Abstract
Background:
Although the management of carotid disease is well established for symptomatic lesions ⩾70%, carotid revascularization for symptomatic low-grade (⩽50%) stenosis is not actually supported by data from randomized clinical trials. Such patients may occasionally have recurrent neurological symptoms despite optimal medical treatment owing to vulnerable plaques. In such cases, carotid artery stenting (CAS) may represent an option for treatment but this has not been tested in clinical trials. This study analyzed early and long-term outcomes of CAS performed in patients with low-grade symptomatic recurrent carotid stenosis.
Methods:
From a prospective registry of 322 carotid revascularization in symptomatic patients, 21 consecutive patients with low-grade symptomatic recurrent carotid stenosis who underwent CAS with proximal cerebral protection device Mo.Ma, after ruling out any other source of cerebral embolization, were involved in the study.
All patients had suggestive evidence of unstable plaque or plaque ulceration.
Results:
Procedural technical success rate was 100%. No 30-day stroke or death occurred, and no patients had recurrent neurological events related to the revascularized hemisphere during follow up. No 30-day local complications were reported. No late carotid occlusions were detected. There was one late death, and no stroke-related deaths. Survival rates were 100% at 1 year and 96% at 3 years.
Conclusions:
This study shows that CAS is a well-tolerated, effective and durable treatment for patients with recurrent symptomatic low-grade carotid stenosis associated with a vulnerable plaque. Patients had excellent protection against further ischemic events and survived long enough.
Keywords
Introduction
Although carotid revascularization is well established for neurologically symptomatic patients with carotid stenosis ⩾70%, 1 carotid endarterectomy (CEA) or carotid artery stenting (CAS) are not recommended, except in extraordinary circumstances, for symptomatic low-grade (⩽50%) stenosis.2–6 Nevertheless, such patients may occasionally have recurrent neurological symptoms despite optimal medical treatment (OMT) owing to vulnerable plaque. 7
Accumulating evidence from recent studies relating plaque stability with symptoms indicates that intraplaque hemorrhage, thrombus formation, and ulceration are consistent with a vulnerable plaque and has a large impact on the risk of ischemic events.8–10 Therefore, the importance of evaluating not only the severity of stenosis but also the vessel wall itself has been widely recognized for the management of carotid stenosis. 11 Thus, several reports have revealed plaque characteristics associated with a high risk of stroke on magnetic resonance imaging (MRI)12,13 and ultrasonography (US) assessment.14–16 In such cases, CAS may represent an option for treatment of recurrent symptomatic low-grade carotid stenosis but has not been tested in clinical trials because of previous failure to demonstrate any benefit of CEA in the setting of low-grade stenosis. 17
On the other hand, proximal endovascular occlusion cerebral protection is an alternative approach that uses balloons to occlude both external carotid artery (ECA) and common carotid artery (CCA) leading to blood flow arrest in the target internal carotid artery (ICA). This technique may provide better protection than distal protection with filters during all procedural steps and may be particularly indicated for lesion at high-risk of embolization, such as those low-grade vulnerable plaques with irregular surfaces and high lipid content. 18
The role of carotid intervention in low-grade stenosis at high-risk of recurrent stroke is not well established. Data regarding this treatment are relatively sparse. It deserves further investigation whether CAS with proximal cerebral protection is a possible treatment option for symptomatic patients with low-grade, but at high-risk of recurrence because of unstable plaque, carotid stenosis that is a refractory to OMT. For this purpose, this study analyzed early and long-term outcomes of CAS performed in patients with low-grade vulnerable symptomatic-recurrent carotid stenosis.
Methods
A prospectively compiled computerized database was queried concerning all patients undergoing carotid revascularization at our institution for recently symptomatic (<6 months) carotid plaque.
Patients and lesion assessment
To be eligible for this study, symptomatic-recurrent patients had to have a recurrent transient ischemic attack (TIA), amaurosis fugax, or minor nondisability stroke in the distribution of the study artery despite OMT and had to have carotid artery stenosis ⩽50%. After their recurrent cerebral ischemic event with plaque instability they entered the study and underwent CAS with proximal cerebral protection device. All patients with low-grade carotid stenosis were checked by a cardiologist to rule out any cardiac source of embolization. A vascular neurologist attributed all cerebral or retinal ischemic events to embolization from an ipsilateral carotid stenosis.
