Abstract
Objective:
Mounting evidence has demonstrated that various chronic inflammatory diseases are associated with incident heart failure (HF). However, there is scarce evidence about the association between primary Sjögren’s syndrome (pSS) and HF. We aimed to explore this association using a nationwide database in Taiwan.
Methods:
We selected patients with incident pSS and no history of HF. Using propensity score matching based on age, sex, cohort entry time, comorbidities, and concomitant medications, cohorts of patients with and without pSS (as controls) were created in a 1:1 ratio and the groups were compared. The cumulative incidence of HF was calculated using Kaplan–Meier estimation. Cox proportional hazard regression analysis was used to measure the hazard ratio (HR) of HF-related hospitalization for the pSS cohort compared with the comparison group.
Results:
A total of 16,466 pairs of patients with pSS and those without pSS were identified. The cumulative incidence of HF-related hospitalization at 3, 5, and 10 years in patients with pSS was 1.05%, 1.89%, and 4.33%, respectively. The risk of HF-related hospitalization was not higher in patients with pSS than in the general population (HR: 0.98, 95% confidence interval [CI]: 0.84–1.14). There was no difference in survival probability after the first episode of HF-related hospitalization between pSS and non-pSS groups.
Conclusion:
Our results suggest that distinct inflammatory spectrums in each chronic inflammatory disease might have differential impacts on cardiac function and subsequent risk of HF. Future studies are needed to elucidate the complex and diverse mechanisms of HF in various chronic autoimmune diseases.
Introduction
Primary Sjögren’s syndrome (pSS) is a chronic immune-mediated inflammatory disorder characterized by hyperactivity of B cells and lymphocytic infiltration in the exocrine glands, leading to symptoms of oral and ocular dryness.1,2 Other common extraglandular manifestations in pSS include tubulointerstitial nephritis, interstitial lung disease, cutaneous vasculitis, peripheral neuropathy, and non-Hodgkin lymphoma.3,4 Cardiovascular complications have been recognized as a major cause of long-term morbidity and mortality in patients with chronic inflammatory diseases, such as rheumatoid arthritis (RA) and systemic lupus erythematosus (SLE), largely because of sustained systemic inflammation.5 –9 Although direct cardiac involvement is rare, patients with pSS were found to be at a higher risk of coronary heart disease in a recent epidemiological study. 10 However, the association between pSS and other cardiac complications apart from coronary disease has been rarely investigated and remains unclear.
Heart failure (HF) is a complex clinical syndrome, and its prevalence is increasing worldwide.11,12 The prognosis is poor once patients are admitted due to acute exacerbation of HF.13,14 In addition to the established role of sustained neurohormonal activation in the pathogenesis of HF, immune system activation and systemic inflammation are known to be important in the initiation and progression of HF.15,16 Several systemic autoimmune diseases, such as systemic sclerosis (SSc), polymyositis/dermatomyositis (PM/DM), and RA, have been demonstrated to be associated with significantly higher risk of HF in the more recently published studies,17 –19 and it has been argued that these chronic inflammatory diseases could be considered as Stage A HF. 20 However, whether patients with pSS have a higher risk of subsequent HF due to the above-mentioned inflammatory conditions is largely unknown because of the lack of large-scale and long-term follow-up studies.
Therefore, we conducted a retrospective cohort study to investigate the possible association between pSS and incident HF using a nationwide population-based database. We also sought to compare the survival rates after HF-related hospitalization between patients with pSS and non-pSS controls.
Methods
Data source
All data in this study were obtained from Taiwan’s National Health Insurance Database (NHIRD). A single-payer and mandatory national health insurance program was initiated in Taiwan in 1995, and over 99% of the citizens have been enrolled in this program until now. The universal insurance program provides a broad range of coverage for healthcare services, including ambulatory and inpatient care, laboratory and radiological examinations, surgical procedures, and a large number of prescription medications. This medical information was collected by Taiwan’s government and was used to construct the NHIRD after a strict process of encrypting personally identifiable information. Therefore, the NHIRD contains significant and valuable medical data and has been used extensively in epidemiological research.4,21,22
Ethical approval
This retrospective cohort study was approved by the Institutional Review Board of National Cheng Kung University Hospital (A-EX-109-017), and informed consent was waived as the database used in the current study was deidentified.
