Abstract
Background:
Early intervention for acute severe ulcerative colitis (ASUC) improves outcomes. Outcomes and healthcare costs for an infliximab-first and colectomy-first approach were compared.
Methods:
This single-center retrospective cohort study of inpatients with steroid-refractory ASUC who received infliximab 5 mg/kg (1–3 doses without maintenance) or initial colectomy between 2004 and 2014 assessed long-term healthcare utilization and direct costs following infliximab or colectomy, using admission coding data until 31 December 2016.
Results:
A total of 118 patients received either infliximab (n = 85, 72%) or colectomy (n = 33, 28%) as initial therapy, with 35(41%) patients eventually requiring colectomy post-infliximab (median 213 days, range [6, 3739]). Median follow up was 7 years [0, 14]. Following infliximab for ASUC, 44% of patients then received antitumor necrosis factor maintenance. After ASUC therapy, length of stay and number of admissions did not significantly differ between groups but higher numbers of complications prompting readmission occurred in the colectomy group (median 4 versus 1, p < 0.001). There were no differences in admissions or total length of stay for patients who had received infliximab first then colectomy versus those treated with colectomy first (median 7.0 versus 4.0, 41.5 days versus 29 days, respectively, each p > 0.05). Total costs were lower at 6 months (mean AUD17,662 versus AUD24,852, p = 0.003), yet were similar at 7 years following an infliximab compared with colectomy approach (AUD72,834 versus AUD59,557, p = 0.23). After infliximab, costs were significantly higher at 7 years with biologic rather than immunomodulator-only maintenance therapy (AUD109,365 versus AUD47,842, p < 0.01).
Conclusions:
In support of current practice, infliximab salvage in steroid-refractory ASUC achieved reduced short-term healthcare costs compared with initial colectomy, though long-term costs were not significantly different.
Keywords
Introduction
Acute severe ulcerative colitis (ASUC), occurs in up to 25% of patients with ulcerative colitis (UC) and requires hospitalization and colectomy in 30% of cases.1,2 Medical salvage therapy with either infliximab or cyclosporine is now standard first-line therapy for patients not responding to intravenous (IV) corticosteroids, 3 but this strategy is expensive, and studies have suggested that infliximab salvage therapy and related medication costs are higher than for early colectomy.4,5 Even the evolving strategy of accelerated infliximab induction has not been found to reduce long-term colectomy rates but is likely to have greater costs.6,7 Conversely, delaying colectomy has been associated with worse outcomes,8,9 thus conceivably the universal use of infliximab, both initially and with ongoing persistence, could result in unnecessary delays in colectomy. Of course there are also risks associated with colectomy, including a 1–3% perioperative mortality rate, plus the risk of future long-term complications and morbidity; for instance with an ileo pouch-anal anastomosis (IPAA) this includes pouchitis and pouch failure, and stomal complications with an ileostomy.10–12
Since the advent of antitumor necrosis factor (anti-TNF) therapy, medications rather than hospitalizations and surgery have become the principal driver of costs in managing UC. 13 Economic modeling studies have yielded mixed conclusions regarding the use of early colectomy and salvage infliximab for ASUC, but uniformly suggest that long-term medical therapy is costly.5,14 Given the variable course of UC, data from real-world cohorts are valuable to assess further the overall health economic impact of infliximab salvage therapy versus early colectomy on long-term costs following ASUC. This study aimed to address this knowledge gap, with the hypothesis that initial infliximab for steroid-refractory ASUC would, in the long term, result in similar healthcare utilization and costs to an early colectomy approach as medication costs would be offset by the costs of greater inpatient healthcare utilization for follow-up surgeries and related surgical complications with early surgery. Comparison of those patients who received infliximab first and then ultimately required colectomy with those who received colectomy first was also of interest to assess for any potential differences in subsequent complications and healthcare costs.
