Abstract
Cystic fibrosis (CF) is caused by two mutations in the Cystic Fibrosis Transmembrane Conductance Regulator (CFTR) gene. In the last years, drugs targeting the underlying protein defect like lumacaftor/ivacaftor (LUM/IVA) or tezacaftor/ivacaftor (TEZ/IVA) and more recently elexacaftor/tezacaftor/ivacaftor (ELX/TEZ/IVA) were admitted. Outcome parameters evaluating therapy response like forced expiratory pressure in 1 s (FEV1), body mass index (BMI) or the efficacy of CFTR function in sweat glands showed improvement in several cases. Other, CFTR biomarkers were analysed rarely. This prospective observational study was aimed at evaluating CFTR function in patients treated with different CFTR modulators together with common valid clinical outcome parameters at standardized appointments (day 0, week 2, 4, 16). We followed four patients with the same mutation (F508del-CFTR), sex, age and disease severity. Monitoring focused on lung function, gastrointestinal aspects and CFTR function of sweat glands, nasal and intestinal epithelium. Sweat tests were performed by pilocarpine iontophoresis. Nasal potential difference (NPD) measured transepithelial voltage in vivo and potential increased when CFTR function improved. Rectal biopsies were obtained for intestinal current measurements (ICM) ex vivo. Intestinal CFTR function was assessed by stimulating chloride secretion with different reagents. Response to CFTR modulators regarding clinical outcome parameters was rather variable. A sweat chloride reduction of 35.3 mmol/L, nasal CFTR rescue of 4.4% and fivefold higher CFTR function in the intestine was seen at week 16 post-LUM/IVA. Due to our monitoring, we identified a non-responder to LUM/IVA and TEZ/IVA. In case of ELX/TEZ/IVA, clinical parameters and CFTR bioassays improved and were concordant. Although our cohort is small, results emphasize that non-responders exist and conclusions could not be drawn if patients were not monitored. Data on CFTR function can confirm or disprove ongoing CFTR dysfunction and might be helpful selectively. Non-responders need other alternative therapy options as demonstrated with ELX/TEZ/IVA.
Introduction
In Caucasians, cystic fibrosis (CF) is the most common life-threatening genetic disease. 1 Nevertheless, CF is defined as a ‘rare disease’. CF is caused by two mutations in the Cystic Fibrosis Transmembrane Conductance Regulator (CFTR) gene on the long arm of chromosome 7. CFTR is a chloride channel, which is activated through a cyclic adenosine monophosphate (cAMP) depending protein kinase A. 2 In detail, CFTR, an ATP-binding cassette transporter C member, comprises two membrane-spanning domains (MSDs), two nucleotide-binding domains (NBDs), and a regulatory domain. 3 It plays an important role in the epithelial ion and water household on the surface of mucous membranes of different organs. CFTR protein is not working correctly due to ‘loss of number’ and / or ‘loss of function’ mutations (e.g. absence, dysfunction). Until now, there are over 2100 different mutations identified and more than 350 are known to cause disease. The most common mutation in patients with CF is the F508del mutation. The prevalence of F508del mutation varies between countries, for example, in the Italian population F508del occurs less often than in the Northern European population 4 and ethnical differences can be found too. 5 F508del belongs to class II mutations. 1 The amino acid phenylalanine on position 508 in the CFTR protein is missing. Therefore, the CFTR protein is misfolded in the endoplasmic reticulum and is not passed to the membrane surface. As a result, CFTR cannot work correctly and will be removed by the proteasome.
New therapy approaches focus on this underlying protein defect and led to the admission of mutation specific CFTR correctors or potentiators like lumacaftor/ivacaftor (LUM/IVA) or tezacaftor/ivacaftor (TEZ/IVA) and more recently with elexacaftor/tezacaftor/ivacaftor (ELX/TEZ/IVA).
