Abstract
According to the US Centers for Disease Control and Prevention (CDC), an estimated 14% of adults in the United States have either been diagnosed with osteoarthritis (OA) or have symptoms suggestive of the disease. The CDC also points out that the incidence of OA has been gradually increasing over the past 30 years. What is more worrisome is that this trend is going to accelerate due to the aging demographics of the United States and the increasing prevalence of obesity seen in the country. The need for better preventive treatments and efficacious therapeutics are direly needed to combat this public health crisis. Among the possible treatments being hypothesized, antioxidant supplementation has become one of the most widely studied over the past decade due to its ability to attenuate reactive oxygen species (ROS) formation within chondrocytes, a critical step in the pathogenesis of this disease. Vitamin C has emerged as among the most promising of the antioxidant group, with many animal and human studies having been conducted in recent years. Although many of the studies have shown encouraging results in terms of preventing OA, others have reached opposite conclusions, thus making the data controversial. However, after reviewing several of these studies, we hypothesize that certain parameters may not have been properly considered during data collection. In the end, more randomized placebo-controlled trials in humans are desperately needed in order to fully understand whether vitamin C therapy is efficacious in treating and/or preventing OA.
Introduction
Osteoarthritis (OA) is one of the most prevalent degenerative diseases affecting over 50 million Americans currently and continues to be the leading cause of disability in the elderly population. 1 A study performed in 2000 found that in individuals over 80 years of age, nearly half of all women and a third of all men experience some degree of symptoms associated with OA. 2 Individuals with OA might experience symptoms of pain, stiffness, loss of function, and movement, all causing difficulty with everyday activities. The debilitating nature of the disease process not only leads to significant psychological suffering due to a devalued self-worth but also contributes to substantial economic losses from decreased productivity and associated medical care. 3 Unfortunately, clinicians expect the number of patients with OA in the United States to continue to increase dramatically over the coming years, largely due to countries rapidly aging demographic and the ever-increasing prevalence of obesity within the country.4,5 Therefore, researchers and clinicians alike are beginning to explore therapeutic options for in the treatment for OA. Although the exact pathogenesis of the disease has yet to be fully elucidated, several factors including age, history of joint injury, weight, genetics, and gender have been shown to increase individual risk of developing OA.6,7 The aforementioned risk factors are predominantly non-modifiable; consequently, current therapy has been limited to symptomatic relief. Therefore, attaining a more thorough understanding of the disease process is necessary to develop an effective, disease-modifying option for OA therapy.
Current treatments for OA primarily provide symptomatic relief with the hopes of controlling the common clinical manifestations of pain, swelling, and loss of function/movement. 8 Oftentimes, conservative measures such as weight loss, muscle stretching, low-impact exercises, and NSAID are used as first-line treatments. 9 Once these approaches fail, more invasive options such as joint injections and joint replacement surgery can be explored. 9 However, these options have significant side effects as well as issues regarding the effectiveness in preventing the progression of the disease.10,11 Overall, symptomatic relief achieved through the use of current approaches does indeed serve a therapeutic role, but only transiently. Therefore, finding more lasting treatment options focusing on disease modification and prevention should be the ultimate goal. Previous research has made it clear that balanced nutrition is an essential factor for maintaining healthy bone and joint structures while also dampening inflammation. Several key nutrients have been closely studied, taking a look at whether supplementation can reduce the progression of OA, as well as prevent it altogether. It is believed that free radical accumulation within human cells is a contributing factor to degenerative changes associated with aging. Specific to musculoskeletal health, reactive oxygen species induced oxidative stress can cause chondrocyte cell death. 12 With that in mind, it would make sense that the presence of anti-oxidants would oppose this joint destructive influence.
In general, antioxidants have demonstrated a protective role in musculoskeletal health in the elderly population.
13
However, studies looking at the efficacy of antioxidant supplementation in reducing the prevalence and progression of OA have produced varied and sometimes conflicting results. Some effectiveness has been demonstrated with the supplementation of glycosaminoglycans (chondroitin sulfate and glucosamine).
14
These substances, along with their antioxidant properties, were shown to help lubricate and alleviate forces translated across joints by promoting cartilaginous repair and regeneration, thus limiting the incidence of OA.15,16 Other well-known antioxidants, such as B vitamins and L-carnitine, have shown no substantial benefit to the patient in terms of OA prevention,17,18 whereas curcumin and L-glutathione have demonstrated some ability to modulate inflammation via downregulating mRNA levels of inflammatory mediators such as IL-1 and matrix metalloproteinase-3 (MMP-3).19–21 Finally, the link between vitamin D levels and the associated risk of developing OA has been studied on multiple occasions.
