Abstract
The most common complication to chronic liver failure is ascites. The formation of ascites in the cirrhotic patient is caused by a complex chain of pathophysiological events involving portal hypertension and progressive vascular dysfunction. Since ascites formation represents a hallmark in the natural history of chronic liver failure it predicts a poor outcome with a 50% mortality rate within 3 years. Patients with ascites are at high risk of developing complications such as spontaneous bacterial peritonitis, hyponatremia and progressive renal impairment. Adequate management of cirrhotic ascites and its complications betters quality of life and increases survival. This paper summarizes the pathophysiology behind cirrhotic ascites and the diagnostic approaches, as well as outlining the current treatment options. Despite improved medical treatment of ascites, liver transplantation remains the ultimate treatment and early referral of the patient to a highly specialized hepatology unit should always be considered.
Keywords
Introduction
Ascites is defined as accumulation of more than 25 ml of fluid in the peritoneal cavity. In Western countries, development of ascites is in 75% of cases due to underlying cirrhosis [European Association for the Study of the Lever, 2010], but other less common etiologies of ascites such as malignancy, congestive heart failure, Budd Chiari syndrome, tuberculosis and pancreatitis should be considered – especially if ascites is the first presenting symptom.
Ascites is one of the most frequent complications to cirrhosis occurring in approximately 60% of patients within 10 years of diagnosis [Gines et al. 1987a]. The development of ascites in the setting of cirrhosis represents a landmark in the natural history of cirrhosis, predicting a poor prognosis with 50% mortality within 3 years [Fernandez-Esparrach et al. 2001; Guevara et al. 2005]. Consequently, occurrence of ascites signifies the need to consider referral for liver transplantation, which remains the ultimate treatment option of cirrhosis. Ascites formation often develops in cirrhotic patients presenting with acute-on-chronic liver failure (ACLF), which is acute worsening of liver function due to a precipitating event, e.g. infection, upper gastrointestinal bleeding, electrolyte disturbances [Angeli et al. 2014].
Portal hypertension is a prerequisite for development of cirrhotic ascites [Ripoll et al. 2007]. Survival of cirrhosis depends mainly on the degree of portal hypertension, the degree of liver insufficiency and the degree of circulatory dysfunction.
The principles behind treatment of ascites include diuretics, paracentesis, insertion of a transjugular intrahepatic portosystemic shunt (TIPS), as well as managing complications to ascites such as spontaneous bacterial peritonitis (SBP). SBP occurs in approximately 25% of patients due to bacterial translocation [Wiest and Garcia-Tsao, 2005; Wiest et al. 2014], which is crossing of gut bacteria or bacterial products from the gut lumen to the blood (and ascitic fluid). Bacterial translocation happens as a result of altered intestinal motility, intestinal bacterial overgrowth, increased intestinal permeability and impaired immune defense mechanisms in the cirrhotic patient [Guarner and Soriano, 2005].
Since ascites signifies a deterioration of renal and circulatory function, these patients are also at high risk of developing severe hyponatremia and hepatorenal syndrome (HRS), which together with SBP represent significant clinical and treatment challenges. HRS and SBP further aggravate the risk of a poor outcome [Gines et al. 1993; Garcia-Tsao, 2001; Gines and Schrier, 2009].
There is a strong rationale for an active approach in the management of cirrhotic ascites as a successful treatment may improve symptoms and outcome.
Pathogenesis of ascites formation in the cirrhotic patient
The pathophysiology behind formation of ascites is complex but three key factors are involved: portal hypertension; splanchnic and peripheral arterial vasodilation; and neurohumoral activation [Bernardi and Caraceni, 2001; Gentilini et al. 2002].
Cirrhotic ascites primarily develops due to impaired renal sodium excretion leading to a positive sodium balance and hence water retention, causing expansion of the extracellular fluid volume. The decreased sodium excretion is predominantly caused by arterial vasodilation, which triggers neurohumoral responses such as the renin–angiotensin–aldosterone system (RAAS) and the sympathetic nervous system (SNS) – responses that cause renal vasoconstriction and sodium retention, and hence development of ascites and edema [Dudley, 1992; Gines et al. 1997].
