Abstract
Background:
Our aim was to systematically review the relationship between iron and incident cognitive decline or dementia from midlife onwards.
Methods:
Systematic review of eligible studies using Medline, Embase and PsycINFO® for the period from 1 January 1986 to 2 December 2016 (CRD42016023800), where study populations had a mean age of over 50 years and were free of cognitive impairment or dementia at baseline. Two authors independently extracted data according to eligibility criteria and assessed study characteristics, quality and outcomes. Disagreement was resolved by discussion.
Results:
A total of 1185 relevant records were identified with 12 full-text articles eligible for review. Six studies were excluded, leaving six texts to be included. Sample size ranged from 90 to 7173, with an average follow up of approximately 11.5 years. Baseline iron measures included brain iron (n = 2), iron-related biomarkers in blood and plasma (n = 2), and iron intake estimates from dietary records (n = 2). Outcomes were dementia incidence (n = 2) and longitudinal outcomes on neuropsychological tests (n = 4). Bias was evident across studies in one or more of the following: recruitment, iron exposure, outcome assessments, potential confounders, missing data or attrition.
Conclusions:
Diversity across the small number of identified studies precludes conclusions regarding the role of iron in cognitive decline or dementia. Our review highlights substantial gaps in the evidence base and the need for more comprehensive, higher quality studies in this area.
Introduction
Older age is associated with increased risk of iron deficiency (ID), elevated body iron stores and increased brain iron levels.1,2 Complex regulatory mechanisms fine-tune iron metabolism and homeostasis for both heme and nonheme iron. Iron has a key role in multiple pathways, for example, as a constituent part of proteins needed for oxygen transport, oxidative phosphorylation, myelin production, and production and breakdown of neurotransmitters.3–5 Consequently there are multiple plausible mechanisms by which iron deficiency or overload may elevate the risk of age-related cognitive decline and dementia. A detailed mechanistic review is beyond the scope of this article, however for recent reviews in this area, see Hare and colleagues 3 and Waldvogel-Abramowski and colleagues 4
Iron deficiency
In older adults, absolute ID is caused by insufficient dietary iron intake, gastrointestinal malabsorption, or increased blood losses attributable to gastrointestinal pathologies. 2 Potential cognitive impacts related to inadequate iron might stem from cerebral hypoxia, 6 insufficient neurotransmitter synthesis 5 or poor myelin integrity. 7
There is also a growing body of evidence that anaemia in older age (defined by haemoglobin level <13 g/dl in men and <12 g/dl in women) is associated with poor cognition, cognitive decline and dementia. 8 ID anaemia accounts for around 16% of total anaemia cases. 9
Iron overload
Body iron levels may be elevated in middle-aged and older adults due to consumption of highly bioavailable forms of iron (supplemental iron and red meat) and of fruit, an enhancer of nonheme-iron absorption (vitamin C). 10 In two meta-analyses of prospective cohort studies, high body iron stores and consumption of heme iron were associated with increased risk of coronary heart disease (21 studies) 11 and type 2 (T2) diabetes (11 studies), 12 which may highlight a further pathway between iron overload and cognitive impairment and dementia.13–15
Increased brain iron
The accumulation of iron in the brain is an established hallmark of ageing. 16 Excess iron stores may increase pro-oxidant reactions and generation of free radicals,1,3 which could contribute to neurodegeneration. In cross-sectional studies, magnetic resonance imaging (MRI) measures of iron are associated with amyloid load, 17 smaller hippocampal volume 18 and poorer cognitive performance.15,19 Hippocampal iron content has also been correlated with cognitive performance and disease duration in Alzheimer’s disease20,21 and MRI-measured iron in the basal ganglia in cognitively normal adults predicted greater rates of cognitive decline and cortical atrophy.22,23 Brain iron deposits have also been positively associated with white matter hyperintensities, suggesting a potential link between brain iron and vascular pathology.15,24
The proportion of the population at risk for cognitive decline and dementia is rapidly increasing, and there is no effective pharmacological cure or preventive intervention. The promotion of clinical understanding through collation of evidence for modifiable risk factors is an important preventive strategy. Iron has been raised as a potentially modifiable risk factor for cognitive decline or dementia but the area is complex. There are multiple ways of measuring both iron and cognition, and to date there is no critical appraisal of the evidence depth, breadth or quality. Our aim was to systematically review and evaluate the current evidence base that relates to the relationship between iron and incident cognitive decline or dementia.
