Abstract
Aim:
To evaluate the renal outcomes and prognostic factors among patients with type-2 diabetes (T2D) and biopsy-confirmed diabetic nephropathy (DN), non-diabetic renal disease (NDRD) and DN mixed with NDRD (MIX).
Design and Methods:
Patients with both T2D and chronic kidney disease (CKD) who underwent renal biopsy between January 2014 and December 2016 were recruited in this prospective observational study. Participants were divided into DN group, NDRD group, or MIX group according to the baseline pathological diagnosis. The primary endpoint was a composite renal event of end-stage renal disease (ESRD) or ⩾ 40% reduction in estimated glomerular filtration rate (eGFR).
Results:
Among the 292 participants included, 153 (52.4%) belonged to the DN group, 30 (10.3%) belonged to the NDRD group, and 109 (37.3%) belonged to the MIX group. During the median follow-up of 27 months, the adverse renal events occurred in 132 (44.2%) patients. Compared with NDRD group, the multiple adjusted hazard ratios (HRs) for renal events in patients with DN and MIX groups were 3.900 (95% confidence interval [CI]: 1.103–13.788) and 2.691 (95% CI: 0.662–10.936), respectively. Baseline lower eGFR (HR: 1.159, 95% CI: 1.060–1.266), severe proteinuria (HR: 2.047, 95% CI: 1.227–3.416), lower hemoglobin (HR: 1.170, 95% CI: 1.008–1.267), and a family history of diabetes (HR: 1.138, 95% CI: 1.008–2.285) were independent predictors for adverse renal outcomes in patients with DN.
Conclusion:
In patients with T2D and CKD, pure DN and MIX group displayed a worse renal prognosis than NDRD group. Worse renal function, severe proteinuria, lower hemoglobin, and a family history of diabetes may be associated with adverse renal outcomes in patients with DN.
Keywords
Introduction
Diabetic nephropathy (DN) is one of the most common complications of type-2 diabetes (T2D) and the leading cause of end-stage renal disease (ESRD) worldwide.1,2 The extensive application of renal biopsy indicates that coincident chronic kidney disease (CKD) in patients with T2D can be DN, non-diabetic renal disease (NDRD), or DN mixed with NDRD. 3 Clinical manifestation and management of CKD vary widely among different pathologic types. However, the results of existing studies on the prognosis of different pathologic types were inconsistent, possibly due to differences in study population, inclusion criteria, and sample size limitations.4–6
Of note, as an invasive diagnostic modality, renal biopsy is still infrequently performed in T2D patients. 7 Diagnostic renal biopsy is usually recommended in patients suspected to have NDRD, especially in those with the presence of large amount of proteinuria, rapid decline of renal function, absence of retinopathy and persistent glomerular hematuria. 8 Most patients diagnosed as DN in previous clinical trials did not receive renal biopsy. However, the diagnosis only based on clinical features might increase the likelihood of misdiagnosing NDRD or the combination of NDRD and DN as “DN.” Therefore, it remains a challenge to evaluate the prognostic predictors beyond pathological classification in patients with T2D and atypical renal damage. Hence, we conducted this prospective observational study to investigate the difference of clinical characteristics and prognosis among biopsy-confirmed DN, NDRD, and DN mixed with NDRD and to identify the baseline risk factors for adverse renal outcomes.
Materials and methods
Research design and participants
Patients with T2D and CKD who underwent renal biopsy in the First Affiliated Hospital of Zhengzhou University between January 2014 and December 2016 were recruited in this study. Inclusion criteria were as follows: (1) baseline age ⩾18 years; (2) clinically diagnosed as type-2 diabetes; (3) clinical manifestations and laboratory examination results suggested the presence of CKD; (4) the renal biopsy had already performed, and the pathological diagnosis was clear. Diagnostic criteria for T2D were based on the recommendations of the American Diabetes Association. 9 CKD was defined as the elevated urinary albumin excretion (albuminuria ⩾ 30 mg/24 h) and/or low estimated glomerular filtration rate (eGFR < 60 mL/min/1.73 m2) sustained for at least 3 months. The exclusion criteria included: (1) patients with type-1 diabetes or other special types of diabetes; (2) patients with incomplete clinical or pathological data; (3) patients who already had an ESRD prior to or near (less than 30 days) renal biopsy; (4) patients with acute kidney injury, polysistic kidney disease, or kidney transplantation; and (5) patients with autoimmune system diseases, malignant tumors, or severe infectious diseases (a flowchart of the study was presented in Figure 1).

