Abstract
Background:
It is still uncertain if a dysregulated expression of activating Fc gamma receptors (FcγRs) is associated with the development of immunoglobulin A nephropathy (IgAN).
Methods:
RNA sequencing was used to determine the mRNA levels of type I FcγRs, which were then verified by quantitative reverse transcription–polymerase chain reaction (qRT-PCR). Commercial ELISA kits were used to detect plasma soluble FcγRIIIb (sFcγRIIIb).
Results:
We first examined the expression of FcγRs genes in 17 patients with IgAN and six healthy controls. The expression of FcγRIa, FcγRIb, FcγRIIa, FcγRIIc, FcγRIIIa, and FcγRIIIb was shown to be higher in IgAN patients. Even without statistical significance, there was a downward trend in FcγRIIb mRNA levels in IgAN. We observed that the expression levels of activating FcγR mRNAs were consistently higher in an independent set of 20 IgAN patients and 20 healthy controls, confirming the RNA-seq results. FcγRIIIb was the IgG receptor with the greatest difference in expression between the two groups (log2 fold-change = 1.82). We observed a much higher percent of FcγRIIIb positive cells in IgAN by flow cytometry. Next, we measured plasma sFcRIIIb levels in 50 patients with IgAN and 50 healthy controls. The findings revealed that the mean sFcγRIIIb level in plasma in participants with IgAN was much higher than that of healthy controls. Increased sFcγRIIIb levels were associated with a substantial increase in body mass index (BMI), lipid levels, serum creatinine level, and a larger percentage of sclerosis compared with lower sFcRIIIb levels. Patients in the group with higher sFcγRIIIb levels were more likely to get glucocorticoid treatment.
Conclusion:
The results demonstrated that the mRNA levels of the activating Fc receptor of IgG were significantly increased in IgAN. Patients with higher plasma sFcγRIIIb levels may have had more severe illness than those with lower levels.
Introduction
Immunoglobulin A nephropathy (IgAN) is the most frequent type of primary glomerulonephritis worldwide, with an estimated 10% of patients progressing to end-stage kidney disease (ESKD) within 10–20 years after diagnosis. 1 Immune complexes (ICs) formed by galactose-deficient IgA1 (Gd-IgA1) and its antibodies (primarily IgG) tend to deposit on the glomerular mesangium, triggering the pathogenesis of IgAN.2–4
One of the molecular mechanisms of autoantibody-induced organ damage is binding the activating Fc region of IgG to the receptors for the activating Fc region of IgG (FcγRs).5–7 ICs containing IgG crosslink the Fc gamma receptor (FcγR) on recruited monocytes, leading to the pathophysiology of autoimmune and inflammatory diseases. 7 Increased FcγRI expression on monocytes has been linked to systemic inflammation and renal injury in patients with systemic lupus erythematosus (SLE). 8 In SLE mice, dysregulated FcRIIA expression by bone marrow cells aggravated lupus nephritis and accelerated mortality, but in healthy mice, FcγRIIA expression was unaffected. 9 On the contrary, the expression of activating FcγRs and their potential role in IgAN remain unknown.
The current study examined the association between type I FcγR levels and IgAN susceptibility and severity.
Materials and methods
Subjects
In this retrospective case-control study, the participants were 50 patients with biopsy-proven primary IgAN from January 2020 to March 2020 as well as 50 healthy controls (HCs) who were age- and sex-matched to the patients. In this study, all patients must have had complete clinical information at the time of kidney biopsy. We included only the individuals whose blood samples were available. The exclusion criteria were as follows: (1) patients with secondary IgAN or with other comorbid renal diseases; (2) patients who had received corticosteroids or immunosuppressive medications; (3) those who lacked clinical or serological data. The creatinine equation developed by the Chronic Kidney Disease Epidemiology Collaboration was used to calculate the estimated glomerular filtration rate (eGFR) 10 in this study. Histological lesions were diagnosed based on the Oxford classification system, 11 which was used to determine their severity. The protocol for the research was approved by the Medical Ethics Committee of the Tianjin Medical University General Hospital and informed written consent was acquired from all the participants.
Our research population was divided into two distinct groups to identify FcγRs mRNA expression: discovery (17 IgAN and 6 HCs) and validation samples (20 IgAN and 20 HCs). As the initial stage, the discovery participants were included in the RNA-seq experiment. Next, the identified receptors were detected in validation samples.
