Abstract
Background:
Hypertension-mediated organ damage (HMOD) is an emerging problem among young adults. The potential role of chronic immune-mediated inflammation in the pathogenesis of HMOD is increasingly being recognized. High-mobility group box 2 (HMGB2) is known for its role in the modulation of innate immunity and exerts signaling functions that affect various inflammatory diseases. However, the association between HMGB2 and HMOD in young adults remains unclear.
Objectives:
The aim of this study was to explore the association between HMGB2 and subclinical HMOD in young adults.
Design:
This is a cross-sectional study.
Methods:
Body composition, carotid ultrasound, carotid-femoral PWV (cf-PWV) measures, echocardiography, serum HMGB2 levels, and serum classic cardiometabolic risk factors were measured in 988 untreated young adults. We estimated the risk related to serum HMGB2 using multivariable-adjusted linear and logistic regression models. Then, we conducted a pathway overrepresentation analysis to examine which key biological pathways may be linked to serum HMGB2 in young adults with HMOD.
Results:
Among the 988 untreated young adults, we identified four distinct hypertension phenotypes: normotension (40.0%), white-coat hypertension (16.0%), masked hypertension (20.9%), and sustained hypertension (23.1%). High levels of serum HMGB2 were related to increased carotid intima-media thickness (cIMT) and left ventricular mass index (LVMI), higher cf-PWV and blood pressure, and a lower estimated glomerular filtration rate (eGFR). Linear regression analysis showed that serum HMGB2 was positively associated with cf-PWV and negatively associated with eGFR in all patients. Multivariate analysis showed that high levels of serum HMGB2 were associated with high odds of subclinical HMOD (damage in at least one organ). Biological pathway analysis indicated that patients with high serum HMGB2 levels had increased activity of pathways, related to endothelial dysfunction, inflammatory processes, and atherosclerosis.
Conclusion:
High serum concentrations of HMGB2 are associated with an increased risk of subclinical HMOD in untreated young adults.
Keywords
Introduction
Hypertension affects up to one in three young adults during their lifetime, and most of these individuals are unaware of their condition.1,2 Hypertension in young adults has attracted increasing concern, as it can increase their risk of cardiometabolic disease and stroke.3,4 Even though hypertension-mediated organ damage (HMOD) commonly appears clinically at midlife and later, its onset can occur in young adulthood.5–7 Subtle HMOD, such as left ventricular hypertrophy (LVH), microalbuminuria, cognitive dysfunction, and thickening of the carotid vessel wall, is detectable early in the development of hypertensive cardiovascular disease (CVD). 8 However, most previous studies of hypertension have focused on older individuals (>50 years),9–11 and few studies have focused on younger individuals with hypertension. Young individuals with hypertension may have distinct characteristics, and the pathophysiologic characteristics of hypertension may differ between older and younger individuals.2–4 Chronic immune-mediated inflammation is increasingly recognized as an important contributor to HMOD. Thus, the early exploration for hypertension-related inflammatory biomarkers in young individuals may provide a more sensitive and adequate measure for identifying early cardiovascular risk, especially for those individuals who are asymptomatic.
High-mobility group box 2 (HMGB2) is a member of the high-mobility group box (HMGB) protein family (HMGB1−4) and is widely distributed in eukaryotic cells. 12 HMGBs are known for their role in the modulation of innate immunity and exert signaling functions that affect various inflammatory diseases. 13 HMGB2 regulates various cellular activities, including transcription, DNA replication, and repair.12,14 Low-grade inflammation is increasingly recognized as a robust mechanism underlying myocardial ischemia (MI) injury.15,16 Among the four HMGB members, HMGB2 is mainly related to arterial injury incurred in the pathophysiology of CVDs, leading to atherosclerosis, MI, heart failure development, and intimal hyperplasia.14,17–19 Recent publications suggest that HMGB2 is associated with MI severity and major adverse cardiac events (MACEs) at 1 month in rats.14,17 Moreover, HMGB2 increases arterial wire injury via reactive oxygen species (ROS) activation, which promotes neointimal hyperplasia in mice. 14 Since most data originate from experimental models, the relationship between serum HMGB2 and subclinical HMOD in young adults remains elusive.