Role of portal hypertension
Ascites only develops if moderate portal hypertension is present. Thus, ascites rarely develops if the post sinusoidal pressure gradient is below 12 mmHg [Casado et al. 1998]. Portal hypertension leads to increased hydrostatic pressure within the hepatic sinusoids, causing transudation of fluid into the peritoneal cavity [Kravetz et al. 2000; Henriksen et al. 2005].
Accordingly, the amount of ascitic fluid produced is determined by the magnitude of hydrostatic pressure. Oncotic dynamics (plasma albumin concentration) play only a minor role, if any role, in the rate of ascites formation [Henriksen et al. 2001; Moore et al. 2006].
Portal hypertension occurs as a consequence of structural changes within the liver in cirrhosis. The degree of portal hypertension is determined by the degree of hepatic vascular resistance and the portal venous inflow. The hepatic vascular resistance is determined by both structural changes such as, for example, fibrosis and regeneration nodules and dynamic changes [Bosch et al. 2003]. The dynamics of the hepatic hemodynamics relate partly to contractile properties of hepatic stellate cells and myofibroblasts [Groszmann et al. 2005; Schuppan and Kim, 2013]. These cells may dynamically regulate sinusoidal tone, and hence portal pressure and flow [Rockey, 2001, 2006]. Moreover, data suggest that defective nitric oxide (NO) production and circulation of endogenous vasoconstrictors such as endothelin-1, angiotensin-II, catecholamines and leukotrienes could further aggravate hepatic vascular resistance [Bosch et al. 2003; Møller and Henriksen, 2005; Iwakiri and Groszmann, 2006].
Role of arterial vasodilation and neurohumoral response
One of the crucial events in the pathogenesis of renal dysfunction and sodium retention is the development of arterial vasodilation. However, the pathophysiological link between portal hypertension and arterial vasodilation still remains to be fully answered. Overproduction of vasodilators due to augmented sheer stress in the splanchnic circulation, defective contractile signaling in smooth muscle cells in response to vasoconstrictor stimulation (also termed ‘vascular hypocontractility’) and neurohumoral signals from the liver to the brain are currently the predominant theories [Ferguson et al. 2006; Iwakiri and Grozmann, 2007; Hennenberg et al. 2008].
The systemic and splanchnic vasodilation seen in cirrhotic patients leads to reduced systemic vascular resistance, decreased effective blood volume, and hence decreased arterial blood pressure.
In order to maintain blood pressure during systemic vasodilation, activation of effective sodium retention and vasoconstricting systems such as the RAAS and SNS are initiated. Nonosmotic release of vasopressin is also commenced [Bernardi and Caraceni, 2001; Møller and Henriksen, 2005; Liu et al. 2008].
See Figure 1 for schematic illustration of the pathogenesis behind ascites.