We excluded patients when cardiogenic or aortogenic embolism remained the possible cause in accordance with assessment by both cardiologist and neurologists.
Carotid plaque morphology and characteristics were analyzed by both carotid US (Philips HD11XE US machine, Philips, Amsterdam, The Netherlands) and MRI. Patients were imaged using 1.5T MR scanners (Signa Horizon EchoSpeed; General Electric Health Care, Milwaukee, Wi, USA) and phased-array surface coils (Pathway MRI, Inc., Seattle, USA). Fast spin-echo-based T1-weighted, proton density-weighted, and T2-weighted images as well as time-of-flight images of bilateral carotid arteries were obtained using a standardized protocol. 19 The scan was centered on the bifurcation of the operative side. In most cases, this scan covered the complete atherosclerotic plaque. Average scan time was 35 to 45 min. All images were obtained using a field of view of 13 to 16 cm, matrix size of 256, slice thickness of 2 mm and two signal averages. A zero-filled Fourier transform was used to reduce pixel size (0.25 × 0.25 or 0.31 × 0.31 mm2) and to minimize partial-volume artifacts.
Plaque morphology in terms of echogenicity, was assessed in a modified version of the classification proposed by Gray-Weale and colleagues 20 and graded from 1 to 4 as echolucent, predominantly echolucent, predominantly echogenic, or echogenic. 19 Unstable plaque was defined on carotid US as echolucent or heterogeneous plaque, with surface irregularities, ulcer, or rupture. Plaque inflammation, adventitial vasa vasorum, intimal angiogenesis, and plaque neovascularization were identified on US as potential indicators of atheroma instability.
Atherosclerotic plaque associated with intraplaque hemorrhage was diagnosed when axial T1-weighred 3-dimensional gradient-echo black-blood MRIs revealed high signals relative to the sternocleidomastoid muscle.21,22 Using axial MRIs of areas with the highest rate of stenosis, we measured relative overall plaque myocardial infarction (MI) signal intensity and compared it with plaque MRI signals as described.23,24
All patients underwent additional non-invasive imaging to exclude tandem lesions and to confirm the decision to perform CAS.
We excluded patients with low-grade carotid stenosis with stable plaques.
We examined the implementation of CAS with proximal cerebral protection device when patients presented with ⩾2 recurrences despite OMT consisting in single-or dual-antithrombotic therapy and statin treatment (Atorvastatin 80 mg), had no other general problems, and consented to endovascular intervention.
Device and procedure description
Activated clotting time was monitored during the procedure and maintained at greater than 250 sec throughout the intervention. The procedure was performed under general anesthesia as usual at our institution for patient comfort and for control by anesthetist to mitigate potential severe bradycardia during stent release.
At the start of the procedure, an 8 to 10 F, 25-cm long, introducer sheath (Terumo, Tokyo, Japan) was inserted in the infrarenal aorta via the common femoral artery. Systemic anticoagulation was obtained with administration of heparin.
Preprocedural angiography of the aortic arch, bilateral CCA, ICA and ECA and intracranial vessels was completed. Angiographic documentation included anteroposterior and lateral plane views of the ipsilateral carotid and cerebral hemispheres. After aortic arch angiography, a selective bilateral carotid artery catheterization was performed using a 5-F JR4 diagnostic catheter advanced over a 0.035-inch soft hydrophilic wire (Standard Glidewire, Terumo).
Once diagnostic angiography was completed, the wire was advanced in one of the ECA distal branches, the diagnostic catheter was advanced in the distal ECA, and then the hydrophilic wire was exchanged for a 300-cm, 0.035-inch stiff wire (Hi-Torque Supracore, Abbott Vascular, Abbott Park, IL, USA).