Selection of patients and comparison group
Patients diagnosed with pSS from 1 January 2000 to 31 December 2013 were identified from the catastrophic illness registry, a subset of NHIRD. Individuals in Taiwan who have been diagnosed with several critical disorders, such as malignancy, end-stage renal disease, or autoimmune diseases, are issued catastrophic illness certificates for exemption from co-payment when they seek medical services related to their underlying catastrophic illness. The approval of specific catastrophic illness certificates is confirmed by associated specialists based on the relevant medical records, results of laboratory and imaging studies, or formal pathology reports. For example, only when the associated symptoms/signs and results of examinations in patients with pSS fulfilled the revised American-European Consensus Group (AECG) classification criteria or the European classification criteria23,24 and were subsequently confirmed by two independent board-certified rheumatologists, would patients be issued a pSS certificate, and this information was recorded in the catastrophic illness registry. Therefore, the reliability and accuracy of this method for identifying patients with pSS were high. The date when the diagnosis of pSS was made for the first time was defined as the index date, and the age of patients with pSS at the index date was documented. Patients aged < 20 years or with a diagnosis of HF preceding the index date were excluded from the analysis. We also searched all available medical records for each patient in the period before the index date to ensure that only patients with incident pSS were included in our study. We excluded individuals who had other autoimmune diseases, such as SLE or RA, to ensure that no patients with secondary Sjögren’s syndrome or overlap syndrome were included in our study.
Propensity-score matched comparison cohort
Individuals who had never been diagnosed with pSS were selected from our database to be included in the comparison cohort. The index dates of patients in the non-pSS comparison cohort were randomly assigned matching to the distribution of those of the pSS cohort. Therefore, the entry times of the case and control cohorts were matched. We then estimated the propensity score through a logistic regression model that was constructed considering the presence or absence of pSS as dependent variables, with age, sex, selected comorbidities, and concomitant medications (type of comorbidities and medications are described below) as explanatory variables. Patients with pSS were matched with the comparison cohort by propensity score in a 1:1 ratio. We implemented the nearest-neighbor matching with a caliper width of 0.1 of the pooled standard deviation of the logit of the calculated propensity score. The other confounders between the disease and control groups were examined using absolute standardized mean difference (ASMD), and ASMD less than 0.1 was considered as an acceptable match. 25
Covariate
We searched the following comorbidities based on the International Classification of Diseases, Ninth Revision, Clinical Modification (ICD-9-CM) codes in the database during the 1-year period preceding the index date in both pSS and comparison cohorts: diabetes (ICD-9-CM code 250), hypertension (ICD-9-CM codes 401–405), ischemic heart disease (ICD-9-CM codes 410–414), dyslipidemia (ICD-9-CM codes 272), atrial fibrillation (ICD-9-CM codes 427.31), chronic kidney disease (ICD-9-CM codes 580–588), COPD (ICD-9-CM codes 491, 492, 496), and peripheral artery disease (ICD-9-CM codes 443). The presence of comorbidities was ascertained by the respective ICD-9-CM codes, recorded in the outpatient claims thrice or more, or in the hospitalization claims at least once.
Information regarding exposure to concomitant medications, which might be associated with the occurrence of HF, was obtained from the prescription records. The drugs considered in the current study included aspirin, nonsteroidal anti-inflammatory drugs (NSAIDs), statins, corticosteroids, digoxin, angiotensin-converting enzyme inhibitor/angiotensin receptor blocker (ACEI/ARB), β-blocker, loop diuretics, and spironolactone. We only captured the use of three β-blockers (metoprolol, carvedilol, and bisoprolol), since other types of β-blockers have not been proven to reduce the risk of hospitalization for HF. Patients were defined as drug users if the corresponding drugs were dispensed for 28 consecutive days or more during the 90-day interval preceding and after the index date, except for NSAID exposure. NSAIDs are frequently prescribed but usually intermittently in patients with chronic inflammatory disease; NSAID use was defined as intake of NSAIDs for three consecutive months or more during the baseline assessment period.