In this study, we aimed to study the relationship between serum HMGB2 and subclinical HMOD in young adults. We further investigated the mediating roles of the pathophysiological mechanisms related to HMGB2 in these associations.
Methods
Study population
This study was a single-center and cross-sectional observational study. Between October 2018 and January 2022, 2,805 consecutive participants (aged 18–45 years) who underwent a national health examination were recruited through an active recruitment strategy at the First Affiliated Hospital of Chengdu Medical College. Based on previous studies, the sample size was calculated with an alpha level of 5% and a power of 80%.20,21 We obtained a minimum sample size of 526 patients. The inclusion criteria for the study were as follows: aged 18–45 years, underwent the usual clinical and laboratory workup, and were willing to sign the informed consent. Exclusion criteria included subjects with long-standing diagnosed but untreated hypertension, secondary hypertension (e.g. primary aldosteronism), antihypertensive drug use, use of lipid-lowering and antiplatelet drugs, severe renal insufficiency or hepatic insufficiency, congenital heart disease, valvular heart disease, cardiomyopathy, previous coronary artery bypass grafting or coronary stent implantation, autoimmune diseases, serious infectious diseases, and subjects with missing laboratory or health examination data. The study was approved by the Ethics Committee of the First Affiliated Hospital of Chengdu Medical College (No: 2020CYFYIRB-BA-97), and participants provided written and informed consent. This study was conducted according to the principles of the Declaration of Helsinki. The exclusion criteria are detailed in Figure 1.

Flowchart showing the enrollment protocol.
Clinical evaluation
At baseline, all participants completed standardized questionnaires6,22,23 (including medical history, medication use, and cardiovascular risk assessment, and this information can be found in the Supplementary material online) and underwent clinical examinations (demographics and laboratory results). The blood pressure (BP) measurement protocol suggested at least 5 min of rest in a seated position followed by two repeated measurements at 5-min intervals.4,24 Clinic BP measurements were consecutively performed 3 times at clinic visits by trained medical staff using an HEM-1000 BP monitor (Omron Healthcare). Ambulatory BP monitoring was measured using the same type of ambulatory BP monitor (ABPM 6100; Welch Allyn) for each participant. According to the European Hypertension Practice Guide 2018, 6 the threshold for an elevated clinic BP is ⩾140/90 mmHg, and the mean ambulatory BP is ⩾135/85 mmHg in the daytime, ⩾120/70 mmHg at night, and ⩾130/80 mmHg throughout the whole day. Based on the results of the clinic BP and ambulatory BP monitoring measurements, four hypertension groups were defined as follows:21,25–27 normotension (NT), individuals with normal clinic and ambulatory BPs; white-coat hypertension (WCHT), individuals with elevated clinic BPs, and normal ambulatory BPs; masked hypertension (MHT), individuals with normal clinic BPs, and elevated ambulatory BPs; and sustained hypertension (SHT), individuals with elevated clinic and ambulatory BPs. This information is shown in Figure 1.