The Mo.Ma system (Invatec, Roncadelle, Italy) was guided over the stiff wire until the radiopaque marker of the distal balloon was located in the ECA, at approximately 1 cm beyond bifurcation and in proximity to or at the superior thyroidal artery. Then the distal balloon was inflated in the ECA and the proximal balloon in the CCA, thus blocking the antegrade and the retrograde flow across the target vessel. Slow inflation of the ECA and CCA balloons was observed under fluoroscopy until occlusion was indicated by the change in balloon shape from circular to cylindrical. Flow cessation was confirmed by stagnation of 5 cc of slowly injected contrast/saline mix and the establishment of carotid artery back pressure. A 0.014-inch wire was then navigated through the ICA stenosis and self-expanding carotid stent was deployed. None of the lesions were predilated before stent deployment. Open Nitinol stents (Acculink, Abbott Vascular) were used in all patients. Before postdilatation of the stent, every patient was administered 1mg of atropine.
After dilation, at least 60 ml of blood was aspirated and filtered through sieves, checking for visible plaque debris. Blood flow was restored only after three consecutive aspirations free of debris, deflating first the distal balloon and then the proximal balloon. The final angiography included ipsilateral biplane carotid and intracranial views.
All subjects underwent neurological assessments after the procedure to determine if any ischemic hemispheric deficits developed. Hemodynamics, including systemic pressure and carotid artery back pressure, obtained through the Mo.Ma device after the proximal balloon inflation, were continuously monitored during the procedure.
Independent neurological examination [National Institutes of Health Stroke Scale (NIHSS), Barthel Index and Rankin score] was conducted at baseline, prior to discharge, and at 30-day follow up by a neurologist. An US examination of the carotid stent site was performed at 30 days post-procedure. Any adverse events were recorded through 30 days post-procedure, and thereafter up to 4 years. All patients were treated with 100 mg of aspirin daily indefinitely and 75 mg of clopidogrel for 1 month post-procedure.
Follow up
Periprocedural neurologic evaluations were conducted preprocedurally, at 24–48 h postprocedurally, and at 1 month, 3, 6 months, and annually. Cardiac enzyme levels were obtained for chest pain lasting >15 min. Electrocardiograms were completed preprocedurally, 24–48 h postprocedurally, and for chest pain lasting >15 min.
Brain imaging (diffusion-weighted MRI) was performed in all patients 24–48 h after CAS. Diffusion-weighted MRI was acquired with sagittal T1 sequences, T2-weighted axial, fluid-attenuated inversion recovery axial, and apparent diffusion coefficient map. In addition, T1-weighted axial and sagittal sequences were conducted after contrast administration. Imaging was performed on a 1.5 T Signa HD unit (General Electric, Buckinghamshire, UK). Postoperative diffusion-weighted MRIs were read by two independent neuroradiologists unaware of the patient’s clinical status and blinded to the timing of the paired scans. Ischemic lesions in the cerebral hemispheres, size, and location were recorded by each reader and compared with that of the second reader. Discrepancies were reviewed and reconciled.
Other complications and events observed during the follow up were recorded in accordance with the guidelines of the Ad Hoc Committee on Reporting Standards for Cerebrovascular Disease, Society for the Vascular Surgery/North American Chapter of the International Society of Cardiovascular Surgery. 25
Endpoints
The primary endpoint was the composite rate of all major adverse events, including death, stroke, and MI, through a 30-day follow up or thereafter up to 4 years. Stroke was defined as a new focal neurological deficit of presumed vascular origin persisting more than 24 h with a neuroimaging study not indicative of a different etiology. A major stroke was a stroke causing a deficit that was present after 7 days and increased the patient’s NIHSS by at least four points. A minor stroke caused a deficit that lasted >24 h, but did not meet the definition of major stroke. A TIA was a focal ischemic neurological deficit of abrupt onset that resolved in less than 24 h. MI was defined as biomarker elevation plus either the occurrence of chest pain or equivalent symptoms consistent with MI or electrocardiogram evidence of ischemia including new ST-segment depression or elevation >1 mm in ⩾2 contiguous leads.
Statistical methods/analysis
Data were reported with standard descriptive statistics. We used Kaplan–Meier survival analysis to estimate long-term mortality, stroke, and MI incidence. For the purposes of this latter analysis, we considered the periprocedural stroke event, which by definition occurred within 30 days of the procedure, as if it had occurred at time 0.
