Abstract
Objective:
We aimed to examine the effect of doxycycline on serum levels of neuron-specific enolase (NSE), a marker of neuronal damage in traumatic brain injury (TBI) patients.
Methods:
Patients were randomly assigned into two groups (n = 25 each) to receive either placebo or doxycycline (200 mg daily), with their standard management for 7 days.
Results:
NSE serum levels in the doxycycline and control groups on day 3 were 14.66 ± 1.78 versus 18.09 ± 4.38 ng/mL, respectively (p = 0.008), and on day 7 were 12.3 ± 2.0 versus 16.43 ± 3.85 ng/mL, respectively (p = 0.003). Glasgow Coma Scale (GCS) on day 7 was 11.90 ± 2.83 versus 9.65 ± 3.44 in the doxycycline and control groups, respectively (p = 0.031). NSE serum levels and GCS scores were negatively correlated (r = −0.569, p < 0.001).
Conclusion:
Adjunctive early use of doxycycline might be a novel option that halts the ongoing secondary brain injury in patients with moderate to severe TBI. Future larger clinical trials are warranted to confirm these findings.
Keywords
Introduction
Traumatic brain injury (TBI) is a major health concern that affects more than 57 million individuals yearly worldwide. 1 TBI pathogenesis is a complex process that results from primary and secondary injuries which lead to neurological deficits. 2 The primary injury is irreversible and results in immediate neuronal cell necrosis; a cascade of cellular and molecular changes, referred as secondary injury, starts later as a complication of the primary insult. Despite exhaustive research, the pathophysiology of the secondary injury is not completely understood. Strong evidence suggests that blood–brain barrier (BBB) disruption, cerebral ischemia and neuroinflammation contribute to the characteristic features of secondary neuronal damage post TBI.3,4 Secondary insult is potentially reversible; however, if untreated, it may lead to progressive white- and gray-matter damage.5–7
Tetracyclines, in addition to their antibiotic properties, have recently been found to have anti-inflammatory8,9 and anti-apoptotic properties.10,11 Doxycycline, a second-generation tetracycline, recently gained particular attention as a neuroprotective drug. 12 Experimental use of doxycycline has been associated with reduced progression and severity of several neurodegenerative diseases: multiple sclerosis, 13 Alzheimer’s, 14 and Parkinson’s disease. 12 In a study by Meli et al. 15 doxycycline improved survival and posed a significant cerebral anti-inflammatory effect in an animal model of meningitis. Limited data are available regarding doxycycline use in TBI. One study showed improved neurological functions in mice treated with doxycycline following TBI; doxycycline preserved the BBB integrity by inhibiting the activity of the matrix metalloproteinase-9, 16 one of the main proteolytic enzymes implicated in BBB disruption. Recently, a randomized controlled trial assessed the effects of minocycline, a tetracycline derivative, on neurological outcomes in patients with brain trauma. Compared with placebo, minocycline significantly reduced serum levels of brain injury biomarkers in those patients. 17 Current therapies targeting the underlying pathophysiology of a TBI are lacking. Doxycycline readily penetrates the BBB. 18 Its affordability and excellent safety profile make it a novel promising option that perhaps attenuates secondary neuronal loss post TBI. 19
Neuron-specific enolase (NSE) is a promising marker of brain damage and recovery after TBI. 20 Inverse correlation between levels of NSE with Glasgow Coma Scale (GCS) have been established. Its validity as a short-term predictor marker after TBI has also been verified.21,22 For the first time in humans, this study aimed to examine the impact of adjunctive short-term use of doxycycline on clinical neurological functions and serum level of NSE in patients with moderate to severe TBI.
Patients and methods
Study design
This study was a single-center, randomized, placebo-controlled, double-blinded study performed in patients suffering from moderate to severe TBI. The study was carried out at Tanta University Hospitals, Tanta, Egypt. Institutional ethical approval of the study protocol was obtained (approval number: 1219PP21). The study protocol was registered prior to patient enrollment at clinicaltrials.gov (NCT04239755). The study was performed according to the Declaration of Helsinki. Written informed consent was obtained from the patients or their legally authorized representatives before enrollment.
Patients and interventions
Patients between 18 and 70 years of age with moderate (GCS 9–12) or severe (GCS 3–8) isolated TBI were assessed for the study eligibility. 23 Inclusion criteria were less than 24 hours (h) since traumatic injury and the ability to tolerate enteral feeding. The exclusion criteria were treatment with doxycycline in the past 10 days, the first dose of study drug unable to be given within 24 h of the primary injury, hypersensitivity to tetracyclines, pregnant/lactating women, autoimmune abnormalities, history of receiving isotretinoin, hepatic or renal dysfunction or known malignancies. Patients were managed according to an institutional protocol based on the Brain Trauma Foundation Guidelines.2,24 Emergency surgical treatment was based on neurologic status and findings on head computed tomography (CT) criteria.