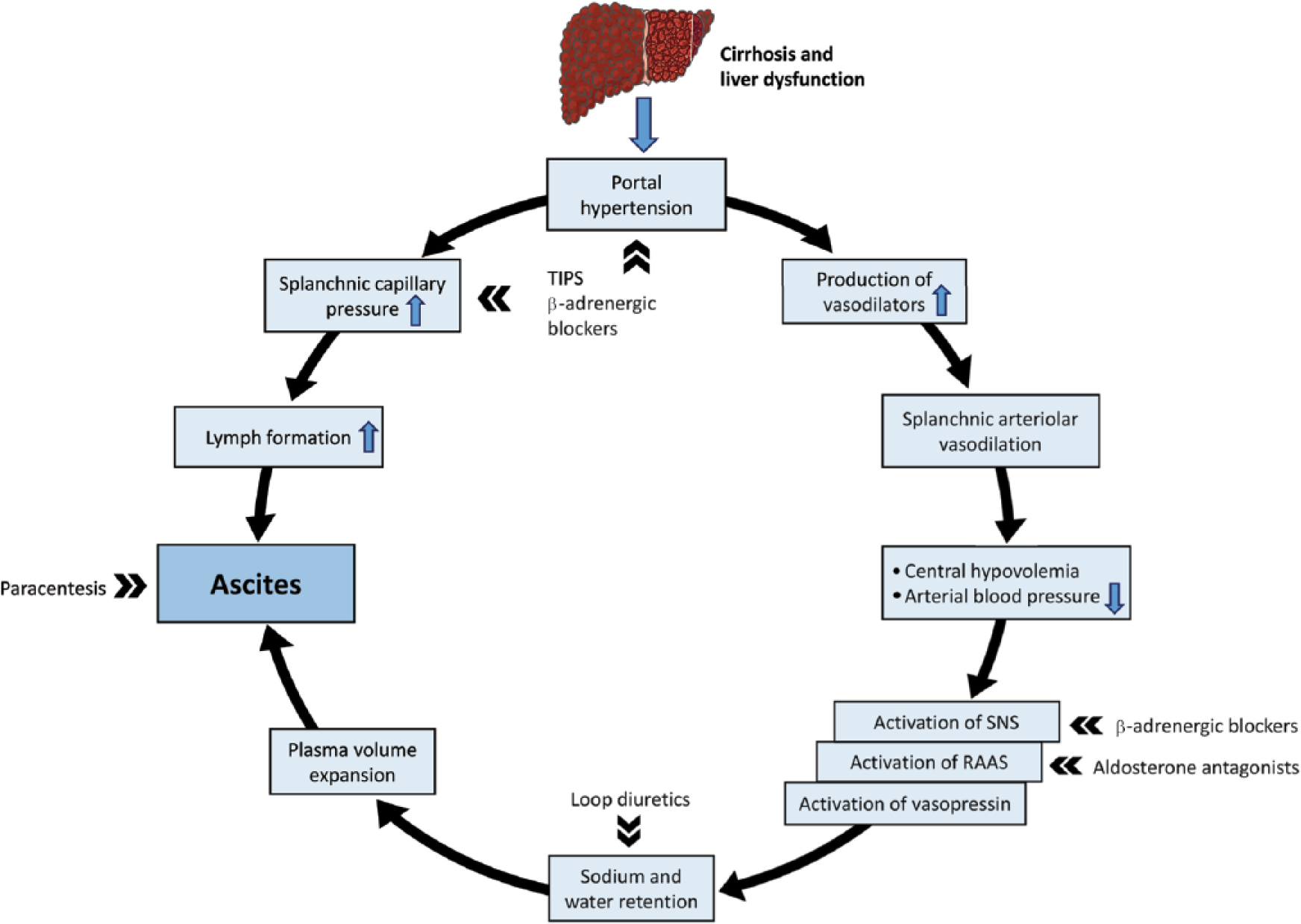
Pathogenesis of ascites formation in the cirrhotic patient.
The above-mentioned mechanisms cause hemodynamic changes and result in a hyperdynamic circulation with an increased heart rate and increased cardiac output.
Renal dysfunction and HRS
Another consequence of the activation of RAAS and SNS is renal vasoconstriction, leading to decreased renal blood flow diminishing the glomerular filtration rate and hence to a progressive impairment of renal function [Dagher and Moore, 2001; Stadlbauer et al. 2008; Gines and Schrier, 2009].
Even in very early stages of cirrhotic portal hypertension, the capacity of renal sodium excretion is compromised and these early signs of renal dysfunction are seen before the development of ascites [Bernardi et al. 1992; Sansoe et al. 1999].
In cirrhotic patients, the free water clearance is dramatically reduced – often below 1 ml/min equivalent to intake of only 1.5 liters of fluid/day before fluid accumulation arises, causing a dilutional hyponatremia (serum sodium <130 mmol/l) [Angeli et al. 2006]. Hypervolemic hyponatremia is associated with decreased survival [Luca et al. 2007; Kim et al. 2008]. The clinical use of vaptans, drugs that cause a selective blockade of V2 receptors and increase water excretion, is currently not routinely recommended in cirrhotic patients. Although successful in increasing serum sodium levels, questions regarding their safety and use in cirrhotic patients have been raised [Dahl et al. 2012].