Methods
Search strategy and selection criteria
The databases MEDLINE, Embase and PsycINFO were searched over a 30-year period from 1986 to 2 December 2016. Because this is a first review of the literature in this area, search terms were chosen to be as broad as possible to prevent inadvertent exclusion of particular cognitive assessment or particular iron measures. Search terms included dementia, alzheimers disease, vascular dementia, multi-infarct dementia, cognit* and iron, heme, haem, ferritin and serum transferrin. See supplementary text for details. Reference lists were screened and experts in the field were consulted. In addition, searches of the Cochrane Library, the ISRCTN Register and the ClinicalTrials.gov website were carried out to look for ongoing or completed relevant trials.
All identified abstracts, or titles, where abstracts were unavailable, were independently read by two reviewers (DH, RP) and a list of potential evidence for inclusion was compiled by the two analysts (DH, RP). These lists were then compared and any differences resolved by discussion. Full text copies of the selected texts were independently read and assessed for relevance by both analysts and in accordance with the following inclusion and exclusion criteria.
Inclusion criteria
Prospective longitudinal studies of primary research (cohorts or clinical trials) reporting on the following:
An assessment of exposure to iron (assessed via biomarkers, e.g. blood, cerebral spinal fluid or neuroimaging) or external sources, for example, diet or iron supplementation.
Evidence, or clear implication, that participants were free of cognitive decline or dementia at baseline assessment.
Use of formal assessment of cognitive function.
Reporting outcomes of cognitive decline or incident dementia.
Adult populations with a mean age over 50 years
Exclusion criteria:
Publications relating only to populations selected on the basis of clinical comorbidity when that comorbidity may also affect cognitive function, for example, requirement for renal dialysis.
Non English publications (in the absence of resources available for translation).
Inclusion of nonadults.
Papers identified as relevant were then independently assessed for quality and data relating to study characteristics, assessment of cognitive function and iron exposure were extracted by each analyst. A formal quality scoring scheme was not used as these lack discriminatory power, however each paper was assessed against the key factors given in Critical Appraisal Skills Programme checklists for evaluating trials and longitudinal studies (Critical Appraisal Skills Programme: http://www.casp-uk.net/ accessed 15th February 2017).