Patients (n = 50) were randomly allocated using computer-generated random sequence in (1:1 ratio) to enroll either in: doxycycline group who received 100 mg doxycycline capsules twice daily, either orally or by feeding tube, in addition to the standard treatment for 7 days; or placebo group who received placebo capsules twice daily, either orally or by feeding tube, in addition to the standard treatment for 7 days.
The first dose was administered within 24 h of the estimated time of TBI. For enteral administration of the study drugs, the powder of capsule was dissolved in 5 mL water and the contents were drawn up to the syringe and immediately administered via the nasogastric tube. The plate and syringe were rinsed with a plenty of water and the flush contents were also emptied into nasogastric tube.
Patient assessment and follow-up
Patient demographics, underlying diseases, drug history, mechanism of injury, vital signs, and GCS values were assessed in all patients at baseline. Additionally, the severity of brain injury was assessed using the Rotterdam CT score based on the admission of CT scan by a neurosurgeon that was blinded to the treatment assignment. A completely normal-appearing scan has a Rotterdam score of 1 and the worse possible score is 6. The GCS scores were evaluated by independent investigators blinded to treatment allocations for 7 days. The Glasgow Outcome Scale-Extended (GOS-E) 25 score was used for evaluation of the recovery level after 3 months, and the overall survival was evaluated 28 days after randomization. In this regard, patients or their first-degree relatives were contacted for assessment of survival and GOS-E score. Outcome assessors and patients were blinded to treatment allocation.
Study outcomes
The primary outcome of the study was the difference between the two study groups in mean NSE serum levels at day 7. Secondary end points of the study included differences between study groups in GCS scores over 7 days, mortality rate at 28 days, and GOS-E level at 3 months. The GOS-E level was defined as a GOS-E of 1–4 (death, vegetative state, and severe disability) or a GOS-E of 5–8 (moderate disability and good recovery). 26
Biochemical analysis
Venous blood samples were collected from all patients to determine NSE levels before administration of study drugs (baseline assessment), 3 days, and 7 days post randomization. The serum was separated by centrifugation (3000 rpm for 10 min), and the isolated serum was immediately frozen at −80°C. NSE levels were measured by enzyme-linked immunosorbent assay using a human NSE EIA kit (Cloud-Clone Corp, USA) according to the manufacturer’s instructions.
Statistical analysis
The required sample size was calculated using G*Power software version 3.1.0 (Institut fur Experimentelle Psychologie, Heinrich Heine Universitat, Dusseldorf, Germany). It was estimated that a total sample size of 40 patients would have a power of 81% to detect a medium to large effect size of 0.94 in the primary outcome measure. Statistical analyses were performed using Statistical Program for Social Sciences (SPSS) software version 26.0 (IBM®, Inc., Chicago, IL). Numerical data were expressed as mean and standard deviation or median and range as appropriate. Qualitative data were expressed as frequency and percentage. Data were tested for normality using Kolmogorov–Smirnov and Shapiro–Wilk test and were found not normally distributed. Comparison between two groups with respect to continuous variables was done using Mann–Whitney test. Regarding assessment within a group, comparisons among baseline, after 3 days, and 7 days were done using the Freidman test. Post hoc analysis was conducted with a Bonferroni correction applied. The Chi-square test or Fisher’s exact test was used to compare between the groups with respect to categorical data. Survival was estimated by Kaplan–Meier survival method and the difference was evaluated by the log rank test. Spearman’s correlation was used to for analysis of the bivariate relationship. All p-values were 2-sided and a value of less than 0.05 was considered significant.
Results
Our study, conducted from February 2020 to October 2020, included a total of 86 patients with brain trauma who were screened for eligibility. Fifty patients fulfilled the inclusion criteria and were randomly allocated to one of the study groups. As shown in Figure 1, 40 patients completed the study and were included in the final analysis. The baseline characteristics and physiological variables were balanced across the two groups. Thirty-five patients (70%) were diagnosed with moderate TBI, whereas 15 patients (30%) were diagnosed as severe cases. Initial injury severity and mean time from injury to randomization were also similar between the two arms of the study (Table 1). Both groups were comparable with respect to the length of intensive care unit stay (p = 0.390).

CONSORT flow diagram showing the flow of patients throughout the study.
Baseline characteristics of all enrolled patients.