ELISA quantification of serum HMGB2
Blood samples were collected at field sites using standard protocols.24,28 The level of HMGB2 in serum samples was measured by an HMGB2-ELISA kit (CSB-EL010560MO, CUSABIO Life Science) according to the manufacturer’s guidelines. 12
Evaluation of subclinical organ damage
Carotid-femoral pulse wave velocity (cf-PWV) is considered the gold standard for arterial stiffness assessment in clinical practice.29–31 cf-PWV was quantified automatically on an applanation tonometry (SphygmoCor, AtCor Medical, Sydney, Australia) device, and three consecutive measurements were performed on each occasion after the participants rested for at least 5 min. Direct carotid–femoral distance was corrected by a factor of 0.8, and the mean value was used, as recommended.29,31,32 Bilateral carotid arteries and echocardiograms were both measured artificially with a Vivid-7 Pro Ultrasound machine (GE Healthcare, USA). An echo-tracking device based on a radiofrequency signal permits us to obtain the presence of atheromatic plaques in the common carotid artery (CCA), the carotid sinus, and the internal carotid artery (ICA).33–35 The probe was first placed on the patient’s left and right upper concave of the clavicle.33–35 Then, it was moved from the proximal end of the CCA to the carotid bifurcation and ICA in sequence in the longitudinal axis views, ensuring that the segment from the CCA 3 cm proximal to the bifurcation to the ICA 1 cm distal to the bifurcation was fully scanned.33–35 The intima-media thickness (IMT) at the anterior and posterior walls of the bilateral carotid arteries 1 and 2 cm proximal to the carotid bifurcation in the CCA and 1 cm distal to the carotid bifurcation in the ICA in the long-axis view of the carotid artery were measured.33–35 The mean value of the 12 IMT measurements in each individual was calculated as carotid IMT (cIMT). 36 Screening for LVH was performed as per the guidelines.33,34 Taken together, all of the above measurements followed a standardized protocol33,34,37,38 and were obtained by an experienced and certified sonographer. According to the current guidelines,6,39–41 subclinical HMOD is defined as echocardiographic evidence of LVH [LVH was defined as left ventricular mass index (LVMI) > 115 g/m2 for men and > 95 g/m2 for women], 33 ultrasound evidence of carotid artery wall thickening (common cIMT > 0.9 mm) or plaques (a plaque was defined as a focal thickening ⩾ 1.5 mm in any segment of the carotid arteries), 33 increased arterial stiffness (cf-PWV ⩾ 10 m/s),38,39 and microalbuminuria [a urinary albumin/creatinine ratio (UACR) ⩾ 2.5 mg/mmol in men or ⩾ 3.5 mg/mol in women] or early-stage renal damage [estimated glomerular filtration rate (eGFR): 30–60 mL/min/1.73 m2]. 40
Pathway overrepresentation analyses
We performed overrepresentation with ClueGO14 (in Gene Ontology biological processes, KEGG and Reactome pathways) using the hypergeometric test and the default Bonferroni step down method for multiple testing corrections (familywise error rate). 42 We performed the whole annotation option as a reference set and reported only biological processes with a corrected P value ⩽ 0.05 as significant.
Statistical analysis
The sample size was calculated using G*Power 3.1 software. Clinical data are summarized as percentages for categorical variables and as the mean ± standard deviation (SD) for continuous variables. Comparisons between all variables from multiple groups were assessed by the chi-square test, the Kruskal–Wallis test, or one-way analysis of variance (ANOVA). Pearson correlations were calculated and linear regression analyses were performed. In addition, the independent association between serum HMGB2 and subclinical HMOD of generalized hypertension phenotypes, adjusted for the identified confounders, was further assessed by multivariate logistic regression analysis. Statistical analyses were performed using SPSS software version 23.0 (SPSS, Inc., Chicago, Illinois, USA), STATA (version 12.0), and Origin 9.0 (OriginLab, USA). Statistical significance was assigned at p < 0.05.
Results
Baseline Characteristics
The characteristics of the study population are displayed in Table 1. From 2018 to 2022, 40.0% (n = 395) of individuals were determined to have a ‘NT phenotype’, 16.0% (n = 158) of individuals had a ‘WCHT phenotype’, 20.9% (n = 207) of individuals had a ‘MHT phenotype’, and 23.1% (n = 228) of individuals had a ‘SHT phenotype’. Compared with the other three hypertension phenotypes, individuals with the SHT pattern had higher body mass index (BMI), waist-to-hip ratio (WHR), heart rate, systolic and diastolic BP, total cholesterol (TC), non-high-density-lipoprotein-cholesterol (non-HDL-C), uric acid, cf-PWV, cIMT, LVMI, and a lower eGFR. They also had a higher prevalence of family history of hypertension, obstructive sleep apnea–hypopnea syndrome (OSHAS), abdominal obesity, and diabetes mellitus. Significant changes in the levels of serum HMGB2 are depicted in Figure 2. The prevalence of HMOD (0, 1, or ⩾2 damaged organs) in subgroups by four quartiles of serum HMGB2 (Q1–Q4 quartile) is shown in Figure 3. Higher levels of serum HMGB2 were related to increased HMOD numbers.
General characteristics of participants stratified into four hypertension phenotypes.