CFTR function can be evaluated by several CFTR bioassays in vivo and ex vivo. The most common one is the sweat test, because according to guidelines6,7 CF is diagnosed by elevated sweat chloride. Sweat is obtained by pilocarpine iontophoresis and concentration of electrolytes can be measured. A high extracellular concentration of sodium and chloride due to decreased reabsorption of NaCl in patients with a CFTR defect can be found. 1 A chloride sweat level of ⩽29 mmol/L is found in healthy individuals, whereas patients with CF have levels ⩾60 mmol/L. Furthermore, other CFTR bioassays should be considered to clarify the diagnosis in patients with inconclusive sweat test. CFTR bioassays like nasal potential difference (NPD) or intestinal current measurements (ICM) can be used for confirmation of CFTR dysfunction. However, these bioassays are more difficult to perform than other electrophysiological measurements (e.g. sweat test). It requires extensive training and specialized equipment listed in the Standard Operating Procedures (SOP) of the European Cystic Fibrosis Society (ECFS) Diagnostic Network Working Group and Clinical Trials Network.8,9 NPD measures the transepithelial potential difference between the Regio respiratoria (inferior nasal turbinate) and a subcutaneous electrode placed at the forearm. 10 In patients with CF a more negative basal potential difference (PD) due to a defective chloride secretion and a hyperabsorption of sodium can be measured. 11 During the measurement, the sodium channel and afterwards the chloride channel will be blocked. A decreased potential difference is pathognomonic for this disease and the function of the chloride channel can be predicted.12–14 The same current measurements as discussed before can be performed on the intestinal epithelium (ICM) ex vivo. 15 After stimulation of CFTR-mediated chloride secretion in ex vivo rectal biopsies, the circuit current (Isc) will be measured as a value of ion transport. In patients with CF only a small amount of chloride will be detected; in healthy individuals, it will be a huge current.16,17
Our observation was aimed at evaluating the effect of different CFTR modulators on CFTR assembly via sweat test, nasal potential difference (NPD) and intestinal current measurements (ICM) as well as common valid clinical outcome parameters (FEV1, LCI2.5%, BMI) in real-time setting.
Subjects
According to her CFTR function in sweat glands, nasal and intestinal epithelium patient 1 was improving with the combination therapy LUM/IVA. Furthermore, an improvement was seen in lung function parameters at week 16. No improvement concerning LCI2.5% or BMI could be found. Slightly lower faecal calprotectin was found after LUM/IVA initiation. Similarly, to the increased faecal calprotectin, C-reactive protein was elevated [0.77–1.84 mg/dl (reference value <0.5 mg/dl)] constantly throughout observational period. Medication response until 16 weeks after first intake of LUM/IVA can be seen in Tables 1 and 2.