Assessment of outcome
The primary outcome was the first hospitalization for HF, which was identified through ICD-9-CM codes 428 from the discharge summary. These codes were previously validated, with a specificity of 97.8% in our database. 26 The secondary outcome was survival rate following the occurrence of hospitalization for HF. Patients were followed until the occurrence of the outcome of interest, death, or the end of the study period (31 December 2013), whichever occurred first.
Statistical analysis
The demographic characteristics of the pSS and non-pSS groups are expressed as mean (standard deviation) or numbers (percentage), as appropriate. The difference between the groups was determined using the student’s t-test or Pearson’s chi-square test, depending on the continuous or categorical variables compared. The incidence rate of HF was calculated as the number of HF events divided by the total follow-up duration of all patients at risk. The cumulative incidence of HF and survival following hospitalization for HF are graphically presented by Kaplan–Meir estimation, and the log-rank test was used to compare the difference between the pSS and non-pSS cohorts. Cox proportional hazard regression models were used to analyze the direction and strength of the impact of pSS on the risk of hospitalization for HF. The assumption of proportional hazards was evaluated by plotting the log[–log(survival)] versus log(time) curve and by conducting the scaled Schoenfeld residuals test; the results did not reveal any evidence of non-proportional hazards.
The likelihood ratio test was used to determine whether a significant interaction effect existed between the pSS and pre-specified subgrouping variables. All statistical analyses were performed using STATA software (version 13.0; StataCorp LLC, College Station, TX, USA), and p values of less than 0.05, were considered statistically significant.
Power analysis
We assumed that an overall event rate of hospitalization for HF in this observational study was 4%, and a hazard ratio (HR) of 1.2 in the pSS group versus non-pSS group. The required sample size was estimated with the use of a two-sided test at a 0.05 significance level and 90% power level. It was determined that a total of 31,610 patients (15,805 pairs of pSS patients and controls) should be included in the current study.
Sensitivity analysis
Three additional sensitivity analyses were conducted to validate the findings of the main analysis. The first was the use of diagnostic codes with high sensitivity (96.3%) for HF (ICD-9-CM: 425.4, 425.9, 402.01, 402.11, 402.91, 404.01, 404.03, 404.11, 404.13, 404.91, 404.93, and 428.xx) to capture the primary outcome 27 instead of diagnostic codes with high specificity in the main analysis. The second sensitivity analysis was the competing risk regression using the Fine and Gray model to derive the HR considering death as a competing event for HF-related hospitalization. 28 The third sensitivity analysis was the estimation of HR after excluding patients who experienced HF events or were followed for less than 1 year; this was to minimize the risk of including prevalent cases of HF in our cohort.
Results
Baseline demographics and characteristics of pSS and comparison group
In all, 19,037 patients with pSS and no history of HF were identified. The average age at diagnosis of pSS was 54.5 ± 13.8 years, and females constituted nearly 90% of the pSS cohort (Table 1). The most common comorbidities at baseline were hypertension, followed by dyslipidemia, diabetes, ischemic heart disease, and other comorbid conditions, accounting for less than 5%. The most commonly prescribed medications were corticosteroids and NSAIDs. After 1:1 propensity score matching, there was a total of 16,466 pairs of subjects; each pair had one subject with pSS and other with no-pSS. The distribution of all potential confounders between the pSS and non-pSS groups were similar, as seen from the results of ASMD (all less than 0.1).
Baseline characteristics of the patients with primary Sjögren’s syndrome (pSS) and the matched groups for comparison.

ACEIs/ARBs, angiotensin-converting enzyme inhibitors/angiotensin receptor blockers; ASMD, absolute standardized mean difference; COPD, chronic obstructive pulmonary disease; NSAID, nonsteroidal anti-inflammatory drug; RA, rheumatoid arthritis; SD, standard deviation.