RNA extraction and RNA deep sequencing
Following the manufacturer’s instructions, total cellular RNA was isolated from CD19 positive B lymphocytes using a miRNeasy Micro Kit (Qiagen, Hilden, Germany). The VAHTS mRNA-seq v2 Library Prep Kit for Illumina was used to prepare the NGS libraries (Vazyme, Nanjing, China). As previously reported, RNA samples in each group were sent for mRNA deep sequencing on an Illumina HiSeq X sequencing platform. Differential expression gene analysis was performed using R v3.2.2. Fold changes of ⩽−1.5 or ⩾1.5 and p < 0.05 were considered significantly differentially expressed.
Peripheral blood mononuclear cells isolation and reverse transcription polymerase chain reaction
To collect about 5 ml of peripheral venous blood, anticoagulated tubes containing ethylenediaminetetraacetic acid were utilized. Total RNA was recovered from peripheral blood mononuclear cells (PBMCs) after they had been separated using a lymphocyte separation medium. The cDNA was generated using a revert first-strand cDNA kit in accordance with the manufacturer’s instructions (Promega, USA). The resultant cDNA was amplified in an Applied Biosystem 7500 Real-Time PCR System using a 20 µl reaction mixture including SYBR Green PCR Master Mix (Roche, USA) and a SYBR Green PCR Master Mix (Roche, USA). Table 1 lists the primer pairs for FcγRs including FcγRIa, FcγRIb, FcγRIIa, FcγRIIb, FcγRIIc, FcγRIIIa, FcγRIIIb, and GAPDH. The fold changes in between patients and controls were expressed by the 2-∆∆CT method.
Primers used to amplify the FcγRIa, FcγRIb, FcγRIIa, FcγRIIb, FcγRIIc, FcγRIIIa, FcγRIIIb, and GAPDH.

Flow cytometry for FcγRIIIb in PBMCs
Human whole blood was collected and centrifuged at 1500 r/min for 10 min to remove plasma. PBMCs in 100 μl phosphate-buffered saline (PBS) were stained with Phycoerythrin (PE)-conjugated mouse anti-human FcγRIIIb (BD Biosciences, USA). The result was expressed as mean fluorescence intensity.
ELISA for FcγRIIIb
Plasma soluble FcγRIIIb (sFcγRIIIb, Abcam, UK) levels were determined using a commercial ELISA kit in accordance with the manufacturer’s instructions. It was determined by measuring the absorbance of the sample at 450 nm using an EL312 Bio-Kinetics microplate reader (Bio-TekInstruments, Winooski, VT).
Statistical analysis
Data with a normal distribution were reported as the mean ± SD, and they were compared using an independent-samples student t-test. Data with a non-normal distribution, on the contrary, were reported as the median (first and third quartiles) and analyzed using the Mann–Whitney U test. For categorical variables, data were evaluated as percentages and compared using an χ2 test to determine significance. Power calculations were used to determine the number of participants required with 0.90 power at an alpha level of 0.05. The threshold of significance was set at p < 0.05. All statistical tests were carried out with the help of SPSS version 16.0.
Results
Differentially expressed FcγR mRNAs in patients with IgAN
To find differentially expressed mRNAs, we first examined the gene expression of type I FcγRs in 17 patients with IgAN and six HCs. According to the findings in Table 2, the expressions of FcγRIa, FcγRIb, FcγRIIa, FcγRIIc, FcγRIIIa, and FcγRIIIb were shown to be higher in patients with IgAN than in HCs. The mRNA level of FcγRIIb in IgAN has been decreasing even without statistical significance. To validate the RNA-seq findings, quantitative reverse transcription–polymerase chain reaction (qRT-PCR) was conducted on type I FcγRs isolated from PBMCs of an independent group of 20 IgAN patients and 20 HCs to identify mRNA levels of type I FcγRs. We observed that the expression levels of activating FcγR mRNAs were consistently higher in patients with IgAN (Figure 1).
The type I FcγRs correlated with the susceptibility of IgAN.

IgAN, immunoglobulin A nephropathy.
