Abstract
Background:
Explicit criteria for potentially inappropriate medications (PIMs) developed for other countries are difficult to apply to a specific territory. This study aimed to update the PIM-Taiwan criteria from a qualitative review of several published PIM criteria, followed by consensus among regional experts in Taiwan.
Methods:
After a review of the literature, we selected four sets of published PIM criteria to construct preliminary core PIMs. The Beers criteria, Fit fOR The Aged (FORTA), and Japan criteria were used for PIMs, without consideration of chronic diseases. The Beers criteria, Screening Tool of Older Persons’ Prescriptions (STOPP) criteria, and Japan criteria were used for PIMs with respect to chronic diseases. We asked experts (n = 24) to rate their agreement with each statement, including in the final PIM criteria, after two rounds of modified Delphi methods. The intraclass coefficient (ICC) was used to examine the reliability of the modified Delphi method.
Results:
Overall, two categories of PIMs were established: 131 individual drugs and 9 drugs with combinations that should generally be avoided; and 9 chronic diseases with their corresponding PIMs that have drug–disease interactions. The ICC estimates for PIMs to be avoided generally were 0.634 and 0.557 (round 1 and 2) and those for PIMs with respect to chronic diseases were 0.866 and 0.775 (round 1 and 2) of the Delphi method, respectively.
Conclusions:
The 2018 version of PIM-Taiwan criteria was established and several modifications were made to keep the criteria updated and relevant. Clinicians can use them to reduce polypharmacy and PIMs among older patients.
Introduction
National Health Insurance in Taiwan is well known worldwide and has a high coverage rate. 1 Therefore, the average years of survival among Taiwanese individuals is increasing under this affordable and well-developed health care system. When people live longer, they frequently have a higher chance of having chronic diseases. In current clinical practice, under the assumption of one guideline that is applied to all adults, 2 multiple medications are more likely to be prescribed for multimorbid patients, because each guideline might recommend an average of three medications.3,4 As the number of medications increases, the incidence of adverse drug reactions (ADRs) and drug–drug and drug–disease interactions increases significantly. 5 ADRs are associated with falls, geriatric syndrome, higher rates of hospitalization, and mortality.6,7 In previous studies, some ADRs were regarded as preventable when medications with high risks of ADRs can be avoided before they are prescribed.
Drugs with a risk of ADRs outweighing clinical benefits, uncertain therapeutic effects, or with safer alternatives for older people are defined as ‘‘potentially inappropriate medications’ (PIMs). 8 Under this concept, explicit criteria are established to discourage the use of PIMs in older people. The first established PIM criteria was the Beers criteria in the United States in 1991. 8 The initial arrangement of this list was not a system-oriented arrangement, and the PIMs were selected from locally available drugs and regarded as inappropriate according to experts opinions. However, it has been updated 9 and applied to clinical practice and many clinical studies to find the associations between PIMs and outcomes over the past two decades. 10 However, the prescription preference of physicians and the drug market varies in different regions of the world. Therefore, regional PIM criteria are preferred, and they have also been developed in many countries including Germany, 11 France, 12 Ireland, 13 Norway, 14 Italy, 15 Thailand, 16 Japan, 17 and Canada. 18
Establishing a new set of criteria is time-consuming, particularly during the literature review process, and relatively few studies have enrolled older people with multiple comorbidities in clinical trials. Since the publication of the Beers criteria in 1991, most regional PIM criteria have been derived from expert’s opinions using the modified Delphi method. 19 Based on regionally available drugs, the consensus among regional experts was obtained using the modified Delphi method. The PIM-Taiwan criteria have been established and have proven their applicability in several cross-sectional studies among older Taiwanese adults.20–22 In comparison with the Beers criteria and PRISCUS criteria, PIM-Taiwan can detect a similar number of PIMs across different populations in Taiwan. PIM users had higher health resource utilization and higher costs of medications20,21 than non-PIM users.
As technology advanced and new results from clinical studies emerged, many new medications were developed after 2010, and some of the statements in the PIM criteria were considered irrelevant or inaccurate. In addition, some older drugs are not available in the market. Therefore, the aim of this study was to establish a new version of the PIM-Taiwan criteria using a two-round modified Delphi method, and intraclass correlations were used to investigate the correlation and agreement among experts’ opinions.