Methods
Setting/recruitment
Given that early colectomy is essentially no longer used as first-line therapy in ASUC, a retrospective analysis comparing healthcare utilization and costs between early colectomy and infliximab for the management of ASUC between 2004 and 2014 was performed. Up until 2014, there was no local government reimbursement for infliximab use in UC, so infliximab and colectomy were each still employed as first-line treatments in ASUC, and so were used to compare the costs of infliximab and early colectomy. Given recent data suggesting that delaying colectomy even for 5 days in ASUC is associated with worse outcomes, 15 the study only included patients treated with first-line colectomy as representative of the early colectomy group, while individuals who received infliximab and had a colectomy at any subsequent time point were included in the infliximab group.
At Eastern Health, a tertiary hospital network in metropolitan Melbourne, Australia comprising over 1000 inpatient beds, multiple data sources including hospital admissions and diagnostic coding, pathology, pharmacy, and inflammatory bowel disease clinic databases were assessed retrospectively and cross-checked in this retrospective, observational study. All cases of ASUC between 1 January 2004 and 1 January 2014, meeting Truelove–Witts criteria 16 on admission and who, having failed at least 48 h of IV corticosteroids (at a dose of 300–400 mg IV hydrocortisone daily or equivalent), were given either one or more infliximab 5 mg/kg IV infusions and/or colectomy as their first-line therapy, were retrieved. Patients were given an infusion of infliximab followed by subsequent infusions at 2 and 6 weeks if deemed necessary. The choice of initial therapy and number of infliximab infusions given was at the discretion of the treating team at the time of presentation.
Follow-up data collection
Long-term follow up was then conducted from the date of initial infliximab or colectomy (i.e. within index admission) for ASUC through to 31 December 2016. Data captured in this follow-up period for each patient included inpatient healthcare utilization (in terms of the total number of inpatient admissions and cumulative total length of stay in days, outpatient visits, and procedure, inpatient, outpatient, and medication costs), and after first-line infliximab, the date of subsequent colectomy, date of death, and/or date of loss to follow up, if these latter variables eventually occurred.
Other data collected included demographics, disease characteristics (Montreal classification, 17 disease duration, Mayo endoscopy score 18 at ASUC presentation), medication use before and after ASUC presentation, serum C-reactive protein (mg/L), albumin (g/L), and abdominal imaging at ASUC presentation. The Oxford index, 19 Sweden index, 20 and Edinburgh predictive index 21 at day 3 of admission or on day of infliximab or colectomy therapy (whichever occurred earlier) were calculated in each patient. Data were only collected and analyzed for our hospital network, so admissions to other hospitals outside the hospital network were not included.
Post-infliximab and/or colectomy complication data
Complications that developed following ASUC therapy with either early infliximab or colectomy were captured through analysis of the International Classification of Diseases (ICD-10) coding diagnoses recorded for all presentations to Eastern Health network hospitals. If multiple complications occurred concurrently during the same presentation, each complication was counted for that admission. Complications were then grouped into four broad categories, including surgical complications, hemorrhagic complications, infective complications, and thrombotic complications and these were compared cumulatively between the two groups. Complications that were assessed are listed in Appendix 1.
