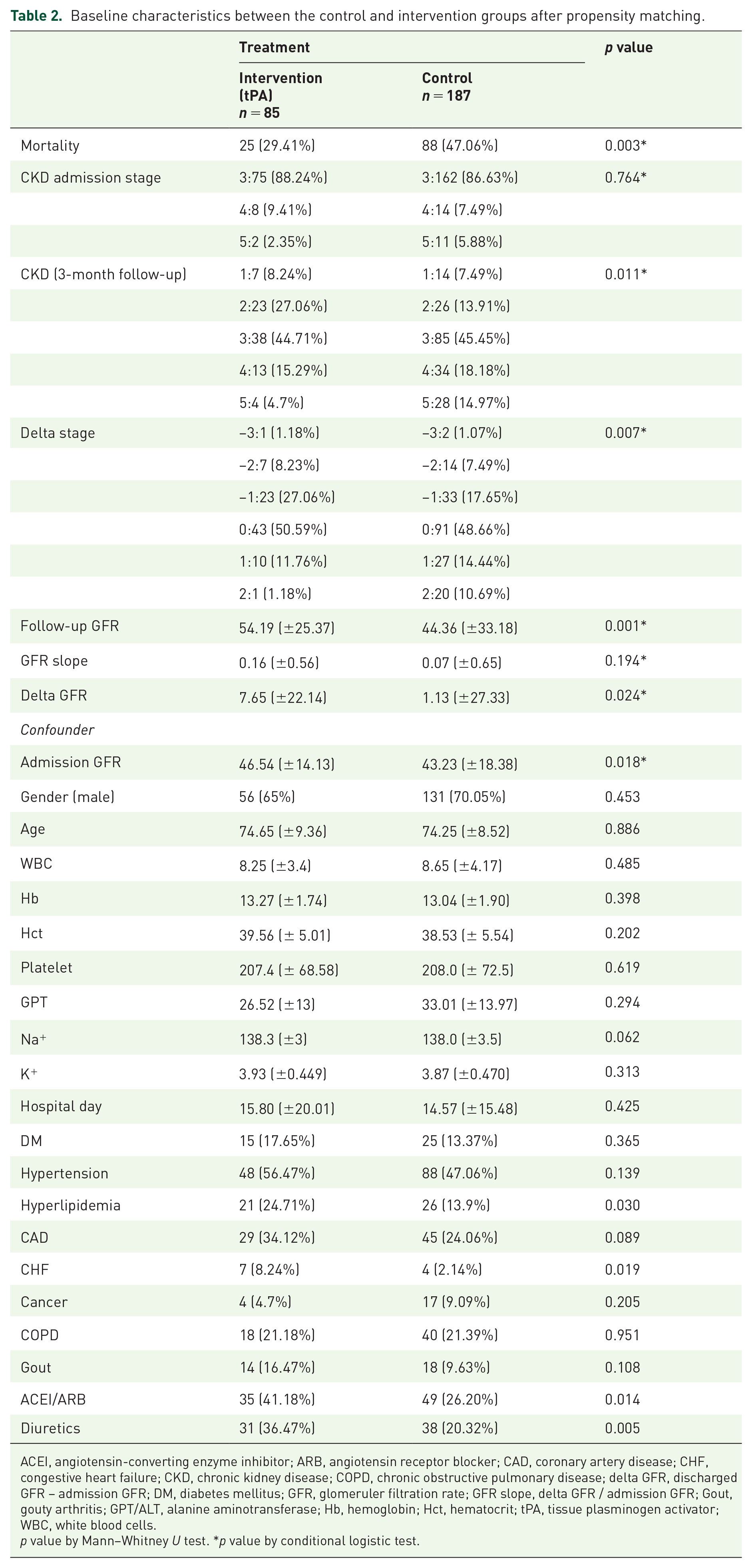
ACEI, angiotensin-converting enzyme inhibitor; ARB, angiotensin receptor blocker; CAD, coronary artery disease; CHF, congestive heart failure; CKD, chronic kidney disease; COPD, chronic obstructive pulmonary disease; delta GFR, discharged GFR – admission GFR; DM, diabetes mellitus; GFR, glomeruler filtration rate; GFR slope, delta GFR / admission GFR; Gout, gouty arthritis; GPT/ALT, alanine aminotransferase; Hb, hemoglobin; Hct, hematocrit; tPA, tissue plasminogen activator; WBC, white blood cells.