Data of several outcome parameters for patient 1 (#; coloured in orange), patient 2 (*; coloured in grey), patient 3 (~; coloured in yellow) and patient 4 (§; coloured in green) during the observational period of LUM/IVA intake (day 0 till week 16) are outlined below. The outcome parameters are divided in lung function data (spirometry, multiple breath washout), gastrointestinal aspects (BMI, faecal chymotrypsin and calprotectin) and CFTR bioassay data (sweat chloride and sodium, NPD scores, ICM outcome parameters).
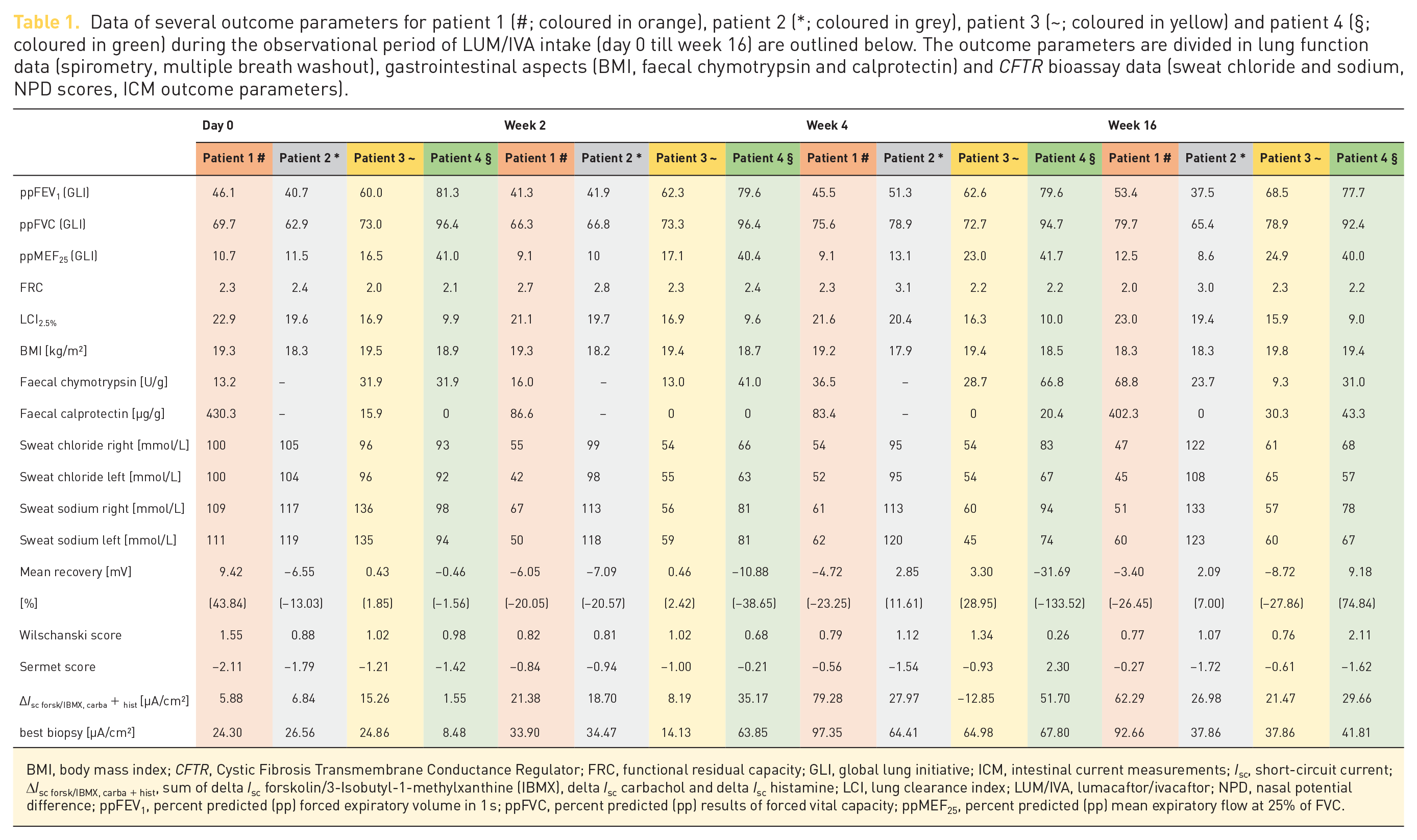
BMI, body mass index; CFTR, Cystic Fibrosis Transmembrane Conductance Regulator; FRC, functional residual capacity; GLI, global lung initiative; ICM, intestinal current measurements; Isc, short-circuit current; ∆Isc forsk/IBMX, carba + hist, sum of delta Isc forskolin/3-Isobutyl-1-methylxanthine (IBMX), delta Isc carbachol and delta Isc histamine; LCI, lung clearance index; LUM/IVA, lumacaftor/ivacaftor; NPD, nasal potential difference; ppFEV1, percent predicted (pp) forced expiratory volume in 1 s; ppFVC, percent predicted (pp) results of forced vital capacity; ppMEF25, percent predicted (pp) mean expiratory flow at 25% of FVC.
Data of several outcome parameters for patient 1 during the observational period of LUM/IVA (day 0 till week 16; #; coloured in orange) are outlined below. After treatment with LUM/IVA for 3 years, ELX/TEZ/IVA was started following the same prospective observational investigation (day 0 till week 16; °; coloured in blue). Results for day 0 of ELX/TEZ/IVA correspond to results three years after LUM/IVA. The outcome parameters are divided in lung function data (spirometry, multiple breath washout), gastrointestinal aspects (BMI) and CFTR bioassay data (sweat chloride and sodium, ICM outcome parameters). NPD measurements were not performed due to technical problems.

BMI, body mass index; CFTR, Cystic Fibrosis Transmembrane Conductance Regulator; ELX/TEZ/IVA, elexacaftor/tezacaftor/ivacaftor; FRC, functional residual capacity; GLI, global lung initiative; ICM, intestinal current measurements; Isc, short-circuit current; ∆Isc forsk/IBMX, carba + hist, sum of delta Isc forskolin/3-Isobutyl-1-methylxanthine (IBMX), delta Isc carbachol and delta Isc histamine; LCI, lung clearance index; LUM/IVA, lumacaftor/ivacaftor; NPD, nasal potential difference; ppFEV1, percent predicted (pp) forced expiratory volume in 1 s; ppFVC, percent predicted (pp) results of forced vital capacity; ppMEF25, percent predicted (pp) mean expiratory flow at 25% of FVC.
