Abstract
Background:
A large proportion of patients with inflammatory bowel disease (IBD) relapse after drug discontinuation despite achieving a stable state of infliximab-induced clinical remission. Resuming the use of the same tumor necrosis factor-alpha (TNF-α) inhibitors in patients who relapse following TNF-α inhibitor discontinuation was suggested as a treatment strategy. We conducted a systematic review and meta-analysis to evaluate the efficacy and safety of infliximab retreatment in patients with IBD.
Methods:
A systematic literature search to shortlist relevant studies was conducted using the MEDLINE, Embase, CINAHL, and SCOPUS databases for studies published from inception to August 2020.
Results:
Nine studies were included in the meta-analysis. The pooled clinical remission rate of infliximab retreatment in patients with IBD was 85% (95% confidence interval (CI), 81–89%) for induction treatment and 73% (95% CI, 66–80%) for maintenance treatment. A clinical remission rate following infliximab reintroduction was achieved in a greater proportion of patients with Crohn’s disease (87%; 95% CI, 83–91%) than in those with ulcerative colitis (78%; 95% CI, 61–91%) for induction treatment, but the difference was not statistically significant. Infusion-related reactions after infliximab retreatment occurred in 9% of patients with IBD (95% CI, 3–16%).
Conclusion:
Infliximab retreatment showed high clinical remission rates with tolerable infusion-related reactions in patients with IBD who achieved remission with initial infliximab treatment but relapsed after its discontinuation. We suggest infliximab as a viable alternative in patients with IBD who previously responded well to infliximab treatment.
Introduction
Inflammatory bowel diseases (IBDs), including Crohn’s disease (CD) and ulcerative colitis (UC), are chronic immune-mediated diseases characterized by uncontrolled inflammation of the gastrointestinal tract.1,2 Therapeutic strategies for the management of IBD include conventional therapies with 5-aminosalicylic acid drugs, corticosteroids, and immunomodulators and biologic therapy with tumor necrosis factor-alpha (TNF-α) inhibitors.3,4
Infliximab, a chimeric monoclonal IgG1 antibody against TNF-α, is one of the preferred treatment options among TNF-α inhibitors5,6 and has proven to be efficacious as induction and maintenance therapy for patients with refractory IBD.7,8 Despite the success of infliximab for achieving remission and improving clinical outcomes, approximately half of patients with IBD who have achieved clinical remission relapse within 1 year after its discontinuation due to patient preference, cost, or potential for adverse effect, and so on. 9 European Crohn’s and Colitis Organization suggested resuming the use of the same TNF-α inhibitors in patients who relapse following TNF-α inhibitor discontinuation as an ‘exit strategy’. 10
Retreatment with infliximab can be considered an alternative for patients with IBD who relapse after discontinuation. However, conflicting results regarding clinical outcomes have been reported after infliximab retreatment,11,12 including the development of antibodies against infliximab, increased risk of severe systemic reactions, and shortened duration of efficacy, which limit its usefulness.13,14 Kugathasan et al 12 reported that infliximab retreatment is associated with high rates of severe systemic reactions in adults and should only be used in safe, well-controlled clinical settings. In contrast, a multicenter study demonstrated clinical remission in 79% of patients with IBD, 11 with adverse events in only 13% of patients following infliximab reinduction, suggesting that infliximab can be safely and effectively reintroduced. 15 Therefore, it remains unclear whether infliximab retreatment is effective and tolerable in patients with IBD who relapsed after its discontinuation. As prospective randomized comparative studies on infliximab retreatment are difficult to conduct due to ethical issues, including those associated with deliberate drug suspension and resumption, it is preferable to collect data from reported studies and investigate its effects using a meta-analysis.
Therefore, in this study, we conducted a meta-analysis and systematic review to evaluate the efficacy and safety of infliximab retreatment in patients with IBD who initially responded to infliximab but relapsed after its discontinuation.