p value by Mann–Whitney U test. *p value by conditional logistic test.
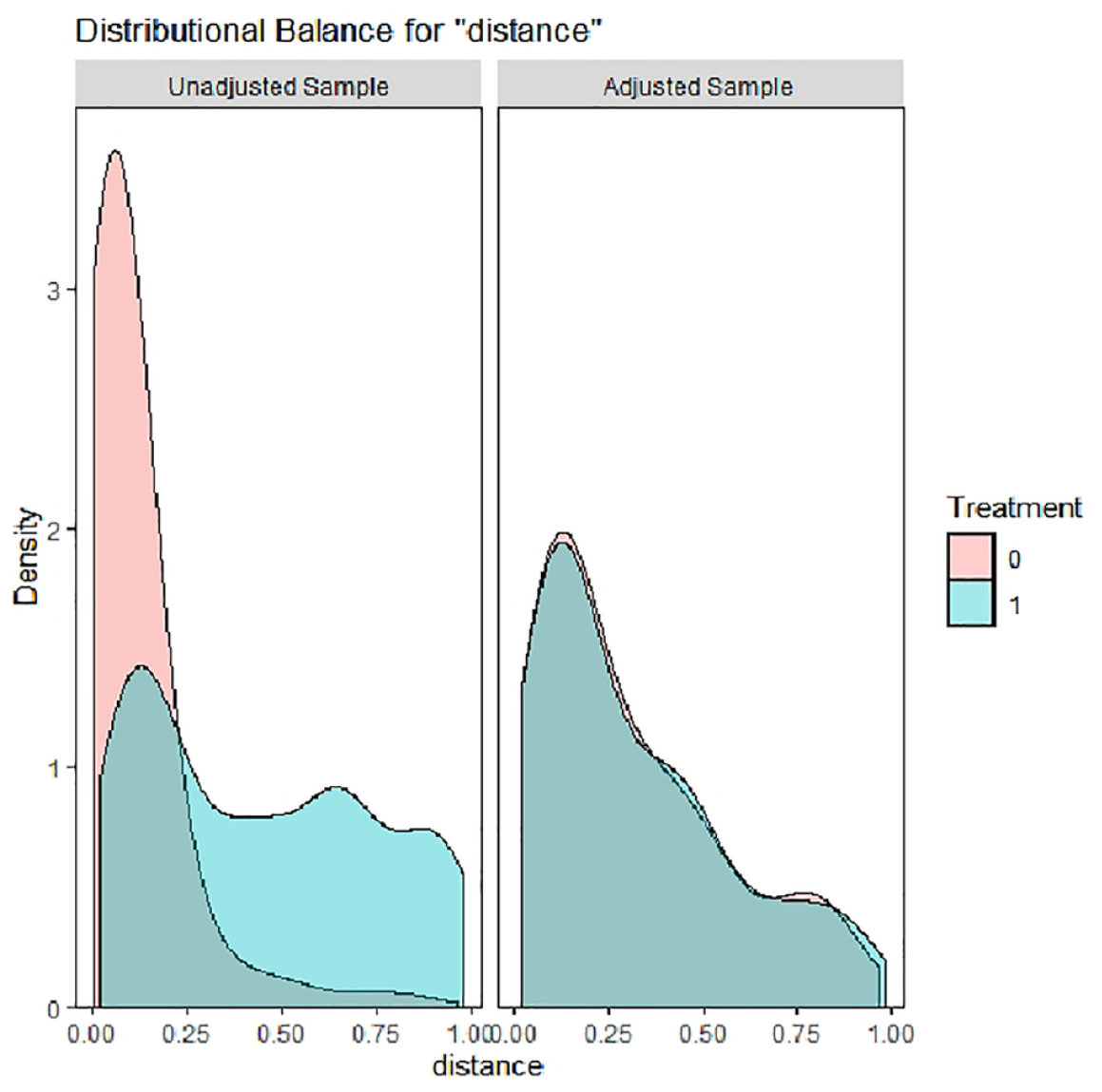
Distribution of propensity score between tPA and non-tPA groups before and after matching.