Costing data
Finally, for each individual patient, the total costs of healthcare were estimated from the perspective of a third-party payer. The estimate was based on reimbursement data for inpatient admissions, medication costs, procedure costs, and the costs of outpatient visits and was calculated from the date of initial ASUC therapy with colectomy or infliximab until the end of the follow-up period. A data analyst from the study hospital network was consulted to determine the method of calculating the hospital costs from a given hospital admission. Each admission was classified based on the Australian Refined Diagnosis Related Groups (version 6.0x) (based on ICD-10-AM classifications), which assigns each admission with a value based on the average cost of the admission, accounting for patient demographics, admission diagnosis, comorbidities or complications, duration of stay, and any significant procedures performed. 22 From these data, a cost-weighted separation (or Weighted Inlier Equivalent Separation [WIES]) was determined, which equated to the amount of funding the hospital received from the government for each admission. The dollar amount of the WIES changes each financial year, so each admission cost was calculated according to the WIES value for 2015/16 to allow costs to be standardized across the follow-up period. 23 For maintenance medications a combination of pharmacy data and outpatient records were utilized to determine long-term medications used for inflammatory bowel disease and the doses and duration of therapy. The cost of each medicine was based on data from the Australian Pharmaceutical Benefits Scheme, available online as of 30 June 2017, and the calculated cost was based on the dose and duration of therapy according to outpatient records. 24 For infliximab, the cost per vial (AUD604.86) was multiplied by the number of vials given per dose then multiplied by the number of doses given to each respective patient. Associated costs for the delivery of infliximab infusions were calculated using averaged WIES data for 2016 and were AUD500.52 per infusion for all patients at the study center and these were added to the cost for patients who received further infliximab. For a patient on maintenance anti-TNF therapy, the daily cost of infliximab or adalimumab was calculated, and this was multiplied by the number of days spent on the medication to determine the total cost. This was then cross-checked with the pharmacy database, with the higher of the costs being used for the study. The number of outpatient visits was determined using hospital records and the cost of each consultation was based on the Medicare Benefits Schedule 2016 (Table 1). 25 Finally, procedure costs were based on coding data and the reimbursement was based on the same Medicare Benefit Schedule, including endoscopic procedures. Surgical procedure costs were included for patients based on coding data. If patients received a staged procedure, then the reimbursement data for each admission and procedure was included. Given the variable length of follow up for each patient, cost data were assessed at different time points across follow up (i.e. 30, 60, 90, and 180 days, 1, 2, 3, 5, and 7 years).
Cost data for items following initial admission with acute severe ulcerative colitis.

MBS, Medicare Benefits Schedule; PBS, Pharmaceutical Benefits Scheme.
Statistical analysis
Statistical analysis was performed using SPSS version 22 (IBM Corp, NY, USA). Normality of data was assessed and for continuous variables relevant to the study aims, according to Shapiro–Wilk test ⩾ 0.05, nonnormality was assumed. Hence nonparametric statistics were used to evaluate data with medians presented and Mann–Whitney tests were used for comparisons. For economic data, two-tailed t tests for comparisons of means were performed given the interest in the total expenditure rather than distribution of results. 26 Nonparametric bootstrapping was performed using a simple sampling method with 1000 samples and a 95% confidence interval to confirm the results of the t test. Proportions were compared using Fisher’s exact tests. A p value < 0.05 was deemed significant throughout the study.
Patients with incomplete data or who were lost to follow up were retained in the analysis based on available data. We performed multiple imputation using monotone predictive mean matching via SAS software (version 9.4, SAS Institute, Cary, NC, USA) for missing cost data at 5 and 7 years (including cost-discounting scenarios) for any reason other than death and compared the results to the cases with available data. Values were assumed to be missing at random. The imputation model included age, gender, disease duration, and total cost values for all time points. There were 25 imputed datasets that included within-imputation and between-imputation estimates. There were no meaningful differences between the results with and without imputation hence we present the available case data.
Ethics approval
The study was approved by the Eastern Health Human Research Ethics Committee (LR44/1011) and was therefore performed in accordance with the ethical standards of the 1975 Declaration of Helsinki and its later amendments.
Results
Characteristics of overall cohort
There were 118 patients who all had presentations for ASUC, each meeting Truelove–Witts 16 criteria on admission and having failed IV steroids within the data-capture period: all had at least one further encounter for which data could be captured and included in the analysis. Of these, 85/118 (72%) received one or more doses of infliximab (5 mg/kg) as initial salvage therapy while 33 (28%) had early colectomy as the initial therapy for ASUC. Median follow up after initial presentation with ASUC was 7 years (range 0, 14 years). A total of 90/118 (76%) had 5 years follow up (72% versus 88% for infliximab and early colectomy, respectively, p = 0.05) and 57% had at least 7 years follow up (51% versus 73%, respectively, p = 0.03). There was no significant difference in median Sweden index score (10.95 versus 8.14, p = 0.19), Edinburgh predictive index (4 versus 2, p = 0.16), or the proportion meeting Oxford index criteria (50% versus 56%, p = 0.85) at day 3 of admission for patients treated with infliximab compared with early colectomy, respectively. Of the 85 patients who received early infliximab, 35 (41%) required a subsequent colectomy during follow up, with the majority (30/35, 86%) occurring within 12 months of admission with ASUC. For patients treated with infliximab, 38 (45%) received one dose, 26 (31%) received two doses, and 20 (24%) received three doses. Further demographic data are presented in Table 2.