Materials and methods
This study followed the guidelines recommended by the Meta-analysis of Observational Studies in Epidemiology (MOOSE) (Supplementary Table 1). 16
Search strategy
The MEDLINE, Embase, CINAHL, and SCOPUS electronic databases were systematically searched for relevant studies published from inception to 15 August 2020. A comprehensive literature search was conducted using a combination of the following keywords and medical subject headings: ‘inflammatory bowel disease’, ‘Crohn’s disease’, ‘colitis, ulcerative’, ‘infliximab’, ‘retreatment’, and ‘reinduction’. The detailed search strategies for each electronic database used in this analysis are provided in Supplementary Table 2. Our search was not restricted to any language.
Study selection
Two authors (S.Y. and Y.K.J.) independently conducted the literature search and followed the study selection protocol. Discrepancies were resolved by consensus. Studies were considered eligible if they met the following inclusion criteria: (1) population: patients diagnosed with IBD; (2) intervention: patients who restarted infliximab treatment after discontinuation of initial infliximab treatment for an interval and had no history of any other biologic therapy; (3) outcome: the proportion of patients who experienced either induction of remission, maintenance of remission, or infusion-related reactions. In this review, remission includes clinical, biochemical, or endoscopic remission defined as a state with completely symptom-free or steroid-free remission, pediatric Crohn’s Disease Activity Index ⩽ 10, Crohn’s Disease Activity Index score of <150, partial Mayo score of ⩽3, C-reactive protein <10 mg/l, or closure of all fistulas based on endoscopic or magnetic resonance examination; 17 and (4) study design: prospective or retrospective controlled or uncontrolled studies excluding case reports. Studies were excluded if they were (1) nonhuman studies, including animal and in vitro studies; (2) available only in the form of abstracts or posters; and (3) reviews, meta-analyses, letters, editorials, or ongoing studies.
Data extraction
The eligible studies were reviewed, and the following data were extracted using a standardized extraction form: first author, publication year, country, study design, number of patients, sex, age, infliximab drug holiday, infliximab retreatment regimen, concomitant medications, follow-up period, definition of clinical outcomes, rates of remission induction at 3 months after infliximab reinitiation, and maintained rates of remission after remission induction, which were assessed 1 year after infliximab reinitiation, incidence of infusion-related reactions, proportion of patients who tested positive for anti-infliximab antibodies, and serum concentration of infliximab.
Assessment of risk of bias
The risk of bias of the included studies was evaluated using a modified Methodological Index for Non-Randomized Study (MINORS), 18 which contains the following eight items designed specifically for noncomparative studies: (1) clearly stated aim, (2) inclusion of consecutive patients, (3) prospective data collection, (4) endpoints appropriate according to study aim, (5) unbiased assessment of the study endpoints, (6) follow-up period appropriate to study aim, (7) <5% loss of patients during follow-up, and (8) prospective calculation of study size. The items were scored as 0 (not reported), 1 (reported but inadequate), or 2 (reported and adequate). The scores were evaluated as follows: 0–4, very low quality; 5–8, low quality; 9–12, moderate quality; and 13–16, high quality. 19 Two authors independently assessed the potential degree of bias, and any disagreement between the authors was resolved by consensus.
Statistical analysis
The efficacy outcomes were the rates of induction of remission at 3 months after infliximab reintroduction and subsequent maintenance of remission at 1 year, which were presented as ‘induction of remission’ and ‘maintenance of remission’, respectively, and the safety outcome was the proportion of patients who experienced any infliximab-related infusion reaction after its reintroduction. The pooled estimates of rates for induction remission, maintenance remission, and infliximab-related infusion reactions were calculated using meta-analyses. Heterogeneity among studies was detected using inconsistency statistics (I2). 20 A random-effects model was used when potential heterogeneity existed (I2 > 40%); otherwise, a fixed-effects model was employed. 21 Moreover, the restricted maximum likelihood method was used to estimate the parameters of a random coefficient model, and the value of tau-squared (τ2) was used to estimate between-study variance. 22
Subgroup and sensitivity analyses were performed to remove heterogeneity and evaluate treatment effects based on the subgroup of patients with different diagnoses and study design. To investigate whether treatment efficacy was reduced according to the duration of infliximab discontinuation, the remission rate was stratified according to the studies in which the minimum duration of infliximab discontinuation was reported as >16 weeks. Moreover, a meta-regression was performed to investigate the association of remission rate and the incidence of infusion-related reactions with the proportion of concomitant immunomodulators. Statistical significance was defined as p < 0.05. The metaprop module in the R, version 3.6.0 (R Foundation for Statistical Computing, Vienna, Austria) statistical software package and the Comprehensive Meta-analysis, version 2 (Biostat, Englewood, NJ, USA) were used for analysis.
