Abstract
Background:
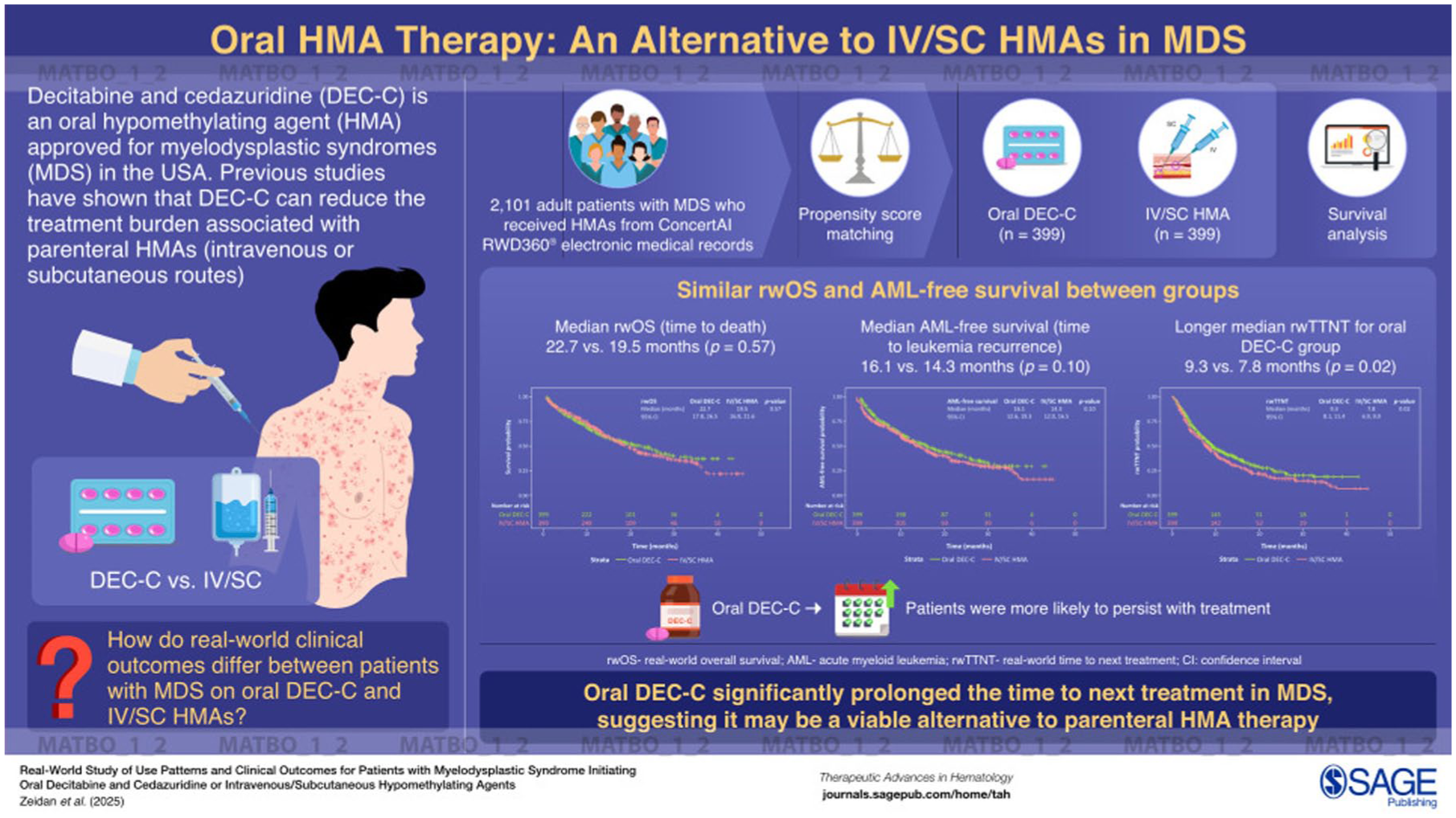
Decitabine and cedazuridine (DEC-C) is the only oral hypomethylating agent (HMA) approved for myelodysplastic syndromes (MDS) in the United States. The other HMAs approved for MDS, decitabine and azacitidine, are administered intravenously (IV) or subcutaneously (SC).
Objectives:
This study examined real-world outcomes among MDS patients treated with oral DEC-C compared to IV/SC HMAs.
Design:
Adult patients diagnosed with MDS who received HMAs as their first treatment on or after July 1, 2020 through February 2024 in ConcertAI’s RWD360® electronic medical records dataset linked to open claims were included in the study.