Significant factors for predicting the 1-year mortality for tPA and non-tPA groups by logistic regression.

CAD, coronary artery disease; CHF, congestive heart failure; CI, confidence interval; delta stage, discharged GFR stage – admission GFR stage; DM, diabetes mellitus; Hct, hematocrit; OR: odds ratio; tPA, tissue plasminogen activator.
The use of ACEI/ARB or diuretics and long-term patient mortality is analyzed in Table 4. It shows they are not statistically significant with ACEI/ARB (p = 0.198), whereas for diuretic, the OR is 1.619 with a p value of 0.025, which indicates higher mortality after long-term administration.
Results of multiple logistic regression analysis of ACEI/ARB and diuretics for the prediction of life situation at 1 year.

ACEI, angiotensin-converting enzyme inhibitor; ARB, angiotensin receptor blocker; CI, confidence interval.
The authors further analyze the datasets with two classifiers, stepwise logistic regression (LR) and decision tree (DT), to explore the significant factors for predicting the 1-year mortality for r-tPA and non-r-tPA groups separately. The results of LR indicates that age, DM type 2, GFR delta stage, and cardiac diseases [congestive heart failure (CHF) or coronary artery disease [CAD]) are significant common factors for both groups. Among them, DM type 2 has an OR of 3.2 and 1.94 for r-tPA and non-r-tPA, respectively, and the worsening of the GFR by one stage will increase the 1-year mortality risk by OR of 1.92 and 1.76 for tPA and non-r-tPA, respectively.
Figures 3 and 4 list the tree structures from DT classifier for non-r-tPA and r-tPA groups. On the top of both trees, delta stage is the most important predictor of the 1-year mortality for non-r-tPA and r-tPA groups. For the r-tPA group, delta stage is a single DT predictor; in fact, patients with delta stage ⩽0 had a 24% mortality rate after 1 year, while that for delta stage >0 it is 75%. For non-r-tPA patients, delta stage, Hct, and age are three important DT predictors, which were similar to the LR predictors. Non-r-tPA patients had mortalities of 30.9% and 66.3% for those with delta stage ⩽0 and >0 respectively.

Tree structure of DT classifier for non-tPA, indicating delta stage as the most important predictor for the survival rate of patients.

Tree structure of DT classifier for tPA, with delta stage as the most important predictor for the survival rate of patients.
By taking the difference of functional assessments at admission and 1-year follow-ups for NIHSS, mRS, and Barthel index, respectively, the authors defined the binary outcomes as value 1 with improvement and as 0 without improvement. Delta stage is to be correlated with the abovementioned three assessment tools. Figure 5 shows the ROC plots on the prediction of improvement of neurological recovery assessment. For NIHSS (a), AUC = 0.83 and optimal criterion with sensitivity = 77.8%, specificity = 80.6%, and cross-validation total accuracy = 79.04%. For mRS (b), AUC = 0.835 and optimal criterion with sensitivity = 84.4%, specificity = 42.5%, and cross-validation total accuracy = 79.02%. For Barthel Index (c), AUC = 0.663 and optimal criterion with sensitivity = 90.2%, specificity = 42.5%, and cross-validation total accuracy = 66.64%.