Baseline demographic features.

There was a total of five deaths during the follow-up period; three in the early colectomy group and two in the infliximab group. There was one death within 30 days of ASUC presentation in the early colectomy group, which occurred after discharge from hospital and the cause was unknown. The remaining four deaths all occurred over 12 months following the initial ASUC admission from unrelated comorbidities.
Of patients who received infliximab first, there was a higher median number of infusions given in the group who received early infliximab without subsequent colectomy than in those who received infliximab and then required a colectomy, although this did not reach significance (2 versus 1, p = 0.16). Similarly, a higher proportion of patients who received infliximab first and who subsequently required colectomy received only a single dose of infliximab (versus two or three doses) compared with those who never subsequently required a colectomy, though again this did not reach statistical significance (56% versus 37%, p = 0.06).
Primary outcome analyses: comparison of inpatient and outpatient healthcare utilization and total direct healthcare costs in the early infliximab versus colectomy groups after presentation of ASUC
Inpatient healthcare utilization was not significantly different between those who received early infliximab compared with early colectomy for ASUC, either in terms of median number of admissions (7.0 versus 4.5 admissions, respectively, p = 0.22), or median inpatient cumulative hospital length of stay (24 days versus 29 days, p = 0.44) over the follow-up period. Furthermore, the median number of outpatient visits after ASUC did not differ between the groups (7.0 versus 6.0, p = 0.77).
The mean total healthcare costs (the combined cost of inpatient hospitalization, medications, procedures, and outpatient visits) were significantly lower for early infliximab patients than for those who had received early colectomy at 30 days and remained lower until 6 months after initial admission with ASUC (mean cost at 30 days AUD10,350 versus