Methods:
We used propensity score matching (PSM) to balance potential confounders. Kaplan–Meier survival analysis was utilized to compare real-world overall survival (rwOS), acute myeloid leukemia (AML)-free survival, and time to next treatment (rwTTNT) among patients treated with oral DEC-C and IV/SC HMAs.
Results:
A total of 2101 patients with MDS were included, with 405 treated with oral DEC-C and 1696 treated with IV/SC HMA. After PSM, there were 399 patients in each treatment group; demographic and clinical factors were well balanced. Patients treated with oral DEC-C had a similar median rwOS (22.7 months vs 19.5 months; p = 0.57) and AML-free survival (16.1 months vs 14.3 months; p = 0.10) compared with patients who received IV/SC HMA; however, there were nonsignificant trends in favor of oral DEC-C. The median rwTTNT was significantly longer for the oral DEC-C patients than for the IV/SC HMA patients (9.3 months vs 7.8 months, respectively; p = 0.02).
Conclusion:
This real-world study is the largest to date to examine clinical outcomes among MDS patients who initiated oral DEC-C compared to IV/SC HMAs. While study results indicate comparable rwOS and AML-free survival among patients treated with oral DEC-C or with IV/SC HMAs, patients treated with oral DEC-C had a significantly longer rwTTNT. These findings support the use of oral DEC-C as an alternative to parenteral HMA therapy.
Plain language summary
Why was the study done? A new type of chemotherapy drug called a hypomethylating agent (HMA) that is taken by mouth (oral) has been approved for patients with myelodysplastic syndromes (MDS). This drug is decitabine and cedazuridine together (DEC-C). Previous HMA treatments had to be given through a patient’s veins (IV) or under the skin (SC). Data collected from patients being treated in the real-world setting, rather than a clinical trial setting, are needed to see whether oral DEC-C can be considered in place of the IV/SC HMAs.
What did the researchers do? The study team looked at electronic medical records in ConcertAI’s RWD360® dataset of adult patients with MDS. The researchers grouped patients by their first HMA treatment received after MDS diagnosis: oral DEC-C or IV/SC HMA. Then the researchers matched the patients based on characteristics such as age, race, and when they received the initial HMA treatment to make sure the groups were similar.
What did the researchers find? Patients who received oral DEC-C had a similar time to death (median 22.7 months versus 19.5 months) and time to leukemia development (median 16.1 months versus 14.3 months) compared with patients who received IV/SC HMAs. However, patients who received oral DEC-C had a longer time until another treatment was needed compared to patients who received IV/SC HMAs (9.3 months versus 7.8 months).
What do the findings mean? The results of this study support the consideration of oral DEC-C as a treatment option in place of IV/SC HMAs for patients with MDS.
Keywords
Introduction
Myelodysplastic syndromes (MDS) are a diverse and heterogeneous group of hematologic syndromes characterized by clonal proliferation, dysplastic cellular morphology, and cytopenias, including anemia, neutropenia, and thrombocytopenia. The age-adjusted incidence of MDS in the United States was 4.0 per 100,000 annually in 2021, with higher incidence among male, White, and older individuals. 1 The etiology of MDS is poorly understood; potential risk factors include prior treatment with radiotherapy or chemotherapy, obesity, smoking, ionizing radiation, and exposure to heavy metals and organic chemicals. 2
Patients with MDS have an increased risk of progression to acute myeloid leukemia (AML). 3 Survival is variable, with a disease course ranging from slow progression to rapid transformation and death. MDS is characterized by multiple epigenetic abnormalities, including the hypermethylation of multiple genes, 4 and the degree of DNA methylation may be prognostic for survival.5,6 Current treatment options include hypomethylating agents (HMAs), which can reverse methylation.