ROC plot on the prediction of improvement of neurologic disability assessments: (a) NIHSS, with AUC = 0.83 and optimal criterion with sensitivity = 77.8%, specificity = 80.6%, and cross-validation total accuracy = 79.04%; (b) mRS, with AUC = 0.835 and optimal criterion with sensitivity = 84.4%, specificity = 42.5%, and cross-validation total accuracy = 79.02%; and (c) Barthel index, with AUC = 0.663 and optimal criterion with sensitivity = 90.2%, specificity = 42.5%, and cross-validation total accuracy = 66.64%.
Discussion
Middle cerebral artery ischemic stroke requires a prompt management, as the rapid cerebral edematous change could potentially hamper the patient’s chance of surviving. Thrombolytic therapy (r-tPA) remains the gold standard treatment of choice when the first line in charge, physician, encounters this kind of situation. However, there is a limited point window (NIHSS: 4–25) that is suitable for qualified r-tPA candidates. Even if r-tPA has been administered, there is still a 6% chance of cerebral hemorrhage, leading to poor long-term outcomes. On top of that, a poor general medical condition (for instance, poor renal function) might further complicate the status of a patient with index middle cerebral artery ischemic stroke.12,13 Based on the thrombolytic treatment inclusion and exclusion criteria published by the AHA/ASA guidelines,1,2 it is stated that patients with poor general medical condition, after the family being well explained, are not eligible for thrombolytic treatment. In addition, patients with poor renal function are not specified, being one of the items listed in the exclusion criteria. The authors believe this research project is a groundbreaking study and the finding of delta stage in the current study can provide first-line doctors with concrete and valuable information, fundamental for their decision-making in treating middle cerebral artery ischemic stroke patients.
CKD generates vascular structure and functional changes, with significant hemodynamic effects, favoring the progression of arterial stiffness. 14 Furthermore, it is well known that arterial stiffness represents an independent risk factor for ischemic stroke, especially in patients with ischemic stroke from large vessel occlusion 15 and also favoring different complications after thrombolysis and influencing the outcome. 16
According to the current study, r-tPA can effectively reduce the overall mortality up to 1 year after the treatment, as well as improve the long-term physical functionality. It is a general belief among doctors to avoiding the administration of r-tPA because, in the presence of already complicated medical conditions, it could increase cerebral bleeding and jeopardize the general medical situation. Surprisingly, the survival rate of patients with r-tPA treatment was 1.5 times greater than those without r-tPA intervention. The NEFRONA study 17 prospectively confirmed a higher prevalence of atherosclerosis and contribution of CKD to subclinical atherosclerosis in patients with moderate CKD. Progression of CKD correlates to progression of atherosclerosis as well. In CKD, the emerging risk factors involved in atherosclerosis include dysregulation of calcium and phosphate metabolism, vascular calcification, inflammation, oxidative stress, oxidized lipid, and endothelial dysfunction. 18
This phenomenon might be determined by the fact that the intravenous route of r-tPA to the major renal artery could possibly canalize the possible atheroma, hence increasing the general renal function and toxin excretion.19,20 Moreover, r-tPA might be theoretically more potent and target the obvious thrombus in the major cerebral artery, leading to a thrombolytic effect and achieving recanalization despite cerebral bleeding occurring case by case. In this study, delta GFR stands out as the most relevant outcome predictor for these groups of victims, compared with other indicators of admission GFR, 3-month follow-up GFR, and slope of GFR. It provides clinicians with clear and simple clinical information regarding the long-term patients’ outcomes. Moreover, DM, if not properly managed, plays a crucial role in determining the subjects’ long-term chance of mortality in both r-tPA and non-r-tPA groups, with OR of roughly 2 and 3 times the death rate.