Incidence rate and relative risk of hospitalized HF
We identified 641 events of incident hospitalization for HF over a median of approximately 4 years. The absolute and relative risks of hospitalization for HF are presented in Table 2. The number and percentage of HF events were similar and observed in 318 (1.9%) and 323 (1.9%) patients in the pSS and non-pSS groups, respectively. The corresponding incidence rates of HF were 4.02 and 4.18 per 1000 person-years in the pSS and non-pSS groups, respectively. Among patients with pSS, the cumulative 1-, 3-, 5-, and 10-year incidences of HF were 0.3%, 1.05%, 1.89%, and 4.33%, respectively. Among the non-pSS cohort, the cumulative 1-, 3-, 5-, and 10-year incidences of HF were 0.27%, 1.10%, 1.89%, and 4.44%, respectively. There was no significant difference in the cumulative HF probability between the pSS and non-pSS groups (Figure 1, p value by log-rank test: 0.75). The Cox regression model revealed that the risk of HF in patients with pSS was similar to that in the non-pSS group (HR: 0.98, 95% confidence interval [CI]: 0.84–1.14).
Incidence rate and risk of HF-related hospitalization in patients with primary Sjögren’s syndrome (pSS) compared with the propensity score-matched control group.

CI, confidence interval; HR, hazard ratio; HF, heart failure.

Cumulative incidence of hospital admission for heart failure in patients with primary Sjögren’s syndrome (pSS) compared with the propensity score-matched control group.
Survival following first HF hospitalization among patients with and without pSS
There was no significant difference in the survival rate after hospitalization for HF between the pSS and non-pSS cohorts (Figure 2, p value by log-rank test: 0.20). Due to the possibility of difference in age and sex distribution between the pSS and non-pSS cohorts at the time of hospitalization for HF, we used Cox regression models adjusting for age and sex to compare the risk of death following HF, and no significant difference was observed (HR: 1.11, 95% CI: 0.89–1.40, p = 0.33).

Cumulative survival after hospitalization for heart failure (HF) in patients with primary Sjögren’s syndrome (pSS) versus non-pSS controls.
Subgroup analysis
Stratified analyses according to age and sex are shown in Table 3. The effect estimates of pSS on the risk of hospitalization for HF were similar across these two pre-determined subgroups. There were also no significant interactions detected using the likelihood ratio test in the male population versus the female population, and the older population versus the younger population (both p values for interaction > 0.05).
Subgroup analysis of the risk for HF by sex and age in patients with primary Sjögren’s syndrome (pSS) and matched cohort. Effect of pSS on the risk of heart failure (HF) across various pre-specified subgroups is presented.

CI, confidence interval; HR, hazard ratio; HF, heart failure.
Adjusted HRs were estimated using a fully adjusted Cox proportional hazards model adjusted for sex, age, comorbidities, and concomitant medications (Table 1), except for the subgrouping variable.
p values for interaction were determined using the likelihood ratio test.
Sensitivity analysis
The results of multiple sensitivity analyses are presented in Table 4. When diagnostic codes with high sensitivity were used to capture the HF events from hospital discharge records, the estimated risk for HF was similar to that seen in the primary analysis (HR: 0.96; 95% CI: 0.83–1.11). Additional sensitivity analysis using the competing risk regression (Fine and Gray’s model) or excluding participants who were followed for less than 12 months also did not show a significant change in HR.
Sensitivity analysis.