With more advanced stages of cirrhosis, progressively increasing sodium and water reabsorption and decreasing renal blood flow and glomerular filtration rate (GFR) often happen in parallel with progression of the liver insufficiency [Wensing et al. 1997]. The progressive fall in renal blood flow and GFR may lead to the development of HRS [Arroyo et al. 2007]. Approximately 20% of cirrhotic patients with refractory ascites progress to HRS. HRS develops in the response to severe liver – and systemic circulatory – dysfunction.
HRS is defined as a functional prerenal failure, unresponsive to volume expansion in the setting of chronic liver disease with ascites. In HRS there are no significant morphological changes in renal histology and these patients display a largely normal tubular function [Gines and Schrier, 2009; European Association for the Study of the Liver, 2010]. There are two types of HRS. Type 1 HRS is a rapidly progressing acute renal failure often precipitated by triggering factors such as SBP, septic insult or variceal bleeding causing a swift deterioration of liver and circulatory function and hence HRS [Follo et al. 1994; Cardenas et al. 2001; Ruiz-Del-Arbol et al. 2005; Krag et al. 2010]. Type 2 HRS is a more chronic form with a more stable renal dysfunction [Arroyo et al. 1996; Gines et al. 2003].
The diagnostic criteria for HRS are illustrated in Box 1 [Salerno et al. 2007b].

Diagnosis of ascites
Suspicion of clinical ascites should be confirmed with abdominal ultrasound. Furthermore, the initial evaluation of a patient with ascites should include medical history, physical examination, and laboratory assessment including electrolytes and liver and renal biochemistry.
Based on these findings the underlying cause of ascites is often obvious. However, if it is the first time the patient presents with ascites, more extensive analysis of the ascitic fluid should be carried out with a view to excluding other causes for ascites formation than cirrhosis and excluding complications such as SBP.
Clinical practice includes determination of the serum albumin ascites gradient (SAAG) (serum albumin concentration–ascites albumin concentration), which can help differentiate between ascites ascribed to portal hypertension and ascites due to other causes. If the SAAG is ⩾1.1 g/dl (or 11 g/l), ascites is most likely due to portal hypertension [Mauer and Manzione, 1988, Runyon et al. 1992].
In general, analysis of the ascitic fluid should always be carried out either by diagnostics paracentesis or, if tense ascites is present, in combination with therapeutic large volume paracentesis (LVP). All patients should be screened for the presence of SBP and ascitic fluid investigations should at least include neutrophil cell count, albumin/protein concentration and ascitic fluid inoculation in blood culture bottles.
An ascitic neutrophil cell count of 250 cells/mm3 (0.25 × 109/l) is diagnostic of SBP and is found in approximately 15% of all patients with cirrhotic ascites admitted to hospital [Caly and Strauss, 1993; Tandon and Garcia-Tsao, 2008].
An ascitic albumin concentration <15 g/l in patients with no prior SBP episodes is associated with an increased risk of development of SBP and these patients should therefore receive prophylactic antibiotics to decrease the risk of SBP episodes [Rimola et al. 2000; European Association for the Study of the Liver, 2010].
If clinical suspicion to other underlying causes of ascites, cytological examination (cancer suspicion), ascitic and blood amylase concentration (pancreatic disease), polymerase chain reaction (PCR) and culture for mycobacteria (tuberculosis) should be carried out.
Treatment of uncomplicated ascites
Patients with cirrhotic ascites often present with concomitant complications such as SBP, hyponatremia and HRS. Patients without these complications are termed as having ‘uncomplicated ascites’ [Salerno et al. 1999].
There is no evidence for treatment of patients with mild ascites (grade 1). Patients with grade 2 ascites do often not acquire hospitalization and can be treated in the outpatient clinic [European Association for the Study of the Liver, 2010].Grading of ascites and treatment overall strategy is depicted in Table 1.