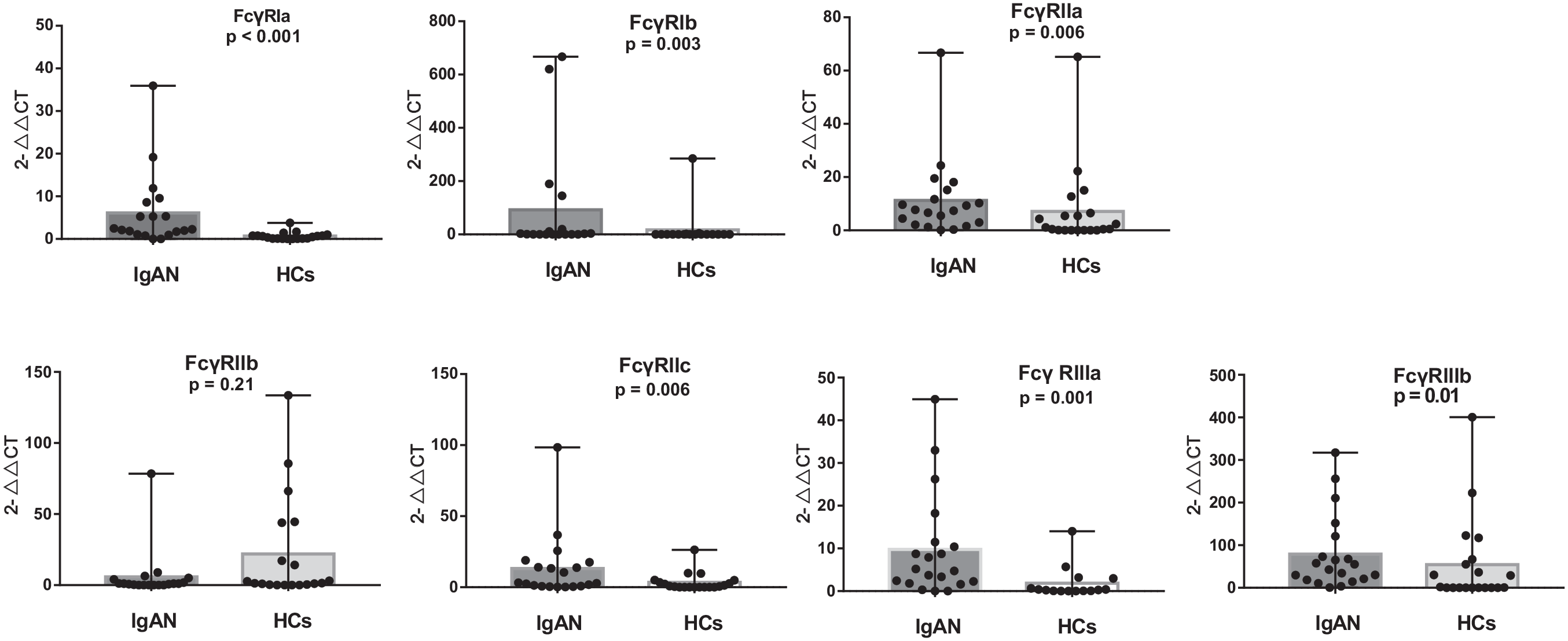
Validation of differential expression of FcγRIa, FcγRIb, FcγRIIa, FcγRIIb, FcγRIIc, FcγRIIIa, and FcγRIIIb in an independent set of PBMCs from 20 patients with IgAN and 20 healthy controls.
Patients with IgAN had high levels of FcγRIIIb
FcγRIIIb was the IgG receptor with the greatest difference (log2 fold-change = 1.82) in expression between the two groups. In the next step, we compared the level of FcγRIIIb in PBMCs by flow cytometry. The results revealed the FcγRIIIb level was increased in patients with IgAN (19.22% ± 6.82% versus 14.01% ± 4.73%, p = 0.02, Figure 2). We also measured plasma sFcγRIIIb levels in 50 patients with IgAN and 50 HCs. The findings revealed that the mean sFcγRIIIb level in plasma in participants with IgAN was 103.42 ng/ml, which was significantly higher than the mean level in plasma in HCs (74.60 ng/ml, p = 0.03, Figure 3).