Flowchart depicting the study design.
The study was complied with the Declaration of Helsinki and was approved by the ethics committee of the First Affiliated Hospital of Zhengzhou University (No. KY-2013-LW-1282). Written informed consent was obtained from each participant.
Calculation of sample size
We performed sample size calculations using PASS 15.0. No published data are available to contribute to estimate the needed sample size of MIX group. Based on previous literature and our experience, it was assumed that the proportion of the DN patients to NDRD patients will be 5:1. The 5-year event rate was assumed to be no more than 10% in the NDRD group and 40% in the DN group. A sample size of 133 patients (22 in NDRD and 111 in DN) provides a statistical power of more than 90% for a K–M analysis of comparison of event rates between DN and NDRD groups at 5% level of significance. A Cox proportional hazard regression-based analysis provides a statistical power of 90% for the noted comparison, assuming the overall event rate in the study is 30.0%.
Demographic and laboratory data collection
The baseline demographic data and laboratory parameters were collected at the time of renal biopsy, including age, gender, body mass index (BMI), duration of diabetes, family history of diabetes, presence of diabetic retinopathy (DR), presence of hypertension, blood pressure (systolic blood pressure and diastolic blood pressure), fasting plasma glucose (FPG), hemoglobin A1c (HbA1c), hemoglobin (Hb), serum albumin, blood lipids (total cholesterol (TC), triglyceride (TG), high-density lipoprotein (HDL), and low-density lipoprotein (LDL)), urinary red blood cell, blood urea nitrogen (BUN), blood uric acid, 24-h urine protein, serum creatinine, and eGFR.
BMI was calculated using the formula BMI = weight (kg)/height (m2). Hypertension was defined as systolic blood pressure ⩾140 mmHg, diastolic blood pressure ⩾90 mmHg, or the use of anti-hypertensive drugs. Other information such as age, gender, medical history, smoking, and alcohol consumption history were collected by self-administered questionnaires. DR was diagnosed by direct ophthalmoscopy performed by an experienced ophthalmologist. Microscopic hematuria was defined as three or more red blood cells per high-power field in a centrifuged urine sample. The eGFR was calculated using the CKD-EPI formula. 10
Renal biopsy and pathological classification
Informed consent was obtained from each patient when the renal biopsy was performed. In our center, renal biopsy was usually indicated in patients with diabetes under the suspicion of NDRD, and the indications mainly including rapid onset of proteinuria, absence of DR, presence of unexplained hematuria, active urinary sediment, and rapid decrease of renal function.
All biopsy specimens were obtained by percutaneous needle biopsy and were routinely processed for light microscopy, immunofluorescence, and electron microscopy. The specimens were evaluated and reported by at least two experienced pathologists independently. DN was diagnosed based on the presence of mesangial expansion, diffuse capillary glomerulosclerosis, presence or absence of Kimmelstiel–Wilson nodules, diffuse thickening of the glomerular basement membrane, and exudative lesions such as fibrin cap, capsular drop, or/and hyaline thrombi. 11 Pathological diagnosis of NDRDs was based on the basic glomerular changes described in the classification of glomerular diseases of the World Health Organization (WHO).
Follow-up and outcomes
After accurate diagnosis by renal biopsy, DN patients received therapies consisting of angiotensin-converting enzyme inhibitor (ACEI) or angiotensin receptor blocker (ARB) as well as blood glucose, blood pressure, and blood lipid management. NDRD patients were given glucocorticoid and/or immunosuppressive agent therapies according to their pathological patterns. Patients in MIX group received comprehensive treatments. Based on the previous literature, a 40% decline of eGFR is a valid and useful surrogate endpoint in clinical research. 12 Therefore, primary endpoint was defined as ESRD (the commencement of renal replacement therapy or eGFR <15mL/min/1.73m2 sustained for at least 30 days), or a 40% decline of eGFR from baseline sustained for at least 30 days. Patients were followed up until June 2020 and censored at the time of death (if before endpoint) or the last eGFR measurement before June 2020. Subsequent follow-up examinations were performed in outpatient clinics at 1, 3, and 6 months after renal biopsy and every 6 months thereafter or in the event of symptoms. The FPG, albuminuria, and serum creatinine were measured. For patients in remote area, phone interviews were conducted and the results of examination in local clinics were recorded. Patients with NDRD superimposed on DN received comprehensive treatments. All patients were followed up for at least 6 months.