Results
The results of the study selection process are shown in Figure 1. We initially identified 459 articles through an electronic database search. After removing duplicates, 246 articles were screened for relevance based on the title and abstract, resulting in the exclusion of 199 articles that did not include patients with IBD, infliximab retreatment, or efficacy or safety outcomes of infliximab or were review, comments, or conference abstracts. Next, 47 relevant articles were assessed for eligibility through full-text evaluation. Finally, nine studies were selected for meta-analysis: five studies involving patients with CD, one involving patients with UC, and three involving both patients with CD and UC.14,15,23 –29 Among the nine studies, eight studies, excluding a study by Rodrigo et al., evaluated the remission rate of infliximab retreatment, and seven studies, excluding the studies by Laharie et al. and Dai et al., evaluated the incidence of infusion-related reactions after infliximab retreatment.

Flow chart of the study selection process.
Table 1 summarizes the characteristics of the included studies. In total, 428 patients, who reinitiated infliximab treatment after discontinuation for a reported median duration of 4–26.7 months, were included in this meta-analysis. Most studies were conducted in Europe, and the remaining two studies by Kang et al. and Dai et al. were conducted in Asia. All retrieved studies were nonrandomized, single-arm, observational studies, five of which were retrospective and four were prospective. Participants in the included studies received an intravenous dose of 5 mg/kg, except for a small proportion (<3%) of participants in one study who were administered an intravenous dose of either 7.5 or 10 mg/kg at 0, 2, and 6 weeks. 26
Characteristics of studies included in the meta-analysis.

CD, Crohn’s disease; CDAI, Crohn’s Disease Activity Index; IBD, inflammatory bowel disease; IFX, infliximab; IMM, immunomodulator; IQR, interquartile range; IV, intravenous; n, number of patients; PCDAI, Pediatric Crohn’s Disease Activity Index; pMayo, partial Mayo; SE, standard error; UC, ulcerative colitis; W, weeks. An ellipsis (. . .) indicates ‘not available’.
Age at diagnosis.
Age at first IFX infusion.
Information on all study participants is presented because there is no separate information available on patients who underwent IFX retreatment.
During IFX discontinuation and retreatment.
Based on the timepoint after patients had discontinued IFX.
Based on the timepoint after patients had been retreated with IFX.
With a cut-off of 3 U/ml.
Since the serum infliximab level was ⩾1 mg/l, 73% of the samples were inconclusive.
The proportion of patients who received concomitant immunomodulator therapy during infliximab discontinuation and retreatment, ranged from 61% to 100%, as reported in five studies (Table 1). The proportion of patients who were antibody-positive for infliximab was measured at cessation and after the initiation of retreatment in three studies (Table 1). However, the detectable level of antibody positivity for infliximab was not consistent among the studies. The proportion of antibody-positive patients for infliximab after the initiation of retreatment was 40% with a cut-off of 3 U/ml in the study by Baert et al.; although the proportion of patients who were antibody-negative for infliximab was 27% in the study by Louice et al., data for the remaining 73% were inconclusive due to the interference of circulating infliximab with level >1 mg/ml. The median serum concentrations (interquartile range) of infliximab reported in the studies by Louis et al. and Baert et al. were 3.7 (1.7–8.0) µg/ml and 5.2 (0.36–17.3) µg/ml, respectively.
