Abstract
Atrial fibrillation (AF) is prevalent in patients with aortic stenosis (AS) undergoing transcatheter aortic valve replacement (TAVR). Depending on the timing of AF detection, it is usually categorized as pre-existing AF or new-onset AF. Antiplatelet therapy, rather than a vitamin K antagonist, may be considered as the primary treatment for patients without an indication for oral anticoagulants who undergo TAVR. However, the optimal postprocedural antithrombotic regimen for patients with AF undergoing TAVR remains unknown. In this review, we briefly introduce the management strategies of antithrombotic therapy and list the evidence from related studies to elucidate the optimal antithrombotic management for patients with AF undergoing TAVR.
Keywords
Introduction
Aortic stenosis (AS) is a common cardiovascular disease in older people with increasing prevalence. 1 Aortic valve calcification is one of the main causes of aortic stenosis. Recent researchers uncovered different mechanisms regarding the osteogenic process of cardiac valves, such as TLR4 stimulation and double-stranded deoxyribonucleic acid (dsDNA) promotion progression.2–4 Elevated homocysteine levels and intestinal microbiota were also shown to be related to cardiac valve calcification.5,6 Transcatheter aortic valve replacement (TAVR) has been considered as the standard care for patients with severe symptomatic AS when surgical aortic valve replacement (SAVR) is deemed prohibitive or high risk.7,8 Recently, several clinical studies have shown that TAVR can be considered for patients with low and moderate risk.9,10
Atrial fibrillation (AF) is the leading arrhythmia in people with heart disease, and there is a growing trend. 11 Many of the risk factors in AF and AS are the same, such as high blood pressure, age, and AS, which can lead to increased pressure of the left ventricle, thereby promoting the incidence of AF. 12 Due to the sharing of multiple risk factors, risks for thromboembolism and bleeding are both increased in AS patients after TAVR. Selecting the appropriate treatment for these patients remains challenging. The optimal antithrombotic regimen for patients with AF undergoing TAVR is currently unknown. In this review, we briefly introduce management strategies for antithrombotic treatment and list the evidence from related studies to elucidate the optimal antithrombotic management for patients with AF undergoing TAVR.
Epidemiology of AF in TAVR patients
Pre-existing AF is frequently detected in patients before TAVR or at the time of eligibility screening for TAVR. Conversely, new-onset AF has been defined as an occurrence after TAVR or SAVR.13–15 AF is present in more than one third of patients undergoing TAVR. Moreover, clinical new-onset AF is up to 36% in patients after TAVR.7,9,16–20 It is noteworthy that, according to a recently study by Kalra et al., the incidence of new-onset AF was significantly higher after SAVR than that after TAVR. 21 More importantly, among TAVR patients, there was a relationship between AF and a high risk of adverse events. Based on the results of Transcatheter Valve Therapy Registry (TVT) and SAPIEN XT Aortic Bioprosthesis Multi-Region Outcome Registry (SOURCE XT) registries, both pre-existing and new-onset AF were related to significantly higher rates of mortality at 1 year.22,23 Similarly, among patients who underwent TAVR, compared with patients without AF, those with new-onset AF have higher risk of stroke, mortality and bleeding rates.24,25
Definition of patients with AF with a high risk of embolism and bleeding
Among AF patients, the CHA2DS2-VASc score (Table 1) has simplified the decision for treatment with an oral anticoagulant (OAC). The 2016 European Society of Cardiology (ESC) guideline recommends estimating stroke, transient ischemic attack, and systemic embolism risk in AF patients based on a CHA2DS2-VASc score of 1 or more for men, and 2 or more for women, and these patients will likely benefit from an OAC.26–28 Bleeding risk assessment should also be part of the clinical decision process. Based on the ESC guideline, several bleeding risk scores have been formulated, such as HAS-BLED, ABC, and ORBIT bleeding scores.26,29–31 The simplest and best validated score is HAS-BLED (Table 1), which reliably predicts the risk of bleeding in patients on an OAC, aspirin, or no antithrombotic treatment. 32
Clinical risk factors for CHA2DS2-VASc ischemic/thrombotic risk score and HAS-BLED bleeding risk score.