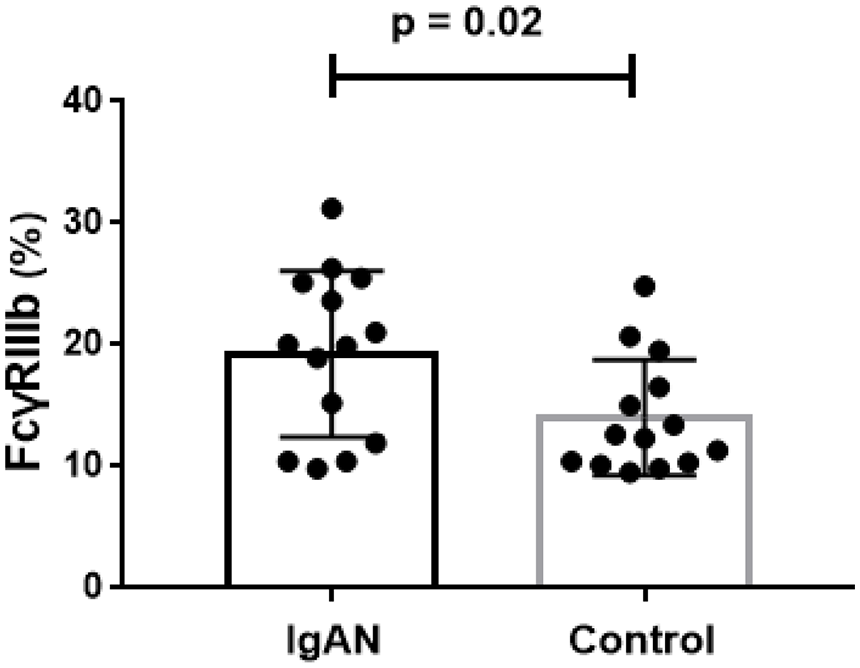
FcγRIIIb levels in PBMCs from patients with IgAN and healthy controls by flow cytometry.

Plasma soluble FcγRIIIb levels in 50 patients with IgAN and 50 healthy controls.
Plasma FcγRIIIb levels correlated with the severity of IgAN
We investigated the link between plasma sFcRIIIb and clinical symptoms and pathological abnormalities in patients with IgAN. Patients were separated into two groups based on the median value of their plasma sFcγRIIIb levels (90 ng/ml). We discovered that those with higher sFcγRIIIb levels (> 90 ng/ml) had significantly higher body mass index (BMI), triglyceride, serum creatinine, and a significantly higher proportion of sclerosis (⩽90 ng/ml), as shown in Table 3. Patients in the group with higher sFcγRIIIb levels got more glucocorticoids than those in the lower sFcγRIIIb group (Table 3).
The baseline data for IgAN patients with lower and higher soluble FcγRIIIb levels.
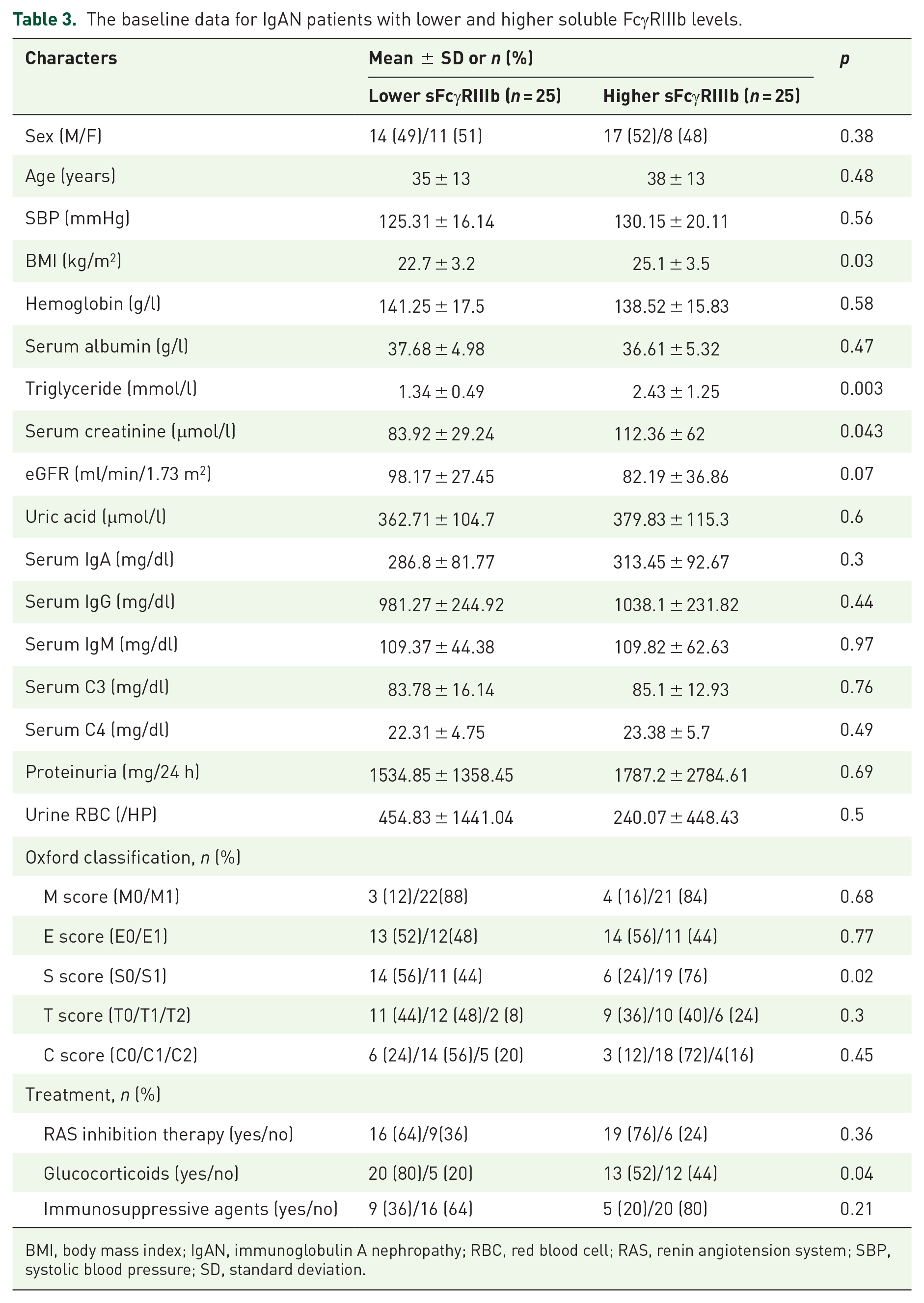
BMI, body mass index; IgAN, immunoglobulin A nephropathy; RBC, red blood cell; RAS, renin angiotension system; SBP, systolic blood pressure; SD, standard deviation.
Discussion
Activating FcγR expression levels in IgAN patients were investigated for the first time in this research. The current research found that increased activating FcγRs mRNA expression was associated with increased IgAN susceptibility. Among them, plasma levels of sFcγRIIIb were shown to be correlated with the severity of IgAN. Following these observations, it was hypothesized that the activating type I gamma receptor for the Fc portion of the IgG molecule is critical in controlling immunological attack in patients with IgAN.