Two HMAs for MDS, decitabine 7 and azacitidine, 8 are administered intravenously (IV) or subcutaneously (SC). While azacitidine is often considered a standard therapy for MDS, many patients discontinue the treatment prematurely, specifically due to treatment failure and adverse events, leading to unfavorable outcomes. 9 About 25%—30% of patients who are treated with IV/SC HMA discontinue HMA treatment before four cycles of treatment, leading to underutilization of HMA treatment.10,11 The main risk factors associated with discontinuation of IV/SC HMA were older age and poor performance status. 10
Decitabine and cedazuridine (DEC-C) is an oral HMA that was approved in July 2020 in the United States for the treatment of adult patients with MDS, including previously treated and untreated, de novo and secondary MDS with specific subtypes and intermediate 1, intermediate 2, and high-risk International Prognostic Scoring System groups. 12 A patient survey reported that many patients found DEC-C to be a convenient mode of HMA administration (83%) and, among those previously treated with IV/SC HMAs, agreed that oral DEC-C interfered less with their daily lives (91%). 13 In a study utilizing claims data, the persistence of IV/SC HMAs and oral DEC-C were similar for the first 6 months of use, but there was longer use of oral DEC-C beyond 6 months (17% vs 25%, respectively). 14 Along with decreased interference with daily activities and potentially better persistence, oral HMAs may also improve outcomes in patients with MDS. In the ASCERTAIN clinical trial, the median overall survival for patients who received oral DEC-C was 31.8 months. 15
Based on previous studies, DEC-C may be an alternative to parenteral HMAs and may be preferred by patients with MDS in the real-world setting due to convenient dosing and improved persistence. 13 However, there is a lack of real-world evidence comparing the effectiveness of oral DEC-C and IV/SC HMAs. This study aimed to describe patient characteristics and examine real-world clinical outcomes among MDS patients treated with oral DEC-C compared to IV/SC HMAs using the ConcertAI real-world electronic medical records (EMR) database.
Methods
Data source
Patients included in this study were from ConcertAI RWD360®, an oncology EMR dataset linked to open claims. Data in the ConcertAI RWD360 dataset are comprised of geographically diverse practice locations within the United States and are primarily from community-based oncology practices. The structured data available include demographic and clinical characteristics, infused and oral therapies, and outcomes. Death data were obtained from multiple sources, including structured EMR data, the Social Security Death Index, third-party obituary sources, and claims. 16
Study population
Patients were eligible for this study if they had a diagnosis of MDS (based on ICD-10 code D46*), were 18 years or older at MDS diagnosis, and received an HMA (either as monotherapy or in combination therapy) as their initial treatment after MDS diagnosis and had to have initiated the HMA after July 1, 2020. Patients were excluded if they had an AML diagnosis at baseline or had other primary cancers (except nonmelanoma skin cancer), had less than 30 days of follow-up (from the start date of HMA to end of record or death), or participated in an interventional clinical trial at any point after their MDS diagnosis. Details on the number of patients within the population meeting these eligibility criteria are provided in Supplemental Figure 1.
