Abstract
Atopic dermatitis is a heterogeneous disease and resists classification. In this review, we discuss atopic dermatitis nomenclature and identify morphologic phenotypes, which will facilitate correct diagnoses and development of treatment strategies. We support using the term ‘atopic dermatitis’ rather than eczema, because it describes the allergic background and inflammation (‘itis’) as drivers of the disease. Atopic dermatitis has many morphologic manifestations that vary by topographic area affected, age, or race and require consideration in differential diagnosis. Different phenotypes based on morphology and topographic location, ethnicity, and age are discussed. A better-defined phenotype identification for atopic dermatitis will facilitate earlier and correct diagnosis of this complex condition and inform selection of the most appropriate treatment choice in an era in which targeted therapies may generate more individualized patient care.
Introduction
Atopic dermatitis (AD) is a chronic inflammatory skin condition affecting both children (15–20%) and adults (2–5%).1–3 Its prevalence is increasing, particularly in lower-income countries, and it occurs in a variety of geographic and ethnic patterns.3,4 Previously classified as either early-onset [i.e. infantile (aged <2 years), childhood (aged 2–12 years), adolescent (aged 12–18 years)], or adult-onset (aged >18 years), a separate subgroup of elderly-onset AD (aged ⩾60 years) has recently been described.5–7 AD can therefore be regarded as a lifelong disease, and clinical and laboratory differences observed in these subtypes contribute to the heterogenous nature of the disease. 8
Traditionally, clinical lesions in eczema are classified as ‘acute’ (oozing, oedema, and erythema) or ‘chronic’ (dyspigmentation, xerosis, and lichenification) (Figure 1). However, as AD is a chronic relapsing condition, both types of lesions can coexist during flares. Pruritus is a hallmark of AD, and excoriations secondary to scratching are often present. Diagnosis of AD is largely based on the morphology and distribution of lesions,2,3,9 but also on the patient’s clinical course, comorbid conditions, and family history. Our understanding of the pathogenesis of AD in part reflects the complex interplay between genetic and environmental factors. 9

Clinical signs of atopic dermatitis.
Many genes have been linked to AD across different populations, but the extent of genetic risk for AD is yet to be fully defined. However, candidate gene studies, genome-wide association studies, and genetic sequencing studies have shown that genetic susceptibility is associated with epidermal barrier dysfunction and type 2 dominated immune responses.9–13 Indeed, type 2 inflammation plays a key role in both acute and chronic phases of the disease.14–17 However, an incomplete understanding of the underlying immunologic mechanisms of AD and its heterogeneous manifestations has resulted in a therapeutic approach based more on disease severity than on phenotype or its underlying specific biologic pathways (endotype).
Objectives
First, we discuss the nomenclature associated with AD. Second, we identify and illustrate different morphologic phenotypes seen in patients of different age ranges or races and in different anatomic locations. To guide appropriate treatment decisions, it is critical that the different AD phenotypes are diagnosed efficiently and correctly and in a timely manner.
AD nomenclature
Atopy, which is Greek for ‘out of place’, entered the medical lexicon in 1923, with ‘atopic dermatitis’ following a decade later, soon after the term ‘atopic eczema’ had first been used.18–21 The American Academy of Asthma and Immunology defines atopy as ‘the genetic tendency to develop allergic diseases such as allergic rhinitis, asthma, and AD’ (or ‘eczema’, depending on the terminology used). 22 Most individuals with AD are sensitized to allergens and have a coexisting allergic disease, and allergen exposure can have an effect on AD symptoms, severity, and flares.
The term ‘dermatitis’ was excluded from early discussions of hereditary and clinical presentation of allergic sensitization because it was considered difficult to define.19,23 An electronic survey of experts from the International Eczema Council led to a consensus (97.2%) on the need to include the prefix ‘atopic’ in all descriptions of the disease, regardless of a preference for ‘eczema’ or ‘dermatitis’. 18 We agree that use of the term ‘atopic’ provides important mechanistic information and helps group AD with other diseases with the same aetiology.
Differences in nomenclature create potential for confusion and for issues with the quality of epidemiologic data 24 given that AD and eczema are represented by different codes in the International Classification of Diseases, Tenth Revision (ICD-10), system. Besides the ICD-10 codes for AD (L20.x; Table 1), other ICD-10 codes can be used in diagnosis.
ICD-10 codes that can be used for AD and its different morphologic phenotypes.