The review was carried out to best practice and in accordance with PRISMA guidelines. 25 Protocol registration no. CRD42016023800.
Results
A total of 2001 records were identified from the searches and a further two from reference screening/expert recommendation; 1185 records remained once duplicates were removed. These were screened and 12 full text articles representing 12 separate studies were reviewed. Six studies were included with the remaining six excluded because they lacked results related to iron exposure (n = 2),26,27 included prevalent cases of cognitive impairment (n = 3),15,28,29 or had a cross-sectional design (n = 1). 30 See Figure 1 for the PRISMA study flow chart.
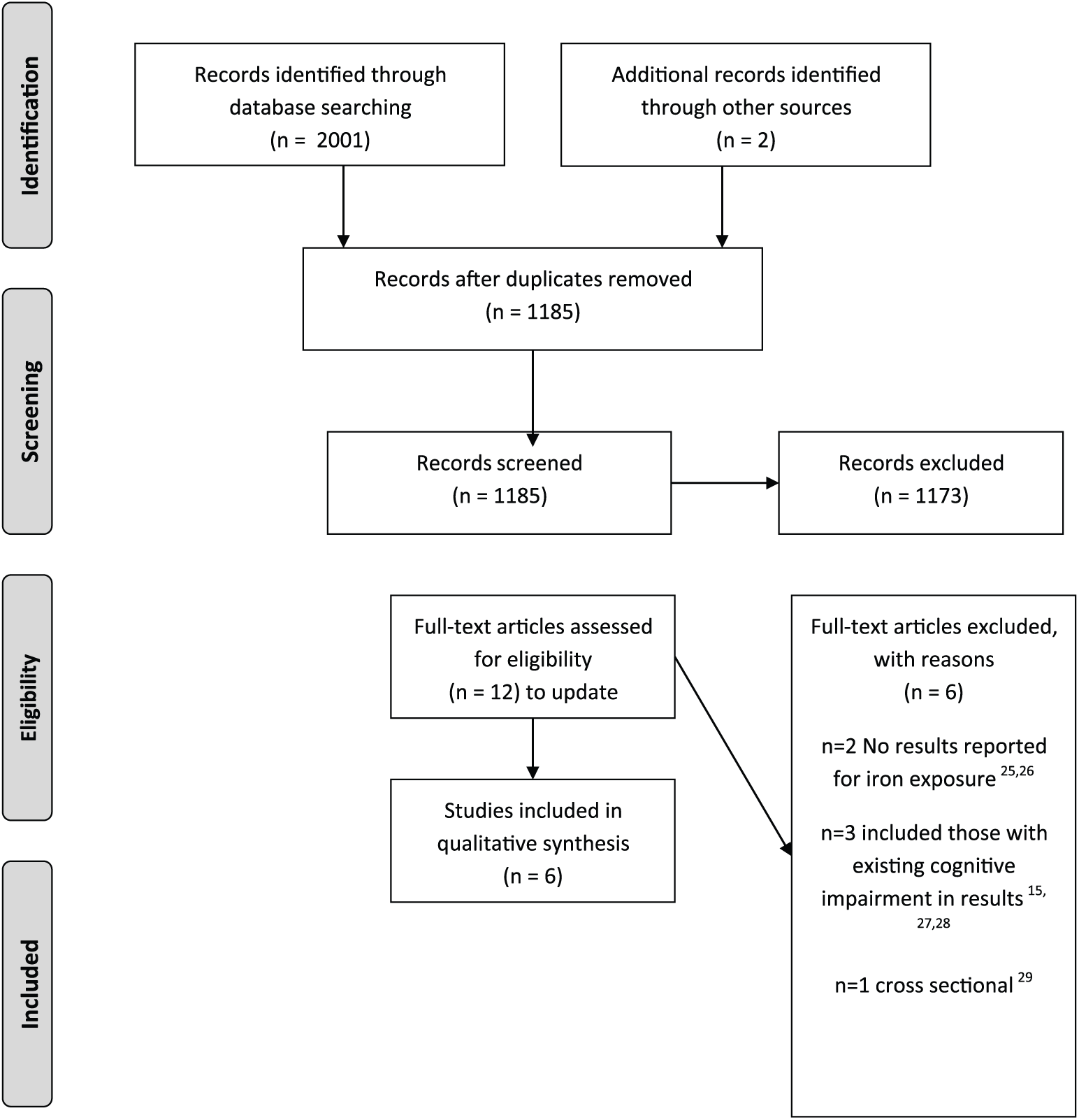
Study selection flowchart.
Study characteristics
Of the studies that were included, four reported results from North American populations (United States of America); two using data from the National Health and Nutrition Examination Survey (NHANES),31,32 one from a Detroit-based cohort study, 22 and one using participant data from the Alzheimer’s Disease Neuroimaging Initiative (ADNI) cohort. 33 The remaining studies were from Australia, a longitudinal cohort study based in Canberra and Queanbeyan, 34 and the Netherlands, a cohort analysis of clinical trial data from the Folic Acid and Carotid Intima-Media Thickness (FACIT) trial. 35 For further study characteristics, see Table 1.
Study characteristics.

N/A, not applicable.
Measures of iron
Studies used widely differing measures of baseline iron (Table 2). Of the six identified studies, two used measures of brain iron22,33 and four used measures of peripheral iron.31,32,34,35 The NHANES 40-plus cohort reported on serum transferrin, 32 the NHANES 60-plus cohort and the Australian cohort study used dietary iron, 34 the FACIT trial collected total iron-binding capacity, transferrin saturation, ferritin and nontransferrin bound iron. 35 Of those that collected data on brain iron, the ADNI cohort used ferritin in cerebrospinal fluid (CSF) 33 and the Detroit study used imaging (MRI). 22 The Detroit cohort and the FACIT trial cohort were the only studies to report assessment of iron at follow up. The FACIT trial collected serum iron, ferritin and nontransferrin bound iron and the Detroit study repeated their baseline MRI scan.22,35
Measurements of iron.

Transferrin: iron transport protein.
Transferrin saturation: the ratio of serum iron to serum concentration of transferrin.
Ferritin: indicator of total body iron stores.
Nontransferrin-bound iron: free iron in the circulation not bound to transferrin.
MRI, magnetic resonance imaging; N/A, not applicable; FFQ, food frequency questionnaire.