FcγRs are now recognized as the important molecules, which are responsible for coupling antigen recognition by IgG antibodies to cellular effector pathways in macrophages, neutrophils, natural killer cells, and mast cells.12,13 They are primarily separated into activating and inhibitory receptors based on their binding stoichiometry and the recognized Fc sites of IgG. The activating FcγR/CD64, FcγRIIa/CD32a, and FcγRIII/CD16 are intracytoplasmic immunoreceptor tyrosine-based activating motifs that bind Fc at its hinge-proximal region and cause monocyte activation following receptor aggregation. 14 FcγRIIb, on the contrary, is characterized by the presence of a tyrosine-based immunoreceptor inhibitory motif and functions as an inhibitory FcγR. 14 It is decided by the balance between activating and inhibiting FcγRs in monocyte-derived DCs. 15 Ravetch and Bolland 16 hypothesized that increased activating FcγR expression or reduced inhibitory FcγR expression might result in the production of pro-inflammatory cytokines and contribute to the development of lupus nephritis. Our findings were consistent with the hypothesis that mRNA levels of activating FcγRs were increased, whereas the expression of inhibitory FcRIIb was decreased in our experiments. In SLE, circulating monocytes activated by ICs may upregulate the surface expression of FcγRI/CD64. 8 Further investigation will be required to identify which factors, such as IgG or IgG-ICs, have an effect on the expression of FcRs.