AUD20,397, p < 0.001, and at 6 months AUD17,662 versus AUD24,852, p = 0.003, respectively). The mean costs increased more rapidly in the infliximab-treated group over long-term follow up, but there were no significant differences from 1 year until 7 years after index admission between patients treated with early infliximab or colectomy (AUD25,406 versus AUD31,799, p = 0.06 and AUD72,834 versus AUD59,557, p = 0.23, respectively). The total healthcare costs are shown in Figure 1(a).
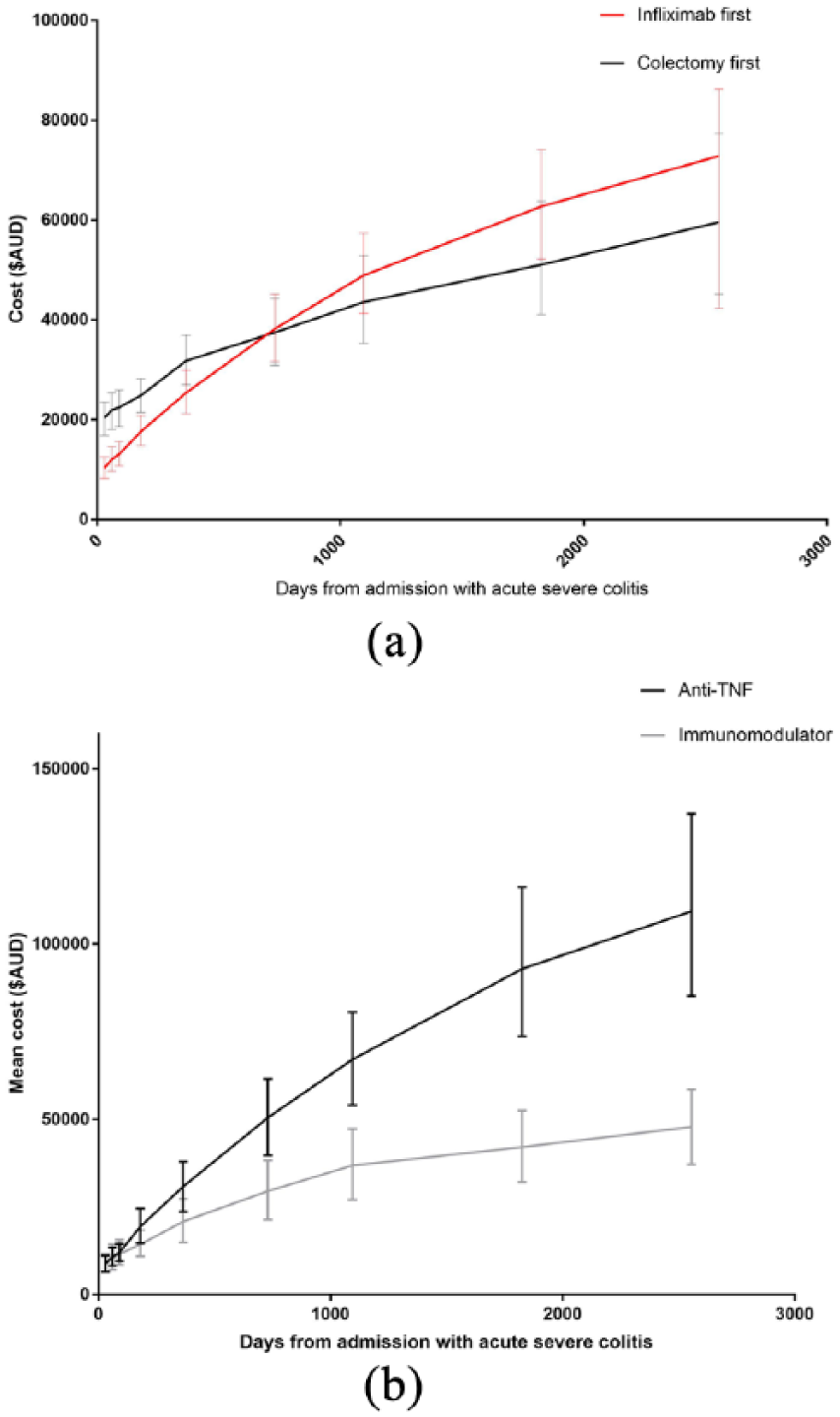
Total mean healthcare costs for patients following admission with acute severe ulcerative colitis based on treatment used. (a) Infliximab versus early colectomy. (b) Immunomodulator versus anti-TNF maintenance therapy following infliximab salvage therapy.
Hospital costs (inpatient hospital costs for all admissions based on reimbursement data) were significantly lower in the first year for patients treated with early infliximab compared with early colectomy (AUD19,505 versus AUD27,583 at 1 year, p = 0.008), but from 2 years to 7 years there was no significant difference (e.g. cost at 7 years AUD44,031 versus AUD45,244, p = 0.85). In contrast, procedure costs were significantly lower for patients treated with early infliximab compared with early colectomy for all durations to 7 years (cost at 7 years AUD2598 versus AUD4097, p = 0.02).
Medication costs remained significantly higher from 30 days until 7 years after index admission for ASUC for patients treated with early infliximab compared with early colectomy first (mean cost at 7 years AUD28,202 versus AUD11,204, p = 0.02).
Outpatient costs were relatively low in both groups and did not significantly differ throughout the follow-up period, with mean costs at 7 years following index admission being AUD681 versus AUD639 (p = 0.74) for the early infliximab and early colectomy groups, respectively. The proportion of total costs in each group is shown in Figure 2.

Percentage of total direct healthcare costs for patients receiving a colectomy first (a) and infliximab first (b) for acute severe ulcerative colitis.