AD, atopic dermatitis; ICD, International Classification of Diseases, Tenth Edition.
Thus, the heterogeneity of AD presentation may be a source of the varied terminology used to describe AD. Consensus within the medical community is necessary to avoid confusion, bias, and errors in epidemiologic data. We argue for the use of ‘atopic dermatitis’ over ‘atopic eczema’ because it more fully captures the inflammatory aetiology of the disease, an important feature when considering use of new targeted therapies. Education of the lay community will be a key next step to ensuring use of consistent terminology.
Diagnosis of AD
The diagnostic criteria used for AD have been thoroughly reviewed by Andersen et al. 19 Diagnostic guidelines from the UK Working Party 25 and the American Academy of Dermatology (AAD)26,27 are established for paediatric and adult diagnoses. These guidelines are refinements of the Hanifin–Rajka criteria, first introduced in 1980 and comprising four major and 23 minor clinical features, of which at least three major and three minor criteria must be present to confirm an AD diagnosis. 28 The UK Working Party Criteria mandate that pruritus must be present along with at least three of five major criteria: history of flexural involvement, history of dry skin, onset of AD before 2 years of age, history of any other atopic condition, and flexural dermatitis. 25 The AAD guidelines comprise essential, important, and associated clinical features. For a positive diagnosis of AD, essential features (pruritus and eczema) must be present, and important features (early AD onset, history of atopy and xerosis) are usually present, lending further support to the diagnosis; evidence of associated features (e.g. facial pallor, lichenification), while suggestive of AD, are not a mandatory requirement. 27 The Millennium Criteria were the first to require the presence of antigen-specific immunoglobulin E (IgE) as a mandatory criterion for the diagnosis of AD. 29 The presence of IgE antibodies is also required for some definitions of atopy, such as that of the World Allergy Organization. 18 However, this criterion is controversial because it excludes as many as two-thirds of patients with AD who do not display this type of response 30 and is generally not used as a diagnostic criterion in practice.
Several challenging clinical phenotypes of AD exist that display peculiar morphologic features or topographic distributions, and some of these phenotypes have difficult-to-discern aetiologies or may mimic or coexist with other dermatologic conditions. These challenging phenotypes can be defined morphologically [nummular dermatitis, prurigo nodularis (PN)-like lesions, erythroderma, lichenified dermatitis, follicular/papular dermatitis and pompholyx (dyshidrosis)]4,31–43 (Figure 2) and topographically (flexural or periorificial or occurring on the face, lips [eczematous cheilitis], eyelid, head and neck, hand and foot, or nipple)34,36,38,39,41–46 (Figure 3). Figures 4 and 5 show histologic samples of AD.

Morphologic phenotypes. (a) Nummular (discoid) dermatitis. (b) Prurigo nodularis. (c) Erythrodermic. (d) Lichenoid dermatitis. (e) Follicular/papular dermatitis. (f) Dyshidrosis or Pompholyx.
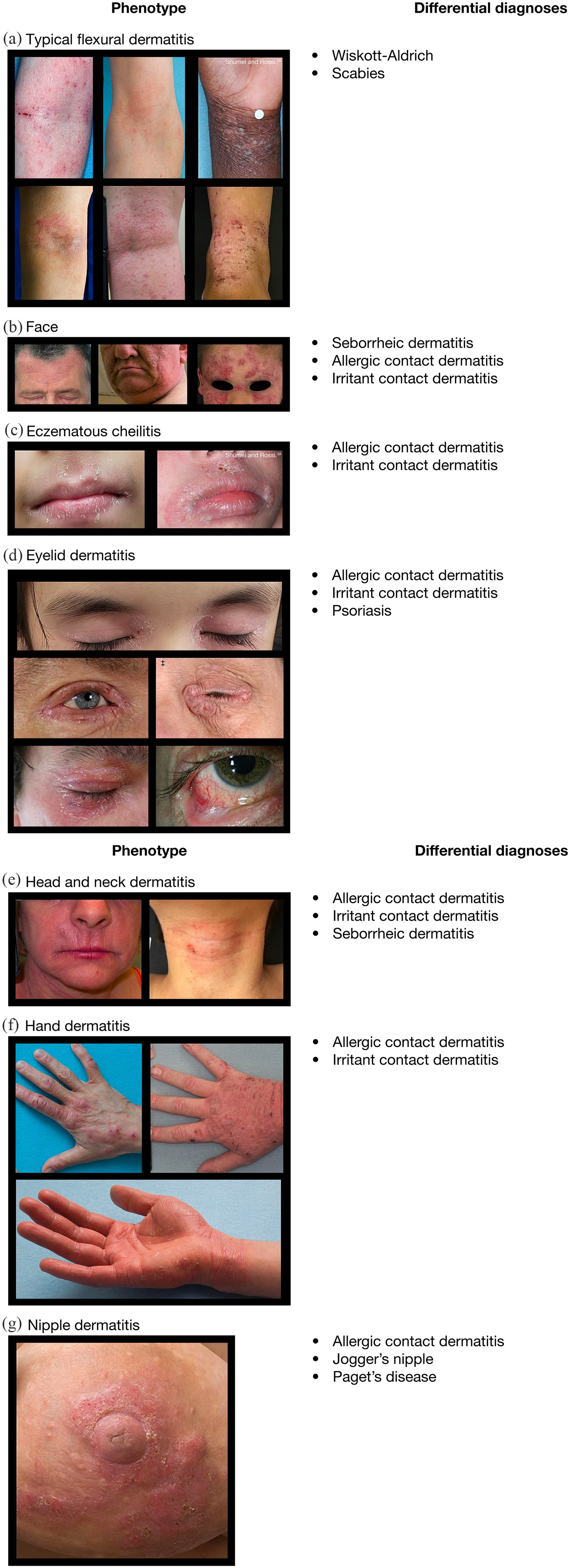
Topographic phenotypes of AD. (a) Typical flexural dermatitis. (b) Face. (c) Eczematous cheilitis. (d) Eyelid dermatitis. (e) Head and neck dermatitis. (f) Hand dermatitis. (g) Nipple dermatitis.

Atopic dermatitis: histology. (a) Normal skin. (b) Atopic dermatitis. (c) Dyshidrosis. (d) Acute dermatitis. (e) Chronic dermatitis.

Histology: subacute chronic dermatitis, psoriasiform dermatitis. (a) Sub-acute-chronic dermatitis. (b) Psoriasiform dermatitis.