Three years after first dose of LUM/IVA a stabilization in values for lung function and BMI was detected, while CFTR function was worse. LUM/IVA was stopped, and ELX/TEZ/IVA was started following the same prospective observational investigations (Table 2).
These findings were comparable to patient 1. But patient 2 did not show a response to the combination therapy with LUM/IVA relating to lung function parameters, BMI, sweat test, NPD. Only a slightly lower LCI2.5% regarding fewer ventilation inhomogeneity and a higher, but not satisfying intestinal CFTR function could be obtained at week 16 compared with day 0. So, due to a lack of improvement in several clinical outcome parameters at week 16 (Tables 1 and 3), patient 2 was categorized as non-responder for LUM/IVA. The combination therapy of LUM/IVA was finally stopped after 1 year of observation and TEZ/IVA was started. The same monitoring for TEZ/IVA was performed (Table 3) but did not lead to acceptable response and non-responder status for TEZ/IVA was verified again. Recently, patient 2 started the intake of ELX/TEZ/IVA and is improving under the combination therapy respecting nearly all evaluated outcome parameters (Table 3).
Data of several outcome parameters for patient 2 during the observational period of LUM/IVA (day 0 till week 16; *; coloured in grey), during TEZ/IVA start (day 0 till week 16; +; coloured in yellow) and during ELX/TEZ/IVA intake (day 0 till week 16; ^; coloured in green) are outlined below. The outcome parameters are divided in lung function data (spirometry, multiple breath washout), gastrointestinal aspects (BMI) and CFTR bioassay data (sweat chloride and sodium, NPD scores, ICM outcome parameters). Faecal chymotrypsin and calprotectin was not analysed in patient 2.

BMI, body mass index; CFTR, Cystic Fibrosis Transmembrane Conductance Regulator; ELX/TEZ/IVA, elexacaftor/tezacaftor/ivacaftor; FRC, functional residual capacity; GLI, global lung initiative; ICM, intestinal current measurements; Isc, short-circuit current; ∆Isc forsk/IBMX, carba + hist, sum of delta Isc forskolin/3-Isobutyl-1-methylxanthine (IBMX), delta Isc carbachol and delta Isc histamine; LCI, lung clearance index; LUM/IVA, lumacaftor/ivacaftor; NPD, nasal potential difference; ppFEV1, percent predicted (pp) forced expiratory volume in 1 s; ppFVC, percent predicted (pp) results of forced vital capacity; ppMEF25, percent predicted (pp) mean expiratory flow at 25% of FVC; TEZ/IVA, tezacaftor/ivacaftor.
Methods
This prospective, single-centre monitoring pilot study was conducted to gain a detailed overview of CFTR function during the administration of different CFTR modulators in real-time settings.
For patients, monitoring was explained in detail and written informed consent was obtained before study start. The Ethical Committee for Human Research at the Medical University of Innsbruck approved this project.
Pulmonary function testing was performed with MasterScreenTM Body (Jaeger® or rather CareFusion®, Hoechberg, Germany) according to the spirometry standards of the American Thoracic Society/European Respiratory Society (ATS/ERS). Percent predicted (pp) results of forced vital capacity (ppFVC), forced expiratory volume in 1 s (ppFEV1), mean expiratory flow at 25% of FVC (ppMEF25), were based on equations of the global lung initiative (GLI by Quanjer et al. 21 ).
Multiple Breath Washout (MBW) techniques were carried out using Exhalyzer® D (Eco Medics AG®, Duernten, Switzerland) with 100% oxygen, obtained functional residual capacity (FRC) and lung clearance index (LCI2.5%).
Gastrointestinal parameters (faecal chymotrypsin and faecal calprotectin) were obtained from stool samples. The activity of chymotrypsin was used as parameter for the intake and resorption of pancreatic enzymes and was determined with Chymotrypsin Activity Kit® (Immundiagnostik AG®, Bensheim, Germany) and measured using Agilent Cary 60 UV-Vis® spectrophotometer (Agilent Technologies®, Santa Clara, California, USA) accordingly to manufacture’s manuals. Normal values are defined >6 U/g. Furthermore, calprotectin (a marker for intestinal inflammation)22,23 was determined with commercially available Enzyme-Linked Immunosorbent Assay (ELISA) Kit Calprest® (Eurospital diagnostic®, Trieste, Italy) and measured by VICTORTM X3 Multilabel Reader (PerkinElmer®, Waltham, Massachusetts, USA) according to manual. Values <50 μg/g stool were considered normal.