The median number of complications requiring rehospitalization was significantly higher amongst patients who received early colectomy versus infliximab first (4.0 versus 1.0, p < 0.001). Also, the proportion of patients experiencing any complications was significantly higher amongst patients treated with colectomy first (94% versus 64%, p = 0.002). Specifically, surgical complications (including one or more of ileostomy malfunction, wound dehiscence, postprocedural bowel obstruction, and wound infection) were significantly higher (median 2.0 versus 0, p = 0.001), while infective, bleeding, and thrombotic complications were comparable between the groups (each p > 0.05). Figure 3 demonstrates the rates of relevant complication captured via ICD-10 coding during re-admissions after the initial presentation for ASUC by group.

Proportion of patients with complications based on ICD-10 coding for initial infliximab and early colectomy groups.
Secondary analyses
Comparison of maintenance strategy for patients after receiving infliximab salvage therapy for ASUC
Of the 85 patients who received early infliximab, 37 (44%) were commenced on maintenance anti-TNF therapy while 34 (40%) received maintenance immunomodulator without biologic therapy. A further six (7%) patients had a colectomy within 30 days of receiving infliximab and thus never received maintenance therapy, while eight (9%) patients had no available data regarding the maintenance strategy utilized. Of those receiving maintenance anti-TNF therapy, 33 (83%) were also on concomitant immunomodulator therapy. In terms of the maintenance anti-TNF therapy employed, 22 (59%) continued on or resumed infliximab and 10 (27%) were prescribed adalimumab, while 5 (14%) patients had both maintenance infliximab and adalimumab at different time points during follow up. The median time to escalation to maintenance anti-TNF therapy was 5 days after admission with ASUC (range 0, 3042 days), that is the majority of patients went straight on to maintenance anti-TNF therapy following induction, then remained on anti-TNF therapy for a median of 1174 days (1, 3739).
There was no significant difference in long-term colectomy rates for patients treated with maintenance anti-TNF therapy versus conventional therapies (33% versus 49% during the follow-up period, p = 0.19).
In those treated with early infliximab, there was no difference in total costs at 12 months following the index ASUC admission between those who went on to maintenance anti-TNF therapy versus immunomodulator therapy, AUD29,657 versus AUD 20,862 (p = 0.08). Subsequently however, total costs were higher amongst patients who received maintenance anti-TNF therapy (total mean costs at 7 years after index admission were AUD109,365 versus AUD47,842, p < 0.01). The cost trends are shown in Figure 1(b). Medication costs were higher in the anti-TNF maintenance group compared with the immunomodulator maintenance group, throughout all follow-up time points from 30 days onwards, with the mean costs (and deltas thereof) increasing from AUD603 versus AUD183 (p < 0.001) at 30 days to AUD47,023 versus AUD12,989 (p < 0.001) at 7 years. Procedure and inpatient costs however did not significantly differ between the two maintenance groups following index admission (p > 0.05 at all time points).
Comparison of outcomes in patients receiving initial infliximab then colectomy versus early colectomy for ASUC
When comparing those who received infliximab first then a colectomy with initial colectomy for ASUC, there were no significant differences in the median total length of stay (41.5 days versus 29 days, respectively, p = 0.11), number of admissions (7.0 versus 4.0, p = 0.24), or number of outpatient visits attended during follow up between the groups (6.0 versus 6.0, p = 0.82) (Table 3). Also, there was no significant difference in the number or rates of complications during follow up between these groups.
Comparison of outcomes for patients treated by early colectomy or colectomy following initial infliximab for acute severe ulcerative colitis.

The mean total costs were comparable for patients treated with early infliximab but requiring eventual colectomy and those treated with initial colectomy at 7 years following index admission (AUD80,811 versus AUD59,557, p = 0.15), and also there were no significant differences in mean inpatient, procedural, or medication costs between the groups.