Grading of ascites and treatment strategy [European Association for the Study of the Liver, 2010].

Dietary salt restriction, restriction of fluid intake
Generally patients should be advised to follow a low sodium intake diet to create a negative sodium balance and hence increased mobilization of the fluid retention. From a clinical point of view, restriction of daily sodium intake to 80–120 mmol (corresponding to 4.6–6.9 grams of salt/day) is possible through a ‘no added salt diet’ and avoidance of preprepared foodstuffs. A more rigorous salt restriction is often intolerable to the patients and also may further harm the poor nutritional status often seen in these patients.
Restriction of fluid intake is generally considered an obsolete treatment possibility in patients with uncomplicated ascites. However, water restriction in patients with ascites and hyponatremia has become standard clinical practice in many centers, although controversy remains as to what is the best treatment of these patients. Fluid intake can rarely be restricted to <1 l/day, which is insufficient to cause fluid loss [Gines et al. 2008] and furthermore the efficacy of water restriction may depend on the level of hyponatremia [Moore and Aithal, 2006]. Thus, more studies are needed to find the best approach and, until then, water restriction should only be used in few, if any, patients.
Diuretics
Diuretics remain the standard treatment of cirrhotic ascites. The effect of the diuretics should in the beginning of treatment be assessed with daily control of bodyweight and close monitoring of creatinine and electrolytes to avoid serious electrolyte disturbances and to reduce the risk of diuretic-induced renal failure.
The diuretic treatment should always be initiated with the administration of spirono-lactone, an aldosterone receptor antagonist. Spironolactone counteracts the effects of the high circulating levels of aldosterone, facilitating the increased reabsorption of sodium in the distal tubules of the kidneys. Thus, spironolactone accelerates the natriuresis and is more effective than loop diuretics in patients with cirrhotic ascites [Eggert, 1970; Perez-Ayuso et al. 1983]. The initial dose should start at 100 mg/day gradually increasing with 100 mg/week until adequate natriuresis is achieved. The effect of spironolactone is seen after 3–5 days of treatment and the maximum recommended dose of spironolactone is 400 mg/day.
Patients with a first episode of grade 2 ascites should initially only be treated with an aldosterone antagonist [Santos et al. 2003; Bernardi, 2010]. However, it is often necessary to add loop diuretics (e.g. furosemide) to counteract the potassium sparing effects of aldosterone antagonists resulting in hyperkalemia.
Also, in case of an insufficient response (defined by reduction in bodyweight of <2 kg/week) to spironolactone alone, loop diuretics should be added in a stepwise manner every 2–3 days to a maximum of 160 mg/day.
In patients with recurrent ascites, studies have concluded that the combination of an aldosterone antagonist and loop diuretics is the preferred regimen since the combination therapy increases the natriuretic effect [Angeli et al. 2010; Bernardi, 2010].
Furosemide alone is not recommended since efficacy in cirrhotic patients is low [Perez-Ayuso et al. 1983].
To avoid intravascular volume depletion and risk of renal impairment, hepatic encephalopathy and hyponatremia, diuretic dosage should be adjusted to a daily weight loss of no more than 500 g/day in patients without peripheral edema and 1 kg/day in patients with peripheral edema [Shear et al. 1970; Pockros and Reynolds, 1986; Gines et al. 2004].
After mobilization of ascites, the least effective dosage of diuretics should be sought to reduce the risk of diuretic-induced complications and to minimize side effects. Furthermore, total alcohol abstinence is a key element in avoidance of recurrence of ascites in alcoholic cirrhosis.
Patients with ascites that cannot be mobilized despite intensive diuretic treatment and sodium restriction diet are termed ‘diuretic resistant’. Approximately, 10% become refractory to diuretic treatment and other treatment modalities should be considered [Moore and Aithal, 2006]. Diuretic intractable ascites is characterized by diuretic-induced complications such as encephalopathy and hyponatremia that preclude the use of an effective diuretic dosage. See Table 2 for the definition and diagnostic criteria for diuretic-resistant ascites and diuretic intractable ascites.