22
Although previous data indicated that low levels of 25-OH vitamin D were associated with a greater risk of OA progression,
17
Felson
Vitamin C
Vitamin C, also known as L-ascorbic acid, is a water-soluble essential nutrient that is not synthesized by most mammals, including humans. Most mammalian species cannot perform the crucial last step of vitamin C biosynthesis, the conversion of L-gulono-G-lactone into the vitamin, due to inactivation of the gene coding for the enzyme gulonolactone oxidase (GULO).16,18 Due to this evolutionary mutation, vitamin C is considered an essential vitamin and thus requires daily supplementation. The clinical manifestations of vitamin C dietary deficiency are known as scurvy and have been thoroughly documented in history, most notably during long ship voyages across the ocean. The sailors that happened to avoid developing scurvy had diets predominantly of fruits and vegetables, thus fulfilling their daily vitamin C requirements. 23 Aside from green vegetables, other dietary sources of vitamin C include citrus fruits and potatoes, with daily recommended doses dependent upon factors such as age, gender, and smoking status. 24
The molecular structure of vitamin C is similar to that of glucose. It can be oxidized to form dehydroascorbic acid (DHA), the preferred form for use by certain cells in the body. 25 The absorption takes place in the ileum of the small intestine via an energy-dependent sodium-dependent transporter, while the transfer of DHA utilizes an energy-independent process of facilitated diffusion. 26 Depending on the amount of vitamin C intake, the kidneys will regulate its excretion and reabsorption rates. 27 Vitamin C’s most important functions include being a key cofactor in many metabolic pathways, of which several promote proper musculoskeletal development and repair in humans. 27 For instance, intracellular vitamin C acts as an essential cofactor for both prolyl hydroxylase and lysyl hydroxylase, two enzymes crucial for collagen synthesis. The vitamin has also been found to play key roles in chondrogenic differentiation and extracellular matrix synthesis. Furthermore, the vitamin acts to promote cell growth through increasing DNA synthesis and differentiation of mesenchymal stem cells to various cell types such as adipocytes, osteoblasts, myoblasts, and chondrocytes.28,29 In addition, studies have illustrated that vitamin C enhances iron absorption in the gut.27,30 Iron is a known cofactor necessary for use in collagen synthesis, further contributing to maintaining musculoskeletal health.
Vitamin C and its transporter
Vitamin C is a highly water-soluble vitamin, which cannot diffuse across the hydrophobic plasma membranes. A specific transport system, sodium-dependent vitamin C transporters (SVCT) exists in the plasma membrane to mediate the entry process. Two subtypes of the sodium-dependent vitamin C transporter (SVCT1 and SVCT2) have been discovered.28,31–33 SVCT1 is expressed in tissues such as liver, kidney, and intestinal epithelial cells, whereas SVCT2 is seen predominantly in tissues such as the brain, heart, and muscles.28,31,32 Our lab has extensively studied these transporters and their essential role in musculoskeletal tissue engineering over the past decade.33–36 We previously demonstrated the expression of SVCT2 in chondrocytes, bone marrow stromal cells, osteoblast, and articular cartilage.33–36 Our laboratory previously reported that the SVCT2 transporter is negatively regulated in human OA tissues and mice aging bones.28,33–36 In addition, it is known that aging, oxidative stress, and inflammation can negatively alter the SVCT2 expression in these tissues.28,37,38 Therefore, we speculate that down-regulation of SVCT2 transport might be an important contributing factor in the pathophysiology of OA.
Vitamin C supplementation for OA
The most important inherent property of vitamin C is its ability to act as a strong antioxidant, with the capability to reduce and stabilize potentially damaging free radical compounds. It is known that a major contributing factor to age-induced cartilage degeneration (OA) occurs due to the formation and accumulation of free radicals (superoxide, hydrogen peroxide, peroxynitrite) within chondrocytes. 39 More specifically, these reactive oxygen species induce genomic instability in human chondrocytes leading to a reduction in viable cartilage. 40 Given this fact, it suggests that antioxidants such as vitamin C could serve a protective function in preventing oxidative stress-induced chondrocyte dysfunction from occurring. To gain a better insight into recently published data, we summarize animal studies, human clinical trials, and in-vitro studies involving vitamin C in the setting of treating and/or preventing OA. This information was obtained by searching for primary literature through PubMed with the search criteria; “Vitamin C,” “Osteoarthritis,” “Treatment,” “Prevention.”
Animal studies
Vitamin C supplementation on prevention and progression of OA
Overall, the clinical studies included in Table 1 show mixed results, thus providing a cloudy picture of the vitamin’s ability to serve a therapeutic role in treating and preventing OA. In the 1990 study, Meacock
Complete list of animal studies involving vitamin C and osteoarthritis used for analysis and commentary.