Cognitive outcome measures
The NHANES cohorts reported on dementia outcomes (Table 3). Data on dementia and Alzheimer’s disease outcomes were collected from death certificates, hospital and nursing home records for the 40-plus cohort. 32 Diagnoses of Alzheimer’s disease for the 60-plus cohort were collected from death certificates only; 31 ; neither carried out cognitive testing. The Detroit cohort, 22 FACIT trial cohort, 35 ADNI cohort 33 and Australian cohort 34 used standard cognitive assessment tools at baseline and follow up.
Measurement of cognitive function.
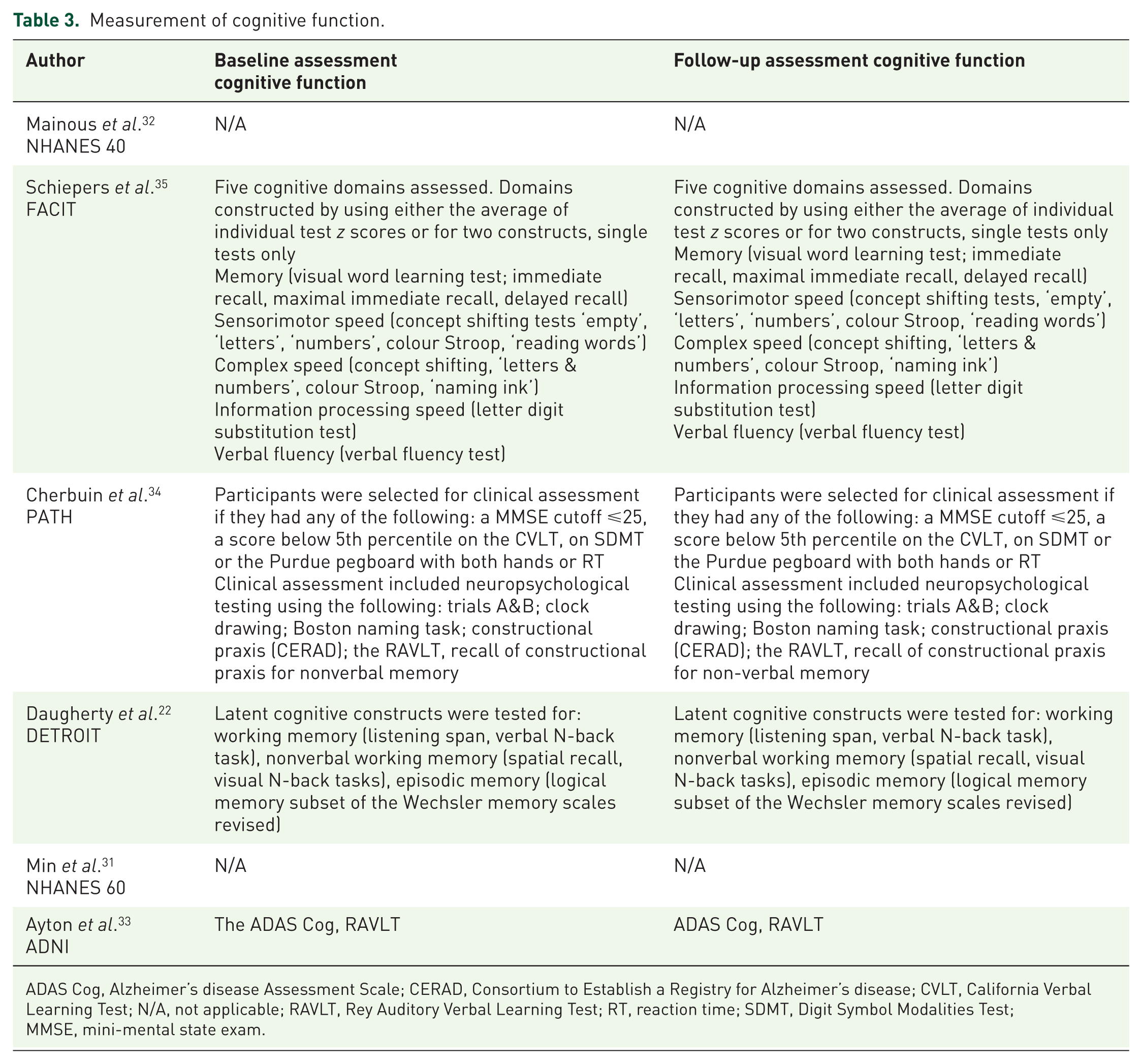
ADAS Cog, Alzheimer’s disease Assessment Scale; CERAD, Consortium to Establish a Registry for Alzheimer’s disease; CVLT, California Verbal Learning Test; N/A, not applicable; RAVLT, Rey Auditory Verbal Learning Test; RT, reaction time; SDMT, Digit Symbol Modalities Test; MMSE, mini-mental state exam.