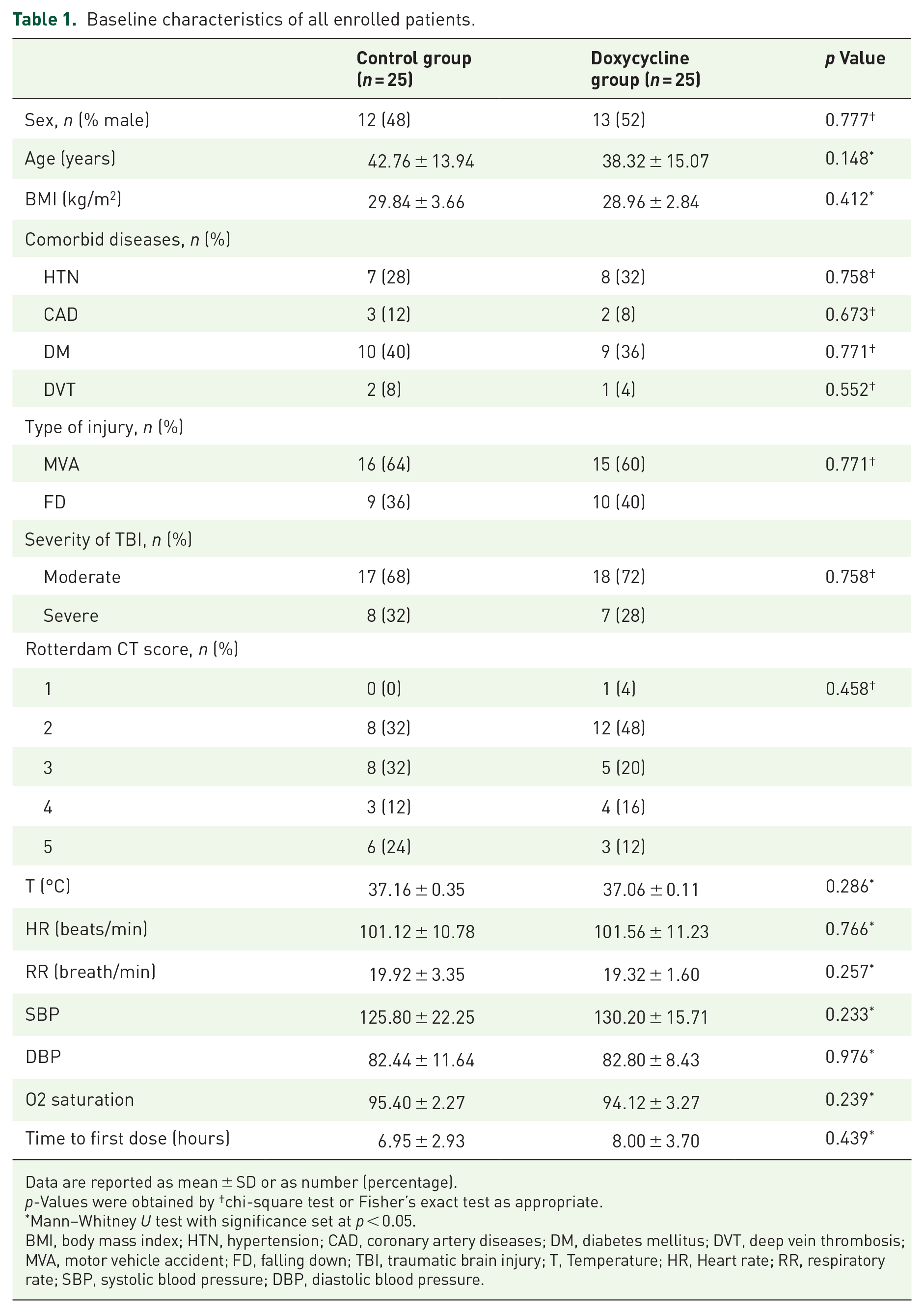
Data are reported as mean ± SD or as number (percentage).
p-Values were obtained by †chi-square test or Fisher’s exact test as appropriate.
Mann–Whitney U test with significance set at p < 0.05.
BMI, body mass index; HTN, hypertension; CAD, coronary artery diseases; DM, diabetes mellitus; DVT, deep vein thrombosis; MVA, motor vehicle accident; FD, falling down; TBI, traumatic brain injury; T, Temperature; HR, Heart rate; RR, respiratory rate; SBP, systolic blood pressure; DBP, diastolic blood pressure.
In each of the study groups, a marked decrease in the mean NSE serum levels was observed overtime as compared with baseline (p = 0.005 in control group versus p < 0.001 in doxycycline group) (Table 2). The comparison of the intragroup changes in the NSE serum levels revealed significant day-to-day reduction in NSE when day 3 was compared with day 7 in both groups (p < 0.001 in both groups). Nevertheless, the NSE levels recorded marked significant decrease at day 7 compared with baseline only in the doxycycline group (p = 0.001) (Figure 2).