No consensus exists on whether some of these challenging phenotypes represent exclusive forms of AD, and this lack of consensus is further complicated by the availability of separate ICD-10 codes for these manifestations (Table 1). There is a need to confirm which of, and when, these conditions should be considered forms of AD and which are independent diagnoses. We propose that nummular dermatitis, PN-like AD, erythroderma, pompholyx, lichenified dermatitis, flexural dermatitis, facial dermatitis, eczematous cheilitis, eyelid dermatitis, head and neck dermatitis (HND), hand and foot dermatitis, and nipple dermatitis should be considered part of the spectrum of AD when other AD features (e.g. typical flexural eczematous lesions), elevated IgE, or atopic comorbidities (history of asthma, rhinoconjunctivitis, food allergy) are present, especially when there is no evidence of other diseases known to cause these phenotypes.
Morphologic AD phenotypes
Nummular or discoid dermatitis
The term ‘nummular’ derives from the coin-like appearance of lesions that form rapidly by the merging of papules and papulovesicles into circinate and ovoid plaques (Figure 2a).34,40 Lesions are typically 1.5–3 cm in diameter and progress from a highly inflammatory acute phase characterized by marked erythema, crusty appearance, and oozing/weeping to a smoother, scaly phase that may become annular because of central clearing and peripheral extension. Lesions are predominantly present on lower extremities but can also appear on upper extremities and the trunk. 47 Nummular dermatitis is the most common atypical morphologic variant of AD, 48 but no agreement exists as to whether nummular dermatitis is AD in all cases. In most cases, nummular dermatitis is idiopathic, and no allergic or irritant cause can be discerned. It is more common in children (although not infants) and adult-onset AD, 49 and coexists in some individuals with typical flexural AD or elevated serum IgE (or both).50,51 As occurs in AD, staphylococcal superantigen-producing Staphylococcus aureus colonization is commonly present in nummular dermatitis. 52 Nummular dermatitis may be considered AD when other features of AD (e.g. typical flexural eczematous lesions), elevated IgE, and atopic comorbidities (history of asthma, rhinoconjunctivitis, food allergy) are currently or have been present and when no evidence exists for other diseases (e.g., stasis dermatitis) that are also known to cause nummular dermatitis.31,32
Prurigo nodularis
PN (Figure 2b) is a condition distinct from AD, but PN secondary to AD can occur. PN is characterized by single to multiple excoriated hyperkeratotic and intensely itchy papules and nodules that occur predominantly on the extremities.33,53 Pruriginous lesions are persistent and tend to be symmetrically distributed in areas accessible to scratching, with normal or lichenified skin between the lesions, and a characteristic ‘butterfly’ sign on the back where no lesions are present in areas inaccessible to scratching. PN is commonly located on the extensor surfaces of the extremities and rarely affects the face. 54 Pruritus may be accompanied by burning, stinging, pain, and other symptoms. There is often neuronal sensitization, demonstrated by allokinesis (light touch-evoked itch) and hyperkinesis (exaggerated itch response to a pruritic stimulus). 55 The key immune mediators and mechanisms behind atopic itch in AD have been reviewed and include histamine, TSLP and type-2 cytokines. 56 The key role of type 2 cytokines in PN is emphasized by the very good therapeutic response to dupilumab. 57
AD has been identified as an underlying or contributing cause in nearly one-half of PN cases.58,59 PN secondary to AD is more common in adults and in individuals of South-East Asian or African origin.4,59,60 In an AD registry study performed in Japan, the prevalence of prurigo nodules in 300 patients with AD was high: 30.9% in patients with moderate AD and 56.3% in patients with severe AD. 61 Itch is a cardinal symptom in AD, and the itch–scratch cycle could lead to secondary PN lesions. Accordingly, PN can coexist with AD or persist after cessation of AD. 33