Statistical analysis
Normality was tested using the Kolmogorov–Smirnov test. Continuous variables were described as means with standard deviations if normally distributed or medians with interquartile ranges if not. Categorical variables were expressed as numbers with proportions. Differences between groups at baseline were compared using one-way analysis of variance (ANOVA) or Kruskal–Wallis H-test as appropriate for continuous variables, and chi-square test or Fisher exact probability test as appropriate for categorical variables. The overall survival was analyzed using the Kaplan–Meier method followed by the log-rank test. Cox regression analysis was used to evaluate the adjusted prognostic difference among three groups and to evaluate the effect of baseline clinical parameters on the renal outcome. Clinical risk factors for predicting adverse renal outcomes were first determined by univariate Cox analysis, age gender, and variables with p < 0.1 were presented for multivariate Cox regression analysis. All analyses were performed with SPSS 24.0 and GraphPad Prism 9.0 Software. Two-sided p < 0.05 was considered statistically significant.
Results
Demographic and clinical characteristics of study cohorts
A total of 440 patients were eligible for enrollment, but 137 patients met the exclusion criteria and 11 patients were lost to follow-up. Finally, a total of 292 patients were considered to be eligible and enrolled in our final study cohort, including 153 (52.4%) in DN group, 109 (37.3%) in MIX group, and 30 (10.3%) in NDRD group. The mean age was 51.6 years and 111 of the patients were female. Median duration of diabetes was 84 months. The mean eGFR was 67.4 mL/min/1.73 m2 and the median protein excretion rate at baseline was 4.29 g/24 h.
Baseline characteristics in different groups are shown in Table 1. Compared with the NDRD group, the incidence of DR, levels of HbA1c and 24-hour urine protein were significantly higher in MIX and DN groups. In addition, patients in DN group had longer duration of diabetes, higher systolic blood pressure, lower levels of Hb and eGFR compared with NDRD and MIX groups (all p < 0.05). There was no significant difference in gender, age, BMI, family history of diabetes, TC, HDL, LDL, and presence of microscopic hematuria among three groups (p > 0.05).
Baseline characteristics of study participants in DN, NDRD, and MIX group.

BMI, body mass index; BUN, blood urea nitrogen; DBP, diastolic blood pressure; DM, diabetes mellitus; DN, diabetic nephropathy; DR, diabetic retinopathy; e-GFR, estimated glomerular filtration rate; FPG, fasting plasma glucose; HbA1c, hemoglobin A1c; HDL, high-density lipoprotein cholesterol; LDL, low-density lipoprotein cholesterol; MIX, diabetic nephropathy mixed with non-diabetic renal disease; NDRD, non-diabetic renal disease; SBP, systolic blood pressure; TC, total cholesterol; TG, triglyceride.
Data are in mean (SD), median (IQR) or n (%). P values were derived from one-way ANOVA (for normally distributed continuous variables), Kruskal–Wallis H-test (for non-normally distributed continuous variables), chi-square test (for categorical variables) or Fisher’s exact test (when the expected value < 5).
P < 0.05 versus NDRD group.
P < 0.05 versus MIX group.
Pathological types and proportions of NDRD
The pathological types and proportions of NDRD detected in this study are shown in Table 2. IgA nephropathy was the most common pathologic type of NDRD, with a total of 51 (37.0%) cases, followed by 44 (31.9%) cases of membranous nephropathy and 11 (8.0%) of minimal change disease. IgA nephropathy was also the most common type in either NDRD group or MIX group, accounting for 10 (33.4%) and 41 (38.0%) cases, respectively.
Pathological types and proportions of NDRD.

MIX, diabetic nephropathy mixed with non-diabetic renal disease; NDRD, non-diabetic renal disease.
IgA nephropathy not included in this part.
Renal outcomes
During the median follow-up of 27.0 (24.8–29.2) months, end-point renal events occurred in 129 (44.2%) patients. Fifty-eight of them received regular continuous renal replacement therapy (CRRT) dialysis after progression to ESRD and 71 had a 40% reduction in eGFR from baseline. The incidence of adverse renal outcomes in DN, MIX, and NDRD groups was 54.2% (n = 83), 36.6% (n = 40), and 20.0% (n = 6), respectively.