Patient outcomes on during study days in the doxycycline and control groups.

Data are reported as mean ± SD, Median (IQR) or as number (percentages) as appropriate.
chi-square test or Fisher’s exact test.
Mann–Whitney U test for comparisons between groups.
Freidman test for Comparisons of groups over time with significance set at p < 0.05.
NSE, neuron-specific enolase; GCS, Glasgow Coma Scale; ICU, intensive care unit; ng, nanogram.

Clustered box plot of serum levels of neuron-specific enolase (ng/mL) for the studied interventions measured at three time points.
At baseline, the mean NSE levels were comparable between the two groups (at day 0) (p = 0.148) (Table 2). However, visual inspection of individual data points reveals a more scattered spread of values and higher NSE median levels in the control group compared with doxycycline group (Figure 2). On day 3 of the study, the mean serum NSE levels were significantly lower in patients allocated to the doxycycline group compared with those allocated to the control group (14.66 ± 1.78 versus 18.09 ± 4.38 ng/mL, respectively; p = 0.008). At day 7, the doxycycline group showed markedly lower NSE levels of 12.81 ± 2.10 ng/mL compared with 16.43 ± 3.85 ng/mL in the control group (p = 0.003) (Table 2). The mean percentage reduction in serum NSE levels from baseline to day 7 was statistically significant, [17.7% in the doxycycline group versus 6.7% in the control group (p = 0.02)]. At the end of treatment period (day 7), the serum NSE levels were within normal limits (⩽12 ng/mL) only in 25% (n = 5) in the control group while it normalized in 45% of patients (n = 9) who received doxycycline (p = 0.185) (Figure 3).

Percentage of patients with normal (⩽12 ng/mL) and high (>12 ng/mL) neuron-specific enolase (NSE) levels in the study groups on different days 7 of the study.
GCS values did not differ between the doxycycline and control group at baseline (p = 0.255) and at day 3 (p = 0.116); yet significant higher GCS scores were recorded at day 7 in the doxycycline group compared with control group (p = 0.031) (Table 2). There was a significant correlation between reduction of NSE serum levels and the rise in GCS scores at day 7 in all patients (r = −0.569, p < 0.001) (Figure 4). Our results revealed that short-term treatment with doxycycline did not improve the functional outcomes at 3 months post randomization. The total number of patients with poor outcomes (GOS-E of 1–4) was deemed comparable between the two study interventions groups (p = 0.736). Survival was analyzed for up to 28 days and was defined as the time (in days) that each patient survived from the day of randomization; death was included as event. The mean survival time was 23.1 days with placebo versus 24.4 days with doxycycline. Both groups were comparable with respect to mortality. Figure 4 shows Kaplan–Meier curves for comparison of time to death.

Correlation between neuron-specific enolase and Glasgow Coma Scale in the studied groups at day 7 (†r= - 0.569; P<0.001).

Kaplan–Meier survival curve represents difference in time to death between the study groups.
Discussion
The main therapeutic goal for patients with TBI is prevention of secondary injury. Currently, available therapeutic options provide only symptomatic relief and unable to halt the progression of secondary injury that occur following TBI. 2 Recent preclinical studies10,16,27 showed doxycycline as a promising choice that ameliorates the secondary brain injury. Hence, this study was designed to clinically investigate the benefit of adjunctive doxycycline use in acute TBI. NSE is an acute neuronal cell body injury marker. A strong relationship between serum levels of NSE and neurological outcomes has been established in patients after TBI. 28 Hence, effect of doxycycline on the serum levels of NSE was taken as the primary outcome. A similar approach was followed to investigate the neuroprotective effects of L carnitine 29 and memantine 30 in patients with TBI.
The current study has shown that serum NSE levels measured on day 7 were significantly reduced in patients who received doxycycline compared with placebo. Significant difference was also found regarding the proportion of patients who achieved normalized NSE levels, favoring doxycycline. At baseline, visually a difference in distribution pattern of NSE levels was observed between the two study groups. The small sample size and the considerable previously reported heterogeneity in both study population 31 and outcome of interest 32 might explain this variability. By utilizing stratified sampling where possible confounders such as age, time of sampling and type of injury could be robustly controlled and should be considered in the future studies. The significantly higher percentage reduction of NSE levels from baseline reported at day 7 in doxycycline-treated patients denotes its potential benefit despite this heterogeneity. This is the first randomized study to examine the potential benefit of doxycycline on serum levels of NSE in patients with TBI; however, our finding is consistent with the conclusions of Koulaeinejad et al., 17 who showed the positive influence of short-term minocycline (7 days) in patients with TBI in a pilot randomized controlled trial. As compared with the placebo group, the serum levels of brain injury biomarkers including NSE markedly decreased in the minocycline-treated group. On the preclinical level, Kovesdi et al. 33 showed that minocycline treatment for 4 days normalized serum and tissue levels of NSE in a rat model.