Definition and diagnostics criteria for refractory ascites in cirrhosis [Salerno et al. 2010].

Therapeutic paracentesis
In patients with large grade 3 ascites or refractory ascites, LVP rapidly removes tense ascites and is the treatment of choice. A patient with tense ascites is seen in Figure 2. LVP should be completed in a single session, with the needle preferably inserted in the left iliac fossa under strict sterile conditions. LVP can often be managed in the outpatient clinic.

Patient with tense ascites.
Several controlled clinical studies have demonstrated that LVP with albumin substitution is rapid, safe and effective [Quintero et al. 1985; Gines et al. 1987b; Salerno et al. 1987]. In general LVP is a low-risk procedure and local complications such as hemorrhage (even though coagulopathy is often evident) or perforation of the bowel are low [Pache and Bilodeau, 2005]. Coagulopathy is not a contraindication for LVP and there are no data supporting the use of prophylactic fresh frozen plasma and platelets before or during LVP.
LVP >5 l should always be followed by intravenous administration of human albumin (HA) to reduce the risk of development of postparacentesis circulatory dysfunction (PPCD) due to rapid decrease in effective arterial blood volume [Gines et al. 1988; Pozzi et al. 1994]. PPCD can cause rapid reappearance of ascites, renal failure and HRS, dilutional hyponatremia and increased mortality risk [Gines et al. 1988, 1996; Sola et al. 1994]. To prevent development of PPCD, 20% HA infusion is recommended after paracentesis at a dose of 8 g/l ascites removed [Runyon, 2009; European Association for the Study of the Liver, 2010]. HA is generally preferred over other plasma expanders, since it is more effective when more than 5 l of ascites is removed [Gines et al. 1996].
However, paracentesis does not address the underlying cause of ascites (renal sodium and water reabsorption) and ascites will re-occur in 93% of patients if treatment with diuretics is not reinstituted [Fernandez-Esparrach et al. 1997].
Treatment of refractory ascites
Refractory ascites is defined as ‘ascites that cannot be mobilized or the early recurrence of which (i.e. after LVP) cannot be satisfactorily prevented by medical therapy’ [Moore et al. 2003].These patients are primarily treated with repeated LVPs. Once ascites has become refractory to diuretics, the median survival is poor – approximately 50% die within the next 6 months. Emerging data suggest that use of nonselective beta-blockers (prevention of oesophageal variceal bleeding) in cirrhotic patients with refractory ascites is associated with poorer outcome and caution regarding nonselective beta-blocker indication and dosage should probably be considered thoroughly in these patients [Sersté et al. 2010; Krag and Madsen, 2014].
Patients with refractory ascites should in general be considered for liver transplantation.
It is a frequent asked question whether diuretics should be continued in patients with refractory ascites. However, discontinuation or at least reduction of diuretics should happen in all patients with diuretic-induced complications such as severe electrolyte disturbances, renal impairment and development of overt hepatic encephalopathy. In the remaining patients, measuring the urine sodium concentration will determine if the diuretic therapy should be discontinued. If the urinary sodium excretion under diuretic therapy is below 30 mmol/day, the natriuretic effect is not sufficient and diuretics might as well be discontinued [Moore et al. 2003].
TIPS
In case of recurrent ascites and requirement of frequent LVPs (>3/month), insertion of TIPS should be considered. TIPS functions as a side-to-side portocaval anastomosis between the high pressure portal vein side and low pressure hepatic vein side, and effectively decompresses the portal system. The decrease in portal hypertension results in a secondary decrease in the activation of RAAS, leading to an increased sodium excretion [Rossle et al. 1998]. Figure 3 depicts a TIPS.

Transjugular intrahepatic portosystemic shunt. The shunt (stent) is inserted between the vena portae and the hepatic vein.
Compared with repeated LVP, TIPS is considered a more effective treatment option for the control of ascites [Rossle et al. 2000; Gines et al. 2002; Salerno et al. 2007a] and complete resolution of ascites is seen in up to 75% of patients [Ochs et al. 1995].