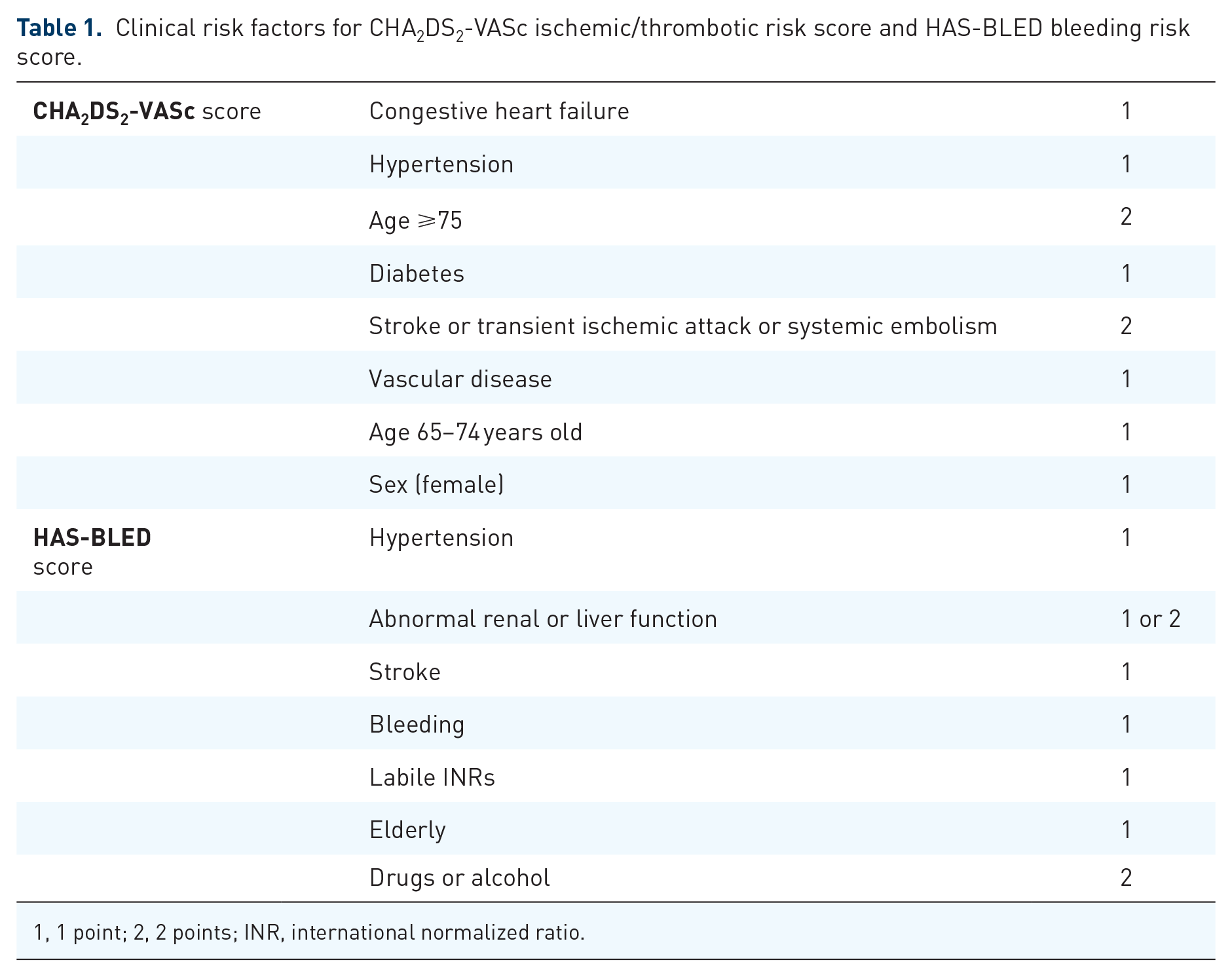
1, 1 point; 2, 2 points; INR, international normalized ratio.