Cohorts were created based on oral DEC-C use or IV/SC HMA use as the first HMA. The index date was defined as the start date of the initial HMA treatment on or after MDS diagnosis, and the type of HMA initiated on the index date was defined as the index HMA. The index HMA episode was defined as the start date of the index HMA drug to the date of the last administration of the index HMA, with no gap of >63 days of the index HMA. The study period was defined as the interval from 12 months before the index date through the end of the patient’s EMR data, data cutoff for the study (February 29, 2024), or death, whichever occurred first.
Variables and statistical methods
Demographic and clinical characteristics
Descriptive statistics were generated for demographic and clinical variables, including means, standard deviations, medians, and minimum and maximum values for continuous variables and frequencies and percentages for categorical variables. To compare these values across the DEC-C and IV/SC HMA treatment groups, significance testing was performed as specified in Table 1. Variables with missing data were reported as “unknown.”
Demographic and clinical characteristics by type of index HMA, pre- and postpropensity score matching.
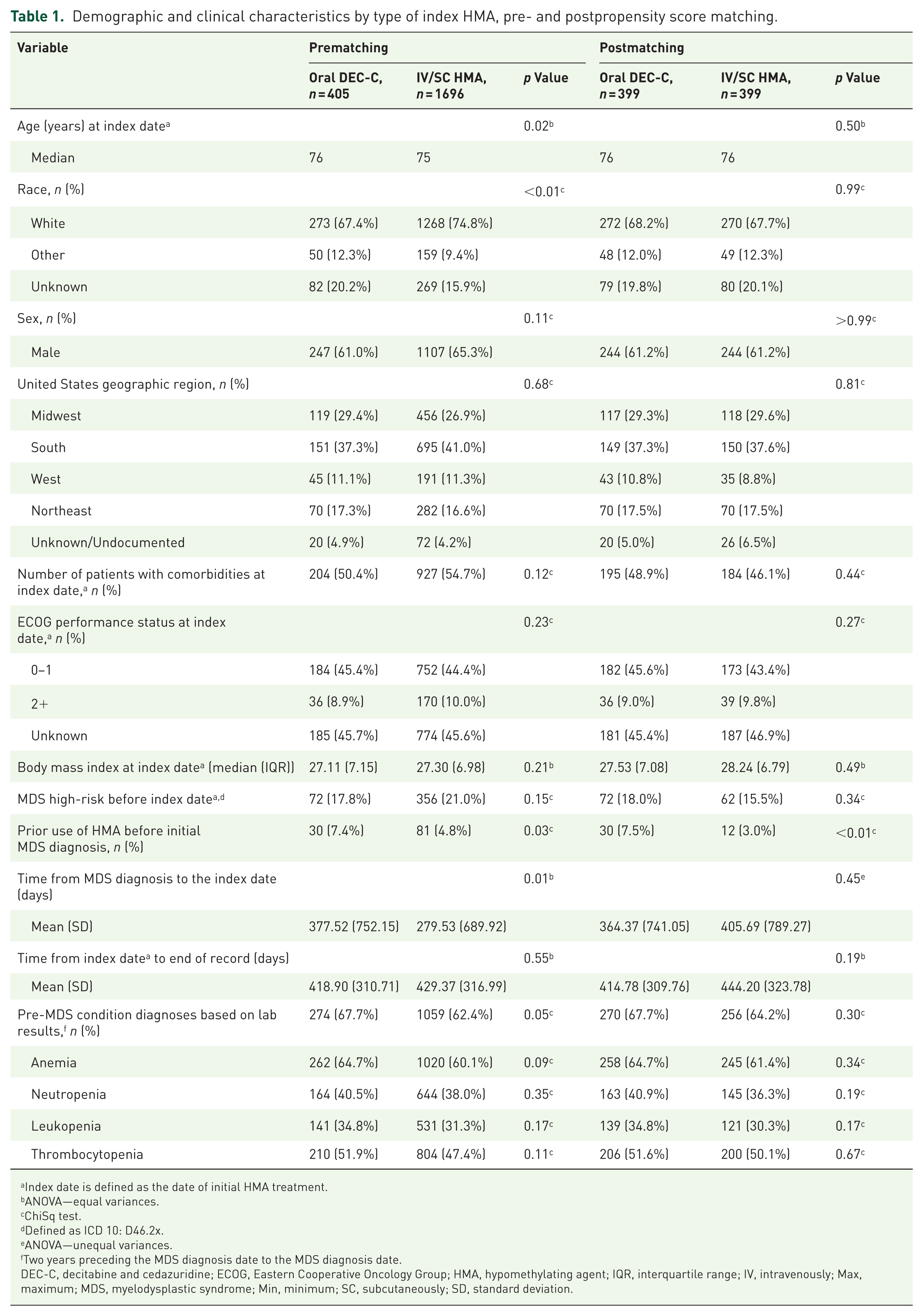
Index date is defined as the date of initial HMA treatment.
ANOVA—equal variances.
ChiSq test.
Defined as ICD 10: D46.2x.
ANOVA—unequal variances.
Two years preceding the MDS diagnosis date to the MDS diagnosis date.
DEC-C, decitabine and cedazuridine; ECOG, Eastern Cooperative Oncology Group; HMA, hypomethylating agent; IQR, interquartile range; IV, intravenously; Max, maximum; MDS, myelodysplastic syndrome; Min, minimum; SC, subcutaneously; SD, standard deviation.
Clinical outcomes analysis
DEC-C and IV/SC HMA patients were compared to evaluate several time-to-event endpoints utilizing Kaplan–Meier analysis. Real-world overall survival (rwOS) was assessed from the index date to the death date, with censoring at the last activity date. Real-world time to AML (AML-free survival) was assessed from the index date to the earlier of AML diagnosis or death, with censoring at the last activity date. Real-world time to next treatment (rwTTNT) was assessed from the index date to the start of the next treatment after discontinuing the HMA or death, with patients censored at the last activity date. Multivariable adjusted Cox proportional hazards models were additionally performed for each time-to-event analysis, comparing DEC-C and IV/SC HMA patients, as detailed in Supplemental Table 1.
Propensity score-matched analysis for clinical outcomes
For a rigorous assessment of the main clinical outcomes, propensity score (PS) matching 17 was performed to address confounding within a Kaplan–Meier analysis. The PS was developed including potential confounders and other covariates. The confounders and covariates are listed in Supplemental Table 2. The DEC-C patients were matched 1:1 with the IV/SC HMA patients based on the PS using a caliper of 0.1 (0.1 times the standard deviation of the PS). Descriptive demographic and clinical characteristics statistics were generated for both cohorts postmatching (Table 1). Using the matched cohorts, DEC-C and IV/SC HMA patients were compared to evaluate the time-to-event endpoints (rwOS, AML-free survival, and rwTTNT) utilizing Kaplan–Meier analysis, as above.
Exploratory analyses
Several exploratory analyses were performed within the full, unmatched cohort. DEC-C patients were compared separately with azacitidine and decitabine patients for rwOS, AML-free survival, and rwTTNT using Kaplan–Meier analysis, as described above.