Methods
Establishment of a preliminary list
The initial literature review was conducted on PubMed from studies published from 1 January 2011 to 1 January 2017. We selected this time limit for literature inclusion because our PIM-Taiwan criteria were published in 2012. Because we could not include some sets of criteria that were published 1 year before the PIM-Taiwan publication year (2012), we searched for studies dated after 2011. The search included the terms (potentially inappropriate medication list [MeSH] OR inappropriate prescribing [All Fields] OR inappropriate prescribing/classification [All Fields] OR inappropriate prescription [All Fields]). The MeSH term ‘potentially inappropriate medications’ was introduced in 2016. Prior to this time, the term ‘inappropriate prescribing’ was used.
As an initial step for the development of PIM criteria in Taiwan, we identified nine sets9,11,13,15,17,23–26 of explicit criteria published in the English language. After criteria were omitted that were developed based on other published criteria, we selected four sets of criteria to develop a preliminary list (Figure 1). A research team was convened, including three geriatricians, one psychiatrist, and two clinical pharmacists, to create a list of preliminary PIMs from the four published criteria. For the preliminary list of PIMs that should be generally avoided, the Beers criteria, Japan criteria, and FORTA criteria were used as references. For PIMs with respect to comorbidity, the Beers criteria, Japan criteria, and Screening Tool of Older Persons’ Prescriptions (STOPP) criteria were used as references. Medications/medication classes that appeared in at least two out of three sets of the criteria were selected and reviewed by the research team. The research team reviewed the concerns regarding these PIMs and approved a preliminary list that consisted of 169 individual drugs and 9 drugs with combinations to be generally avoided and 9 statements for drug–disease interactions.

Flow diagram of the systemic literature review and selection of explicit criteria.
The availability of medications in Taiwan listed in the preliminary PIM list was confirmed using the medication database from the National Health Insurance Administration. We identified generic names and the Anatomical Therapeutic Chemical (ATC) Classification System code for each medication. When a set of medication classes, rather than an individual medication, was considered potentially inappropriate, we identified all available medications in Taiwan belonging to this set of medication classes. For anticholinergic drugs, we reviewed the literature to define the strong anticholinergic drugs that were given a score of at least two on the ‘Anticholinergic Risk Scale’.9,27
Modified Delphi method
A group of 24 experts from different specialties (geriatricians, neurologists, psychiatrists, cardiologists, pulmonologists, gastroenterologists, urologists, and clinical pharmacists) was invited to develop a consensus using the modified Delphi methods from the preliminary PIM list. Briefly, this method is a form of communication among these experts with a structured questionnaire to reach a consensus decision. All nongeriatric experts were selected because they are experienced and skilled in the principles of prescribing for older people and have reputations in their specialties in Taiwan. Hard copies of the questionnaire were mailed to all of the experts. A brief introduction of the modified Delphi method and scoring methodology was also given. Then, the experts were asked to rank each statement according to its degree of inappropriateness using a 5 point Likert scale that ranged from 5 points (strongly agree) to 1 point (strongly disagree). In addition, the experts could add suggestions for PIMs that were not listed originally or remove PIMs in the preliminary list. The first round of the questionnaires was scored by each expert by October 2017. PIMs with a mean Likert scale ⩾3.5 were scored again by November 2017. During round two, we also gave the mean score for each statement, and all experts could find the opinion of other experts in round one. We also welcomed the suggestions of new statements or alternative therapies from the experts. The PIM list was finalized from medications/medication classes with mean Likert scale scores ⩾3.5 after two rounds of rating. We welcomed the suggestions of new statements or alternative therapies from the experts, and the alternative therapies or suggestions for each PIM to be generally avoided were finalized by four clinical pharmacists. Finally, the research team confirmed that all PIMs were available in Taiwan, and that all of the concerns and alternative therapies/suggestions were optimized.
Theory and calculation
The resulting data generated by the hard copies of the survey were imported into Microsoft Excel (Microsoft Office 2013) for analysis. Descriptive statistics were used to measure the consistency of each measurement. The intraclass correlation coefficient (ICC) estimates and their 95% confidence intervals were calculated using Stata statistical package version 13 (Stata, College Station, TX, USA) based on a mean-rating (k = 24), consistency, and a two-way mixed-effect model.