Erythroderma
Erythroderma (Figure 2c), also known as exfoliative dermatitis, is the presence of erythema on >90% of the body surface area. Erythroderma typically begins with the appearance of erythemato-pruritic lesions of varied primary morphology, most often on the head, trunk, and genital region, and rapidly spreads to all or most of the body within days or a few weeks. The palms of the hands and soles of the feet tend to be spared, along with the nose (nose sign) in some cases.62,63 Scaling of the skin follows, with large scales in acute cases and small scales in chronic cases. 62 Erythrodermic AD is more common in adolescents and adults (aged 12–60 years) in East Asia, particularly those with a longer disease course.4,64,65 Erythroderma is not specific to AD and a differential diagnosis must consider numerous causes, but AD has been reported to be the underlying cause of erythroderma in 5%–24% of cases. 66 Erythrodermic AD is a serious condition because it is associated with a high rate of hospitalization, skin infections, and potential life-threatening complications. 67
Lichenified dermatitis
Lichenified dermatitis (Figure 2d) refers to a thickening of the skin, which appears elevated, with accentuated creases and a leathery appearance due to prolonged scratching and rubbing. In an analysis of AD clinical trial data, lichenification was found to be more common in patients from South-East Asia or Africa than in Caucasian patients. 4 The condition is most common in young people (median age 7 years). 35 Along with pronounced greater lichenification, individuals from Africa or of African descent can present with perifollicular accentuation and post-inflammatory depigmentation, which can complicate the diagnosis. 60 People from Asia or of Asian descent with AD exhibit increased epidermal hyperplasia (increased epidermal thickness and Ki67 counts) on lesional biopsy specimens, which is associated with increased gene expression of T helper (Th)17- and Th22-related markers.60,65
Follicular/papular dermatitis
Follicular and papular dermatitis is a morphological subtype more commonly identified in darker skin phototypes (Figure 2e). An analysis of AD feature prevalence reported among clinical studies by region found that studies from Africa reported a higher prevalence of papular lichenoid lesions (22%) compared with the overall prevalence from all studies (22%). 4
Topographical phenotypes
AD typically affects the face, neck, and flexural zones. 27 Many of the morphologic variants show predisposition for certain areas of the body. Some topographical phenotypes of AD are described as follows.
Typical flexural dermatitis
A tendency for AD to affect flexural areas was first noted in Roman Emperor Augustus by Suetonius,68–70 and remains a prominent feature of both US and UK diagnostic guidelines.12,25,27,64,71 The signs most frequently seen with flexural involvement are erythema, oedema or papulation, excoriation, lichenification, and oozing or crusting (Figure 1). Flexural areas typically affected by AD include the neck, cubital and popliteal fossae, wrists, and ankles (Figure 3a).
The extent of flexural involvement appears to vary with disease course and differs by racial origin. Flexural involvement is more common in patients with early-onset AD and a chronic persisting course. 72 There may be sparing of flexural involvement in patients aged >60 years. 5 Whereas flexural involvement is common in adolescent and adult Caucasians, extensor involvement appears to be more common in infants and Asians and can occur in those of African descent35,43,73,74 (Figure 3a, right two panels).