BMI, body mass index; cf-PWV, carotid-femoral pulse wave velocity; cIMT, carotid intima-media thickness; CRP, C-reactive protein; DBP, diastolic blood pressure; eGFR, estimated glomerular filtration rate; HDL-C, high density lipoprotein-cholesterol; HMOD, hypertension-mediated organ damage; LDL-C, low density lipoprotein-cholesterol; LVH, left ventricular hypertrophy; LVMI, left ventricular mass index; MHT, masked hypertension; NT, normotension; OSAHS, obstructive sleep apnea–hypopnea syndrome; SBP, systolic blood pressure; SHT, sustained hypertension; TC, total cholesterol; TG, triglycerides; UACR, urine albumin-to-creatinine ratio; WCHT, white-coat hypertension; WHR, waist-to-hip ratio.
Values are M ± SD or n (%).
A value <0.05 was considered statistically significant.
p < 0.05, the WCHT group versus the NT group; #p < 0.05, the MHT group versus the NT group; △p < 0.05, the SHT group versus the NT group.

The value of serum HMGB2 in different hypertension phenotypes.

Proportion of individuals with 0, 1, or ⩾ 2 types of HMOD by four quartiles of serum HMGB2 (Q1–Q4 quartile).
Analysis of associations
Regarding subclinical HMOD indices, the Pearson correlation analyses using scatter plots are illustrated in Figure 4. In all patients, serum HMGB2 was positively correlated with cf-PWV [r = 0.376; p < 0.001; Figure 4(a)], cIMT [r = 0.361; p < 0.001; Figure 4(b)], and LVMI [r = 0.494; p < 0.001; Figure 4(c)], and it was negatively correlated with eGFR [r = −0.469; p < 0.001; Figure 4(d)]. Results of serum HMGB2 and other variables are shown in Table 2. No correlation was observed when the analysis was performed separately for the different hypertension phenotypes.

Scatter plots were used to illustrate the association between serum HMGB2 and subclinical HMOD indices (a–d) in young adults.
The correlation between serum HMGB2 level and cardiovascular risk factors.

BMI, body mass index; CRP, C-reactive protein; HDL-C, high density lipoprotein-cholesterol; LDL-C, low density lipoprotein-cholesterol; OSAHS, obstructive sleep apnea–hypopnea syndrome; WHR, waist-to-hip ratio.
Pearson correlation analysis was used to investigate the correlation between serum HMGB2 and cardiovascular risk factors. Correlation coefficients and the corresponding p values were shown in the table.
A p value of <0.05 was considered statistically significant.
In all patients, the unadjusted linear regression illustrated that subclinical HMOD parameters, including cf-PWV, LVMI, cIMT, and eGFR were significantly associated with serum HMGB2 levels. After adjusting for cardiovascular risk factors, only cf-PWV (β = 0.043) and eGFR (β = −0.428) remained significant. The results are shown in Table 3. No association between subclinical HMOD parameters and serum HMGB2 levels was observed when the analysis was performed separately for the different hypertension phenotypes.
The linear association of HMOD with serum HMGB2 level.

cf-PWV, carotid-femoral pulse wave velocity; cIMT, carotid intima-media thickness; eGFR, estimated glomerular filtration rate; HMOD, hypertension-mediated organ damage; LVMI, left ventricular mass index; UACR, urine albumin-to-creatinine ratio.
Linear regression analysis was conducted to investigate the association of serum HMGB2 and subclinical HMOD. Logarithmic transformation was applied to serum HMGB2 level to comply normal distribution. Regression coefficients (β) and the corresponding p values derived from different models were presented in the table.
Model a was not adjusted.
Model b was adjusted for age, gender, body mass index, waist-to-hip ratio, and smoking.
Model c was adjusted for obstructive sleep apnea–hypopnea syndrome, abdominal obesity, total cholesterol, non-high-density-lipoprotein-cholesterol, uric acid, familial history of hypertension, and the variables in Model b.
A p value of <0.05 was considered statistically significant.