According to the ‘Human Subjects Research Act’ in Taiwan, this study did not involve obtaining, investigating, analyzing, or using human specimens or an individual person’s biological behavior, physiological, psychological, genetic, or medical information. Therefore, this study was not human subject research. In the legislation of the Research Ethics Committee of National Taiwan University Hospital, the study did not need ethical approval as it was not human subject research. The objectives of the Delphi method were presented to all experts, their agreement and availability to participate were obtained (consisted of replying positively by email to the invitation sent by the research team). The information that this study generated was used for consensual quality criteria only and can be publicly accessible and there is no reasonable expectation of privacy.
Results
The preliminary list consisted of 169 individual drugs and 9 drugs with combinations, for example, ‘promethazine, combinations’ (ATC code R05FA02) to be avoided generally and 9 statements for drug–disease interaction. A total of 29 individual drugs were not available in 2018. PIMs in the first round (n = 9) with mean Likert scale scores <3.5 were not entered into the second round. The response rate of the hard copies of the questionnaire was 100% in two rounds of the Delphi method. After two rounds of the modified Delphi method, the final PIM-Taiwan criteria included 131 individual drugs, 9 drugs with combinations to be avoided generally (Table 1), and 9 statements for drug–disease interaction. When an entire medication class was considered to be PIMs, all individual drugs available in the class are listed in Table 1. We designated the symbol marker† for drugs that were already listed in the 2012 PIM-Taiwan criteria. Alternative therapies or suggestions for PIMs were created to prevent the general use of PIMs. When there was no optimal alternative medication for certain PIMs, we recommended other nonpharmacological therapies. For example, antipsychotics for behavioral problems of dementia or delirium should be avoided among older adults because the mortality rate is higher for antipsychotic users. Therefore, nonpharmacological options (e.g. behavioral interventions, such as psychosocial interventions, reality orientation, and physical activity) are suggested first for the behavioral problems of dementia or delirium. 28 Otherwise, PIMs should be used only for a short duration for those with appropriate indications. Table 2 summarizes drug–disease interactions, which included individual medications or medication classes that should be avoided in patients with corresponding chronic diseases. The entire medication class should be considered inappropriate if patients have a corresponding chronic disease. Table 3 lists 131 individual drugs in 10 medication classes overall. The ATC codes, medication/medication classes, and reasons for inclusion are provided. The statements and drugs have been removed since the 2012 version of PIM-Taiwan were enumerated (Table 4). The entire medication class of muscle relaxants was removed from PIM-Taiwan.
2018 version for potentially inappropriate medication use in persons aged ⩾65 years of age (PIM-Taiwan) independent of diagnoses or chronic diseases.

Chlordiazepoxide is classified as a benzodiazepine, and it is available in Taiwan by combining clidinium, anticholinergics or antacid as a single pill for gastrospasm or gastritis.
Hydroxyzine is classified as a first-generation antihistamine, but it can be used for sedation and anxiolysis.
Identical medications listed in the 2012 version of PIM-Taiwan criteria.
NSAID, nonsteroidal anti-inflammatory drug; PIM, potentially inappropriate medication; SSRI, selective serotonin reuptake inhibitor; TCA, tricyclic antidepressant.
2018 version for potentially inappropriate medication use in persons aged ⩾65 years of age (PIM-Taiwan): drug–condition interactions that may cause the exacerbation of chronic diseases.

CNS, central nervous system; NSAID, nonsteroidal anti-inflammatory drug; PIM, potentially inappropriate medication.
Individual medication list for medication classes in Table 2 for drug–condition interactions that may cause the exacerbation of chronic diseases.

NSAID, nonsteroidal anti-inflammatory drug.
Medications or criteria removed since the 2012 PIM-Taiwan criteria.

PIM, potentially inappropriate medication, 2012 PIM-Taiwan criteria. 29
For drug–disease interactions, the entire criterion was removed, including blood clotting disorders or anticoagulant therapy, chronic constipation, glaucoma, sleep apnea syndrome, and urinary incontinence. Statements were removed for benzodiazepines for patients with chronic obstructive pulmonary disease and benzodiazepines for patients with cognitive impairment or dementia.