For assessment of agreement of US with MRI in identifying relevant plaque characteristics for dichotomous measures we used percent agreement and Κ statistics. Reliabilities and Κ statistics below 0.4 were characterized as poor agreement, 0.4 to 0.75 as fair to good, and above 0.75 as excellent. 26 Reliability was estimated for the entire sample.
Ethics
All patients included in this study provided specific written informed consent for inclusion in the study. This study was performed in compliance with the Declaration of Helsinki and was approved by the local ethics committee of Getafe University Hospital (P103/2009).
Results
Between January 2010 and December 2012, 322 consecutive recently symptomatic patients undergoing carotid revascularization were recruited into this prospective cohort. A total of 59 CASs were performed at our institution in 49 patients for symptomatic significant carotid lesion based on the North American Symptomatic Carotid Endarterectomy trial (NASCET) 1 and American Heart Association (AHA) Guidelines 20062 and their subsequent updates until later AHA/American Stroke Association Guidelines 20143 recommendations. Additionally, 21 patients underwent 21 CAS for recurrent symptomatic ⩽50% carotid stenosis despite OMT and formed our study population. None of the CAS procedures considered here were aborted or incomplete, and none of the patients were refused CAS for technical reason emerging during the intervention. The media of the procedure and carotid artery clamping time was 45 ± 2.3 and 5.96 ± 0.74 min, respectively.
The preoperative characteristics of the patients are summarized in Table 1.
Patients demographics, risk factor, clinical presentations, and perioperative medical treatment.
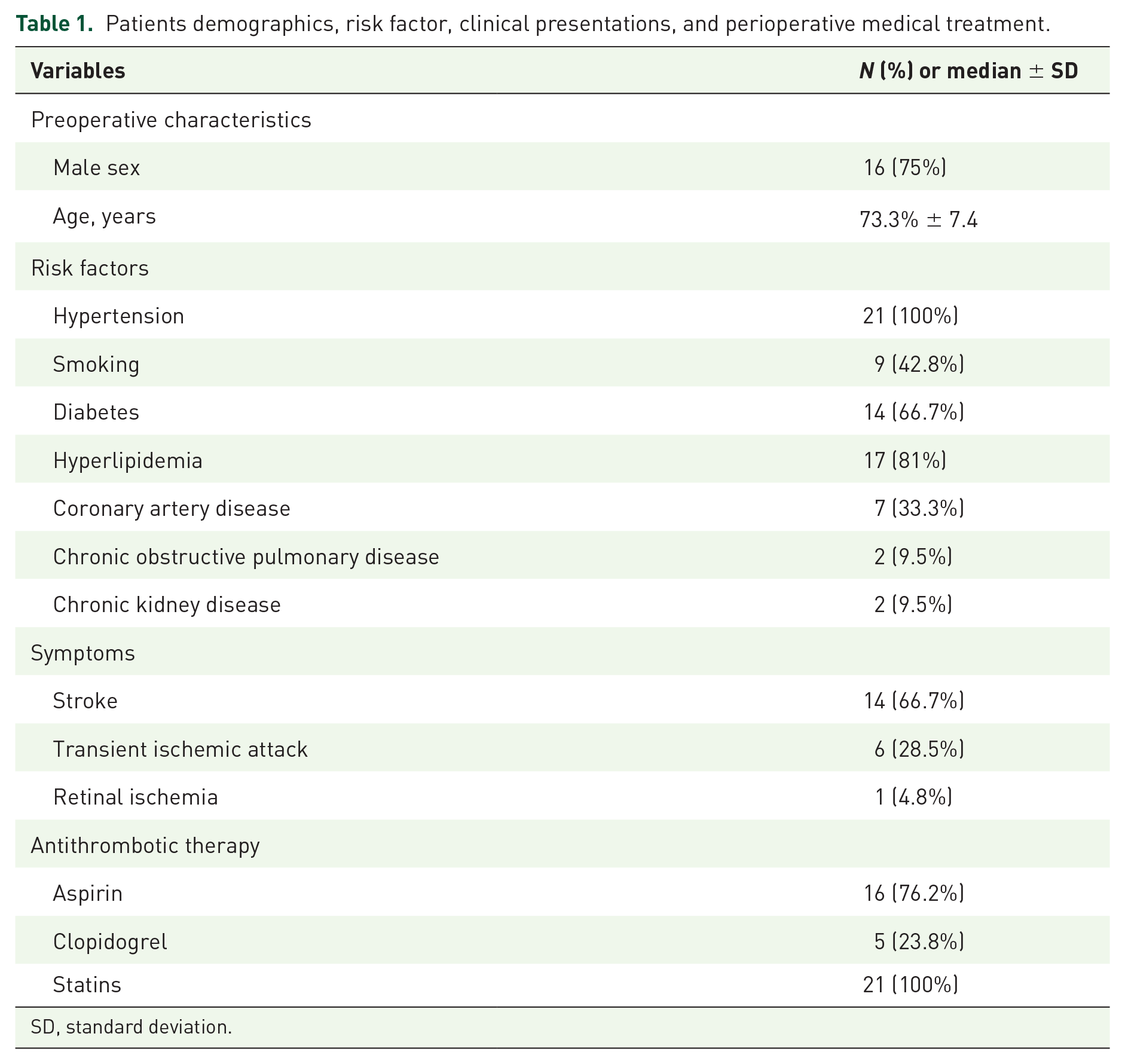
SD, standard deviation.
Perioperative (30 day) results
No perioperative strokes, deaths, cerebral hemorrhages, or TIA ipsilateral to the revascularized hemisphere occurred in this series. No perioperative local complications associated to the procedure were reported.
Long-term outcomes
Long-term follow up was obtained in all patients [median, 26 months, interquartile range (IQR) 17–37, mean 30.3 ± 30.1; range 6–78 months]. No patients had any recurrent neurologic event relating to the revascularized hemisphere.