Association between iron measures and cognitive outcomes as reported by the included studies
Overall the results were variable across studies (Table 4):
In the NHANES 40-plus cohort (n = 6427), an association was found between co-occurring high levels of transferrin (TS) and cholesterol, and increased risk of Alzheimer’s disease [both TS and cholesterol at the 75th percentile, hazard ratio (HR) 3.19, 95% confidence interval (CI) 1.31–7.75]; but no association between transferrin alone and Alzheimer’s disease 32 (transferrin >75% compared with < 75% Alzheimer’s disease: HR 1.47, 95% CI 0.82–2.63).
In the FACIT trial (n = 800) the potential of folic acid supplementation to modify the effects of iron measures on cognitive change was tested prior to examining associations between serum iron parameters and longitudinal cognitive functioning. No associations were found. When stratifying for status as a blood donor, higher iron status was associated with less decline in sensorimotor (parameter estimate = 0.005, p = 0.010) and word fluency tasks (parameter estimate = 0.006, p = 0.012). 35
The Australian PATH cohort study (n = 1354) found an increased risk of cognitive impairment with higher levels of iron intake (HR 1.54, 95% CI 1.03–2.29 for transition from normal cognition to mild cognitive impairment (MCI), per 1 mg iron) and the potential for an interaction with sex such that higher intake may be associated with lower risk in women but higher risk in men. 34
The PATH study reported a mean iron intake of 18 mg per day, more than double the daily intake of 8 mg recommended by the Australian National Health and Medical Research Council (NHMRC) for those in their sixties.
The Detroit cohort (n = 78) (examining the hippocampus, caudate and putamen) reported no association between baseline iron or change in iron and episodic memory or nonverbal working memory, with the exception of baseline iron in the caudate nucleus which was associated with less improvement in verbal working memory, β –0.18 (p = 0.01). 22
The NHANES 60-plus cohort (n = 4639) found no association between Alzheimer’s disease related death and dietary iron intake (p = 0.1022) but an increased risk of Alzheimer’s disease mortality found for those with low haemoglobin (HR 8.4, 95% CI 1.4–50.8). 31
The study using the ADNI cohort (n = 43) reported an increased risk of decline associated with higher levels of ferritin, but suggested that the result may be confined to those with the apolipoprotein (APOE) Ɛ4 allele. For those who were APOE Ɛ4 positive, Alzheimer’s disease Assessment Scale (ADAS Cog) β 0.09 [standard error (SE) 0.04], p = 0.02, Rey Auditory Verbal Learning Test (RAVLT) β –1.49 (SE 0.4), p < 0.001 compared with APOE Ɛ4 negative, ADAS Cog β –0.04 (SE 0.016), p = 0.02, RAVLT nonsignificant. 33
Results reported by included studies.

AD, Alzheimer’s disease; ADAS Cog, Alzheimer’s disease Assessment Scale; APOE, apolipoprotein E; BMI, body mass index; CI, confidence interval; CSF, cerebrospinal fluid; HR, hazard ratio; NS, nonsignificant; RAVLT, RAVLT, Rey Auditory Verbal Learning Test; SE, standard error; MCD, mild cognitive disorders; MCI, mild cognitive impairment.
Study quality
All studies reported a clear research question (Table 5). However, studies utilized diverse designs with the potential for bias in recruitment, measures of iron exposure, outcome assessment, potential confounders, missing data or attrition. The applicability of three studies was limited by recruitment from specific population groups; one from an ongoing longitudinal study of ageing, 22 one from the ongoing ADNI study 33 and one from a clinical trial population. 35 The other three studies used representative population samples from Australia 34 and the USA.31,32 However, Mainous and colleagues 32 (n = 6427) selected a cohort aged from 40 years upwards, thus potentially minimizing the potential to observe meaningful cognitive change. Four of the studies reported on populations with a mean age over 60 years, that is those more likely to manifest cognitive change,31, 33–35 and recruited large numbers (NHANES 60 n = 4639, PATH N = 1354, FACIT n = 800), with the exception of the ADNI study (n = 43). None focused solely on older adults and one, the Daugherty imaging study, included a wide age range from 19 to 77 years (n = 78), which would have included a varied population; in particular, pre- and postmenopausal women. 22 Furthermore, full details of statistical methods and adjustment for potential confounders was limited and inconsistent, and three studies lacked adequate adjustment for the confounding variables likely to influence cognitive function.22,31,32
Assessment of study quality.