Comparison of outcomes in patients receiving early colectomy based on type of surgery performed
Of patients who required a colectomy, 47% (32/68) had an IPAA (14/32 of colectomy first versus 18/36 of infliximab first group, p = 0.61), while the remaining 53% (36/68) had a colectomy with end ileostomy. The majority who had an IPAA had a two-stage procedure (26/32, 81%).
Procedure costs became higher at 1 year following index admission for patients treated with IPAA compared with other colectomy (AUD3588 versus AUD2117, p = 0.01), and remained significantly higher at 7 years after index admission (AUD6232 versus AUD3498, p = 0.01).
Hypothetical comparison of costs when incorporating cost reductions in anti-TNF therapy availed by introduction of biosimilars
The medication costs used in our initial analysis were based on the cost of therapy at current pricing, however with biosimilar infliximab recently available and the imminent arrival of adalimumab biosimilars, we performed a hypothetical adjustment of the model based on cost reductions of 50% and 70% in anti-TNF therapy. As presented in Figure 4, there was no significant difference in costs compared with an early colectomy approach after 7 years follow up, but there was a significant reduction in the mean cost of patients treated with initial infliximab by AUD10,925 (p > 0.05).

Cost trend over time based on a reduction in the cost of anti-TNF therapy.
Discussion
This study provides comprehensive, long-term follow-up data of patients treated for steroid-refractory ASUC in a real-world setting. It supports the current widely adopted practice of infliximab as first-line salvage therapy, showing it to be effective in achieving a durable, sustained efficacy and reducing rehospitalization for complications following ASUC, with no apparent detriment to outcomes in those who, despite infliximab, still required subsequent colectomy compared with those treated with early colectomy alone. Moreover, long-term cost analysis suggested that, on average, initial infliximab did not result in higher healthcare costs than an early colectomy approach, with higher medication costs partially offset by lower inpatient and procedure-related costs. This is an important and novel finding, particularly given that almost half of the patients receiving infliximab required maintenance anti-TNF therapy over a long median follow-up period of 7 years, yet even with this long-term anti-TNF use, costs remained comparable with those for early colectomy patients.
Furthermore, this metric is likely to favor anti-TNF therapy even more in future given the arrival and growing utilization of biosimilars, leading to drug cost savings of a minimum of 30% for health payers worldwide.27,28 For example, in Japan, biosimilar infliximab was released at a 67% unit price reduction compared with reference infliximab. 29 Locally at time of writing, the study hospital pharmacy purchases biosimilar infliximab at a 59% reduction to the Pharmaceutical Benefits Scheme reference price for infliximab.
An important caveat of this ‘real-world’ study is that there were no local guidelines dictating the frequency of infliximab infusions in ASUC, so decisions regarding the number of infliximab infusions and timing of these were at the clinician’s discretion. Generally, patients who were not responding clinically to the initial dose of infliximab were given one or two further doses at weeks 2 and 6. Infliximab dosing was restricted to 5 mg/kg per dose and dose intensification was not utilized in this cohort.
As demonstrated here, where only an additional 7% required colectomy after 1 year, many studies have demonstrated that most colectomies occur within 12 months of ASUC presentation, including the ACT1/2 extension study with only 1% requiring a colectomy between year 1 and year 3 of follow up. 30 Therefore, it is plausible that long-term maintenance anti-TNF therapy is actually not necessary for all patients following ASUC with many patients ably managed on maintenance immunomodulator therapy following initial infliximab induction. As shown in our study as in multiple previous studies, the long-term costs associated with anti-TNF therapy are a major component of total healthcare costs with the growing trend for intensive medical management of UC despite apparent reductions achieved in hospitalizations. Hence, a more nuanced approach to anti-TNF maintenance and preferential utilization of thiopurines as maintenance therapy where possible might be a potential mechanism to reduce these costs.31,32 In addition, the advent of anti-TNF biosimilar agents should also impact overall costs, with mean total costs estimated to reduce by over AUD10,000 in this study.