The number of cycles of the index therapy was assessed for the DEC-C and IV/SC HMA patients. Records with a <14-day gap (treatment start date compared to the previous treatment end date) were considered the same cycle. For records with treatment duration >28 days, the records were further separated into multiple cycles based on 28-day cycle length. Persistence was defined as completing five or more cycles of the index HMA therapy within 140 days of the index date. Patients had to have sufficient follow-up (at least 140 days after the index date) to be included in this analysis. Persistent DEC-C patients were then compared with nonpersistent for rwOS using Kaplan–Meier analysis, as described above.
All analyses were performed using SAS version 9.4 (SAS Institute Inc., Cary, NC, USA) and R version 4.4.1 with a two-sided p value <0.05 considered statistically significant. Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) cohort reporting guidelines were used during the preparation of the manuscript. 18
Results
There were 2101 patients with MDS treated with HMAs as the first treatment post-MDS diagnosis, with 405 treated with oral DEC-C and 1696 treated with IV/SC HMA (Table 1). At the index date, patients treated with oral DEC-C had a median age of 76 years compared with 75 years for patients treated with IV/SC HMA (p = 0.02). Over 65% of patients treated with IV/SC HMA were male compared to 61% of patients treated with oral DEC-C (p = 0.11). The most common US geographic region for both groups was the South (oral DEC-C: 37.3% vs IV/SC HMA: 41.0%; p = 0.68). Patients who received oral DEC-C had a significantly longer time from initial MDS diagnosis to index date compared with patients who received IV/SC HMA (mean 377.5 days vs 279.5 days, respectively; p = 0.01). Patients who received oral DEC-C had a significantly higher use of HMA before the initial MDS diagnosis compared with patients who received IV/SC HMA (7.4% vs 4.8%, respectively; p = 0.03).
There were additional statistically significant differences in baseline characteristics comparing oral DEC-C to IV/SC HMA patients, including race, and diagnosis of a pre-MDS condition (anemia, neutropenia, leukopenia, or thrombocytopenia). After PS matching, there were 399 patients in both groups, and the baseline covariates were well-balanced overall (Table 1). The number of patients with unknown values for baseline covariates are included in Table 1.
Within the overall study, 830 of the 1696 IV/SC HMA patients and 176 of the 405 DEC-C patients died during follow-up. The median rwOS for the overall sample was 19.3 months, and patients who received oral DEC-C had numerically longer median rwOS compared to those treated with IV/SC HMAs (23.2 months vs 19.0 months), but the difference was not statistically significant (p = 0.24; Figure 1(a)).
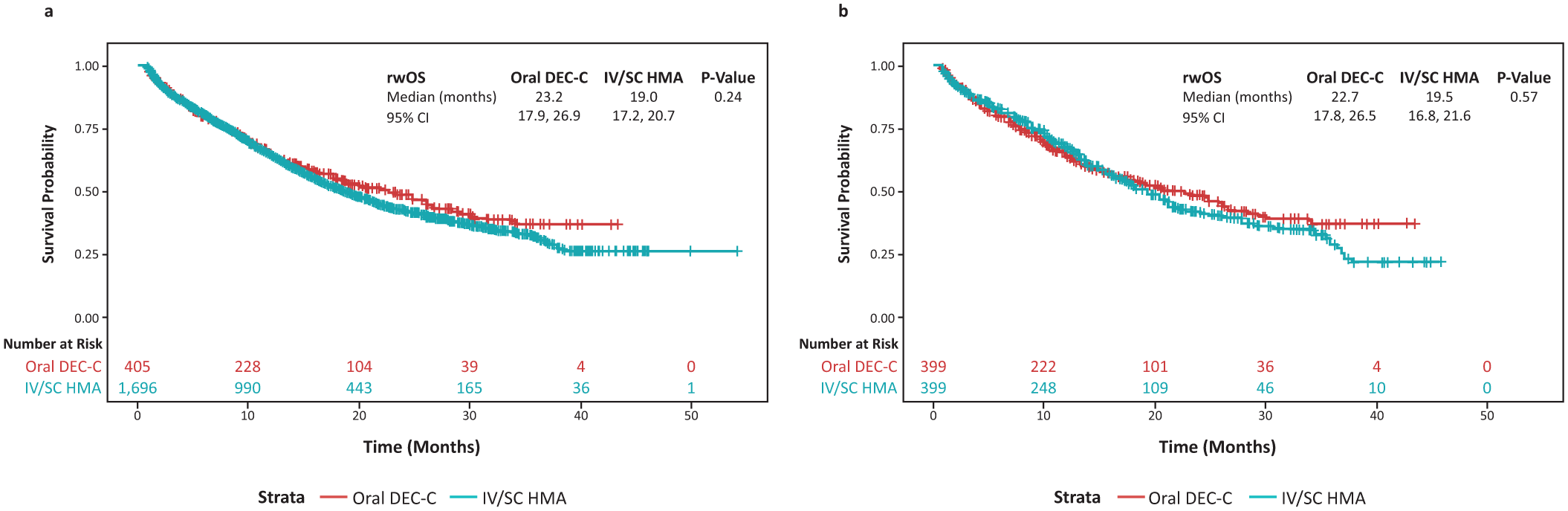
Real-world overall survival by HMA treatment: (a) pre- and (b) post-propensity score matching.