No late carotid occlusions or moderate restenosis (between 50% and 70%) were detected. One late death (4.8%), not stroke-related, was reported 15 months after CAS. The patient died of MI (none had coronary disease at the time of CAS or perioperative cardiac event). Kaplan–Maier life-table analysis showed that survival rates were 100% at 1 year [95% confidence interval (CI), 1.14–1.35] with 21 patients at risk and 96% at 3 years (95% CI, 3.6–5.2) with 20 patients at risk.
Plaque characteristics
Definitions and reliability estimates for categorical imaging plaque characteristics are show in Table 2. Agreement of US with MRI in identifying relevant plaque characteristics ranged from fair to good.
Definitions and reliability of agreement between US and MRI in relevant plaque characteristics-categorical variables.

MRI, magnetic resonance imaging; SE, standard error; US, ultrasound.
Discussion
The present study shows that patients with low-grade carotid stenosis developing medically refractory ischemic events in the presence of unstable plaque, identifiable using non-invasive diagnostic tools, may be treated with CAS with proximal carotid occlusion cerebral protection demonstrating be a well-tolerated, effective, and durable procedure in such cases. No major ischemic events or deaths occurred perioperatively, and no patients had recurrent cerebrovascular events related to the revascularized hemisphere or died of stroke during the follow up. No late carotid occlusions or moderate restenosis were detected. Nevertheless, on the basis of current guidelines, 27 which focus mainly on the severity of carotid lumen narrowing, carotid revascularization would not be recommended in such symptomatic patients due to the minimal balance between benefits and risk of CEA.1–3,6
Patients with low-grade but symptomatic carotid stenosis represent a challenge to the clinician.27–29 Overwhelming evidence from trials does not support CEA to treat such patients 6 and favor medical management as the best treatment modality.2,3 However, high-risk carotid plaques have been observed in patients with low-grade carotid stenosis who may present with recurrent symptoms.12,30,31 Surgical options may be considered when patients have ischemic symptoms from carotid plaque despite OMT, presumably from serial episodes of plaque embolism. 7 The patients described in this series were selected from treatment owing to recurrent ipsilateral neurological symptoms due to plaque embolism or the presence of high-risk carotid plaque features,12,13,32 in the absence of an evident cardioembolic source of these events. All the patients had received OMT, which had failed to resolve their symptoms.