Survival analysis of different pathological groups
Kaplan–Meier analysis showed that patients in NDRD group had a better renal prognosis than pure DN (log-rank χ2 = 14.639, p < 0.001) or MIX (log-rank χ2 = 5.967, p = 0.015) group. However, patients in DN group and MIX groups shared similar prognosis (log-rank χ2 = 3.480, p = 0.062). The median (95% confidence interval (CI)) event-free survival was 23.0 (18.6–27.4) months for DN group and 33.0 (25.8–40.2) months for MIX group, whereas the median event-free survival of NDRD group has not been reached. The survival curves stratified according to the renal pathology are shown in Figure 2.
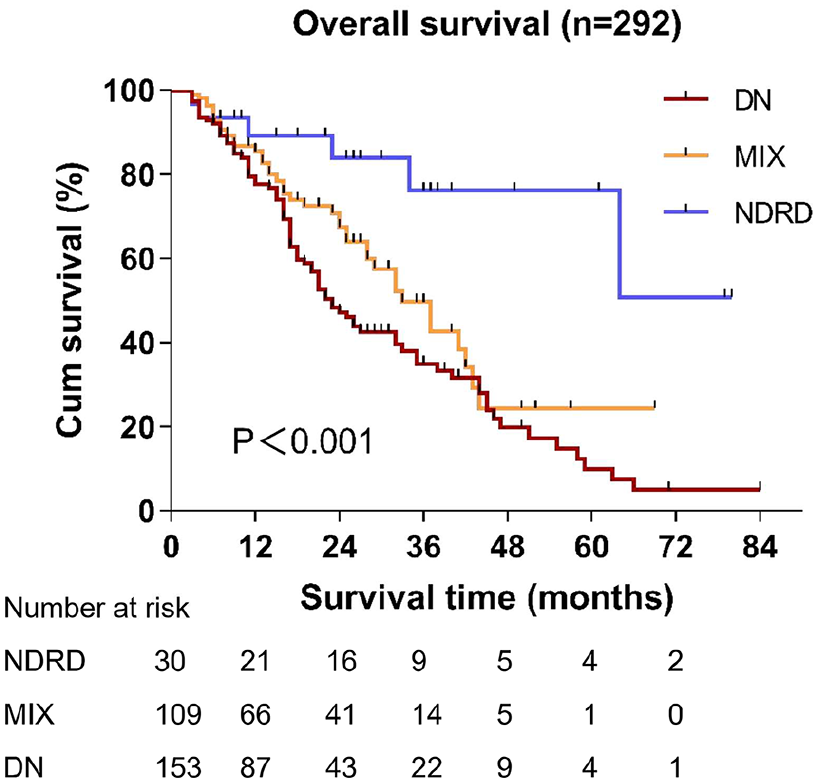
Kaplan–Meier curves for cumulative renal survival rates stratified according to renal pathology. DN versus MIX, log-rank χ2 = 3.480, p = 0.062; DN versus NDRD, log-rank χ2 = 14.639, p < 0.001; MIX versus NDRD, log-rank χ2 = 5.967, p = 0.015.
Multivariate analysis was performed by Cox regression. Compared to NDRD group, patients in DN group had a significantly greater risk of adverse renal outcomes in the fully adjusted model (hazard ratio [HR] = 3.900, 95% CI = 1.103–13.788). The same trend was observed in patients of MIX group, although this difference did not reach statistical significance (HR: 2.691, 95% CI: 0.662–10.936; Figure 3).

Associations between renal pathology and renal outcomes in patients with T2D. Hazard ratios (HRs) and confidence intervals (CIs) were calculated via the Cox proportional hazards model. Model 1: adjusted for age, gender, smoking, drinking, duration of diabetes, family history of diabetes, presence of diabetic retinopathy, and hypertention. Model 2: adjusted for Model 1 as well as systolic blood pressure, hemoglobin, HbA1c, serum albumin, triglyceride, low-density lipoprotein, and microscopic hematuria. Model 3: adjusted for Model 2 as well as eGFR and 24-h urine protein.