However, the effect of TIPS on survival in patients with refractory ascites has not been clearly demonstrated. Three meta-analyses have all failed to demonstrate a difference in survival between TIPS and LVP groups [Albillos et al. 2005; Deltenre et al. 2005; Saab et al. 2006]; one meta-analysis showed a trend towards reduced mortality for TIPS patients and one showed increased transplant-free survival in the TIPS groups [D’amico et al. 2005; Salerno et al. 2007a]. Post-TIPS mortality is predicted by the indication for the TIPS procedure; patients with refractory ascites have decreased survival compared with the group of patients with variceal bleeding as the main indication for the TIPS procedure [Angemayr et al. 2003]. In general post-TIPS mortality risk is primarily predicted by high bilirubin levels (>3 mg/dl) [D’Amico et al. 2005; Gerbes et al. 2005] and high MELD score (Model of Endstage Liver Disease) [Angemayr et al. 2003].
An important challenge with the insertion of TIPS is the increased risk of development of hepatic encephalopathy which occurs in 25–30%, the risk increasing with age [Sanyal et al. 1994; Casado et al. 1998]. Previous episodes of hepatic encephalopathy are a contraindication for insertion of TIPS. TIPS can be considered in patients with refractory ascites with >3 LVPs/month, but appropriate risk assessment prior to TIPS is essential.
Treatment of complicated ascites
Complicated ascites is defined as ascites with complications of one or more of the following: SBP, HRS and Hyponatremia. Management of Hyponatremia has previously been mentioned briefly.
SBP
All patients with cirrhosis and ascites are at risk of developing SBP. A routine cell count from the ascitic fluid obtained either at diagnostic or therapeutic paracentesis should always be done to rule out SBP.
Symptoms of SBP include abdominal pain, fever and vomiting. Diagnosis should also be suspected in patients with worsening of liver function, hepatic encephalopathy, renal failure and/or gastrointestinal bleeding but SBP is frequently asymptomatic.
Previously, mortality exceeded 90% but with early diagnosis and current treatment strategies mortality has been reduced to approximately 20% [Garcia-Tsao, 2001; Tandon and Garcia-Tsao, 2008].
Inoculation of ascitic fluid in blood culture bottles often display Escherichia coli and Streptococcus species [Tandon and Garcia-Tsao, 2008], although ascites culture is negative in as many as 60% of patients, despite both clinical signs of SBP and neutrophil cell count >250 cells/µl. These patients should still be treated as having SBP [Rimola et al. 2000].
Treatment of SBP
Since antibiotic resistance is an increasing challenge, ascitic fluid and blood inoculation + assessment regarding susceptibility to antibiotics prior to antibiotic treatment should always be performed. Empiric antibiotic treatment should be started immediately after diagnosis of SBP. Third generation cephalosporins such as cefotaxim cover most causative organisms and are often the treatment of choice [Felisart et al. 1985; Rimola et al. 1995]. Administration of 2 g intravenously twice a day is often preferred; a 5-day therapy is as effective as a 10-day therapy [Runyon et al. 1991]. Alternatively piperacillin/tazobactam could be considered [Novovic et al. 2012].
Patients with SBP and signs of hepatic dysfunction (bilirubin > 66 µmol/l) or signs of renal impairment should further be given HA at 1.5 g albumin/kg in the first 6 hours followed by 1 g/kg on day 3 since HA infusion reduces the risk of HRS [Sort et al. 1999]. Furthermore, the beneficial effect of administering HA to the cirrhotic patient is probably also attributable to the albumin binding capacity of circulating pro-inflammatory and bacterial products such as prostaglandins (PGE2) and lipopolysaccharide. Consequently HA may play an important role as modulator of the systemic and organ related inflammation evident in patients with decompensated cirrhosis [Arroyo et al. 2014; O’Brien et al. 2014].
The risk of re-infection is high and secondary prophylaxis with, for example, oral norfloxacin 400 mg daily, significantly reduces the risk of future SBP episodes [Gines et al. 1990].