Evaluating the stroke risk by the degree of stenosis alone may not suffice; additional parameters are needed to better identify subgroups of high-risk patients who would benefit more from revascularization.7,28,30 Plaque morphology, regardless of lumen narrowing, seems to influence the likelihood of adverse ischemic events as well as being a valid predictor of ipsilateral stroke, the unstable plaque being an intriguing entity that has been inconclusively studied with contradictory results.8–11
Given the imperfect correlation between the severity of carotid stenosis and ischemic brain events, identification of a vulnerable carotid plaque may lead some to opt for CAS rather than medical therapy, or even more for CEA rather than CAS, or even for a reversal of flow neuroprotection device rather than a filter wire which involves traversing the lesion. However, studies evaluating the modification of the therapeutic strategy according to the instability of the carotid plaque in low-grade lesion, as assessed by the aforementioned techniques, are lacking in the literature.
US evaluation combined with non-invasive cerebral imaging studies has proved valuable for characterizing plaque and assessing stroke risk.14–16,19 All of our patients’ low-grade carotid plaques showed signs of instability.
The theoretical benefit of CAS in the setting of a symptomatic plaque with low-grade stenosis is reconstruction of the endoluminal surface and restoration of laminar flow. 33 The perioperative risk of CAS for low-grade carotid stenosis is theoretically low, as the usual intraoperative problems associated with high-grade stenosis, such as plaque embolization and perioperative MI, are not encountered. This is due to a relative patent carotid artery at site of stenosis, allowing uncomplicated passage of stenting devices and not typically requiring balloon angioplasty. As a result, plaque emboli are minimized during placement of the stent, and limited barotrauma to the carotid bulb theoretically minimizes the risk of perioperative MI.
Despite the favorable outcomes of our study, carotid stent thrombosis is a potential detrimental, although very rare, complication that needs to be considered in the decision to perform CAS in such patients. Moulakakis and colleagues reported an incidence of 0.5–0.8% that can result in thromboembolic events and affect the immediate survival in case of no-prompt restoration of flow. 34
Proximal embolic protection offers obvious benefits, as no device needs to cross the region of stenosis before cerebral protection. The Mo.Ma device provides embolic protection by endovascular balloon-occluding both the ECA and CCA leading to a cessation of flow in the target vessel. 35 Favorable results of Mo.Ma have been reported in nonrandomized studies and single-center experiences36,37 with 30-day stroke, MI or death rates of 2.7% (30-day major stroke rate of 0.9%).
Reimers and colleagues’ multicenter study including 157 patients reported a 30-day stroke rate and mortality of 5.7%. 36 Similarly, data from the Proximal Protection with the Mo.Ma Device During Carotid Stenting study including 262 patients who underwent CAS under proximal protection with the Mo.Ma device demonstrated a 30-day major stroke rate of 0.9% and major adverse cardiac or cerebrovascular event rate of 2.7%. 38
Ballota and colleagues 29 reported their series involving 57 consecutive patients undergoing CEA for symptomatic low-grade carotid disease over 5 years. Carotid plaque morphology and characteristics were analyzed using contrast-enhanced carotid ultrasonography and additional non-invasive imaging, including MRI angiography.
Patients with low-grade stenosis who suffer recurrent TIA/minor strokes in the early time period after the index symptom (whilst on OMT) will be highly likely to have acutely disrupted plaque with overlying thrombus.10,11 These patients may also have evidence of antiplatelet resistance. 17 Accordingly, a small but highly selected group of patients might benefit from carotid revascularization. To date, there are no externally validated algorithms for identifying patients with ‘high-risk for stroke’ ⩽50% stenosis who would benefit from CEA/CAS. 27
Our study has several limitations. First, although the analysis was prospective, the sample was relatively small and concerned a single institution. Second, we are aware that the absence of perioperative and late major or minor neurologic complications or stroke-related deaths among our patients might denote relative lack of power, limiting the reliability of the results. Finally, the possibility of incurrence in a selection bias and the lack of a comparator group limit the soundness of our results.
In conclusion, this study shows that CAS is a well-tolerated, effective and durable treatment for patients with recurrent symptomatic low-grade carotid stenosis associated with vulnerable plaque. Patients had excellent protection against further ischemic events and survived long enough.
Footnotes
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of interest statement
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