AD, Alzheimer’s disease; ADAS Cog, Alzheimer’s disease Assessment Scale; BMI, body mass index; NHANES, National Health and Nutrition Examination Survey; FFQ, food frequency questionnaire.
The measurement of iron was similarly varied. None of the studies attempted to take account of prior iron exposure and iron status at follow up was assessed only in the FACIT trial 35 and the Detroit Aging study. 22 Exposure measures were selective, with no overlap and no combined dietary and biomarker assessments. Only two studies measured brain iron levels.22,33
Measurement of cognitive function was also limited, both in breadth and sensitivity. Outcome assessments were selected from standard test batteries for four of the studies.33–36 The two studies using the NHANES database collected outcome data from death certificates31,32 and healthcare records, 32 thus raising the possibility of misclassification and under reporting due to missed cases in the community, lack of high-quality diagnoses in nursing home environments and less than rigorous death certificate coding.
The interpretation of study results is also limited by the varying research questions and statistical focus in the different studies. For example, the publication by Mainous and colleagues aimed to look at cholesterol in combination with transferrin rather than using iron measures alone, 32 and the publication by Min and colleagues focused on haemoglobin, folate and vitamin B12, reporting on iron only in passing as a covariate and as part of the baseline characteristics. 31 None of the studies focused on a potential U-shaped relationship with iron and cognitive outcome. Participant numbers, and subsequently the numbers per cell in analyses were also particularly small in some studies; in the study by Min and colleagues only 49 participants from 4688 had died from Alzheimer’s disease at follow up. 31 Daugherty included only 125 participants 22 and in the study by Ayton and colleagues there were only 90 participants in the cognitively normal group. 33 Finally, participant dropout and missing data31,34 further limit the interpretation of the results.
Discussion
To our knowledge, this is the first systematic review of the evidence relating to iron, cognitive decline and dementia. Overall, the available studies were inconsistent in terms of length of follow up, composition of study population, percentage female or male, age of study sample, iron assessment, outcome measure and results. Two studies assessed the influence of dietary iron in relatively similar age groups, one (the USA NHANES 60-plus cohort 31 ) found no association with dementia. The other (the Australian PATH study 34 ) found an increased risk of cognitive decline associated with greater iron intake, possibly more relevant in men.
Several of the studies did report a stronger relationship in a particular subgroup, although the subgroups also varied. The older NHANES cohort found a relationship between higher transferrin and increased risk of Alzheimer’s disease in those with higher cholesterol. 32 In the ADNI cohort the relationship between higher cerebrospinal ferritin and cognitive decline was strongest in those who were also APOE Ɛ4 positive, 33 and in the FACIT trial cohort, higher serum iron levels were associated with less decline in sensorimotor speed and word fluency in those who did not donate blood. 35
A recent comprehensive review of iron deficiency and risk of cardiovascular disease included 13 longitudinal studies of serum iron, 15 studies of ferritin and 14 of transferrin saturation and total iron binding capacity, but also observed heterogeneity and inconclusive findings across studies. Although they concluded that ‘extreme conditions of iron deficiency as well as iron overload were associated with increased cardiovascular disease risk’. 37
Limitations associated with our review and its conclusions stem mostly from the paucity of studies rather than the methods used for the review process. It is possible that in restricting the language to English and in excluding study populations with serious clinical comorbidities, potentially relevant evidence has been overlooked. However, including populations with specific comorbidities where the comorbidity itself may influence iron metabolism, risk of mortality and risk of cognitive decline would have been to address a different research question. The lack of evidence, and the varying study designs and populations used within the available evidence base, present a more serious limitation.