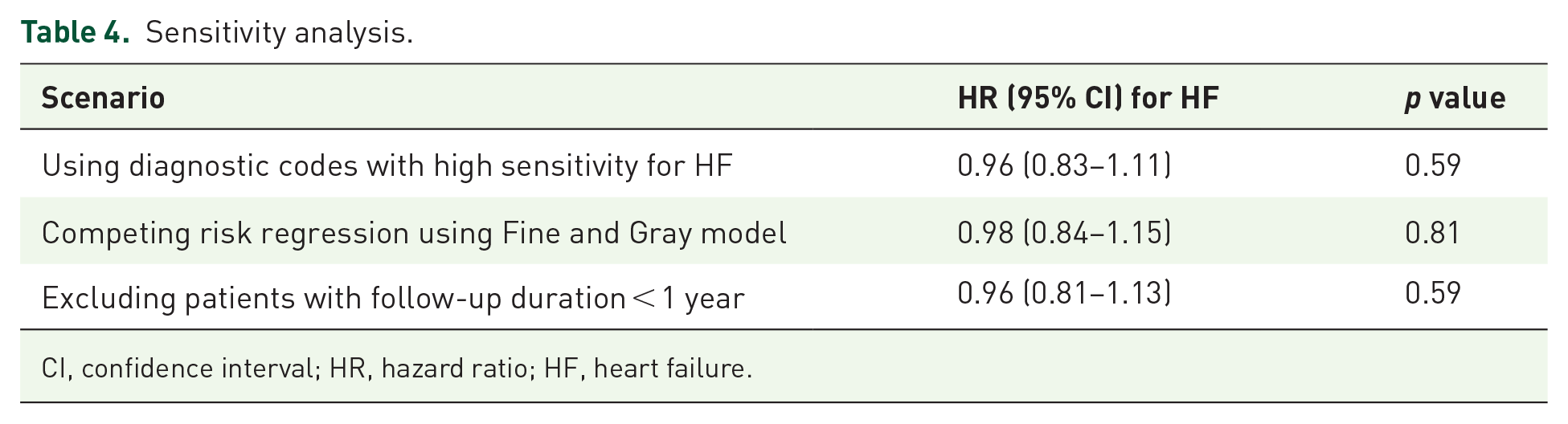
CI, confidence interval; HR, hazard ratio; HF, heart failure.
Discussion
To the best of our knowledge, this is the first nationwide cohort study to investigate the association between pSS and incident HF. The study showed that the risk of hospitalization for HF was not higher among patients with pSS compared with propensity score-matched individuals without pSS. Subgroup analysis according to age and sex also yielded consistent results. Moreover, there was no difference in survival following the episode of HF requiring admission between patients with and without pSS.
Activation of the innate and adaptive immune system has been recognized to be associated with the development and progression of HF in recent years, particularly HF with preserved ejection fraction. 16 The results of a recent randomized trial showed that treatment with canakinumab, a monoclonal antibody against interleukin-1β, was associated with a significant reduction in HF-related hospitalization and mortality, 29 suggesting that systemic inflammation plays a crucial role in the pathogenesis of HF and is not only a biomarker of tissue damage. Several large-scale observational studies published recently revealed that individuals with chronic inflammatory diseases, including PM/DM, SSc, SLE, and RA, were at an increased risk of incident HF,17 –20 providing further evidence of a link between inflammation and onset of HF. Although pSS shares some clinical and immunological features with SLE or RA,30,31 we failed to demonstrate that the risk of hospitalization for HF was higher in patients with pSS than in the general population after accounting for multiple confounders. One possible explanation is that, unlike SLE and RA, pSS often progresses slowly and has a more indolent disease course. 32 The need for intensive immunosuppressive therapy and dosage of corticosteroid was also less compared to that required in other autoimmune diseases, 33 suggesting that the inflammatory burden is lower in pSS and the subsequent negative effects on the heart caused by systemic inflammation is less pronounced. Another explanation is that the relationship between inflammation and HF is complex, and the role of different immune cells in the development of HF might not be equal. Sustained B-cell activation and dysregulation are the hallmarks of pSS. 1 However, the exact role of B cells in chronic HF-associated myocardial inflammation remains largely unknown and poorly described. 16 In contrast, there have been several evidences demonstrating the increase and activation of T cells in the hearts of patients with HF or murine models of chronic HF.34 –36 Previous studies in patients with non-ischemic HF have revealed recruitment of T cells in the heart and increased infiltration of CD3 + T cells in the left ventricle compared to that in healthy participants.36,37 Therefore, distinct inflammatory profiles and different adaptive immune responses in patients with pSS compared to those with other autoimmune diseases characterized by T cell dysregulation might have a differential impact on the myocardial function and subsequent development of HF.