ICC estimates for PIMs to be avoided generally were 0.634 (95% confidence interval 0.540–0.716, p < 0.001) and 0.557 (95% confidence interval 0.444–0.657, p < 0.001) in rounds one and two of the Delphi method, respectively (Table 5). The ICC estimates for PIMs considering chronic disease were 0.866 (95% confidence interval 0.760–0.939, p < 0.001) and 0.775 (95% confidence interval 0.599–0.898, p < 0.001) in rounds one and two of the Delphi method, respectively. This result indicated that there was moderate or good reliability in experts’ measurements for each PIM statement.
Summary of interrater reliability statistics for medications considered to be potentially inappropriate.
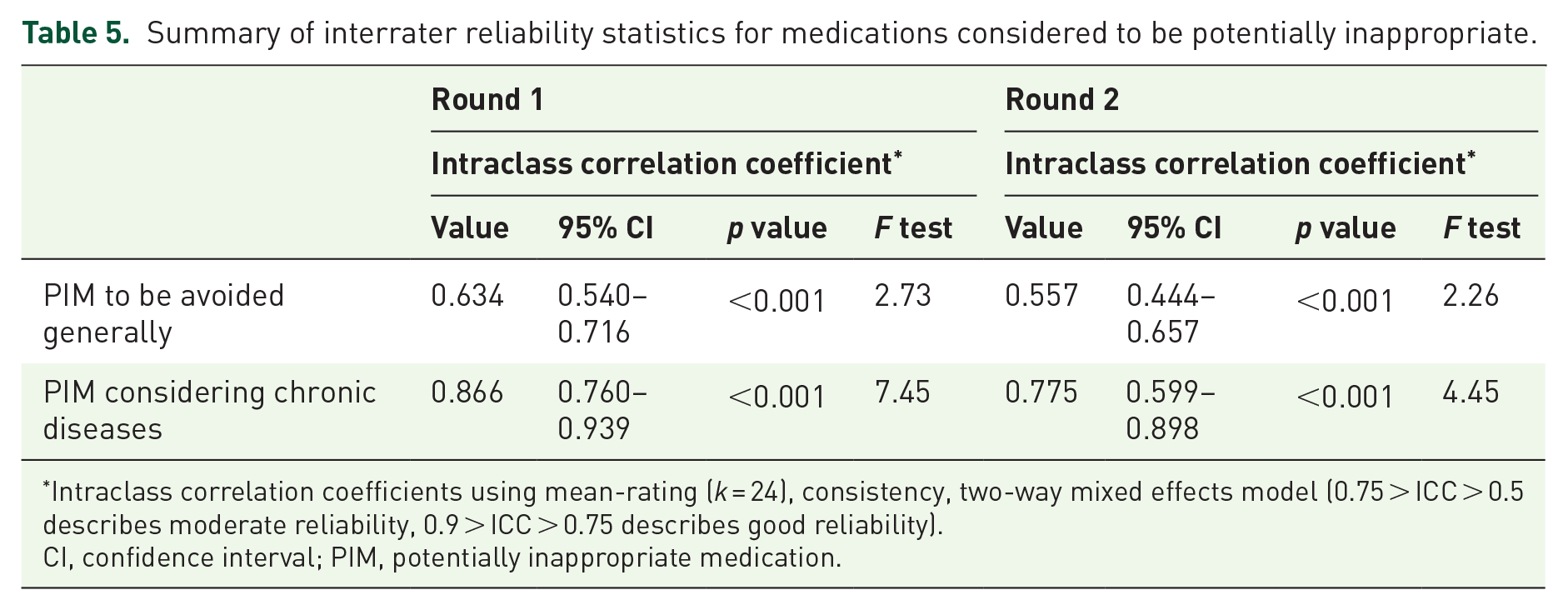
Intraclass correlation coefficients using mean-rating (k = 24), consistency, two-way mixed effects model (0.75 > ICC > 0.5 describes moderate reliability, 0.9 > ICC > 0.75 describes good reliability).
CI, confidence interval; PIM, potentially inappropriate medication.
Discussion
Explicit criteria with a listing of potentially inappropriate criteria have been established since 1991. These criteria have been applied to discourage the use of high-risk drugs among older people and have been used as indicators for prescription quality. The first version of the PIM-Taiwan criteria was published in 2012. Our methodology has been used as a reference to develop regional PIM lists because the literature review processes were dynamic and complex. After the PIM-Taiwan criteria were published, more than half of the regional PIM criteria were established using prior PIM lists.