Significant possible predictors (as shown in Table 1) identified by univariate analyses and common cardiovascular risk factors (as shown in Table 1) were further assessed by multiple logistic regression analysis (Figure 5). In all patients, multivariate analysis showed that smoking, WHR, non-HDL-C levels, serum HMGB2 levels, the presence of abdominal obesity, and OSAHS were associated with an increased probability of subclinical HMOD (damage in at least one organ).
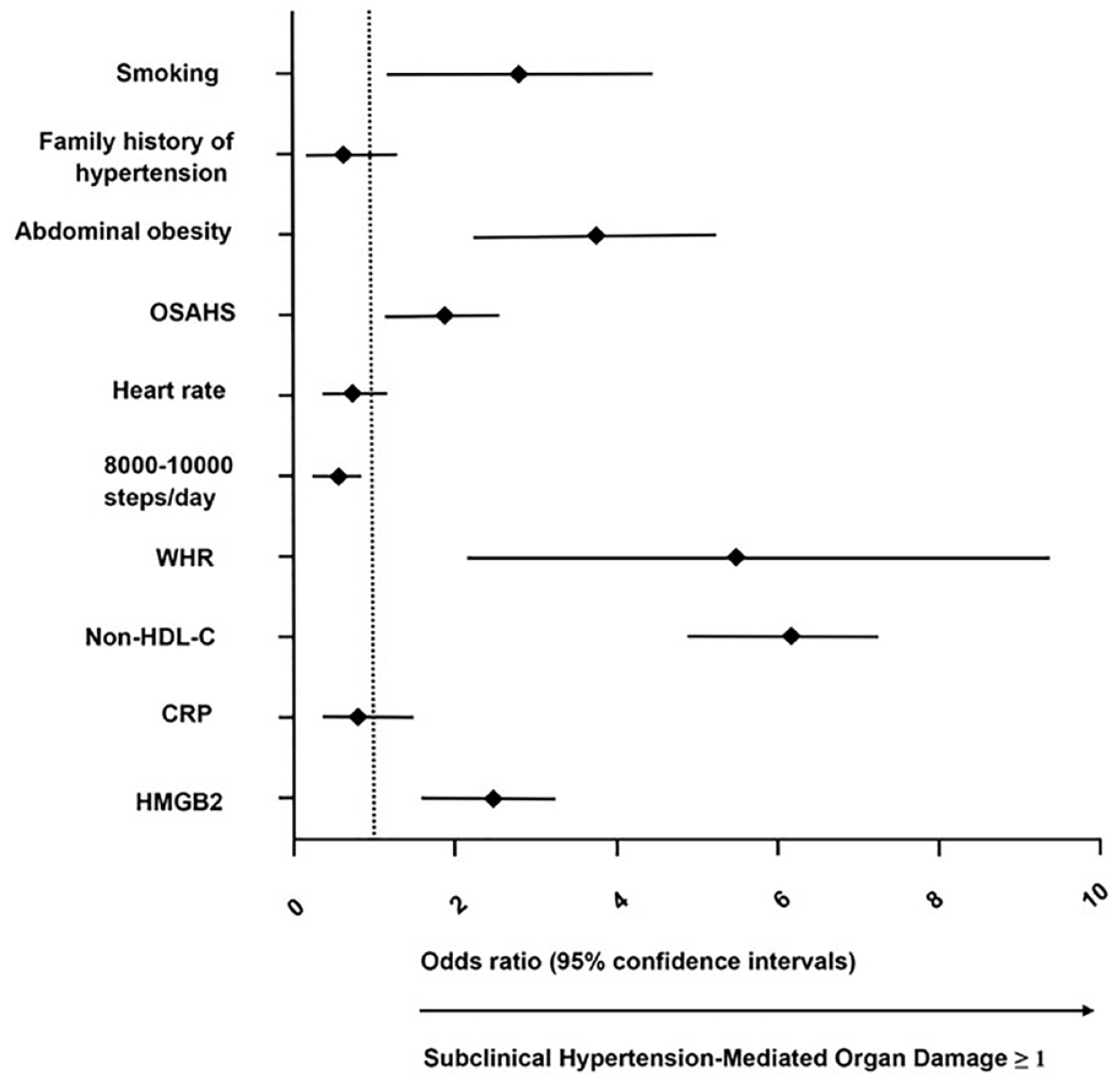
Forest plot showing results of multivariate analysis of serum HMGB2 and other markers.