Face
Facial involvement (Figure 3b) is common in AD, not only in children and infants, 27 but also in adolescents and adults. In many individuals, AD may involve the face alone; in infants aged <1 year, AD is typically restricted to the face, where it is characterized by erythematous and highly pruritic lesions alongside moist, oozing papulovesicles that sometime form crusts or scales45,73 and can be concentrated in the perioral region,5,75 on the cheeks, or, in some cases, the scalp. 73 Periorificial lesions of the head tend to occur in AD during childhood. 5
Eczematous cheilitis
Eczematous cheilitis (Figure 3c) can be a manifestation of AD and presents as red, dry lips, and sometimes with a median fissure of the lower lip, alongside lateral fissures (angular cheilitis). 76 In nearly one-fifth of cases, cheilitis is a manifestation of AD. 77 Chronic cheilitis can be the result of exposure to irritants or allergic contact dermatitis to sensitizers such as nickel, fragrances, Balsam of Peru, chromium salts and manganese salts present in cosmetics, dental materials, and oral hygiene products. 77 In contrast, angular cheilitis (Figure 3c) is usually infectious in nature.
Eyelid dermatitis
Substantial eyelid involvement was highlighted as a key feature in the first description of AD, but eyelid involvement is common in several forms of dermatitis (Figure 3d).19,73 Itching, burning, and crusting of the eyelids is common, and scaling may occur at the base of the eyelashes, along with depigmentation, lichenification, and loss of lashes.39,78 AD is associated with ocular diseases, including conjunctivitis and cataracts.79,80 Inadequate treatment of eyelid dermatitis, blepharitis, or periocular eczema can lead to severe chronic inflammation with scarring and subsequent complications like ectropion or entropion as well as to ocular surface diseases such as keratoconjunctivitis (Figure 3d).
Similar to eczematous cheilitis, eyelid dermatitis is a common manifestation of AD.45,64 Eyelid dermatitis is more common in childhood and may also be more common in people of Asian descent, particularly females.4,81 Onset after 6 months of age and a personal history of atopy are the main risk factors for eyelid dermatitis in AD. 82 Of note, long-term use of potent topical steroids to treat eyelid dermatitis can cause eyelid skin atrophy due to the thin skin of the eyelid, as well as serious adverse events such as glaucoma and cataracts, which are also associated with use of systemic corticosteroids.83–85
Head and neck dermatitis
In adolescents and adults, facial AD, often seen as xerotic, scaly, erythematous, and sometimes lichenified plaques affecting the face and neck and eventually the scalp, is one of the causative factors of HND (Figure 3e).
There are many other potential causes for HND, such as aeroallergen sensitization, allergic contact dermatitis to topical products, rosacea, seborrheic dermatitis, topical corticosteroid (TCS) withdrawal syndrome, and sensitization to Malassezia furfur,37,38,76,81,86 and these may coexist with AD in the same person.
Allergic contact dermatitis (ACD) commonly involves the lips, eyelids, and neck, among other regions, and may be mistaken for or complicate AD. 87 The delayed hypersensitivity reaction observed with ACD following prior sensitization to an allergen results in highly pruritic erythematous plaques. AD treatments may be implicated in ACD, and allergens with higher rates of sensitization among patients with AD include formaldehyde releasers, metal allergens, cocamidopropyl betaine, and fragrances. ACD should also be investigated in paediatric patients; although rates of sensitization are comparable with the general population, ACD is a highly relevant diagnosis in children, and paediatric skin with an immature barrier function is more susceptible to toxicity. 88 In a UK retrospective case study of 114 children aged 3–15 years who were patch-tested over a 3-year period, findings on patch test were positive in 33% of patients with facial dermatitis, 40% with perioral dermatitis, and 86% with eyelid dermatitis. 89 Paediatric patients with moderate-to-severe and early-onset AD have a higher incidence of sensitization to topical products, most commonly to emollients and antiseptics. 90 Sunscreens can also be a cause of ACD on the face and neck, and both sensitization and photosensitization should be investigated when suspected.91,92
HND can be induced by TCS withdrawal. In particular, overuse of potent TCS on the face can lead to many adverse events upon withdrawal, including burning, itching, and facial redness.93,94 A retrospective analysis of 55 people in Australia suspected to be undergoing TCS withdrawal found that 100% of patients experienced redness and itch. Of these patients, TCS were initially prescribed for AD (76%), contact dermatitis (15%), or other rashes (9%). 93