Within the matched cohorts, 198 of the 399 IV/SC HMA patients and 174 of the 399 DEC-C patients died during follow-up. rwOS was not significantly different in patients treated with DEC-C and patients treated with IV/SC HMA (median 22.7 months vs 19.5 months, respectively; p = 0.57; Figure 1(b)). While the 1-year survival rate was higher among IV/SC HMA patients than DEC-C patients (68.2% vs 64.6%, respectively), the 2-year survival rate was higher among DEC-C patients than IV/SC HMA patients (48.5% vs 42.5%, respectively; Supplemental Table 3).
In the overall study, 452 of the 1696 IV/SC HMA patients and 71 of the 405 DEC-C patients progressed to AML. In the AML-free survival analysis, 1011 of the 1696 IV/SC HMA patients and 207 of the 405 DEC-C patients progressed to AML or died. The overall median AML-free survival was 13.7 months, and the DEC-C cohort had a significantly longer AML-free survival compared to the IV/SC HMA cohort (16.5 months vs 13.3 months; p = 0.01; Figure 2(a)). Within the matched study, 101 of the 399 IV/SC HMA patients and 71 of the 399 DEC-C patients progressed to AML. In the AML-free survival analysis, 236 of the 399 IV/SC HMA patients and 205 of the 399 DEC-C patients progressed to AML or died; here the AML-free survival was still numerically longer among DEC-C patients compared to IV/SC HMA patients (median 16.1 months vs 14.3 months, respectively), but this difference did not reach statistical significance (p = 0.10; Figure 2(b)). DEC-C patients had a higher AML-free rate than IV/SC HMA patients at 1 year (57.1% vs 55.5%) and 2 years (40.7% vs 34.0%, respectively; Supplemental Table 4).
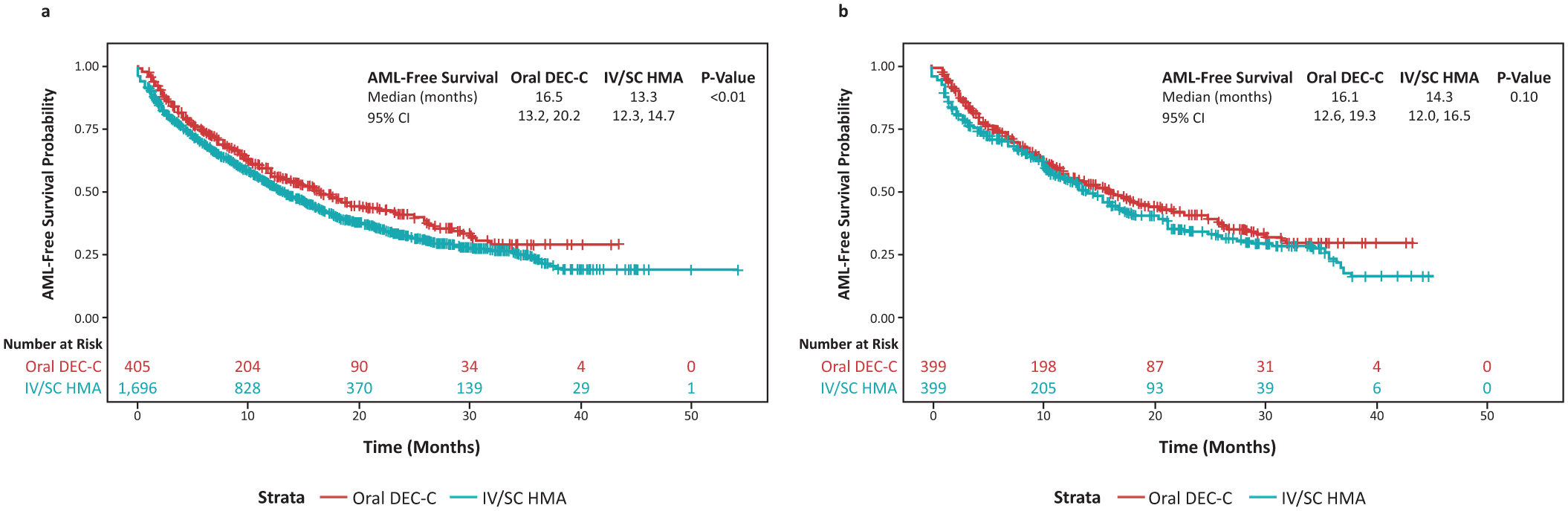
Real-world AML-free survival by HMA treatment: (a) pre- and (b) post-propensity score matching.