Multiple studies have shown the association between renal function and post-ischemic stroke outcomes. Patients with reduced GFR and increased serum creatinine and proteinuria are linked with increased risk of neurologic deterioration, functional outcome, or mortality.20,21 A large-scale study using the Taiwan Stroke Registry also found GFR on admission has an elevated risk of mortality in 1-month and 1-year follow-up.22,23 In a recent meta-analysis, moderate-to-severe CKD is associated with worse functional outcomes in 3 months. 24 Although the general trend is consistent, these studies utilized different equations for the calculation of GFR. 25 The authors decided to use, instead of GFR on admission, the delta GFR or CKD stage for the evaluation of association with functional outcomes and mortality.
It would be interesting to see whether antihypertensive drugs can have a long-term impact on these groups of people. The authors find that diuretics might worsen the general outcome, with an OR of 2 times of chances leading to mortality up to 1-year follow-up. The possible explanation for this might be that for poor renal outcome patients, diuretic could reduce the extracellular fluid aggregation that clinically manifests as lower limb edema. Misuse of this type of drug could cause imbalanced human body electrolytes, potentially leading to cardiac arrhythmia of sudden death if not properly used. By contrast, ACEI/ARB shows no statistically significant values (p < 0.05), indicating that this type of drug is less harmful and in accordance with the current stroke AHA/ASA guidelines. Despite this, careful usage is still needed, and individualized treatment should be arranged.
In addition, as we collect patients with r-tPA and non-r-tPA, intra-arterial thromboectomy (IA) procedures have not been included, and for bridging therapy (IA ensued after intravenous thrombolytic therapy) patients, the outcome is still unknown. Though not collecting IA or bridging therapy patients, based on the research datasets, the general survivorship after r-tPA is roughly 70–80% (20–30% of death rate followed up to 1 year). Compared with other published literature, the death toll is fair. 26
The data of this study were collected and crosschecked with CCH computer-based 2000 system to ensure the dataset’s accuracy. Furthermore, the collected datasets were analyzed by the same specialist to avoid treatment methodology–associated variability. Moreover, all the participants were followed up for more than 1 year, and the outcome assessment tools were conducted by the same case manager to align with the dataset’s accuracy and consistency. The weakness of this study lies in its limited extent and reduced temporary duration of merely 1 year after thrombolytic treatment therapy, which is retrospective in nature. Additional prospective and clinical trial studies are needed to validate the current findings. All the collected cases belong to the same ethnicity; therefore, the findings are suggested to be applied to the same ethnic background. General external application should be interpreted carefully. The study focused only on stroke patients with evident large-artery stenosis; for those with other types of stroke, the results of the current project could not applied and required further investigation. Furthermore, IA procedure was not included in the current project, and the bridging effect and outcome assessment of this group of patients could not be obtained. In addition, pretreated patients’ general health status and post-thrombolytic treatments such as physical rehabilitation and antiplatelet therapy (aspirin or clopidogrel) might potentially confound the functional outcomes measured by the NIHSS, Barthel Index and mRS; the results of the current investigation should be interpreted carefully.
Conclusion
Thrombolytic treatment has a great capacity to reduce middle cerebral artery ischemic stroke patients’ mortality, even when presenting with underlying impaired renal function. Change of CKD stage (delta stage) can serve as an important predictor of mortality for both the r-tPA and non-r-tPA patients. Furthermore, long-term use of diuretics could be potentially harmful to the stroke patient with underlying impaired renal functionality. Finally, delta stage correlates well with clinical neurological functionality assessment (NIHSS, Barthel Index and mRS) and is capable of serving as a crucial surrogate to early predict patients’ long-term outcome as well as aid clinical decision-making
Footnotes
Acknowledgements
Our team would like to thank Miss Giulia Mengato for manuscript proofreading.
Author’s note
Ping-Fang Chi is also affiliated to Department of Post Baccalaureate Medicine, College of Medicine, National Chung Hsing University, Taichung, Taiwan; Department of Hospitality Management, MingDao University, Changhua, Taiwan.