In addition, recent reports have suggested an association between treatment of AD with the biologic dupilumab and development of new regional dermatoses, with a predilection for the face, head, and neck regions.95–97 This rare phenomenon has yet to be fully clinically characterized and a unifying phenotype has not been identified. These reports may represent disparate rashes with multiple underlying aetiologies. It is possible that dupilumab treatment potentially unmasks previously undiagnosed ACD or seborrheic like-dermatitis, which had been kept at bay prior to dupilumab initiation. A recent case report suggests that topical ketoconazole is successful in treating facial rashes refractory to dupilumab. 98
Elevated serum levels of Malassezia-specific IgE are characteristic of HND in contrast to seborrheic dermatitis or pityriasis versicolor.99–101 In 2000, Devos and Van der Valk investigated whether positive prick tests to P. ovale were associated with a specific localization in the head and neck region in a cohort of 589 patients. They found that serum levels of Malassezia-specific IgE were elevated in all patients with AD and HND but in only 13.6% of patients with AD without HND. 100
A positive response to oral itraconazole may support the clinical diagnosis of HND. Randomized, placebo-controlled trials have shown significant clinical improvement of suspected HND in patients with AD treated with systemic antifungals.102–104
Hand and foot dermatitis
AD of the hand (Figure 3f) presents usually as xerotic, scaly, lichenified, and fissured skin, mainly on the dorsal hands. 105 This phenotype predominates in adulthood, although specific manifestations do occur in children (juvenile palmoplantar dermatitis or dermatitis plantaris sicca).4,73 In adults, the lifetime prevalence of hand dermatitis is 15%, 106 and it is twice as common in females as in males.106,107 In a study of Swedish children, approximately 70% of those with hand dermatitis at age 16 years had a history of AD, and the risk of hand dermatitis was greater in children with persistent or more severe AD. 108 In a French study that evaluated the benefit of therapeutic patient education in 72 adults with chronic hand eczema, 43.7% had a personal history of AD. 109 A retrospective review and clinical examination of 725 German adolescent and adults with AD found that two-thirds of patients whose dermatitis developed after infancy and then persisted into adulthood had hand dermatitis. 75 A systematic review and meta-analysis of 35 studies in 12 countries showed that patients with AD have a 3- to 4-fold increased incidence of hand dermatitis when compared with controls, 110 and that hand dermatitis in schoolchildren with AD frequently affects only the dorsa. 111
Hand dermatitis is often associated with occupational or domestic exposure to irritants and can be aggravated by daily tasks that necessitate contact with water, such as dishwashing, laundry, and childcare. However, a history of childhood AD appears to be the main risk factor for development of hand dermatitis. 105 Approximately 29% of people with hand dermatitis have a history of AD. 112 A Norwegian study found that 90% of school children with hand eczema also reported AD, which suggests that hand eczema in children is closely associated with AD. 111 In most cases, distinguishing between contact dermatitis and AD of the hand caused by endogenous factors is impossible by clinical presentation alone, and patch testing is warranted to determine whether any concomitant contact dermatitis is present. Nevertheless, the presence of lesions at the wrist is indicative of an underlying endogenous cause. 105 A 7-year follow-up study in patients with hand dermatitis found that 45% of patients also had concomitant AD lesions on other parts of their body during the follow-up period. 112
Unlike hand dermatitis, foot dermatitis is predominantly caused by allergens in footwear. 113 Foot dermatitis may also be associated with AD. 113 In most cases, the condition is found concomitantly with hand dermatitis. 114 A dorsal pattern similar to that found in hand dermatitis suggests an allergic aetiology. 115 The prevalence of foot dermatitis in patients with AD is approximately 30%. 116
Juvenile palmoplantar dermatitis is a distinct subtype of AD. As the name suggests, it affects the palms and plantar surfaces of the hands and feet, rather than the dorsal surfaces. In affected children, symptoms are most common in winter and tend to resolve between the ages of 12 and 16 years.64,117
Dyshidrosis, also called dyshidrotic eczema or pompholyx, may be a clinical phenotype of AD that manifests as vesicles and blisters on the palms and soles; it accounts for <10% of all cases of hand and foot dermatitis 35 (Figures 2f, 6). This condition is very itchy and disabling. Complications can lead to or include secondary infection.118,119

Most common phenotypes of hand eczema: dyshidrotic and hyperkeratotic.