Patients with low ascitic albumin concentration <15 g/l and severe liver disease without previous SBP episodes should be considered for primary prophylactic treatment with, for example, norfloxacin 400 mg daily, which reduces risk of SBP and improves survival [Fernandez et al. 2007].
Hepatorenal failure
Type 1 HRS is a rapidly progressing acute renal failure evolving in the setting of a precipitating factor, further augmenting circulatory dysfunction, whereas type 2 HRS is a more slowly progression of renal failure seen in patients with refractory ascites.
Early diagnosis of HRS 1 is important to improve short-term survival, which untreated is of only 1 month. Median survival of type 2 HRS is 6 months [Alessandria et al. 2005]. See Table 1 for diagnostic criteria.
Treatment of hepatorenal failure
Patients with suspicion of HRS 1 should be monitored closely by urine output, fluid balance, arterial blood pressure and vital surveillance. Infections should be treated and diuretics should be discontinued.
The specific treatment of HRS 1 should be aimed at decreasing splanchnic arterial vasodilation, decreasing central hypovolemia and increasing renal blood flow. Administration of vasoconstrictor drugs such as terlipressin, which acts as a vasopressin analogue, stimulates the splanchnic vasopressin V1a receptors. Terlipressin induces constriction of the extremely dilated splancnic vascular bed, thereby improving the central underfilling and increasing the arterial blood pressure [Moreau and Lebrec, 2006; Gines et al. 2007]. Terlipressin can only be administered intravenously and is started at a dose of 1 mg/4–6 hours and increased to a maximum of 2 mg/4 hours, if there is no reduction in serum creatinine of at least 25% on day 3 compared with serum creatinine baseline value. Treatment is effective in 40–50% of patients and is associated with increased short-term survival [Gines and Schrier, 2009; Gluud et al. 2010] Patients receiving terlipressin should be monitored for development of cardiac arrhythmias, acute myocardial infarction, and signs of splanchnic or digital ischemia.
HA should in addition be administered with 1 g/kg the first day followed by 20 g twice a day [Ortega et al. 2002]. Treatment with terlipressin and albumin should be continued until normalization of serum creatinine (below 133 µmol/l) is achieved. If HRS 1 redevelops, treatment can be repeated [European Association for the Study of the Liver, 2010].
Hemodialysis (in combination with terlipressin) is frequently used to control azotemia and electrolyte balance before eventual liver transplantation, which should be considered in all patients with HRS who have no contraindications to the procedure [Runyon, 2009].
Terlipressin does not seem to have a sustained effect in type 2 HRS [Gluud et al. 2012].
Use of albumin dialysis [molecular adsorbent recirculating system (MARS)] in patients with ACLF has failed to show improved survival in randomized controlled trials [Banares et al. 2013].
Despite treatment, the prognosis of HRS remains poor with a median survival of all patients with HRS of only 3 months [Gines and Schrier, 2009].
Perspectives and conclusion
Management of cirrhotic ascites and its complications remain an everyday clinical challenge for hepatologists. Although advances in medical therapy have been made, the development of ascites is still associated with poor prognosis and markedly increased mortality.
Adequate management of ascites is important since this not only improves quality of life in these patients, but also decreases risks of complications such as SBP. Several up-to-date clinical guidelines are available [Runyon, 2009; European Association for the Study of the Liver, 2010].
Future strategies should approach target different aspects of the pathophysiological process resulting in splanchnic arterial vasodilation, central hypovolemia and decreased arterial blood pressure. Development of long-acting systemic vasoconstrictors should be encouraged. Optimization of the treatment strategies will hopefully result in better management of ascites with fewer side effects and complications leading to an improved survival.
All patients with ascites should be considered as liver transplant candidates, since liver transplantation still remains the ultimate treatment for these patients.
Footnotes
Conflict of interest statement
The authors declare no conflict of interest in preparing this article.
Funding
SM has received a grant from the Novo Nordisk Foundation.