AA, Ascorbic Acid; KO, Knockout; MIA, Monosodium iodoacetate; OA, osteoarthritis; OARSI, Osteoarthritis Research Society International; PHD, prolyl hydroxylase domain-containing protein; SOD, superoxide dismutase; STR, Strain of mice; SVCT, sodium-dependent vitamin C transporters; VC, vitamin C.
Vitamin C injections on the reduction of oxidative stress parameters
More recently, studies looking at the efficacy behind intra-articular injections of vitamin C in conjunction with other inflammatory mediating compounds have been performed. In 2018, a study investigating whether injections of vitamin C and ferric chloride affected OA progression in surgically induced osteoarthritic mice was performed.
48
Two key features, osteophyte formation, and joint space narrowing, were monitored to quantify the progression of OA in the mice. After 12 weeks of injections, data showed substantial reductions in OA progression within the experimental group mice when compared to the control mice. This allowed the authors to reach the conclusion that intra-articular injections of vitamin C/FeCl3 effectively delayed OA progression in mice.
48
Yao
Human clinical studies
Vitamin C supplementation in osteoarthritic patients
Although no double-blind placebo-controlled human clinical trials have been performed to date, there have been many prospective and cross-sectional studies looking at how vitamin C intake might affect the incidence and progression of OA (Table 2). In one of the largest longitudinal studies published to date, the relationship between vitamin C supplementation and its effects on human knee OA development and progression was investigated. The 2011 study included over 1023 patients aged 40 years or older, and with the use of an MRI, the OA status of each individual was determined.
40
Ten years later, each participant was subject to a second MRI to determine the progression of OA, as well as a detailed questionnaire aimed at determining daily vitamin C intake. Results showed that participants who engaged in supplementation of the vitamin were 11% less likely to develop OA when compared to those who did not supplement. However, results also demonstrated no significant difference in OA progression between the two groups. Thus, Peregoy and Wilder
40
concluded that vitamin C acted in a chondroprotective manner leading to the prevention of OA, but had no effect on delaying the progression of the disease once established. The same mixed results pertaining to the antioxidant therapy were reproduced in Joseph
Complete list of human studies involving vitamin C and osteoarthritis used for analysis and commentary.
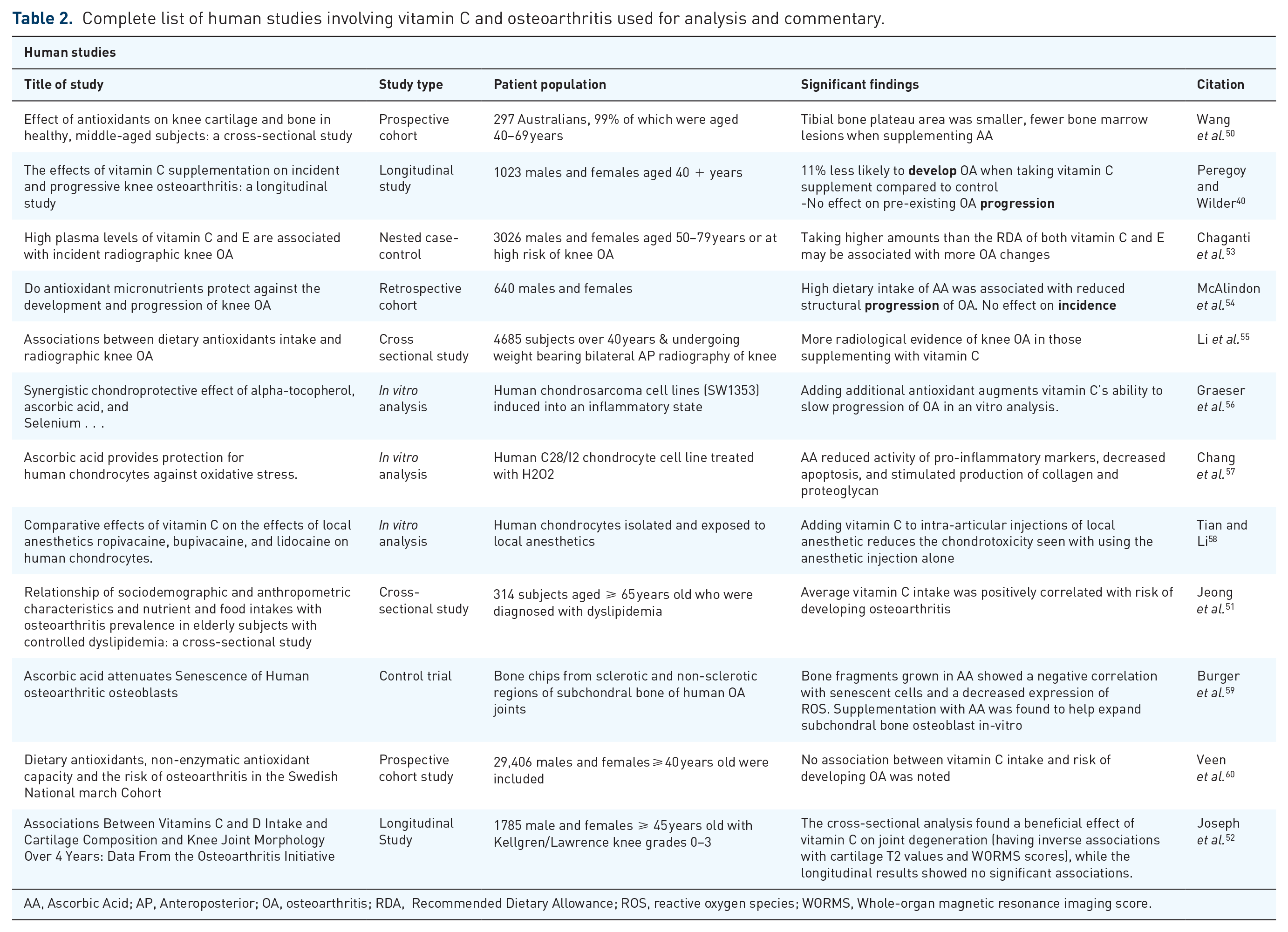
AA, Ascorbic Acid; AP, Anteroposterior; OA, osteoarthritis; RDA, Recommended Dietary Allowance; ROS, reactive oxygen species; WORMS, Whole-organ magnetic resonance imaging score.
While the multiple studies mentioned above have provided data illustrating vitamin C’s chondroprotective ability, several other studies published in the literature have reached the opposite conclusion.53,55,60 No association between vitamin C intake and risk of developing OA was noted in the large prospective cohort study conducted by Veen
In vitro studies involving vitamin C for cartilage repair
To gain further insight into the mechanism by which vitamin C works, several
Discussion and suggestions for future research