Subgroup Analysis
Subgroup analyses according to the presence/absence of arterial stiffness (cf-PWV > 10 m/s), LVH, carotid artery wall thickening, and early-stage renal damage are summarized in Table 4. After multivariable adjustment, we found that serum HMGB2 predicted higher odds of arterial stiffness (cf-PWV > 10 m/s) [odds ratios (OR): 1.450; 95% confidence interval (CI): (1.142, 1.868); p = 0.014], carotid artery wall thickening (OR: 2.043; 95% CI: (1.709, 2.458); p = 0.003), LVH (OR: 2.841; 95% CI: (1.965, 4.203); p = 0.002), and early-stage renal damage (OR: 2.931; 95% CI: (2.616, 3.387); p < 0.001). No correlation was observed when the further subgroup analysis was performed separately for the different hypertension phenotypes.
Subgroup analyses according to the presence/absence of arterial stiffness (cf-PWV > 10 m/s), LVH, carotid artery wall thickening, and early-stage renal damage.

cf-PWV, carotid-femoral pulse wave velocity; cIMT, carotid intima-media thickness; CRP, C-reactive protein; eGFR, estimated glomerular filtration rate; HDL-C, high density lipoprotein-cholesterol; HMGB2, high-mobility group box 2; LVH, left ventricular hypertrophy; LVMI, left ventricular mass index; OSAHS, obstructive sleep apnea–hypopnea syndrome; WHR, waist-to-hip ratio.
In all patients, subgroup analyses according to the presence/absence of arterial stiffness (cf-PWV > 10 m/s), LVH, carotid artery wall thickening, and early-stage renal damage. We used cf-PWV, LVMI, cIMT and eGFR as the dependent variables with HMGB2, smoking, WHR, non-HDL-C levels, the presence of abdominal obesity, and OSAHS as covariates for multivariate adjustments.
p < 0.05, and a p value of <0.05 was considered statistically significant.
Table 5 shows the odds of subclinical HMOD by subgroup of hypertension phenotypes. In multivariable-adjusted models, including serum HMGB2, individuals with SHT had odds ratios of 2.392 (95% CI: 1.367–3.868), 2.942 (95% CI: 1.576–5.498), 2.127 (95% CI: 1.505–3.029), and 2.160 (95% CI: 1.144–4.055) for arterial stiffness (cf-PWV > 10 m/s), carotid artery wall thickening, LVH, and early-stage renal damage, respectively, compared with individuals with NT. Figure 6 illustrates the adjusted odds of having subclinical HMOD in 1 or ⩾2 organs according to subgroups determined by quartiles of serum HMGB2 (Q1–Q4 quartile) level.
The odds of subclinical HMOD by subgroup of hypertension phenotypes.

HMGB2, high-mobility group box 2; HMOD, hypertension-mediated organ damage; LVH, left ventricular hypertrophy; MHT, masked hypertension; n/N, number of individuals with organ damage/number of individuals in category; NT, normotension; OR, odds ratio; SHT, sustained hypertension; WCHT, white-coat hypertension.
Multivariable-adjusted model is adjusted for age, gender, body mass index, waist-to-hip ratio, smoking, obstructive sleep apnea–hypopnea syndrome, abdominal obesity, total cholesterol, non-high-density-lipoprotein cholesterol, uric acid, and familial history of hypertension.
p < 0.05, and a p value of <0.05 was considered statistically significant.

The adjusted odds of having subclinical HMOD in 1 or ⩾2 organs according to subgroups determined by quartiles of serum HMGB2 (Q1–Q4 quartile) level.
Pathway overrepresentation analyses
The differential protein expression patterns showed that serum HMGB2 regulated the inflammatory response and the diverse downstream effectors by interacting with its receptors, including interleukin (IL)-6, receptor for advanced glycation end products (RAGE), toll-like receptor (TLR)-2, TLR-4, and bone morphogenetic protein receptor II (BMPR2) (Figure 7). Pathway overrepresentation analysis revealed that patients with high serum HMGB2 levels had increased activity of pathways related to endothelial dysfunction, inflammatory processes, and atherosclerosis (Figure 8).