Clinical remission
Eight studies involving 358 patients reported clinical remission rates following infliximab retreatment in patients with IBD. The pooled remission rate after infliximab retreatment was 85% (95% confidence interval (CI), 81–89%; I2 = 5%; τ2 = 0.0003, p = 0.39) for induction treatment (at least 3 months) and 73% (95% CI, 66–80%; I2 = 30%; τ2 = 0.0023, p = 0.23) for maintenance treatment (at least 1 year) (Figure 2). The heterogeneity was low. Upon further subanalysis of induction treatment, a higher remission rate following infliximab reinduction was observed in patients with CD (87%; 95% CI, 83–91%; I2 = 4%; τ2 = 0.0003, p = 0.40) than in those with UC (78%; 95% CI, 61–91%; I2 = 46%; τ2 = 0.0137, p = 0.13), but the difference was not statistically significant (Figure 3). The numbers of patients with CD and UC were 272 and 48, respectively.

Forest plot of remission (a) induction and (b) maintenance rates following infliximab retreatment for patients with inflammatory bowel disease.

Forest plot of remission induction rates after infliximab retreatment in (a) patients with Crohn’s disease and (b) patients with ulcerative colitis.
When the results were stratified by studies wherein patients restarted infliximab after a minimum drug holiday of ⩾16 weeks, the remission induction rate for patients with IBD was 84% (95% CI, 78–89%; I2 = 0%; τ2 = 0, p = 0.57) as reported in two studies.15,23 The results of meta-regression showed that remission induction rates in patients with IBD did not significantly differ according to the proportion of concomitant immunomodulators (Supplementary Figure 1). Moreover, when the results were stratified by study design in the sensitivity analysis, the remission induction rates for patients with IBD, CD, and UC were similar between prospective and retrospective studies (Supplementary Table 3).
The effects of infliximab level on the clinical remission rate were reported only in the study by Baert et al., in which trough levels of infliximab greater than 2 mg/ml were associated with higher remission rates. Biochemical or endoscopic examinations were performed in combination with the assessment of clinical remission in the three studies.14,25,27 However, examinations were conducted for only some participating patients, and biochemical or endoscopic remission rates were not reported.
Infusion-related reactions
Seven studies involving 368 patients with IBD were included to evaluate the proportion of patients who experienced infusion-related reactions after infliximab retreatment. Infusion-related reactions were observed in 9% of patients with IBD (95% CI, 3–16%; I2 = 77%; τ2 = 0.0173, p

Forest plot of incidence of infusion-related reactions that occurred in patients with inflammatory bowel disease after infliximab retreatment.
The incidence of severe infusion-related reactions, which were defined as reactions necessitating discontinuation of the infusion owing to significant dyspnea or hypotension was reported in five studies as follows: 0% in three studies and 4% (1/32) and 12.5% (16/128) in two studies, respectively. Most infusion-related reactions were acute and resolved with discontinuation of infliximab infusion, intravenous antihistamines, and steroids. The incidence of hospitalization related to infusion-related reactions has not yet been reported.