Sweat tests were performed with pilocarpine iontophoresis. Two impregnated swabs with pilocarpine were placed on smooth and hairless region (i.e. forearm). Electrodes were added to receive continuous current flow (from cathode to anode). As a result, pilocarpine molecules migrated in the cutis and stimulated the sweat glands production. The amount of pilocarpine, current (1–4 mA) and duration of impact time are standardized. After 5 min swabs were removed. Sweat was collected for half an hour in a Macroduct® Sweat Collection System (Model 3700, Wescor®, Logan, Utah, USA). Afterwards, sweat was collected in an Eppendorf tube. For quality reasons, sweat test was performed on right and left arm. For measurement of chloride concentration, a Chloridmeter CM20 (Gonotec®, Berlin, Germany) and for sodium a Flame Photometer PFP7/C (Jenway®, Stone, UK) were used.
For NPD performance, we used a Calomel / Agar bridge. A double lumen catheter of Marquat Genie Biomedical® (Boissy-Saint-Léger, France) was placed on the nasal epithelium, resting against the surface of the target epithelium, and acting as an exploring electrode, whereas a 23G butterfly needle of Becton Dickinson® (Franklin Lakes, New Jersey, USA) filled with 3% agar and ringer solution was placed on the upper arm serving as reference electrode. 24 The bioelectric potential can be measured by using a high-impedance voltmeter [Power Lab® 8/35 with a BMA-200® AC/DC portable preamplifier and an ISO-Z® isolated head stage of AD Instruments® (Oxford, UK)] between these two electrodes [connected to REF 401 reference electrodes by Radiometer Analytical® (Villeurbanne, France)]. Measurements were performed according to the SOP 8 on both nostrils. Changes in potential difference (mV) by different solutions were recorded continuously by Lab Chart® [AD Instruments® (Oxford, UK)] and can be sorted out at defined points of interest. Closed loop offset initial and final as well as finger PD (pre, between nostrils and post) assess as quality criteria.
To evaluate the change in membrane potential and the function of the chloride channel,
were calculated. Values were rated as pathological if recovery is > −8 mV in total and < −50%. Furthermore, a Wilschanski score > 0.70 and Sermet score < 0.27 were pathognomonic for CFTR dysfunction.
The same current measurements as discussed before can be performed on the intestinal epithelium (ICM) ex vivo. 15 Therefore, biopsy samples of the rectum were extracted freshly by a suction biopsy device [Trewavis Surgical® (Victoria, Australia)] with a defined suction pressure of 9 psi/60 kPa for the least invasive procedure. According to the SOP 9 at least four biopsies were collected. Rectal biopsies were obtained at day 0 as well as 5 h after last dose of LUM/IVA at week 2, 4 and 16, respectively.
The biopsies are given in Meyler buffer solution (128 mmol/L NaCl, 20.2 mmol/L NaHCO3−, 20.2 mmol/L Na2HPO42−, 0.4 mmol/L NaH2PO4−, 4.7 mmol/L KCl, 1.0 mmol/L MgCl2, 1.3 mmol/L CaCl2, 10 mmol/L HEPES; pH 7.4, osmolarity 300 mOsm) shifted with
Subsequently, the voltage electrodes were placed close to the tissue and the current electrodes were attached at distance.
A system equilibration time of nearly 20–30 min was considered. PD offset should be nearly 0 mV and final fluid resistance compensation values should range 250–350 Ω for the 1.5 mm diameter aperture slider in open-circuit mode. After tissue equilibration for around 5 min in open-circuit mode, the system was switched to short-circuit mode, basal PD (values around 0 mV) and basal resistance (Rt; range of 15–30 Ω × cm2) of tissue were recorded. Several different specific stimulators of the chloride secretion 25 were added in an orderly manner to the apical (mucosal) and basolateral (serosal) bathing solutions according to the SOP. 9
The short-circuit current (Isc) [μA/cm2] as a value of ion transport was amplified with a Multi-Channel Voltage Current Clamp and four preamplifiers [all supplied by Physiologic Instruments® (San Diego, USA)] and recorded by Lab Chart® [AD Instruments® (Oxford, UK)] through the whole process of stimulation of CFTR-mediated chloride secretion.