The reduced level in the NSE has strong plausibility, as doxycycline contributes to the hallmarks of secondary neuronal insult: neuroinflammation and BBB disruption.3,4 Jantzie and Todd 34 showed that a single dose of doxycycline given before brain injury significantly reduced the proinflammatory cytokine levels for 7 days post-injury in an animal model. Likewise, the neuro anti-inflammatory action of doxycycline has been recently shown in several trials.35,36 Doxycycline also has a BBB-stabilizing effect. This influence could be explained by its inhibitory effect on matrix metalloproteinase 9, the main enzyme implicated in BBB disruption, 37 that has been previously shown both clinically 38 and experimentally. 39
Although NSE levels have been identified as strong predictors of mortality among patients with TBI, the current study demonstrated that doxycycline did not improve the survival of patients. Additionally, the proportion of patients with severe disability as measured by the GOS-E (1–4) deemed comparable between both arms of the study after 3 months. For safety concerns, we administered the minimum recommended antibiotic dose (100 mg doxycycline twice daily). Furthermore, this study aimed primarily to investigate the impact of the short-term use of doxycycline on NSE, one of the acute brain injury biomarkers. Hence, the short duration and the small dose used might explain our findings regarding lack of effect on mortality and functional neurological outcomes. It is possible that higher doses or use for longer periods are needed for doxycycline to significantly improve the survival and the long-term neurological outcomes in patients with TBI.
Therapeutic strategies affecting gut microbiota such as antibiotics offered a neuroprotective benefit by targeting the gut–microbiota–brain axis in different neurological conditions. 40 The relationship between neurological degeneration induced by TBI and the gut microbiome represents an area of great development. 41 Apart from doxycycline’s BBB-stabilizing action and its neuro anti-inflammatory effect, doxycycline-induced modifications of the gut microbiota might potentially serve as a therapeutic target to prevent secondary injury in brain-injured patients. Future studies with longer treatment courses are needed to investigate how modulation of gut microbiota could potentially enhance the long-term neurological outcomes and quality of life for patients with TBI.42,43
The positive influence of doxycycline on NSE biomarker serum level was coupled with significantly higher GCS score in the doxycycline group at the end of the treatment period. Our results showed significant negative correlation between NSE and GCS evaluated at day 7. This agrees with the findings from previous studies.44,45 Contrary to our findings, the results of Naeimi et al. 46 found direct association between the levels of NSE and the pathological findings shown by the CT scan but failed to establish significant correlation between the NSE and GCS. Lack of correlation with our results might be attributed to the difference in the patient population, as all their enrolled patients had severe TBI.
As doxycycline has a safe pharmacologic profile, it is worthy of being reexamined in future clinical trials with a particular emphasis on its dosing regimen and duration. Further studies are also warranted to examine whether the positive impact of doxycycline would be maintained in other areas of brain injury such as stroke and brain hemorrhage.
The current pilot study aimed mainly to explore for first time the potential therapeutic opportunity of doxycycline in patients with TBI. The neurological outcomes could be affected by some confounders such as age, comorbidities, or gender. This study was limited by the small sample size. The simple randomization employed in our study hindered our ability to examine how these confounders might potentially impact the study outcomes. Neither the precise mechanism for secondary brain injury nor the optimum regimen for doxycycline use as a neuroprotective drug is known.
Conclusion
We concluded that early administration of doxycycline might have a neuroprotective effect in patients with TBI. Doxycycline has shown a significant reduction in the NSE serum levels, a neuronal damage biomarker. Favorable associated short-term outcomes on the GCS scores have also been shown. Considering the long history of safe use of doxycycline in clinical settings, future larger clinical trials with stratified subsets of participants and long-term outcomes are necessary to confirm these findings. Use of different dosing regimens is also warranted to explore the potential magnitude of benefit from doxycycline in halting the progression of neurodegeneration in patients with TBI.
Footnotes
Acknowledgements
The authors are grateful to all study participants and to the medical staff in Emergency Medicine and Traumatology Department – Tanta University Hospitals for their help and valuable contributions.
Conflict of interest statement
The authors declare that there is no conflict of interest.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