Differential protein expression in four distinct hypertension phenotypes patients.

Pathway enriched in HMGB2 correlated genes (adjusted p < 0.1).
Discussion
This study, for the first time, examined the relationships between serum HMGB2, an important signaling molecule, and several indicators of subclinical HMOD in untreated young adults. Consistent with our hypotheses, young adults with higher levels of serum HMGB2 showed a greater burden of subclinical HMOD, as indicated by a greater cIMT and LVMI, higher cf-PWV, and a lower eGFR level. Importantly, the associations remained significant after adjusting for serum HMGB2 and other cardiovascular risk factors, which implied that serum HMGB2 was related to the tissue characteristics of the artery wall independent of the major cardiovascular risk factors.
Experimental studies indicate that serum HMGB2 enhances ischemic injury.14,17 As a key mediator and booster of MI reperfusion injury, serum HMGB2 increases ROS production by activating the RAGE receptor, which contributes to MI injury in rats. 17 HMGB2 administration stimulates injury-induced neointimal hyperplasia in C57BL/6 mice; conversely, such pathological features are markedly attenuated in HMGB2–/– mice. 14 Indeed, ROS are known to augment p47phox phosphorylation, resulting in HMGB2-induced effects in vivo and in vitro.14,17 Moreover, decreased levels of HMGB2 in cardiomyocytes may predispose mice to heart failure progression after pressure overload predominantly via the attenuation of the left ventricular contractile ability. 19 Consistent with previously published data, increased serum HMGB2 levels are associated with MI severity and MACEs at 1 month. 14 In addition, serum HMGB2 levels are robustly related to the presence and severity of in-stent restenosis (ISR) in humans. 17
Our study yielded several main findings. First, only 23.1% of the participants were really hypertensive in our study. This result is largely consistent with those of Mahajan et al., 2 who found that isolated systolic hypertension was identified in one of four young adults in China, most of whom remained unaware of having hypertension. Regarding the independent predictors of hypertension progression in multivariate analysis (including smoking, 8,000–10,000 steps/day, abdominal obesity, OSAHS, WHR, and non-HDL-C levels), our results are mostly consistent with those of previous studies.5,43–47 Moreover, our data provide further support for serum HMGB2 as a novel risk biomarker of subclinical HMOD and reaffirm its potential as a therapeutic target in young adults with hypertension. Prior studies indicate that inflammation and hypertension are physiologically interrelated.48,49 Inflammation, one of the key mechanisms of endothelial dysfunction and arterial damage, combines these risk markers with vascular disease, arterial stiffness, and aging.48,50 The presence of hypertension in early adulthood increases the risk of all cardiovascular outcomes in middle age or later life. 51 Our findings therefore pave the way for future basic experimental research to evaluate whether any observed clinical benefit is due to a slowing of hypertension progression at a young age by lowering serum HMGB2 levels. In addition, our findings suggest that serum HMGB2 was an inflammatory biomarker of subclinical HMOD, as only HMGB2 remained significant compared with C-reactive protein (CRP) in multivariate analysis. Although CRP is the most prevalent quantitative measure of inflammation (in conditions such as hypertension, vascular dysfunction, and metabolic syndrome) in clinical research,52,53 it is frequently virtually undetectable in young healthy persons. Previous studies have reported that HMGB2 is notably more stable over time than CRP in young individuals.14,54 Second, consistent with our hypotheses, young hypertensive adults with high levels of serum HMGB2 showed a greater atherosclerotic burden, as indicated by a greater cIMT and higher cf-PWV. In a review of the role of HMGBs in CVD, findings from a large-scale assessment suggest that HMGB1 is a vital inflammation driver for vasoconstriction because it has a regulatory effect on BP and homeostasis. 