Hyperkeratotic, dry fissured hand eczema, pulpitis, and nummular hand eczema are other clinical manifestations of hand dermatitis (Figure 6). 120
Nipple dermatitis
AD-related nipple dermatitis presents symmetrically, typically involving both the nipples and areolas (Figure 3g). 64 Nipple dermatitis can be aggravated or triggered by breastfeeding121,122 and is found in 11–23% of individuals with AD, most commonly in postpubertal girls and young adults.4,114 Despite being described as a minor diagnostic feature of the Hanifin–Rajka criteria, 30 the specificity of nipple eczema as a symptom of AD remains unclear.27,29,123,124
AD in the elderly: a new phenotype
Phenotyping and stratification of AD type by both age-related clinical picture and age at onset have been reviewed by Bieber and colleagues. 5 Elderly-type AD (AD in patients aged ⩾60 years) has been suggested to be a distinct age-related phenotype alongside previously recognized infantile, childhood, and adult phenotypes.5–7,125
As in adult-type AD, lichenified lesions are common, but tend to be localized to the cubital and popliteal extensor areas rather than the flexural areas, as is typical of adult-type AD.5,126,127 Three forms of elderly-type AD are apparent – elderly onset, relapsing, and continuous – and are subdivided by age at onset.126–132 A comparison of studies carried out in various geographic locations indicates that the prevalence of AD in those aged >60 years is relatively stable at 2–5%.127,130,133 Immune system changes due to aging (immunosenescence), such as increased type 2 cytokine production [mainly interleukin (IL)-4 and IL-13], may play a key role in the pathogenesis of elderly-onset AD,127,134 as they do in chronic idiopathic pruritus of the elderly. 135 Additional considerations in the elderly include changes in the composition of the cornified envelope with aging that may lead to impaired barrier function, 136 and the effects of medications, including calcium channel blocker-induced dermatitis 137 and statin-exacerbated xerotic/asteatotic dermatitis. 138
Guidelines are lacking on the differential diagnosis of AD versus other skin conditions in the elderly, and diagnosis relies on persistence of symptoms for 6 months after excluding other causes such as comorbidities, treatment-related adverse events, cutaneous T-cell lymphoma, and nonbullous variants of bullous pemphigoid.51,126,127,139
Treatment of atopic dermatitis
Preventive skin hygiene, including barrier-stabilizing topical treatment, hydration, and monitoring and treatment for superinfections are critical in the care of patients with AD. Guidance exists on the treatment of AD in adults and children, 140 and typical treatment strategies for AD include treating visible skin lesions with topical anti-inflammatory medications such as corticosteroids and calcineurin inhibitors in combination with frequently applied emollients to minimize the impact of skin barrier dysfunction.140,141 TCS are an effective first line of anti-inflammatory treatment when applied to skin lesions of patients with AD, and they and are associated with minor side effects.99,140 Steroid-sparing topical agents that are licensed for the treatment of AD include the calcineurin inhibitors tacrolimus ointment and pimecrolimus cream and the phosphodiesterase 4 inhibitor crisaborole; these are preferred for sensitive locations and are used commonly for HND, 142 with narrowband UVB phototherapy as another approved option. Patients with severe AD can also be treated with traditional systemic immunosuppressive medication, such as cyclosporine A or oral glucocorticoids, with off-label use of azathioprine, methotrexate or mycophenolate mofetil sometimes considered. 140 However, use of these systemic immunosuppressants can be limited by their adverse effects and tolerability, particularly for long-term treatment.27,142,143 Novel biologic therapies include the approved therapy dupilumab, which targets the underlying inflammatory mechanism of AD by selectively blocking type 2 inflammation.144–147 Many novel systemic and topical treatments are also under investigation, including the JAK inhibitors baricitinib, upadacitinib, and abrocitinib, the dual JAK-SYK inhibitor ASN002, as well as antagonists of histamine and TSLP, which are each implicated in the pathogenesis of AD. 148 The implementation and success of these agents in treating AD relies, however, on further elucidation of the various phenotypes and appropriate diagnosis of the disease.