Vitamin C remains at the top of the list in terms of future treatments options for OA due to its well-tolerated biophysical profile, cheap cost, and easy administration; whether that be via the sole supplementation of the substance or in conjunction with other pharmacologic compounds seen in several studies mentioned above. Two important points can be noted from our literature search, with the first being that the most recent data pertaining to the therapeutic potential of vitamin C in attenuating progression and prevention of OA are mixed and thus remain controversial. While many studies in animal models and humans generally point to vitamin C supplementation as an efficacious treatment in the prevention of OA, other studies have concluded that excess vitamin C intake may lead to detrimental effects within the body, including increased inflammation and even increased progression of OA.40,41,43,44,46,50,51,53–55,60 The second is that there does seem to be a general consensus around an optimal dosage for the substances to have a maximal effect.44,46
Based on our and other published studies, we hypothesize that several important factors have not fully been considered in data collection and analysis, possibly resulting in the discrepancies seen in several of the data sets. These factors include (a) not taking the complexities of the SVCT1/2 transporter system expression into account in human clinical studies and (b) not properly accounting for the vitamin C biosynthesis mechanism in animal models (e.g. mice/rats). Therefore, the outcomes of such animal models do not translate effectively to the human condition. Moreover, we speculate that the data obtained from several of the human studies listed in Table 2 may be inconclusive due to not fully considering the altered regulation of the SVCT transporter during diseased conditions. Recent reports in various cells and tissue types, including cartilage, have shown that vitamin C transporter is dysregulated during pathophysiological conditions such as OA.36,63,64 In such a condition, vitamin C uptake during supplementation or injection will not be able to act intracellularly as an antioxidant, but rather stay extracellularly where the vitamin can aid in ROS generation ultimately leading to further damage of surrounding tissues.65,66
Conclusion
In conclusion, critical barriers in the development of an effective treatment option remain due to a poorly translatable humanized animal model for testing, dysregulation of vitamin C transporter in the osteoarthritic tissue, and an incomplete understanding of human OA pathophysiology. Given this fact, performing studies in the murine model do not translate well to human application. Thus, future human studies are essential, especially double-blind randomized control trials, to properly quantify the effectiveness of vitamin C treatment and identify the ideal amount of antioxidant supplementation needed to achieve the greatest response within an individual.
Footnotes
Acknowledgements
We would like to thank the Department of Orthopedic Surgery, Department of Medicine, and Division of endocrine for their support. Burton Dunlap and G. Taylor Patterson are equal contribution.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work is funded by the Department of Orthopedic Surgery, Department of Medicine, and Division of endocrine, Augusta University.
Conflict of interest statement
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Consent statement and ethical approval
This study is based on published data (PubMed search). A consent statement and ethical approval are not required for the current study.