A recent systemic review and meta-analysis also investigated the burden of cardiovascular morbidity in patients with pSS, which, in contrast to our study, reported that the risk of HF in pSS to be 2.54 times the risk in the general population. 38 High heterogeneity was present in the three studies included in this meta-analysis when analyzing the risk of HF in pSS, and their results were mainly driven by the study conducted by Vassiliou et al. 39 (odds ratio of HF: 5.77 in pSS versus controls). In the study by Vassiliou et al., 39 echocardiography was used to detect subclinical abnormalities in pSS patients with no overt cardiac disease or clinical symptoms; however, in our study, using clinically evident HF requiring hospitalization was the main outcome. In addition, while Vassiliou et al. only reported the number of patients with mitral, aortic, and tricuspid regurgitation, these patients were counted as having HF in the meta-analysis by Beltai et al. 38 Such a definition of HF may overestimate the association between pSS and HF. Similarly, in the study by Chiang et al., 40 only the number and percentage of patients with valvular heart disease were reported. These patients were also considered as having HF in the meta-analysis by Beltai et al. 38 However, another multicenter retrospective cohort study from Italy did not demonstrate a higher prevalence of HF in pSS than in controls; 41 this is consistent with our findings.
Several secretagogues, such as pilocarpine, have been frequently used for the relief of oral dryness in patients with pSS. 42 While a small double-blind trial reported that palpitations were noted in 13% patients undergoing pilocarpine treatment, no evidence of arrythmia was observed in that trial.43,44 On the other hand, there was a animal study that showed that pilocarpine may exert antiarrhythmic effect via stimulating muscarinic acetylcholine receptors in the cardiac tissue. 45 Other side effects of pilocarpine on the cardiovascular system included vasodilatation, possibly a consequence of generalized parasympathomimetic stimulation. 46 Therefore, the long-term impact of pilocarpine on the development of overt cardiovascular diseases or HF remain to be elucidated. We did not capture the prescription records of pilocarpine in our study, and whether it would have influenced our results is unclear and requires further investigation.
Our study had several strengths. First, using a comprehensive nationwide database allowed us to capture a large number of unselected patients with pSS and to follow them for a long period with minimal drop-out, which reduced the risk of selection bias to a minimum compared to that in community or hospital-based cohort studies. Second, the correctness and accuracy of both the exposure and outcome variables in our study were validated. Thus, we believe that our results are unaffected due to misclassification bias. Third, HF hospitalization was used as our primary outcome instead of the diagnosis of HF in the ambulatory setting, which could minimize the risk of detection bias. Briefly, detection bias may occur when physicians are more alert and suggest more investigations to search for asymptomatic or subclinical disorders during the follow-up of patients with pSS. However, HF hospitalization is an acute event and is a more objective evaluation than the outpatient diagnosis of HF, and it is associated with disease progression and prognosis. Moreover, the data of date of hospitalization for HF are likely to be more precise than that of diagnosis during outpatient care, which is particularly important as we used Cox regression to model the time-to-event data. Therefore, using this approach enabled our study to be immune to detection bias.
Some limitations of our study should be acknowledged. First, information about some potential environmental or lifestyle risk factors for cardiovascular diseases, such as socioeconomic status or smoking, was not available in our database. Second, detailed symptoms/signs and relevant tests in patients with pSS were not recorded. Therefore, we could not evaluate the disease activity and severity of pSS and its impact on the incidence of HF-related hospitalization. Third, due to the lack of results of echocardiography and biochemical data, differentiating between the phenotypes of HF in terms of ejection fraction status or severity of HF in patients experiencing HF events was difficult.
Conclusion
Although increasing evidence in recent years has revealed that a number of chronic inflammatory diseases are associated with incident HF, we failed to demonstrate this association in patients with pSS based on a largely unselected cohort. Our findings imply that the degree of inflammation and difference in inflammatory profiles between different autoimmune diseases might have a distinct impact on the onset and progression of HF. Further studies are required to elucidate the complex and diverse mechanisms of HF in various chronic inflammatory diseases.
Footnotes
Author contributions
Availability of data and materials
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was partly supported by Kaohsiung Chang-Gung Memorial Hospital grant: CORPG8K0131 and by grant NCKUH-11003017 from the National Cheng Kung University Hospital, Tainan, Taiwan.
Conflict of interest statement
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics approval and consent to participate
The Institutional review board of National Cheng Kung University Hospital approved this study (A-EX-109-017). Informed consent was not required because the datasets were devoid of personally identifiable information.