Conclusions
Lack of consensus on AD terminology may lead to confusion and result in erroneous data and flawed epidemiologic assumptions. Efforts continue to address this issue, and the increasing use of the consensus term ‘atopic dermatitis’ in the literature and in medical records, may be a sign of progress. Wider use of the term ‘atopic dermatitis’ for diagnosis should help generate more accurate epidemiologic data and improve evaluation of disease burden, outcome measures, and reimbursement decisions, as well as development of new treatments.
AD is a heterogeneous condition that continues to frustrate attempts at classification based on age, morphology, and topographic location. This review considers the clinical characteristics of morphologically (nummular dermatitis, PN-like lesions, erythroderma, pompholyx, papular/follicular and lichenified dermatitis) and topographically defined (flexural, facial, including eczematous cheilitis and eyelid dermatitis, head and neck, hand and feet, or nipple) phenotypes of AD, which we consider to be a practical guide to discerning the distinct manifestations of this heterogenous disease. The subtyping of AD by phenotype may be important to identify the more appropriate treatment for patients, and a protocol has been developed for a systematic review of phenotypes in AD. 149 Given the diversity of AD phenotypes, this reference will facilitate earlier and correct diagnoses of AD, leading to better treatment decisions and management of the disease.
Footnotes
Acknowledgements
Microphotographs in Figure 5 have been generously provided by Dr. Maria Teresa Fernández-Figueras, Department of Pathology, Hospital Universitari General de Catalunya, Universitat Internacional de Catalunya, Sant Cugat del Vallès (Barcelona), Spain. The authors especially thank the patients who provided written agreement for the use of their photographs in scientific publications, and Noah Levit (Regeneron Pharmaceuticals, Inc.) for critical feedback on the manuscript.
Author contributions
GG conceived the manuscript, MdB, ABR, KK, and AB performed literature review references, ABR analyzed data, ABR, GG, MdB, VA, MD, and LP provided clinical photos, MdB and ABR contributed to writing. All authors reviewed and approved all versions of the manuscript.
Conflict of interest statement
Dr. Giampiero Girolomoni has been principal investigator in sponsored clinical trials and received personal fees from AbbVie, Abiogen, Almirall, Amgen, Biogen, Boehringer Ingelheim, Bristol-Myers Squibb, Celgene, Celltrion, LEO Pharma, Lilly, Menlo Therapeutics, Merck, Merck Sharp & Dohme, Novartis, OM Pharma, Pfizer, Regeneron, Samsung, Sandoz, Sanofi Genzyme and UCB Pharma.
Dr. Marjolein De Bruin-Weller has been principal investigator, advisory board member, and consultant for Regeneron, Sanofi Genzyme; principal investigator, and advisory board member for AbbVie, Pfizer, and Leo Pharma; and advisory board member for Lilly, UCB, Galderma, and Janssen.
Dr. Valeria Aoki has received honoraria for lecturing from Sanofi.
Dr. Kenji Kabashima has received consulting fees, honoraria, grant support, and lecturing fees from Japan Tobacco, LEO Pharma, Maruho, Mitsubishi Tanabe, Ono, Procter & Gamble, Sanofi, Taiho, and Torii.
Dr. Mette Deleuran has received research support, honoraria for lecturing and is on consulting/advisory board agreements with AbbVie, Almirall, Galapagos, LEO Pharma, Lilly, Meda Pharma, Pfizer, Pierre Fabre, Regeneron, and Sanofi Genzyme.
Dr. Luis Puig has received research support, honoraria for lecturing, and is on consulting/advisory board agreements with AbbVie, Almirall, Amgen, Baxalta, Biogen, Boehringer Ingelheim, Celgene, Gebro, Janssen, LEO Pharma, Lilly, Merck Serono, Merck Sharp & Dohme, Pfizer, Mylan, Novartis, Roche, Sandoz, Samsung Bioepis, Sanofi, and UCB. Dr Puig is also an Associate Editor of Therapeutic Advances in Chronic Disease and, therefore, the peer review process was managed by alternative members of the Board and the submitting Editor had no involvement in the decision-making process.
Dr. Ashish Bansal is an employee and shareholder of Regeneron Pharmaceuticals, Inc.
Dr. Ana B. Rossi is an employee and may hold stock and/or stock options in Sanofi Genzyme.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Research sponsored by Sanofi and Regeneron Pharmaceuticals, Inc. Editorial assistance was provided by Lauren D. Van Wassenhove, PhD, and Carolyn Ellenberger, PhD, of Excerpta Medica, funded by Sanofi Genzyme and Regeneron Pharmaceuticals, Inc., according to the Good Publication Practice guideline.