Baseline predictors for adverse renal outcomes
The results of multivariate Cox regression analysis suggested that baseline lower Hb (HR: 1.414, 95% CI: 1.094–1.828, p = 0.008) and lower eGFR (HR: 1.161, 95% CI: 1.005–1.357, p = 0.048) were independent risk factors for adverse renal outcomes in MIX group. In the pure DN group, patients with lower eGFR (HR: 1.122, 95% CI: 1.005–1.253, p = 0.040), severe proteinuria (HR: 2.025, 95% CI: 1.063–3.855, p = 0.032) and a family history of diabetes (HR: 1.771, 95% CI: 1.008–3.111, p = 0.047) had greater risk of adverse renal outcomes. Due to the small sample size, Cox regression analysis was not performed in NDRD group.
The baseline parameter and prognosis of DN and MIX were similar, thus, the two groups were combined. Multivariate regression results showed that baseline lower eGFR (HR: 1.159, 95% CI: 1.060–1.266, p = 0.001), severe proteinuria (HR: 2.047, 95% CI: 1.227–3.416, p = 0.006), lower Hb (HR: 1.170, 95% CI: 1.008–1.267, p = 0.037), and a diabetes family history (HR: 1.138, 95% CI: 1.008–1.285, p = 0.046) were independent risk factors for endpoint events in the combined cohort of DN and MIX groups (Table 3).
Relationship between baseline characteristics and renal outcomes in different groups.

CI, confidence interval; DM, diabetes mellitus; DN, diabetic nephropathy; e-GFR, estimated glomerular filtration rate; HR, hazard ratios; MIX, diabetic nephropathy mixed with non-diabetic renal disease.
Multivariate HR was adjusted for gender, age, and parameters with p < 0.1 in univariate regression analysis; in MIX group, the multivariate Cox regression model includes age, gender, hemoglobin, eGFR, and 24-h urine protein; In DN group, the multivariate Cox regression model includes age, gender, DM family history, hemoglobin, hypoalbuminemia, eGFR, and 24-h urine protein; In DN + MIX group, the multivariate Cox regression model includes age, gender, DM family history, hemoglobin, hypoalbuminemia, eGFR, and 24-h urine protein.
Discussion
In this study, patients with type-2 diabetes and CKD were divided into pure DN, pure NDRD, or a combination of both based on the pathologic results of renal biopsy. The results revealed that the pathological classification was significantly associated with renal outcomes, independent of baseline clinical parameters, such as HbA1c, eGFR, and 24-h urine protein. Among patients with DN, individuals with a DM family history, lower Hb, lower eGFR, and severe proteinuria were prone to have adverse renal outcomes.
Previous biopsy-confirmed studies indicated that the prevalence of NDRD ranged widely from 33% to 76% among patients with T2D.13–15 In our study, biopsy-proven NDRD with or without DN accounted for 47.6% of all cases, which is consistent with previous studies. Interestingly, we also observed a relatively high incidence of NDRD superimposed on DN, which presented in more than one-third (37.3%) of T2D patients. Overall, NDRD is not uncommon in patients with T2D and CKD. Differences in pathologic results may be due to variation in biopsy policy, ethnic/racial, geographic location, and sample size.
Our results revealed that at baseline, patients in DN group had longer duration of diabetes, poorer blood pressure and glycemic control, more severe renal impairment, and higher incidence of DR compared with those in NDRD group, which is consistent with previous studies.2,3 However, clinical indicators as DR and duration of diabetes, which are well-known predictors for DN in type-1 diabetes, cannot always accurately distinguish the pathologic types of CKD in type-2 diabetes, especially in the cases of NDRD coexisting with DN. 6 Therefore, renal biopsy should be recommended in patients with type-2 diabetes and CKD to confirm the diagnosis and give appropriate treatment as soon as possible.
Previous studies have reported a worse renal prognosis in patients with DN than in those with NDRD,16,17 whereas the results of prognostic difference between DN and MIX group are still inconclusive. Zhuo et al. 18 found that patients in MIX group present with a more rapidly progressive renal failure than pure DN group. On the contrary, some studies found that patients in MIX group had even superior renal prognosis compared with pure DN patients.5,6 It is worth mentioning that most previous cohorts only have less than 10% patients in MIX group, with the course of diabetes significantly lower than that in DN, which might lead to inaccurate conclusions. Our results showed that after adjustment of baseline parameters including duration of diabetes, HbA1c, DR, eGFR, and 24-h urinary protein, patients with pure DN were almost four times more likely to have adverse renal outcomes compared to NDRD. Patients with pure DN also demonstrated a worse renal outcome compared with MIX group, although the prognostic difference was not statistically significant in the fully adjusted model.