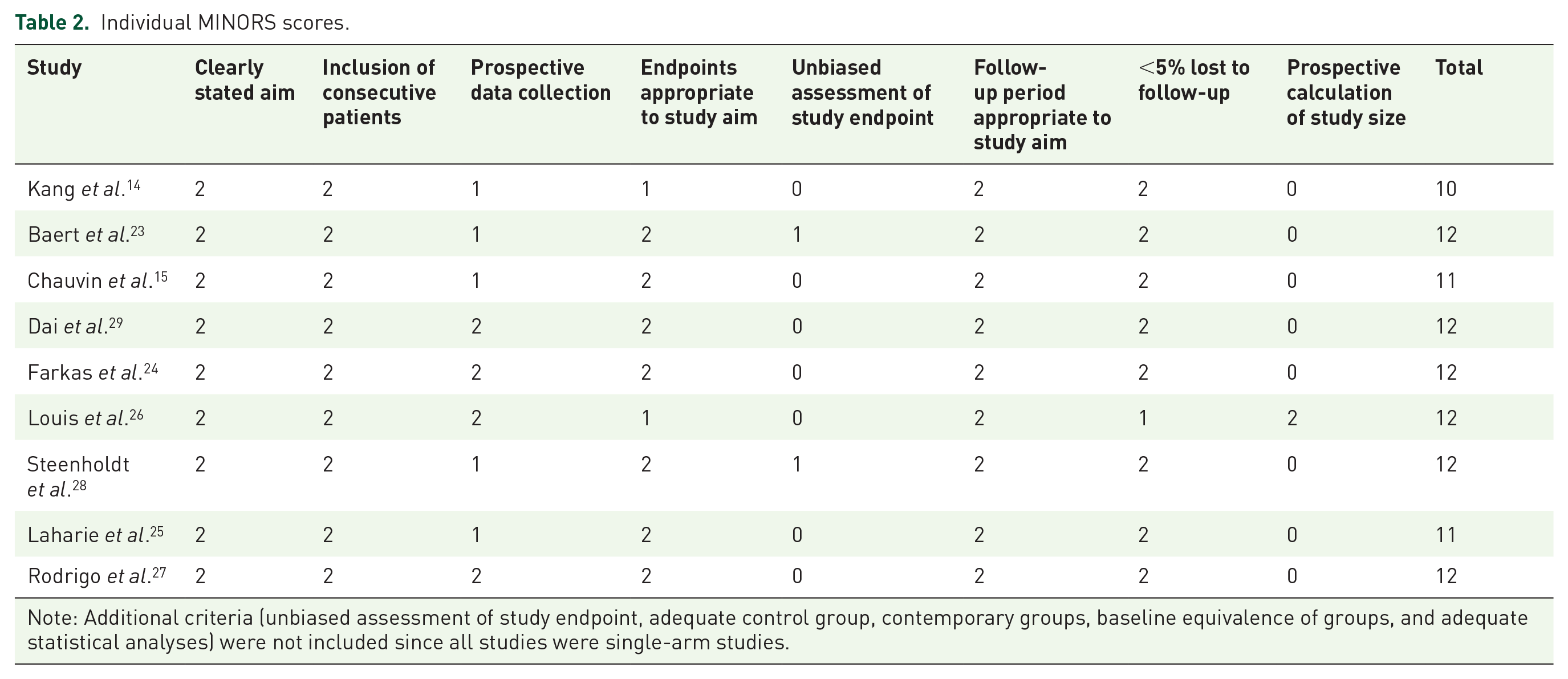
Risk of bias
The overall risk of bias of the included studies ranged from 10 to 12, indicating moderate quality (Table 2). All included studies showed a low risk of bias in the following domains: clearly stated aim, inclusion of consecutive patients, follow-up period appropriate to study aim, and
Individual MINORS scores.
Discussion
This meta-analysis evaluated the efficacy and tolerability of infliximab retreatment for the induction and maintenance of remission in patients with IBD. There is an urgent need for more data to establish standard practice guidelines for clinical remission following infliximab retreatment, as the available data are insufficient. A previous meta-analysis by Gisbert et al. 30 found that clinical response to retreatment with the same TNF-α inhibitors was favorable. Compared to their review, our data, including results from five additional studies, demonstrated an acceptable rate of infusion-related reactions as well as favorable rates of the induction and maintenance of remission after infliximab reinduction in patients with IBD. This study suggests that retreatment with infliximab could be a viable alternative to other TNF-α inhibitors for achieving clinical remission in patients with IBD who relapse after discontinuation.