For evaluation, the sum of delta Isc forskolin/3-Isobutyl-1-methylxanthine (IBMX), delta Isc carbachol and delta Isc histamine (∆Isc forsk/IBMX, carba + hist) were calculated reflecting the function of the chloride channel. 26 In patients with CF only a small amount of chloride was detected (low ∆Isc forsk/IBMX, carba + hist), in non-CF a huge current comparatively was detected (high ∆Isc forsk/IBMX, carba + hist). Minso et al. 20 set the ∆Isc forsk/IBMX, carba + hist cut-off level with 39 µA/cm2 for detecting CF.
Measurements of NPD and ICM are undertaken throughout only a few CF centres in Europe. We implement NPD and ICM for 5 years now and established a CFTR function laboratory the results of which we discuss regularly with another CF reference centre.
For gaining high-quality data, NPD and ICM data of healthy individuals (n = 4) as well as results of CF patients without CFTR modulator (day 0; n = 4) were compared with literature and are presented in the supplementary material.
Discussion
Defining a prognosis for patients with CF is exceedingly difficult due to the huge geno–phenotype variability. 27 Even with the same mutation (e.g. F508del), the clinical outcome can range from severe progression and involvement of several organs to mild courses. 28 With conservative symptomatic therapy including inhalation, sports, nutrition and medication the progression of illness can be influenced effectively. New developed therapies started with the goal to find at least a causal determined therapy approach with mutation specific therapies in the last years. This goal was reached partially by the admission of CFTR modulator therapies. F508del leads to a reduction in CFTR processing and transport to the cell surface. 1 Therefore, addressing the underlying cause of disease in patients homozygous for this mutation is complex. This is expected even more from CFTR modulator therapies. Our observation shows that even with the same mutation (F508del-CFTR), sex, age and disease severity, the clinical outcome after starting the combination therapy with LUM/IVA ranges from nearly complete treatment response (patients 1 and 3) and organ-specific rescue of CFTR function (patient 4) to non-responding therapy outcome (patient 2).
The results of the pivotal registration studies for LUM/IVA showed a mean relative difference of FEV1 between active treatment and placebo of 4.3–6.7% at week 24, 29 whereas another study by Graeber et al. 30 only found a change of 2.27% after 8–16 weeks. In fact, the difference between receiving LUM/IVA or placebo regarding FEV1 was little, 29 smaller than expected and comparable to other therapy approaches like long-term exercise for 6 months independent of strength or endurance training. 31
We found a difference in FEV1 comparing day 0 and week 16 of 7% in our observation of LUM/IVA therapy. Focusing on TEZ/IVA in patient 2, a benefit in lung function parameters was seen during the observational period, but it was not constant (Table 3). Whereas our observation of ELX/TEZ/IVA response showed a huge improvement in lung function parameters even 2 weeks after initiation and further improvement could be achieved 16 weeks after start in patients 1 and 2 (Tables 2 and 3). So, it might be enough to focus on lung function parameters as primary endpoint to interpret the effectiveness of ELX/TEZ/IVA therapy in patients with impaired lung function. Yet, other clinical outcome parameters than the most common FEV1 might be necessary in selected cases to evaluate therapy response and predict clinical outcome.
A more sensitive parameter for early decline of lung function and an indicator for ventilation inhomogeneity or air trapping is the lung clearance index (LCI2.5%). 32 An Irish working group showed even lower median LCI2.5% levels post-LUM/IVA treatment and demonstrated the positive effect of a LUM/IVA therapy in adolescence. 33 These results were consistent with our findings for LUM/IVA, although our patient cohort was older than the Irish one, because nearly every patient (despite patient 1) had a lower LCI2.5% at the end of the observation. Focusing on the results of all analysed parameters of patient 1 (improving in all 3 CFTR function bioassays sweat test, NPD and ICM as well as FEV1 during the study period), LCI2.5% is not the most sensitive outcome parameter for LUM/IVA therapy evaluation as it is suggested by this working group, although it might be helpful in patients with a FEV1 ⩾80% for detecting early lung disease progression. 34 In patient 2, no improvement of LCI2.5% could be achieved by TEZ/IVA, but post-ELX/TEZ/IVA treatment an obvious decline in LCI2.5% was seen. However, if a CFTR modulator therapy is started early in life and LCI2.5% is low, structural pulmonary damage could be avoided or development might be reduced and delayed than without modulator therapy.