Limitations in the assessment of iron
None of the studies used a fully comprehensive assessment of iron status. This is important because cerebrospinal fluid and blood reporters of iron only have a modest association, 29 blood iron markers do not predict MRI measured brain iron content 38 and, in older adults in particular, levels of the peripheral iron reporters, ferritin and haemoglobin, may also signify peripheral inflammation such as anaemia of chronic inflammation, 39 or indeed noniron-deficient anaemia. In the case of the latter, tissue deposition of iron may occur even when presenting with low iron markers in the blood. For example, the iron-trafficking disease, aceruloplasminemia, results in tissue iron deposition and lower blood iron markers because iron cannot efficiently be exported out of the cell. 40 If cellular iron trafficking, rather than iron exposure, is the lesion that results in brain iron elevation in Alzheimer’s disease, it is possible that depletion of iron in the blood accompanies iron elevation in tissues. Furthermore, only two of the studies assessed dietary iron and no studies evaluated the impact of iron supplementation. No studies directly followed a potential link between iron intake and iron blood or neuroimaging biomarkers and subsequent cognitive function, and none attempted to evaluate iron impact by baseline iron level or by clinically relevant categories such as iron deficiency anaemia or iron deficiency no anaemia. We were unable to identify any trials of dietary iron exposure, or of iron supplementation, in normal or iron-deficient adults in mid or late life and risk of cognitive decline.
Limitations in the assessment of outcome
Cognition is complex and multifaceted and it is possible that iron status differentially affects change or decline across the spectrum of ability domains. Clearly, in the cognitively healthy a more comprehensive and psychometrically driven approach to assessing cognitive change is necessary if the impact of iron status on cognitive decline is to be more fully understood. The use of medical and death certificate records for dementia diagnoses in the NHANES analyses presents a further limitation since their accuracy depends upon robust and comprehensive assessment and record keeping.
Gaps in the evidence base and future work
Our systematic review of the current evidence base relating to iron and cognitive function and dementia found a sparse and limited field of evidence. Much more work is needed to evaluate the links between iron intake, brain deposition, ageing and cognitive function over time. In particular, a detailed focus is needed on regional brain iron and related cognitive abilities with multiple follow ups, MRI measures or historical health measures taking closer account of potential causal and associative relationships, including key confounding measures. Obvious examples include ensuring that analyses with ferritin (also an acute phase protein) are, at minimum, adjusted for other inflammatory markers, and taking account of obesity and its association with iron dysregulation. Multimodal approaches should also be used, such as using CSF iron biomarkers in combination with MRI measures of brain iron. Larger, longer and more thorough epidemiological studies are also needed to fully explore the potential pathways as is a greater emphasis on collection of biomarkers to allow more accurate evaluation of the homeostatic iron picture in its entirety, taking account of the varied iron types and storage mechanisms. This would also be an important part of the safety considerations prior to conducting any randomized clinical trials of iron supplementation or iron chelation. In particular, we recommend that future studies in this area focus on reducing risk of bias derived from varied participant populations; that is, select those of a particular age range and stratify by sex. Studies should collect at least two measures of cognitive function over a minimum follow up of 12 months to allow assessment of change; when possible, two matching iron measures should be taken at the same interval, ideally using imaging and linking region of interest to the cognitive domain under assessment. This should be supplemented by peripheral measures, including inflammatory markers; when possible, additional data on usual diet, and clinical and cardiovascular history should be obtained.
Conclusion
The current evidence base is sparse and limited. The diversity of the existing evidence limits interpretation and applicability by either healthcare providers or researchers and precludes any conclusions relating to the relationship between iron and cognition. Much more work is needed before we can develop our understanding of the role of iron in cognitive function.
Supplemental Material
SUPPLEMENTARY_MATERIAL – Supplemental material for More evidence is needed. Iron, incident cognitive decline and dementia: a systematic review
Supplemental material, SUPPLEMENTARY_MATERIAL for More evidence is needed. Iron, incident cognitive decline and dementia: a systematic review by Diane E. Hosking, Scott Ayton, Nigel Beckett, Andrew Booth and Ruth Peters in Therapeutic Advances in Chronic Disease
Footnotes
Author contributions
RP: study concept; RP, DH, AB: study design; RP, DH, SA: data analysis; RP, DH, SA, NB: data interpretation. All authors contributed to manuscript preparation and revision, and approved the final version. No sponsor had any role in the design, methods, subject recruitment, data collection, analysis or preparation of the paper.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grant CE110001029 from the Australian Research Council (DH) and the Dementia Collaborative Research Centre for Early Diagnosis and Prevention; a member of the NHMRC National Institute for Dementia Research (RP).
Conflict of interest statement
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