Antithrombotic management after TAVR in patients with AF
In patients with no indication for OACs, current TAVR guidelines are mainly based on experts’ opinion and recommended dual antiplatelet therapy (DAPT) for the first 1–6 months, followed by lifelong low-dose aspirin, although in cases of low bleeding risk, the use of vitamin K antagonists (VKA) may be reasonable.8,33 However, data about antithrombotic strategies for patients with AF undergoing TAVR are rare. Due to the difficulty in balancing the ischemic and bleeding risk, the management of these patients remains challenging. Therefore, choosing the optimal antithrombotic treatment should not only be based on the transcatheter valve used but also on the individual patient’s previous history and risk factors for bleeding and thrombosis. 34
Anticoagulant therapy
Life threatening or major bleeding and stroke are the major clinical events in TAVR patients. 35 Therefore, how to select the appropriate antithrombotic treatment for these patients is crucial. In addition, subclinical leaflet thrombosis occurrence is more common in transcatheter than in surgical valves. Chakravarty et al. found that anticoagulation with OACs, but not antiplatelet therapy (APT), effectively prevents and treats subclinical leaflet thrombosis. 36 More recently, Rashid et al. showed that leaflet thrombosis increased the risk of cerebrovascular events. 37 Therefore, the reduction of stroke with OACs may be due to a reduction in leaflet thrombosis.
Based on the results of the GALILEO (Global Study Comparing a Rivaroxaban-based Antithrombotic Strategy to an Antiplatelet-based Strategy after Transcatheter Aortic Valve Replacement to Optimize Clinical Outcomes) trial, in patients with no indication for OACs after successful TAVR, an OAC (rivaroxaban) was shown to be associated with worse clinical outcomes than an antiplatelet-based strategy. 38 However, in TAVR patients with AF, anticoagulant therapy is designed to reduce the risk of thrombotic events. 39 A previous study sought to evaluate the management patterns and clinical outcomes of patients with new-onset AF who underwent TAVR. Patients were followed for 1 year. They found that patients with new-onset AF discharged without an OAC had a lower risk of systematic bleeding; however, the incidence of mortality or stroke after TAVR was significantly increased. 23 This study indicated that without treatment with an OAC, there may be an increased risk of ischemic/thrombotic events. Furthermore, in patients with AF, VKA is indicated, as it is more effective than single antiplatelet therapy (SAPT) and DAPT regarding primary stroke prevention with an acceptable risk of bleeding.8,33,40–42 However, currently available studies, limited by their underpowered and mainly observational natures, have not elucidated whether OAC monotherapy [that is, a VKA or a direct-acting oral anticoagulant (DOAC)] or APT in addition to an OAC after TAVR is more appropriate. Nicolas et al. found that VKA therapy without additional antiplatelet treatment is effective and safe in AF patients undergoing TAVR by comparing the incidence of bleeding complications, postprocedural strokes, overall thromboembolic events, intracardiac thrombi, transcatheter heart valve thrombosis, all-cause mortality and a combined endpoint in the first 6 months. Furthermore, compared with patients being treated with either single or DAPT in addition to a VKA, the incidence of major bleeding complications was significantly lower in the VKA monotherapy group. 43 Similarly, another earlier multicenter study, comprising 621 patients with AF undergoing TAVR, showed that adding an antiplatelet drug to a VKA has few clinical benefits in terms of bleeding, regardless of the type of bleeding. Patients were followed for 13 months, and there were no significant differences in the incidence of major adverse cardiovascular events, stroke, and death. However, a significantly higher risk of major bleeding was found in the multiple antithrombotic therapy group (24.4% versus 14.9%; 95% confidence interval: 1.05–3.28; p = 0.04), when compared with VKA monotherapy. 44 These two studies suggested that the consistent efficacy of VKA monotherapy, VKA monotherapy treatment may be an optional in clinical practice.