The limitations of this study include the retrospective nature of data acquisition, the variable follow-up time, and the lack of randomization, each of which could have lead to unknown confounders skewing the results. We attempted to account for missing data through multiple imputation analysis and did not find any difference in our results, thus this is less likely to significantly affect the results. In addition, some cases of fulminant colitis will require colectomy regardless of whether infliximab is given or not, with some patients in our cohort requiring colectomy only days after initial infliximab therapy. Nevertheless, the historical nature of this cohort allowed the comparison of an initial infliximab and initial colectomy approach with reasonable sample sizes per group whereas nowadays, as is the current trend elsewhere, very few patients do not receive infliximab first, thus the second group is no longer available in sufficient numbers for comparison. Another potential issue is potential selection bias. The colectomy-first group had a higher proportion with extensive colitis (E3) on initial demographic features, however the fact that other demographic features did not significantly differ suggests that this was not the primary reason for the differences in outcomes between the groups. Previous baseline models that have been shown to predict colectomy rates in ASUC, including the Oxford index, Edinburgh predictive index, and Sweden index, were similar between the groups, implying no significant underlying differences in disease characteristics or severity.
In an era of cost containment and growing concerns for the sustainability of exponentially increasing biologic usage worldwide, this study is the first to our knowledge that shows the consistent, durable reduction in long-term healthcare utilization achievable with an infliximab-first approach in ASUC. This therefore provides real-world clinical data that goes some way to addressing important issues for health payers, clinicians, and patients alike. For payers, these data suggest that despite the high drug costs of infliximab, the even higher costs of inpatient hospitalization (including procedures) are largely offset by the former. For clinicians, current practice is further confirmed by this study, and reassured by the implication that even in those who still require a subsequent colectomy, infliximab does not appear to increase the risk of postoperative complications requiring hospitalization. Finally, for patients, an initial infliximab approach is intuitively preferable, with added advantages shown here in that subsequent colectomy was avoided long term in almost half, with a lower likelihood of future hospitalizations even if colectomy is ultimately required.
Footnotes
Appendix
Complications assessed in this study following ASUC admission as per ICD-10 coding analysis of all patients to end of follow-up period.

| ICD-10 Coding | Description of complication |
|---|---|
| Z93.2 | Ileostomy/colostomy status |
| Z48.8 | Surgical care follow up |
| T81.41 | Wound infection |
| Y83.3 | Stoma related |
| I80.* | Phlebitis of lower limb vessel |
| Y83.2 | Surgical anastomosis related |
| R10.4 | Abdominal pain not otherwise specified |
| Y83.8 | Other surgical procedures |
| K91.4 | Ileostomy malfunction |
| E86 | Volume depletion |
| N17.9 | Acute kidney injury |
| Z43.2 | Attention to ileostomy |
| K66.0 | Peritoneal adhesions |
| T81.3 | Wound dehiscence |
| D50.9 | Iron deficiency anemia |
| I26.9 | Pulmonary embolism |
| K62.4 | Stenosis of the rectum or anus |
| D62 | Posthemorrhagic anemia |
| K56.0 | Ileus |
| T81.2 | Accidental puncture/laceration during procedure |
| Y42.0 | Steroid side effects |
| K61.* | Rectal/perianal abscess |
| I97.8 | Postprocedure disorder of circulation |
| K65.* | Peritonitis/intra-abdominal sepsis |
| K91.3 | Postprocedure bowel obstruction |
| J18* | Pneumonia |
| A41.9 | Sepsis |
| R50.9 | Fever (unspecified) |
| K91.8 | Postprocedural disorders of the digestive tract |
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: AV receives financial support through an Australian Government Research Training Program Scholarship.
Conflict of interest statement
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: DRvL has served as a speaker and/or received travel support from Takeda, Ferring, and Shire. He has consultancy agreements with AbbVie, Janssen, and Pfizer. He received research funding grants for investigator-driven studies from Ferring, Shire, and AbbVie. AV has received financial support to attend educational meetings from Ferring, Norgine, Takeda, and Shire.