Focusing on other clinical endpoints like BMI, previous studies for LUM/IVA,29,35 found an improvement in BMI in their patients and an increase in BMI is a possible and sometimes desired side effect. In contrast to literature,29,35 BMI showed no constant improvement in our patients when evaluated pre- and post-LUM/IVA. A reason for that might be that our patients had an appointment with dieticians at study start to avoid an excessive increase in body weight. Therapeutic interventions were set where BMI was already over the target range.
Furthermore, faecal calprotectin – a marker of inflammation in the intestinal tract22,23 – showed an improvement in intestinal inflammation focusing on Ivacaftor 36 and more recently LUM/IVA lead to lower faecal calprotectin levels in French adolescents. 37 Elevated concentrations are associated with disease severity of CFTR mutation, pancreas insufficiency and progression. 38 In fact, F508del homozygous subjects have higher faecal calprotectin than patients with other mutations. 39 In our cohort, only patient 1 showed increased faecal calprotectin at day 0. Nevertheless, no obvious modification of faecal calprotectin levels after LUM/IVA initiation were found in our small cohort, even though our cohort was older than the French patients 37 and elevated concentrations are associated with disease progression. 38
In further studies, it is necessary to evaluate the therapy response by focussing on pathophysiological processes due to CFTR functioning tests, 40 especially for LUM/IVA and TEZ/IVA treatment. We found a huge drop in sweat chloride concentration when comparing the results before LUM/IVA start and at week 2 or at week 4 (Table 1). Focussing on long-term effects, there is a reduction of 35.3 mmol/L analogue at week 16 of LUM/IVA treatment. Comparing our results with those of Graeber et al., 30 we even achieved higher CFTR rescue focussing on sweat gland function. An obvious therapy effect concerning sweat chloride concentration was seen when ELX/TEZ/IVA was started in patients 1 and 2 (reduction of 50.5 mmol/L; Table 2 and 59.5 mmol/L; Table 3) consistent with reductions of sweat chloride concentration of 61.0 mmol/L according to literature. 41 Guimbellot et al. 42 argued that the sweat gland epithelium is not affected by sequelae of the defect such as inflammation or tissue destruction and therefore represents an objective parameter to measure CFTR dysfunction. However, CFTR function in sweat gland is not correlating with CFTR rescue in nasal or intestinal epithelium. 30 In contrast, the NPD measurements are influenced by inflammation of the mucosa, nasal polyps and cooperation of the patient. Comparing NPD parameters (e.g. basal PD) our results match with data in the literature. Although histological fundamentals 43 may conclude that the epithelium of the Regio respiratoria is correlating with the bronchus epithelium, 44 it is not predictable if there is a correlation of an improvement in NPD with lung function parameters. 45 However, our results of patient 1 show that there were lower NPD scores after the intake of LUM/IVA, but the lung function parameters did not improve before week 16 and even first declined. Moreover, CFTR rescue in the nasal epithelium is specified by Graeber et al. 30 with 10.2% after 8–16 weeks, whereas we only found 4.4% at study end point with LUM/IVA treatment, even though there was a huge improvement in CFTR function at first (Table 1). Graeber et al. 30 did not measure CFTR function at defined study points. Consequently, the comparability of their results lacks due to missing data. The long-term effect on the nasal epithelium by the CFTR modulator therapy with LUM/IVA in our pilot study was moderate. These findings are confirmed by the modifications of lung function parameters. However, results of patient 2 suggest that post-ELX/TEZ/IVA treatment NPD scores were in a normal range (Table 3). These results regarding NPD parameters post-ELX/TEZ/IVA were confirmed by the study of Graeber et al. 41 focusing on more than 100 patients.
Furthermore, the effect on CFTR function by modulator therapy could be evaluated by ICM and showed an improvement of 17.7% evaluating the response of ∆Isc forsk/IBMX, carba + hist before and after initiation of LUM/IVA therapy. 30 Although our study sample was small, significant changes of ∆Isc forsk/IBMX, carba + hist in the best responding biopsy were found. An obvious changing of CFTR function in the intestine (best responding biopsy) post-LUM/IVA treatment was found at week 4 (fivefold higher Isc forsk/IBMX, carba + hist; Table 1) as well as post-ELX/TEZ/IVA intake (Tables 2 and 3). In contrast to literature, 41 patient 2 showed no improvement post-TEZ/IVA treatment and highest ∆Isc forsk/IBMX, carba + hist was seen at week 2 (Table 3). ICM is one of the most sensitive outcome parameters to evaluate CFTR function and restoration of CFTR function due to modulator therapy.