Recently, some research fields are emerging. A DOAC may be considered if OACs are indicated and without contraindications. In a second observational study by Nicolas et al., similar results were shown for VKA and DOAC in patients with concomitant indications for OAC undergoing TAVR. 45 Similarly, 962 patients who underwent TAVR and were discharged on DOACs (n = 326) or VKAs (n = 636) were followed for 1 year, and there was no significant difference in the incidence of bleeding events, but ischemic events with DOACs need further study. 46 This study provided a new idea for TAVR with anticoagulant therapy for AF. More recently, Butt et al. further indicated that treatment with DOACs was safe and effective. They found that treatment with both VKA and DOACs among patients with AF after TAVR is comparable regarding major bleeding, mortality, and arterial thromboembolism. 47 DOACs may play an increasingly important role in clinical practice. Nevertheless, related studies are limited at present. Consequently, large randomized clinical trials related to antithrombotic therapies are needed to confirm this finding.
Antiplatelet therapy and other combinations of antithrombotic therapy
APT rather than an OACs may be considered a primary treatment for patients without AF, or another indication for OACs, undergoing TAVR. However, for patients with AF, the situation is different. In each of these intersecting scenarios, the decision-making process regarding antithrombotic treatment has many challenges. Recently, the PARTNER II registry (n = 1621), in which patients with existing AF and a CHA2DS2-VASc score ⩾2 following TAVR. Kosmidou et al. aimed to explore the patterns of antithrombotic treatments and their impact on long-term outcomes. Their results suggested that when APT was implemented for at least 6 months, the incidence of stroke was significantly reduced when compared with no APT or no OAC. Therefore, OACs alone are not sufficient in the prevention of stroke. 48 However, the study was limited by the retrospective data analysis, single-center observational design, and lack of an external core laboratory adjudicating the events. Furthermore, international normalized ratio (INR) levels were not monitored through the study, and subtherapeutic INR levels may have influenced the results. Indeed, there are no large randomized clinical data supporting their conclusions. In contrast, previous research has shown that the addition of APT to an OAC in TAVR patients with AF increases the risk of bleeding, and it was unlikely to confer any additional clinical benefits after TAVR when compared with patients treated with VKA alone.43,44 Similarly, the propensity-score-matching data from the ITER registry showed that patients treated with VKA plus aspirin reveal a similar risk of valve dysfunction, with higher risk of bleedings at 30 days compared with patients treated with VKA alone (11.4% versus 4.8%, p < 0.001). After 30 days, major bleeding (4.8% versus 2.9%, p = 0.36) was higher but not significant in the VKA-plus-aspirin group. 49 More recently, the PoPular TAVI trial demonstrated that clopidogrel on top of OAC provided no clear benefit with a significant increased risk of bleeding compared with OAC alone at 30 days and 1 year. 50 Therefore, excessive antithrombotic treatment may lead to adverse results. According to Genereux et al.’s research, when a bleeding event occurred in patients with concomitant AF, they observed that 1-year mortality after TAVR increased by 50%. 51 Based on the previously mentioned analysis, the assessment of optimal antithrombotic therapy in these patients should be carefully considered. Given the risk of bleeding, additional APT in these patients should not be treated routinely.