Without adding new areas to our PIM criteria, the 2018 version of the PIM criteria only has two categories of PIMs, including PIMs that are generally avoided (Table 1) and those considered potentially inappropriate in certain chronic diseases (Table 2). After the PIM-Taiwan criteria were updated, many medication classes and drugs that are newly available in Taiwan have been added to the 2018 version. In contrast, some medications and drug–disease interaction statements were removed (Table 4). The user-friendly characteristics of the statements were retained, including alternative therapies or suggestions for PIMs in Table 1, and the concern and the ATC code for each PIM.
The experts who participated in the establishment of the most frequently published PIM criteria were geriatricians, psychiatrists, and clinical pharmacists. In this study, we invited many experts from other specialties including gastroenterology, cardiology, pulmonary medicine, neurology, and urology, to reflect the multidisciplinary perspectives. With a multidisciplinary approach, the rating of some PIMs in the preliminary list can be more heterogeneous, particularly for medications commonly prescribed in certain specialties. In Table 1, the quetiapine and insulin sliding scale was not considered to be potentially inappropriate. Quetiapine has a less negative influence on mortality than do other antipsychotics. In addition, the majority of its adverse drug events were preventable. Although we did not regard it as a PIM in the PIM criteria, physicians still need to monitor its adverse events and avoid long-term use. For the sole use of sliding scale insulin, we could not conclude from current systemic reviews that using other strategies can reduce hyperglycemia and prevent hypoglycemia. Therefore, we did not include this statement as PIM. In Table 2, all experts agreed that these medication classes were potentially inappropriate in their corresponding chronic comorbidities.
After we published our strategy to establish a new country- or region-specific PIM criteria, 29 several sets of criteria were established using a similar methodology. If we use all the newly published sets of criteria to establish our preliminary lists, we find that some of the PIMs are duplicated, because they may be derived from other criteria, such as the Beers criteria. Therefore, we only selected those established based on the results of a literature review and a subsequently modified Delphi method. As a result of this strategy, in Table 1, two classes of medications (barbiturates and muscle relaxants) that were regarded as PIMs in the first version of our criteria were removed. In Table 2, five chronic diseases (blood clotting disorders or anticoagulant therapy, chronic constipation, glaucoma, sleep apnea syndrome, and urinary incontinence) were removed, because these diseases were not restricted to older adults. These disease–medication interactions should be considered among the general population without regard to age.
Polypharmacy has been shown to be associated with adverse outcomes, high healthcare costs, and poor quality of life. The avoidance of potentially inappropriate medication is an important strategy to prevent adverse drug events and to deprescribe for older adults. In the systemic review, deprescribing seems to be an effective strategy to reduce mortality in nonrandomized studies. However, when studies aim to investigate the effect of implicit and explicit tools on the outcome of reducing PIMs, the benefit of interventions was not clear. When using implicit criteria, pharmacists or other clinicians need more knowledge and clinical experiences to identify target medications for deprescribing. In addition, clinicians may identify different targets for deprescribing without a clear reference. In contrast, the use of explicit criteria is straightforward and simple. Therefore, PIM criteria can be an important tool to reduce PIM-related adverse events. After our PIM-Taiwan criteria are updated, more PIMs could be identified. 22 However, the number of individual drugs and statements increased when PIM lists were updated, and it would be a barrier for clinicians to apply these tools efficiently in clinical practice. With the assistance of e-prescribing or computerized physician order entry systems, the positive effect of reduced PIMs has been demonstrated in several studies. 30
The first disadvantage of this tool was that PIMs were derived from existing criteria, and new findings from recent clinical trials were not fully reviewed. Our criteria only listed drugs to be avoided and not those that should be initiated among older adults. Second, for ease of application in clinical practice, we did not use the complex classification system adopted by the 2015 version of the Beers criteria including PIMs to be used with caution, drug–drug interactions, and dose consideration with varying levels of kidney function in older adults. Third, all medications listed in these criteria were available in the medication database from the National Health Insurance Administration in Taiwan. Therefore, the criteria should be modified before applying them in other countries.
Conclusion
In conclusion, a 2018 version of the PIM-Taiwan criteria was developed through a systemic method that could be replicated by other groups. Several important modifications were made to maintain the relevance and usefulness of the criteria. Its user-friendly characteristics will help clinicians to reduce polypharmacy and PIMs among older patients.
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the National Taiwan University Hospital Research Program (106-S3483).
Conflict of interest statement
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