Retreatment with infliximab has been limited in clinical practice owing to concerns, such as the occurrence of severe systemic reactions and reduction of efficacy.12,13,31 In a prospective clinical study, 21% of patients with CD experienced severe systemic reactions following infliximab retreatment. 12 In addition, antibodies against infliximab have been detected in 7–30% of patients following retreatment with infliximab. 32 According to the US Food and Drug Administration labeling information, an increased incidence of antibodies against infliximab was detected in patients with CD after infliximab reinfusion following a drug holiday of >16 weeks. 33 In addition, patients who tested positive for antibodies are likely to experience a lower benefit owing to a faster clearance and lower concentration of the drug than those who are antibody-negative to infliximab.13,31,33 Therefore, the risk of infusion-related reactions and reduced efficacy in patients treated with infliximab have been predicted to be associated with the formation of neutralizing antibodies and degradation of therapeutic antibodies.13,31,34
Contrary to the concern that infliximab retreatment may be associated with unfavorable remission rates owing to drug-induced immunogenicity, this study revealed relatively favorable remission induction rates after infliximab retreatment. The induction of remission after 3 months of infliximab retreatment was achieved in 87% of patients with IBD, which is consistent with a previous meta-analysis demonstrating remission induction in 88% of patients after the reintroduction of discontinued TNF-α inhibitors. 9 However, in that study, two TNF-α inhibitors, infliximab or adalimumab, were administered to the patients; therefore, the results cannot be extrapolated to infliximab alone. Moreover, remission rates did not differentiate between induction and maintenance therapies in that study. 9
In this study, a higher remission induction rate following infliximab retreatment was observed in patients with CD (88%) than in those with UC (81%), although this finding was not statistically significant. Similar findings have been reported in previous meta-analyses evaluating clinical remission in patients with IBD receiving TNF-α inhibitors including infliximab and adalimumab.9,30 However, the results of this study should be interpreted with caution, as the number of patients with UC was small and the 95% CIs overlapped.
According to the pooled rate from two studies involving a small sample size of 154 patients in our meta-analysis results, the number of patients who maintained remission in patients responsive to infliximab induction therapy was decreased. Although only a few studies have focused on maintaining the induction of remission in infliximab retreatment, this tendency of decreasing rate of remission maintenance over time is consistent with the findings of previous randomized controlled trials.8,35
The remission induction and maintenance rates of this study should be interpreted considering the concomitant use of immunomodulators. Although a subanalysis in this study did not show the combined effect of immunomodulators, several studies have shown that concomitant treatment with immunomodulators increases the clinical remission rate of infliximab treatment.35 –37
This study suggests that infliximab retreatment was well tolerated with a comparable rate of infusion-related reactions with that reported in previous studies. The pooled rate of infusion-related reactions following infliximab retreatment was approximately 9%, similar to that reported in previous studies, ranging from 2% to 27%.38 –41 However, immunomodulators were concomitantly used with infliximab in most studies included in our meta-analyses. Accordingly, the effect on immunomodulators should be considered when interpreting the efficacy and safety of infliximab retreatment. The effectiveness and safety of combination therapy with biologic and immunomodulators have been evaluated in several studies.35,36,42
As the therapeutic strategies for IBD have changed, the importance of biologics used in IBD patients has substantially increased. Meanwhile, the concerns on the risk of immunogenicity of biologics associated with nonremission and loss of disease control have been raised, the concomitant use of immunomodulators has been suggested as a treatment option for reducing the risk of immunogenicity.43,44 Several studies have shown that the concomitant use of immunomodulators has a protective effect against anti-infliximab antibody formation and reduces the incidence of infusion-related reactions.35,36,42 Notably, continuing the use of immunomodulators during drug holidays was reported to be associated with reduced immunogenicity when biologics were reintroduced. 44 Because four studies that did not provide detailed information on the concomitant use of immunomodulators during the drug holidays and retreatment period were not included in our meta-regression analysis, the exact effectiveness and safety of infliximab retreatment, excluding the effect of immunomodulators, cannot be determined from our results. Therefore, further studies comparing the effect of immunomodulator use at different time periods in infliximab retreatment are needed.
This study had several limitations. First, this meta-analysis included single-arm studies, wherein it is difficult to demonstrate the efficacy of a treatment in the absence of a comparison group and assess the practical benefits of clinically relevant endpoints. Second, the included studies were heterogeneous in terms of follow-up duration, drug holiday duration, and definition of clinical remission, which may have affected the results of this meta-analysis. Third, subanalyses based on the proportion of patients who tested positive for antibodies against infliximab and serum concentration of infliximab could not be performed owing to the sparsity of data. Fourth, endoscopic remission is an important endpoint in determining the efficacy of IBD treatment. In this study, we were unable to assess mucosal healing or endoscopic remission because most of the included studies lacked endoscopic measurements after infliximab retreatment. Furthermore, the overall risk of bias for the included studies was moderate, and the risk of bias in the unbiased assessment of the study endpoint and prospective calculation of the study size was high.