55 Moreover, in a cohort study, serum HMGB1 levels, along with cIMT, were substantially increased in hypertensive patients, 56 yet the authors did not focus on the role of serum HMGB2. Third, in the current study, we observed that patients with high serum HMGB2 levels had increased activity of pathways related to endothelial dysfunction, inflammatory processes, and atherosclerosis. Indeed, pathway overrepresentation analysis revealed that serum HMGB2 regulates the inflammatory response and the diverse downstream effectors by interacting with its receptors, including IL-6, RAGE, TLR-2, TLR-4, and BMPR2. To the best of our knowledge, these findings are the first to comprehensively evaluate inflammatory pathways related to HMGB2 regulation and subclinical HMOD in young adults. Fourth, in our analysis, perhaps not surprisingly, a high level of serum HMGB2 was associated with poor kidney function and an increased risk of left ventricular diastolic dysfunction. These results are partially consistent with those of Liu et al., 17 who found that HMGB2 induces a network of pathogenic mechanisms, such as inflammation, apoptosis, and autophagy abnormalities, collectively contributing to the progression of MI injury. However, their study was not designed to assess the major contribution of HMGB2 to the promotion of hypertension. These findings further support the idea that in young adults with hypertension, serum HMGB2 drives CVD risk by promoting the progression of vascular and renal inflammation, providing a potential mechanistic explanation for the association between serum HMGB2 and target organ damage. Fifth, WCHT has been shown to promote the aggravation of coronary artery stenosis and cardiac arrhythmia,21,25 and MHT is accompanied by an increased risk for HMOD. 26 Therefore, special attention should be given to these hypertension phenotypes, as they may not be benign. Hence, our findings increase the credibility of hypertension phenotypes categorized by clinic and ambulatory BP monitoring measurements and can facilitate a deeper comprehension of HMOD in young adults.
The strengths of this study include a comprehensive and assessable set of cardiometabolic risk predictors. In addition, our study design facilitated the assessment of associations between serum HMGB2 and several factors of subclinical HMOD among young individuals before the onset of clinical manifestations of hypertension. Taken together, our findings advance previous knowledge by demonstrating that low-grade inflammation quantified by serum HMGB2 is a hallmark of subclinical HMOD in young adults.
Limitations
Our study has limitations that deserve consideration. First, it was a single-center and cross-sectional observational study, so uncontrolled and residual confounding effects might exist. In addition, the sample size was comparatively small. Unfortunately, the relationship between serum HMGB2 and subclinical HMOD in young adults may be limited to untreated or certain ethnic populations; therefore, further large-scale studies involving various treatments (e.g. antihypertensive drugs) or ethnic groups are needed to confirm this conclusion. Second, it was difficult to differentiate whether an increased level of serum HMGB2 represents a causal risk factor for hypertension progression itself. Indeed, it is more likely to be a biomarker that increases in response to other upstream causal pathways. Third, given that HMGB2 and HMGB1 share >80% sequence homology, 14 we did not measure serum HMGB1 levels in the study population. Importantly, information on the role of HMGB2 in hypertension is relatively limited compared with that on the role of HMGB1. Moreover, high sensitivity CRP (hsCRP) might improve cardiovascular risk assessment, as it is a sensitive assay that can detect extremely low levels of CRP in young individuals. 57 Fourth, although the enrichment/depletion analysis indicated that the association between the identified pathways and the upregulated biomarkers was appreciable, whether the number of biomarkers was sufficient to comprehensively populate the pathways is still uncertain.
Conclusion
Among the untreated young population, high serum HMGB2 levels were associated with a higher risk for future HMOD progression, regardless of adjustment for other prevailing CVD risk factors. These findings support the hypotheses that serum HMGB2 is part of an important inflammatory pathway that may contribute to adverse subclinical HMOD risk profiles in young adults. This provides a missing link supporting the usefulness of HMGB2 as a novel potential therapeutic target related to hypertension.
Supplemental Material
sj-docx-1-taj-10.1177_20406223221135011 – Supplemental material for Association between high-mobility group box 2 and subclinical hypertension-mediated organ damage in young adults
Supplemental material, sj-docx-1-taj-10.1177_20406223221135011 for Association between high-mobility group box 2 and subclinical hypertension-mediated organ damage in young adults by Jindong Wan, Gang Liu, Siwei Xia, Sen Liu, Yi Yang, Dan Wang, Jixin Hou, Xiaozhen Dai, Peng Zhou and Peijian Wang in Therapeutic Advances in Chronic Disease
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