In fact, conventional outcome parameters (like sweat chloride concentration, FEV1, LCI2.5% and BMI) are usually reliable and valid for the evaluation of treatment response, but in the case of non-responders an improvement of clinical parameters is lacking. No correlations of ppFEV1 or BMI with CFTR bioassays were found recently, indicating inadequate outcome parameters for detecting response to modulators at the level of the underlying defect. 41 Nevertheless, CFTR function tests are performed in very few centres and cannot be offered to every patient, which causes a huge limitation. Nonetheless, functional CFTR biomarkers focusing on different organs (e.g. NPD, ICM) can help clinicians potentially to distinguish responders from non-responders especially post LUM/IVA or -TEZ/IVA treatment. Therefore, it should be considered in selected cases for gaining additional information on pathophysiological conditions. For example, if there is no response to LUM/IVA or TEZ/IVA treatment CFTR bioassays should be performed before switching to ELX/TEZ/IVA. Furthermore, we recommend repeating CFTR bioassays after the start of ELX/TEZ/IVA intake, if an improvement of FEV1 lacks.
So, these diagnostic tests are not only recommended for confirming or excluding a CF diagnosis, 11 but they might also be an important tool in clinical trials and for the prediction of patient’s outcome. 46 Due to lacking data on long-term effects, Rubin et al. 47 recently published their modelling study and their analysis showed that the combination therapy of LUM/IVA increased the survival with higher lung function and lower risk of lung transplantation.
However, to account for the long-term consequences, we focused on improvement of clinical outcome parameters and negative side effects. Due to the small number of patients included in our monitoring study no significant alterations either in CFTR biomarkers nor in conventional clinical outcome parameters could be found after LUM/IVA. Furthermore, results emphasize that non-responders for LUM/IVA and TEZ/IVA exist, and such conclusions could not be drawn, if there would not be a strict and regular monitoring emphasizing on several outcome parameters.
So, in case of non-responders (like patient 2), LUM/IVA and TEZ/IVA treatment should be stopped due to possible side effects and cost-effectiveness ratio. Consequently, treatment might be switched to other available highly efficient medication therapies.
Conclusion
This is the first personalized monitoring study of different CFTR modulator efficacy at standardized appointments (day 0, week 2, 4 and 16), which has focused on several routine clinical outcome parameters as well as CFTR bioassays. We showed that there are differences of CFTR function before and after starting the intake of CFTR modulators as well as organ-specific changes due to the therapy. Even with the same mutation (F508del-CFTR), sex, age and disease severity, the clinical outcome ranges from nearly complete treatment response (patients 1 and 3) and organ-specific rescue of CFTR function (patient 4) to non-responding therapy outcome (patient 2). In fact, conventional used outcome parameters (like FEV1 and BMI) are usually reliable and valid for the evaluation of CFTR modulator response, but in the case of non-responders (especially post-LUM/IVA or -TEZ/IVA treatment) clear improvement of clinical parameters is lacking and data on CFTR function of different organs (NPD, ICM) can confirm or disprove ongoing CFTR dysfunction. Non-responders for ELX/TEZ/IVA were not found in our cohort. Therefore, functional CFTR biomarkers should be considered in selected cases for gaining additional information on pathophysiological conditions.
Nevertheless, real-life modulator treatment protocols may need to be adapted individually according to observational results (e.g. therapy discontinuation, switching of CFTR modulators).
Therefore, several clinical and functional biomarkers on multiple defined occasions could be helpful to evaluate individual treatment response in each patient treated with modulators.
Supplemental Material
sj-docx-1-taj-10.1177_20406223221108627 – Supplemental material for Personalized medicine with drugs targeting the underlying protein defect in cystic fibrosis: is monitoring of treatment response necessary?
Supplemental material, sj-docx-1-taj-10.1177_20406223221108627 for Personalized medicine with drugs targeting the underlying protein defect in cystic fibrosis: is monitoring of treatment response necessary? by Katharina Niedermayr, Verena Gasser, Claudia Rueckes-Nilges, Dorothea Appelt, Johannes Eder, Teresa Fuchs, Lutz Naehrlich and Helmut Ellemunter in Therapeutic Advances in Chronic Disease
Footnotes
Acknowledgements
The authors thank the patients with cystic fibrosis for their participation in this observational pilot study. Furthermore, we thank Mrs Nikelwa Theileis, MA, for proofreading.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