Infliximab retreatment resulted in favorable remission induction and maintenance rates with tolerable infusion-related reactions in patients with IBD who achieved remission with initial infliximab treatment but relapsed its discontinuation. Thus, infliximab can be considered a potential retreatment option in patients with IBD who previously responded well to infliximab treatment. Further studies evaluating the long-term outcomes of infliximab retreatment are needed to compare its effects with those of conventional therapies in patients with IBD.
Supplemental Material
sj-docx-1-taj-10.1177_20406223211041927 – Supplemental material for Efficacy and tolerability of infliximab retreatment in patients with inflammatory bowel disease: a systematic review and meta-analysis
Supplemental material, sj-docx-1-taj-10.1177_20406223211041927 for Efficacy and tolerability of infliximab retreatment in patients with inflammatory bowel disease: a systematic review and meta-analysis by Seungwon Yang, Siyoung Yang, Young Kwon Jo, Seungyeon Kim, Min Jung Chang, Junjeong Choi, Jae Hee Cheon and Yun Mi Yu in Therapeutic Advances in Chronic Disease
Supplemental Material
sj-docx-2-taj-10.1177_20406223211041927 – Supplemental material for Efficacy and tolerability of infliximab retreatment in patients with inflammatory bowel disease: a systematic review and meta-analysis
Supplemental material, sj-docx-2-taj-10.1177_20406223211041927 for Efficacy and tolerability of infliximab retreatment in patients with inflammatory bowel disease: a systematic review and meta-analysis by Seungwon Yang, Siyoung Yang, Young Kwon Jo, Seungyeon Kim, Min Jung Chang, Junjeong Choi, Jae Hee Cheon and Yun Mi Yu in Therapeutic Advances in Chronic Disease
Supplemental Material
sj-docx-3-taj-10.1177_20406223211041927 – Supplemental material for Efficacy and tolerability of infliximab retreatment in patients with inflammatory bowel disease: a systematic review and meta-analysis
Supplemental material, sj-docx-3-taj-10.1177_20406223211041927 for Efficacy and tolerability of infliximab retreatment in patients with inflammatory bowel disease: a systematic review and meta-analysis by Seungwon Yang, Siyoung Yang, Young Kwon Jo, Seungyeon Kim, Min Jung Chang, Junjeong Choi, Jae Hee Cheon and Yun Mi Yu in Therapeutic Advances in Chronic Disease
Supplemental Material
sj-tif-1-taj-10.1177_20406223211041927 – Supplemental material for Efficacy and tolerability of infliximab retreatment in patients with inflammatory bowel disease: a systematic review and meta-analysis
Supplemental material, sj-tif-1-taj-10.1177_20406223211041927 for Efficacy and tolerability of infliximab retreatment in patients with inflammatory bowel disease: a systematic review and meta-analysis by Seungwon Yang, Siyoung Yang, Young Kwon Jo, Seungyeon Kim, Min Jung Chang, Junjeong Choi, Jae Hee Cheon and Yun Mi Yu in Therapeutic Advances in Chronic Disease
Footnotes
Author contribution
Y.M.Y. and J.H.C. conceptualized and designed the study and prepared the manuscript. S.Y. (Seungwon Y.) performed the data analysis and drafted the manuscript. S.Y. (Siyoung Y.) conducted a literature search and performed data extraction and analysis. Y.K.J. conducted a literature search and performed the data extraction. S.K. performed the data analysis, interpreted the data, and revised the manuscript. M.J.C. and J.C. interpreted the data and reviewed the manuscript. All authors reviewed, amended, and approved the manuscript for submission.
Conflict of interest statement
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: S.Y. has been employed by Celltrion, a biopharmaceutical company that produces infliximab. The other authors declare no conflicts of interest.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