Gene polymorphism studies have provided more evidence for the involvement of FcγRs in the pathogenesis of IgAN. Zhou et al. 17 explored genetic correlations between the FcγR gene locus in whole blood and susceptibility to IgAN. There were 15 gene variations within the FcγR gene locus detected by the researchers, and they were shown to be associated with IgAN susceptibility. Two genes FcγRIIa and FcγRIIb were found to be shared genes between IgAN and SLE. 18
It is particularly interesting to investigate the involvement of FcγRs in IgAN due to their expression in myeloid cells and B cells, and the fact that they were the effector molecules in mediating antibody/IC effects. The activation of FcγRs triggers the activation of pro-inflammatory signaling pathways. 16 The downstream effects of activating FcγRs include antibody-dependent cell-mediated cytotoxicity (ADCC), antibody-dependent cellular phagocytosis (ADCP), leukocyte activation, and pro-inflammatory cytokine release. Chronic inflammation is often seen in IgAN patients, particularly in the circulation and kidney.19,20 Mucosal infection seems to have an important role in the development of Gd-IgA and IgAN, notably in the phenotype of episodes of visible hematuria, according to an increasing body of evidence. Gd-IgA1-IgG deposits in the mesangial area of glomeruli and stimulates the inflammatory cytokine production and complement activation. However, it is unknown whether IgG or IgG-ICs binding to activating FcγRs results in cellular proliferation and the production of inflammatory mediators, which may contribute to kidney injury in IgAN patients. More study is required to confirm the underlying role of FcγRs in the development of IgAN.
FcγRIIIb, also known as CD16b, is a low-affinity receptor for aggregated IgG or IgG-containing ICs abundantly expressed on human granulocytes. 21 Excessive neutrophil activation and the production of large quantities of cytolytic and pro-inflammatory chemicals are possible consequences of the interaction of IgG-containing immunoglobulins with FcγRIIIb, which may result in severe tissue damage.22,23 The CD16b receptor is released from the surface of leukocytes when they are activated in an inflammatory environment. 24 Many investigations have shown a significant association between CD16b gene polymorphisms and SLE susceptibility.21,25–27 There was a statistically significant rise in the levels of soluble CD16b (sCD16b) in urine in lupus nephritis patients. 28 In the current investigation, we observed a much higher percent of FcγRIIIb positive cells in IgAN by flow cytometry. It was discovered that the plasma sFcγRIIIb level in IgAN patients was increased. A greater sFcγRIIIb level was associated with higher BMI, lipid, serum creatinine, and sclerosis proportions in patients, as well as increased chances of being exposed to glucocorticoid medications. IgAN susceptibility and severity might be predicted by the increased expression of FcγRIIIb/CD16b, which could be employed as a biomarker.
However, there were certain limitations to the research conducted. First, the small sample size precluded further investigation into the course of IgAN patients. Second, due to a scarcity of ELISA kits for other soluble IgG receptors, we could only detect sFcγRIIIb levels in the bloodstream. Third, there is a lack of evidence regarding CD16b cleavage in vivo. Whether the interaction of IgG-containing ICs with FcRIIIb induces proteolytic shedding of sFcγRIIIb into the circulation, resulting in the formation of new circulatory Gd-IgA1-IgG-FcRIIIb complexes, is unknown. To understand more about the precise role of FcγRs in IgAN, additional research in gene-targeted animal models or cell lines would be required.
Conclusion
The mRNA levels of the activating Fc receptor of IgG were found to be higher in IgAN than in HCs. Patients with higher plasma sFcγRIIIb levels may have had more severe illness than those with lower levels. Future research could focus on the involvement of FγcRs in the pathophysiology of IgAN.
Footnotes
Acknowledgements
The authors thank all the study subjects for their participation.
Ethics approval and consent to participate
All subjects provided written informed consent. The study protocol was approved by the Institutional Ethical Committee of Tianjin Medical University General Hospital (IRB2020-KY-155).
Consent for publication
Not applicable.
Author contributions
Availability of data and materials
Raw data used during this study are available from the corresponding author on reasonable request for non-commercial use.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study is supported by the National Natural Science Foundation (grant nos. 81600553 and 82000669).
Conflict of interest statement
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