Previous studies have shown that anemia is significantly associated with the presence and progression of DN.19–21 Patients can have anemia even at the early stage of nephropathy before the decline of eGFR. Especially in those with proteinuria and renal function decline, the decrease of Hb is more remarkable. 21 In this study, we found that baseline hemoglobin level was an independent predictor for adverse renal outcomes in patients with DN, for every 10 g/L decrease of hemoglobin, the probability of end-point renal events was increased by 17% after adjustment. In recent years, it is believed that the decrease of hemoglobin in diabetic patients may be related to insufficient EPO production, shortened erythrocyte life span, autonomic nerve dysfunction, systemic microinflammatory state, and iron metabolism disorder.22–26 These findings suggest that the changes of hemoglobin should be paid enough attention in patients with DN. Whether a positive treatment of anemia can improve the prognosis of DN remains to be further confirmed by prospective interventional studies.
Familial clustering has been identified to be a susceptibility factor for the development of DN. 27 A recent study also showed that a family history of diabetes is an independent risk factor for rapid decline of eGFR in patients with DN. 28 We found that patients with a family history of diabetes are more likely to have adverse renal outcomes, suggests that the genetic background and lifestyle of diabetes may be related to the rapid decline of renal function in patients with DN.
Previous large clinical trials have confirmed that intensive glycemic control can delay or reduce the risk of microvascular complications of T2D including DN, whether in early or intermediate stage.29,30 However, we found no significant correlation between baseline serum glucose levels and renal prognosis after adjustment for confounding factors. This may be attributed to the fact that baseline HbA1c and FPG may not accurately reflect individual glycemic status, especially glycemic variability during the whole follow-up period. On the contrary, a variety of clinical conditions, such as hemoglobinopathies, anemia, and uremia will impact the value of HbA1c, independent of glycemia. 31
As a prospective study, this study contains certain strengths. Each participant was well-defined by renal biopsy at baseline and was divided into DN, MIX, NDRD groups based on pathology results, which avoided misdiagnoses caused by relying only on clinical features. There are also some limitations. First, the prognosis of MIX and NDRD groups was related to the type of NDRD, but we could not perform subgroup analysis due to the small sample size. Second, we were unable to obtain the exact therapeutic regimen during follow-up. Third, the urinary protein excretion of patients included in this study is relatively high and more than 50% of the cohort progressed to renal endpoints within a median follow-up of 27 months. Whether the findings can be generalized to early-phase patients with microalbuminuria is still unclear. In addition, the follow-up period was relatively short. Therefore, larger studies with a broader population and longer follow-up period are needed to confirm and validate the findings of this study.
Conclusion
The prognosis of pure DN might be similar to DN mixed with NDRD, and both of them were worse than that of pure NDRD. A family history of diabetes, lower hemoglobin, worse renal function, and severe proteinuria were independent predictors for endpoint events in patients with DN. Assessment of these indicators might identify HR groups with adverse renal outcomes and further shed new light on diagnosis, treatment, and preliminary evaluation of prognosis in patients with both T2D and CKD.
Footnotes
Acknowledgements
NJ and MP have contributed equally to this work and share first authorship.
Author contributions
Research idea and study design were contributed by YZ, NJ, and MP; acquisition of the data was performed by ZC, SL, XD, CQ, SH, XY, and LZ; analysis and interpretation of the data were performed by NJ, MP, YS, and CS; drafting of the manuscript was performed by NJ and MP; critical revision for intellectual content was performed by FG, HZ, JW, LW, HJ, FH, GQ, and YZ; technical and material support was provided by YZ and GQ; all authors read and approved the final version of the manuscript. NJ and MP contributed equally.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the National Natural Science Foundation of China (grant nos. 81800734 and U2004116), the National Key Research and Development Program of China (grant no. 2017YFC1309800), and the Key Project of Medical Science and Technology of Henan Province (grant no. SB201901046).
Conflict of interest statement
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
This study was reviewed and approved by the ethics committee of the First Affiliated Hospital of Zhengzhou University (approval no. KY-2013-LW-1282). All procedures were performed in accordance with the Declaration of Helsinki.