Most patients (1245/1696 IV/SC HMA; 257/405 DEC-C) had a treatment following their index HMA treatment. The median rwTTNT was 9.4 months for the oral DEC-C cohort and 7.4 months for the IV/SC HMA cohort (p < 0.01; Figure 3(a) and Supplemental Table 5). After PS matching, 294/399 IV/SC HMA and 252/399 DEC-C patients had a treatment following the index HMA treatment. DEC-C patients had a significantly longer median rwTTNT (9.3 months) than IV/SC HMA patients (7.8 months; p = 0.02; Figure 3(b) and Supplemental Table 5).

Real-world time to next treatment by HMA treatment: (a) pre- and (b) post-propensity score matching.
Exploratory analysis
Exploratory analyses were performed within the full, unmatched cohort. Clinical outcomes were compared between DEC-C patients separately with azacitidine and decitabine. Patients who received oral DEC-C had numerically longer median rwOS compared to those treated with azacitidine (23.2 months vs 19.5 months), but this difference was not statistically significant (p = 0.36). Patients who received oral DEC-C also had numerically longer median rwOS compared to those treated with decitabine (23.2 months vs 17.2 months), but this difference was not statistically significant (p = 0.12). Patients who received oral DEC-C had numerically longer median time to AML-free survival compared to those treated with azacitidine (16.5 months vs 14.7 months), but this difference was not statistically significant (p = 0.10). Patients who received oral DEC-C had a significantly longer median time to AML-free survival compared to those treated with decitabine (16.5 months vs 11.0 months; p < 0.01). For rwTTNT, patients who received oral DEC-C had significantly longer median compared to those treated with azacitidine (9.4 months vs 8.3 months; p = 0.01). Patients who received oral DEC-C also had a significantly longer median rwTTNT compared to those treated with decitabine (9.4 months vs 6.1 months; p < 0.01).
DEC-C patients had significantly better persistence compared to IV/SC patients. In the first 140 days after the index, the DEC-C cohort had a higher proportion of patients who received at least five cycles of index treatment (49.5% vs 41.8%; p < 0.01; Figure 4). Among the DEC-C patients, 157 were defined as persistent while 160 were nonpersistent. DEC-C persistent patients had longer median rwOS compared to the nonpersistent DEC-C patient group, but results were not statistically significant (median 29.4 months vs 26.9 months, respectively; p = 0.99).

Number of cycles of initial HMA treatment by type of HMA.
Discussion
This real-world study compared the clinical outcomes of patients with MDS treated initially with oral DEC-C and IV/SC HMAs. We observed that rwOS was comparable among patients treated with oral DEC-C and IV/SC HMAs; however, those treated with oral DEC-C had longer rwTTNT. While not reaching statistical significance, AML-free survival was consistently numerically longer among patients treated with DEC-C.
This study adds to the literature by being among the first and largest to examine clinical outcomes in MDS patients who initiated treatment with oral DEC-C compared to IV/SC HMAs in the real-world setting. We used PS matching to create more equivalent patient populations by balancing the patient demographics and clinical characteristics while examining these clinical outcomes.
The median overall survival for oral DEC-C was lower than that reported in the ASCERTAIN clinical trial—22.7 months vs 31.8 months. This difference in reported overall survival is likely due to the strict inclusion criteria for the clinical trial. The median overall survival was, however, very similar for oral DEC-C patients in this study as previously reported in the real-world setting—22.7 months vs 21.6 months in a prior study. 19 Our study also reports a median time to AML or death of 16.1 months for patients who received oral DEC-C and 14.3 months for patients who received IV/SC HMA. This result is slightly longer than the previously reported median time to AML or death of 12.1 months in a phase II study involving patients who received either oral DEC-C or IV decitabine. 20 While our study reports that patients who received oral DEC-C had a longer time to AML or death compared with those who received IV/SC HMA, the difference between the two groups is not statistically significant when analyzing the matched cohorts. This is likely due to the small number of events and insufficient follow-up period for long-term outcomes.