Coronary artery disease (CAD) exists in approximately 70% of patients undergoing TAVR. Previous reports suggested the use of APT is more than an OAC because of the multiple comorbidities in these patients.9,16–18 In patients with AF and stable CAD, previous research has shown that adding an antiplatelet agent to an OAC brings few clinical benefits. Adding APT did not reduce the risk of coronary artery events or stroke; on the contrary, it significantly increased the risk of major bleeding. 52 Thus, for patients with AF and stable CAD who underwent TAVR, VKA monotherapy may seem to be an appropriate choice. However, according to clinical data, approximately one half of TAVR patients with CAD exhibited multivessel disease. 53 Considering the high risk of coronary artery events, we often choose an APT in combination with an OAC for treatment, although this strategy is empirical and may increase the risk of bleeding.54,55 According to the recommendations of 2017 ESC, which related to patients with AF undergoing percutaneous coronary intervention, 56 triple antithrombotic therapy may be considered in patients with AF who undergo TAVR and have strong indications for DAPT, such as recent acute coronary syndrome (ACS) and extensive or recent coronary stenting, but it should be kept to the shortest possible duration, with dual antithrombotic therapy as an alternative. 57
Left atrial appendage closure (LAAC) is mainly applicable to patients with AF who have a high risk of bleeding or a contraindication to OACs. It has been proven safe and effective.58,59 Similarly, a pilot study showed that concomitant TAVR and LAAC were feasible and safe. 60 Nevertheless, evidence is scant in the TAVR field and no strongly reliable recommendations can be made so far. LAAC might be a potential alternative to OACs, especially in patients with a high risk of bleeding.
Future
According to current research, anticoagulant therapies have high reliability in patients with AF undergoing TAVR. Traditional anticoagulants, including VKA, are proven to be safe and effective. No new oral anticoagulants have been found to have adverse effects in patients adopting the previously mentioned therapy. Furthermore, DOACs, such as apixaban, have been exploited recently and may be more desirable than VKA, 61 whereas no abundant large randomized clinical research exists currently. There are still uncertainties in both the clinical implementation and of the duration of triple therapy, double therapy, or use of DOACs (including the use of the various types of medicine) for patients with AF. The vast majority of studies concentrated on comparisons between empirical therapeutic protocols, most of which are based on local conventions and patient characteristics. Future, rationally designed research should put more focus on determining and clarifying a universal antithrombotic strategy in this population. Several randomized clinical trials are currently ongoing in an attempt to answer the question of the best antithrombotic regime: (a) AVATAR (Aortic Valve Replacement versus Conservative Treatment in Asymptomatic Severe Aortic Stenosis) [Clinical Trials.gov identifier: NCT02436655] where researchers compare clinical outcomes of selective aortic valve replacement with conventional treatment and a watchful-waiting strategy; (b) ENVISAGE-TAVI AF (Edoxaban Compared with Standard Care After Heart Valve Replacement Using a Catheter in Patients With Atrial Fibrillation) [Clinical Trials.gov identifier: NCT02943785] where a DOAC (edoxaban) is compared with VKA; (c) ATLANTIS (Anti-Thrombotic Strategy after Trans-Aortic Valve Implantation for Aortic Stenosis) [Clinical Trials.gov identifier: NCT02664649] where a DOAC (apixaban) is compared with VKA. Though, the ATLANTIS trial was not designed specifically for patients undergoing TAVR with AF, its result may add further evidence on the role of DOAC following TAVR.
Conclusion
In the absence of large randomized clinical trials, we rely on evidence reported in populations other than the TAVR population or on local conventions and patient characteristics. Awaiting the results of ongoing trials, VKA monotherapy should be considered in TAVR patients with AF. Adding SAPT on top of VKA seems more appropriate in patients with recent ACS or recent coronary stenting who have a high ischemia or thrombosis risk. A DOAC may be considered if an OAC is indicated in the absence of contraindications. LAAC might be a potential alternative to OACs, especially in patients with a high risk of bleeding. A proposed individualized treatment scheme based on the evidence described previously is shown in Figure 1.

Potential antithrombotic therapy options in patients with AF undergoing TAVR.
Footnotes
Conflict of interest statement
The authors declare that there is no conflict of interest.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: this project was partly supported by the National Natural Science Foundation of China (grant number 81770386); the Science and Technology Program of Guangzhou (grant number 201804010086); the Youth Science and Technology Innovation Talent of Guangdong TeZhi Plan (grant number 2019TQ05Y136), and Frontier Research Program of Guangzhou Regenerative Medicine and Health Guangdong Laboratory (grant number 2018GZR110105001).