The significantly longer rwTTNT noted for oral DEC-C patients could be due to the longer persistence of treatment observed for patients who received oral DEC-C compared with IV/SC HMA, which was noted in this study and a previous study. 14 We observed that oral DEC-C patients were more likely to persist with treatment beyond four cycles of treatment than IV/SC HMA patients. The persistence may in part be explained by prior findings that patients on oral DEC-C who had previously received IV/SC HMAs felt that oral DEC-C interfered less with their daily life and that it improved their quality of life. 13 Oral DEC-C has also been shown to improve patients’ reported quality of life by reducing the time spent receiving treatment in a healthcare facility and by allowing for more independence. 13 Since DEC-C is administered orally, it may be more convenient for patients to pursue long-term treatment than with an IV/SC-administered drug. The convenience of oral DEC-C may also improve adherence in patients with disease symptoms or comorbidities which would make receiving IV/SC HMAs more logistically challenging. Our group has also previously shown a reduction in time toxicity (which measures time spent by patients in healthcare settings) associated with the use of oral DEC-C compared to parenteral HMAs. 14 In this study, we also observed that, while not statistically significant, persistent patients had numerically better overall survival compared with nonpersistent patients. The link between persistence and improved survival should be explored further in future larger studies.
Our study has several strengths, including the large sample size with long-term follow-up and the wider representation (compared to clinical trials) of the general US population with MDS who are receiving treatment primarily in community settings. In addition, PS matching was utilized as a robust method to address confounding. In this study, we were able to examine not only rwOS but also time to AML diagnosis, a meaningful time point for MDS.
Our study also has important limitations, however, including the reliance on structured EMR data where missing data could cause potential misclassification of patients. Additionally, structured EMR data contains limited information related to the risk status of patients with MDS, an important driver of outcomes in this patient population. This study relied on the identification of high-risk MDS through ICD-10 diagnosis codes. The structured EMR data also do not contain information on data such as cytogenetics found in unstructured EMR data. Additionally, structured EMR data do not contain information regarding complications, infections, transfusion/need dependency, or therapy-related MDS, so these topics could not be evaluated in this patient population. Finally, this study reflects the treatment practices within the oncology practices in the United States that are a part of the ConcertAI network and may not be generalizable to different patient populations. Future studies are needed with larger sample sizes and longer follow-up periods to explore the correlation between persistence and rwOS as well as the inclusion of data from unstructured documents to analyze outcomes by risk status.
Conclusion
This real-world study is among the first and largest to date that examines clinical outcomes and real-world use patterns among MDS patients who initiated treatment with oral DEC-C compared to IV/SC HMAs and utilized PS matching to control for confounding while examining clinical outcomes. While the study results indicate comparable rwOS and time to AML-free survival among patients treated with oral DEC-C and those treated with IV/SC HMAs, patients treated with oral DEC-C had a significantly longer rwTTNT and longer persistence of treatment. Along with our previous findings, these findings strongly support the use of oral DEC-C as an alternative to parenteral HMA therapy.
Supplemental Material
sj-docx-1-tah-10.1177_20406207261421326 – Supplemental material for Real-world study of use patterns and clinical outcomes for patients with myelodysplastic syndrome initiating oral decitabine and cedazuridine or intravenous/subcutaneous hypomethylating agents
Supplemental material, sj-docx-1-tah-10.1177_20406207261421326 for Real-world study of use patterns and clinical outcomes for patients with myelodysplastic syndrome initiating oral decitabine and cedazuridine or intravenous/subcutaneous hypomethylating agents by Amer M. Zeidan, Ruizhi Zhao, Jon G. Tepsick, Ziyu Lan and Tehseen Salimi in Therapeutic Advances in Hematology
Supplemental Material
sj-docx-2-tah-10.1177_20406207261421326 – Supplemental material for Real-world study of use patterns and clinical outcomes for patients with myelodysplastic syndrome initiating oral decitabine and cedazuridine or intravenous/subcutaneous hypomethylating agents
Supplemental material, sj-docx-2-tah-10.1177_20406207261421326 for Real-world study of use patterns and clinical outcomes for patients with myelodysplastic syndrome initiating oral decitabine and cedazuridine or intravenous/subcutaneous hypomethylating agents by Amer M. Zeidan, Ruizhi Zhao, Jon G. Tepsick, Ziyu Lan and Tehseen Salimi in Therapeutic Advances in Hematology
Footnotes
Acknowledgements
The authors would like to thank Kathryn Penney (ConcertAI) and Yuexi Wang (ConcertAI) for their contributions in drafting and editing this manuscript. Part of this research was presented as a poster at the ASH Annual Meeting and Exposition, December 7–10, 2024.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
